Abstract
Stroke is a significant health concern impacting society and the health care system. Reperfusion therapy for acute ischemic stroke and standard rehabilitative therapies may not always be effective at improving post-stroke neurological function, and developing alternative strategies is particularly important. Vagus nerve stimulation (VNS) is a treatment option currently approved by the Food and Drug Administration (FDA) for intractable epilepsy, refractory depression, primary headache disorders, obesity, and moderate to severe upper-limb motor dysfunction in chronic ischemic stroke patients. Moreover, VNS has demonstrated potential efficacy in various conditions, including autoimmune diseases, disorders of consciousness, Alzheimer’s disease, Parkinson’s disease, traumatic brain injury, stroke, and other diseases. Although the popularity and application of VNS continue to increase rapidly, the field generally lacks a consensus on the optimal stimulation parameters. The stimulation parameters for VNS are directly related to the clinical outcome, and determining the optimal stimulation conditions for VNS has become an essential concern in its clinical application. This review summarizes the current evidence on VNS for stroke in preclinical models and clinical trials in humans, paying attention to the current types and stimulation parameters of VNS, highlighting the mechanistic pathways involved in the beneficial effects of VNS, critically evaluating clinical implementation challenges and proposing some suggestions for its future research directions. Achieving safe and effective clinical transformation of VNS requires further animal and clinical studies to determine the optimal stimulation parameters and therapeutic mechanisms.
Introduction
Stroke is a significant health concern impacting society and the health care system. Numerous individuals suffering from a stroke experience long-term neurological dysfunction, which adversely affects their well-being as well as the broader socioeconomic landscape. The number of stroke patients worldwide is projected to reach approximately 200 million by 2050. 1 Ischemic stroke (IS) accounts for approximately 87% of all strokes. 2 Notably, current treatments for acute ischemic stroke (AIS), such as intravenous thrombolysis and mechanical thrombectomy, 3 have certain limitations in their clinical application, as only 10% of patients can receive these treatments within the appropriate treatment time window. 2 These limitations have contributed to the high mortality and disability rates associated with stroke. Additionally, approximately 60% of stroke patients experience only partial recovery or no recovery at all within 6 months. 4 These findings indicate that reperfusion therapy in AIS and standard rehabilitative therapies may not always be effective at improving post-stroke neurological function, and developing alternative strategies is particularly important. Therefore, it is imperative to explore and devise novel supplementary treatment methods that can effectively enhance neurological function recovery in patients with stroke and can be seamlessly incorporated into established protocols such as thrombolysis, thrombectomy, and rehabilitative therapies.
Neuromodulation techniques are being used more frequently in clinical and fundamental neuroscience. Vagus nerve stimulation (VNS) is a well-established form of neuromodulation that has been extensively utilized in the treatment of neurological diseases and has been demonstrated to be a safe and effective treatment method over a significant period of time. VNS is currently primarily used for the clinical management of refractory epilepsy, but its potential applications extend far beyond that. The intricate and extensive fiber connections established by the vagus nerve with the central and peripheral nerves make VNS a promising prospect for diverse clinical applications. It is worth noting that VNS is a treatment option currently approved by the Food and Drug Administration (FDA) for intractable epilepsy, 5 refractory depression, 6 primary headache disorders, 7 and obesity. 8 Moreover, VNS has demonstrated potential efficacy in various conditions, including autoimmune diseases, 9 disorders of consciousness, 10 Alzheimer’s disease, 11 Parkinson’s disease, 12 traumatic brain injury (TBI), 13 stroke, 14 and other diseases. Further investigations into VNS and its implementation in other fields are anticipated.
Given that VNS can target multiple neuroprotective and neuroplasticity pathways, it holds great potential for treating IS. The FDA approved VNS for treating moderate to severe upper-limb motor dysfunction in patients with chronic IS in 2021, 15 which supports using VNS as a new therapeutic option for limb paralysis caused by IS. The study conducted by VNS-REHAB 15 revealed that participants who underwent VNS along with rehabilitation showed significant improvements in upper-limb function compared to those who received rehabilitation and sham stimulation. In the VNS group, almost half of the participants achieved clinically meaningful improvements, approximately twice as many as in the control group. Moreover, the improvement in Fugl-Meyer Assessment-Upper Extremity (FMA-UE) scores corresponded to the improvement observed in the Wolf Motor Function Test (WMFT) scores, and various quality-of-life rating scales also indicated more remarkable improvement in the VNS group.
Current clinical studies of VNS primarily focus on the recovery of neurological functions after chronic or subacute stroke.15-18 However, an increasing number of animal studies have shown that VNS is effective in reducing the infarction volume of stroke, promoting neurological function recovery, and improving motor and cognitive dysfunctions after stroke.19-23 Although the clinical application of VNS for stroke treatment has advanced rapidly, several scientific research issues still require further investigation. These issues mainly include the underlying mechanisms of VNS and the consistency of stimulation parameters in stroke treatment. It is recommended that future studies prioritize investigation of the potential mechanisms of VNS, determination of the optimal stimulation parameters for stroke treatment, and expansion of its clinical application.
This review briefly summarizes the application and potential mechanism of VNS in stroke, paying particular attention to the various stimulation parameters used in preclinical and clinical studies related to stroke, aiming to lay a groundwork for future research.
Anatomy and function of the vagus nerve
Vagus nerve is the 10th pair of cranial nerves and is classified as a mixed nerve. It controls the activities of all visceral organs except the adrenal gland and plays a crucial role in digestion, metabolism, respiration, cardiovascular regulation, emotional and immune regulation. 24 The vagus nerve is the longest and most widely distributed cranial nerve and is a major component of the parasympathetic nervous system. The nerve originates from a series of rootlets in the medulla oblongata of the brainstem, passes through the jugular foramen, enters the carotid sheath, travels along the posterior aspect of the internal carotid artery and internal jugular vein to the root of the neck, and extends to the thorax and abdomen, innervating the corresponding organs and terminating the colon. 25 The two sides of the vagus nerve play distinct roles in controlling the heart. The right vagus nerve primarily influences the sinoatrial node, while the left vagus nerve plays a vital role in the atrioventricular junction. Since the sinoatrial node governs heart rate, stimulating the right vagus nerve is not advisable in humans or large animals due to its impact on cardiac function. Conversely, this concern does not apply to rodents. 26 However, a trial involving 59 patients with chronic heart failure comparing left vs right implanted VNS demonstrated that both left and right VNS are not only equally effective but also equally safe. 27 The vagus nerve is composed of 80% afferent fibers (sensory fibers) and 20% efferent fibers (motor fibers). 28 In the region around the jugular foramen, there are two vagal ganglia, namely the superior ganglion (jugular ganglion) and the inferior ganglion (nodose ganglion), with the cell bodies of the vagal afferent fibers mainly located in the latter ganglion. 29 The auricular branch of the vagus nerve (ABVN) is the only afferent branch of the vagus nerve that exists on the surface of the body, and it provides innervation to the external auditory canal, inner tragus, and skin around the cymba concha. Specifically, the ABVN completely innervates the cymba concha of the outer ear. 30
The afferent fibers of the vagus nerve transmit signals from organs like the heart, lungs, trachea, liver, and gastrointestinal tract to the nucleus of tractus solitarius (NTS) in the brainstem. The NTS then projects these signals directly or indirectly to various key areas of the brain, including the locus coeruleus (LC), dorsal raphe nucleus (DRN), amygdala, hippocampus, and cerebral cortex.
31
In contrast, the efferent fibers transmit parasympathetic cholinergic signals from the nucleus ambiguus and dorsal motor nucleus (DMN) to target organs such as the spleen, digestive tract, and heart.
32
Consequently, this circulatory pathway of the vagus nerve serves as an important connection for bidirectional signal transmission between the brain and the periphery (Figure 1). Pathway of the vagus nerve for bidirectional signal transmission between the brain and the periphery. The afferent vagus nerve conveys signals from peripheral organs to the nucleus of the tractus solitarius (NTS) in the brainstem, from which these signals are subsequently projected to various key areas of the brain, including the locus coeruleus (LC) and others. In contrast, the efferent vagus nerve transmits central signals from the dorsal motor nucleus (DMN) to peripheral organs.
According to the classical Erlanger/Gasser classification, the vagus nerve consists of A, B and C fibers with corresponding conduction velocities. 7 Among the afferent fibers, C fibers are thin and unmyelinated, Aδ fibers are thin but myelinated, and Aβ fibers are thicker and myelinated. Among the efferent fibers, Aα fibers are the thickest myelinated axons of α-motoneurons and specifically innervate the pharyngeal and laryngeal muscles, and B fibers are small and myelinated and are responsible for carrying parasympathetic inputs to visceral organs.29,33 A, B, and C fibers differ in nerve conduction velocities and activation thresholds. Specifically, Aβ fibers have the lowest threshold and are activated first, while C fibers require a higher current intensity for activation. 34 Studies have demonstrated that VNS therapeutic effect are most likely associated with the electrical activation of A and B fibers but not C fibers. 35 Non-invasive transcutaneous VNS primarily targets A fibers and has been shown to reduce seizures. 36 Assessing the physiological effect of VNS in patients, Banzett et al 37 found instances of hoarseness linked to Aα fibers, while cardiovascular effects associated with B fibers and throat burning sensations, coughing, alterations in breathing patterns related to C fiber. Consequently, understanding the anatomy of the vagus nerve and the characteristics of vagus nerve conduction velocities and discharge thresholds is essential for the successful implementation of VNS therapy.
Classification of VNS
At the end of the 19th century, the American neurologist Corning first proposed the concept of VNS, which involved combining a carotid artery compression instrument with transcutaneous vagus nerve electrical stimulation and cervical sympathetic nerve stimulation to treat epilepsy. 38 However, this technique was not mature and was not fully developed or widely adopted by neurologists. In 1988, Penry JK et al 39 performed the world’s first VNS surgery on an epilepsy patient, which significantly reduced the frequency of seizures. After more than 30 years of development and continuous technological advancements, the safety and effectiveness of VNS have been validated, and various types of VNS devices have emerged, including invasive VNS (iVNS) and non-invasive VNS (nVNS).
iVNS is performed by surgically wrapping electrodes around the vagus nerve in the neck and connecting the electrodes to a pulse generator implanted below the clavicle with an extension wire. 16 This system delivers intermittent electrical pulses to the vagus nerve, stimulating the function of related mechanisms. iVNS can provide long-term stable stimulation and more substantial stimulation effects, and personalized treatment can be achieved by adjusting parameters such as current intensity, frequency, pulse width, and duration.
While iVNS has benefits, there are certain limitations to its use. As an alternative, nVNS, also known as transcutaneous VNS (tVNS), may be a more suitable option due to its less invasive nature, consistent therapeutic effects, and relatively lower costs. Two types of tVNS devices are available: transcutaneous cervical VNS (tcVNS) and transcutaneous auricular VNS (taVNS). tcVNS involves the application of two skin electrodes to the neck overlying the vagus nerve for transcutaneous stimulation, typically administered through a handheld device such as GammaCore or ElectroCore. This method has been approved by the FDA for the acute treatment of migraine as well as the acute treatment and prevention of episodic cluster headache. 40 Although this approach is convenient, it has some adverse effects. To stimulate the vagus nerve, tcVNS requires a relatively stronger stimulus intensity to penetrate the skin barrier. As a result, the stimulation field in the neck is diffuse, which may lead to stimulation of the non-vagus nerves in the neck and the efferent neck fibers. taVNS involves the combination of acupuncture and VNS techniques, 41 utilizing electrodes with adjustable parameters that are placed on the skin surface to stimulate the ABVN with electrical impulses, such as the NEMOS. In a functional magnetic resonance imaging (fMRI) involving 37 healthy subjects, Yakunina et al 42 investigated the blood oxygen level dependent (BOLD) signal following transcutaneous stimulation of the auricular branch of the vagus nerve at four sites: the inner tragus, inferoposterior wall of the ear canal, cymba con;chae, and earlobe. Region-of-interest (ROI) analysis revealed that stimulation of the cymba conchae significantly activates the NTS and LC, thereby engaging the vagus nerve pathway to a greater extent than stimulation at other sites. Consequently, the cymba conchae is considered the most effective anatomical site for stimulation. taVNS has been shown to be generally safe, with only mild and temporary adverse effects, and has also been shown to be effective for a range of ailments, including epilepsy, major depressive disorders, insomnia, glucose metabolic disorders, pain, stroke, post-stroke rehabilitation, anxiety, fear, cognitive impairment, cardiovascular disorders, tinnitus, Prader-Willi syndrome, and COVID-19. 43
Potential mechanisms of VNS in stroke treatment
Figure 2 depicts potential mechanisms of VNS in stroke. Research on the use of VNS for treating intracerebral hemorrhage (ICH) is currently limited. Animal experiment has shown that VNS can improve functional recovery after ICH by reducing inflammation, promoting neuroplasticity, and facilitating vascular regeneration and repair in the brain.
44
An observational clinical study suggested that VNS may have a positive impact on recovery after ICH.
14
VNS may also have the potential to improve patients’ cognitive, motor, and language functions and to reduce psychological symptoms such as depression and anxiety.
45
However, these findings require validation through large-scale randomized controlled trials. In addition to potentially reducing inflammation and promoting neuroplasticity, VNS may benefit ICH patients by regulating the autonomic nervous system and enhancing cerebral blood flow (CBF) and oxygen metabolism.
46
It is crucial to highlight that current research on VNS for ICH treatment is still relatively limited, necessitating further large-scale clinical and animal studies to confirm its efficacy and mechanisms and determine the optimal treatment plans and indications. Potential mechanisms of vagus nerve stimulation (VNS) in stroke. It can be broadly summarized as the following processes: inhibition of oxidative stress and reduction of excitotoxicity, decrease of the neuroinflammatory response, protection of the blood-brain barrier (BBB) and reduction of cerebral edema, inhibition of nerve cell apoptosis, enhancement of neuroplasticity, suppression of cortical spreading depolarization, and promotion of neurogenesis and angiogenesis.
The mechanism of VNS for treating IS can be divided into two main categories: neuroprotection during the early stage of focal ischemia and neuroplasticity in the later stage of stroke. Neuroprotection aims to reduce neuron death in the ischemic penumbra, while neuroplasticity focuses on maximizing the repair of ischemic damage. Neuroprotection has been suggested to enhance the ability of neural cells to withstand cerebral ischemia. Potential neuroprotective therapies act by interrupting or reversing detrimental processes such as oxidative stress, excitotoxicity, inflammation, apoptosis and mitochondrial dysfunction. 47 Stroke-induced damage to the neurovascular unit can lead to impaired synaptic connections and neuronal processes. Possible mechanisms of VNS-induced neuroplasticity in IS range from activity-dependent rewiring and the strengthening of synapses to the formation of new neurons and blood vessels. 48 Although the precise mechanism through which VNS improves the prognosis of IS remains unclear, it can be broadly summarized as the following processes: inhibition of oxidative stress and reduction of excitotoxicity, decrease of the neuroinflammatory response, protection of the blood-brain barrier (BBB) and reduction of cerebral edema, inhibition of nerve cell apoptosis, enhancement of neuroplasticity, suppression of cortical spreading depolarization (CSD), and promotion of neurogenesis and angiogenesis.
Inhibition of oxidative stress and reduction of excitotoxicity
Oxidative stress is an imbalance between the production and elimination of cellular free radicals. After ischemia, the excessive release of excitatory neurotransmitters like glutamate results in the overproduction of free radicals or reactive oxygen species, and subsequent reperfusion leads to the further production of large amounts of reactive oxygen species, both of which are key players in ischemic brain injury. 49 VNS markedly reduced ischemia-induced glutamate release and decreased reactive oxygen species production by decreasing blood flow to the hippocampus during reperfusion. 50 Additionally, in a separate study, the concentration of the inhibitory neurotransmitter GABA in the cerebrospinal fluid (CSF) of VNS-treated patients was notably increased, and GABA can reduce excitotoxicity, which may also contribute to the protective effect of VNS on cerebral ischemic tissues. 51
Decrease of the neuroinflammatory response
Neuroinflammation is recognized as a critical component of stroke pathophysiology, affecting both the acute and chronic phases of the disease. It can worsen brain damage, cause secondary injury, hinder tissue repair and recovery, and contribute to post-stroke complications. VNS inhibits the activation of inflammatory cells such as microglia and suppresses cytokine production, thereby preventing cytokine-induced tissue damage in various pathological conditions. 52 The cholinergic anti-inflammatory pathway (CAIP) is a neuroimmune pathway involving the vagus nerve that was recently shown to modulate inflammatory responses in the central nervous system (CNS) and to play a significant role in regulating neuroinflammation following IS. 53 After cerebral ischemia, VNS can trigger the CAIP, prompting T cells to release acetylcholine, which interacts with the α7 nicotinic acetylcholine receptor (α7nAchR) on the surface of inflammatory or immune cells, ultimately decreasing the release of pro-inflammatory factors.54,55 The α7nAchR is a key target for the anti-inflammatory effects of VNS in IS. 56 A recent study suggested that the neuroprotective impact of VNS on stroke could be linked to the activation of the α7nAchR anti-inflammatory pathway and the suppression of inflammatory responses. 57 In a systematic review of meta-analysis examining the effect of VNS on inflammatory cytokines, which included 36 clinical studies, it was revealed that anti-inflammatory effects of VNS for cytokines in several reports. However, the meta-analysis revealed that, despite a numerical reduction in most cytokines, this reduction did not achieve statistical significance for either long-term or short-term stimulation. Only the subgroup analysis of 4 long-term stimulation studies with acute inflammation was significant: VNS decreased CRP significantly more than sham stimulation. 58
Protection of the blood-brain barrier and reduction of cerebral edema
Stroke can result in BBB disruption. A preclinical study demonstrated that VNS can help restore BBB function after stroke. 23 tcVNS induced neuroprotective effects by reducing the BBB transfer rate and serum IgG leakage, protecting vascular tight junction proteins, and decreasing the expression of matrix metalloproteinase-2/9 in reactive astrocytes around compromised vessels in the model of IS. 23 Disruption of the BBB and serum protein extravasation can lead to vasogenic brain edema. 59 VNS-mediated reductions in cerebral edema have been demonstrated in TBI rats,60,61 suggesting that VNS also plays an important role in regulating IS-induced cerebral edema, but this finding requires validation.
Inhibition of nerve cell apoptosis
Ischemia-reperfusion (I/R) injury induces the upregulation of apoptotic factor expression and Ca2+ overload in neurons, thereby triggering the mitochondrial apoptotic pathway. Caspase-3 is a key protease in the mitochondrial apoptotic pathway, and its expression increases with prolonged ischemic duration. The results from one study demonstrated a notable decrease in Caspase-3 levels in the VNS treatment group compared to the control group, indicating that VNS may be able to suppress the apoptotic response and safeguard ischemic neural tissue. 62
Enhancement of neuroplasticity
The enhancing effect of VNS on neuroplasticity has been consistently demonstrated. 63 A preclinical study suggested that VNS combined with rehabilitation can triple synaptic connectivity in damaged corticospinal tract networks. 64 This specific type of neuroplasticity is believed to be triggered by VNS-induced molecular neuronal mechanisms, such as the activation of the noradrenergic, cholinergic, and serotonergic systems. 65 Brain plasticity plays a crucial role in spontaneous recovery after severe CNS damage, suggesting that targeted interventions for neuroplasticity are essential for expediting recovery from stroke. 66 Furthermore, evidence indicates that VNS may influence memory processes by affecting neuroplasticity in the hippocampus of rats. 67 These findings support the theory that VNS can enhance neuroplasticity, which is thought to underlie motor and cognitive recovery following ischemic injury.
Suppression of cortical spreading depolarization
Recurrent CSD waves negatively affect neuronal survival and outcomes after AIS. CSD, a powerful depolarization wave originating from the ischemic penumbra, gradually spreads through the gray matter, leading to arterial constriction in the affected brain region. This process imposes significant metabolic demands, potentially leading to imbalances between oxygen supply and demand, exacerbating post-stroke symptoms and increasing the risk of secondary brain injury. 68 Some studies have even proposed CSD as a potential treatment target, offering a novel therapeutic approach to safeguard vulnerable nerve cells. In a rat stroke model, both iVNS and nVNS significantly reduced the frequency of peri-infarct CSD compared to sham VNS, without affecting relative blood flow changes, blood pressure, heart rate, or respiratory rate. 69 The inhibitory effect of VNS on CSD was also observed in an experimental model of migraine.70,71
Promotion of neurogenesis and angiogenesis
A significant body of evidence suggests that enhancing the regenerative capabilities of nerve cells and blood vessels can facilitate the restoration of neurological function in stroke patients. 72 VNS has the potential to increase the levels of various neurotransmitters and growth factors crucial for neurotrophic processes in the brains of rats, thereby fostering the growth of nerve cells. Recent studies have demonstrated that taVNS boosts the expression of angiogenic factors, triggers endothelial cell proliferation, stimulates angiogenesis, enhances microvessel density around the ischemic area, and ultimately reduces infarct size while improving neurological symptoms.73,74
Stimulation parameters of VNS
Stimulation parameters are essential factors that influence the efficacy of VNS. The selection of stimulation parameters directly impacts the therapeutic outcome, with changes potentially leading to improved efficacy. Since the first VNS implantation in 1988, research has focused on identifying the most effective combination of stimulation parameters to achieve optimal therapeutic results. The parameters of VNS mainly include stimulus intensity, pulse width, stimulus frequency, stimulation and interval time (on-off time), and duration, which determine the type and number of nerve fibers that can be stimulated. In order to activate selectively targeted nerve fibers, further understanding of stimulation parameter settings is needed, which are mainly set to achieve effective stimulation and maximize therapeutic advantages.
VNS outputs electrical pulse waveforms as direct current (DC) square waves (Figure 3). Visual representation of direct current (DC) square wave parameters of vagus nerve stimulation (VNS). These parameters mainly include stimulus intensity, pulse width, stimulus frequency, stimulation and interval time (on-off time), and duration.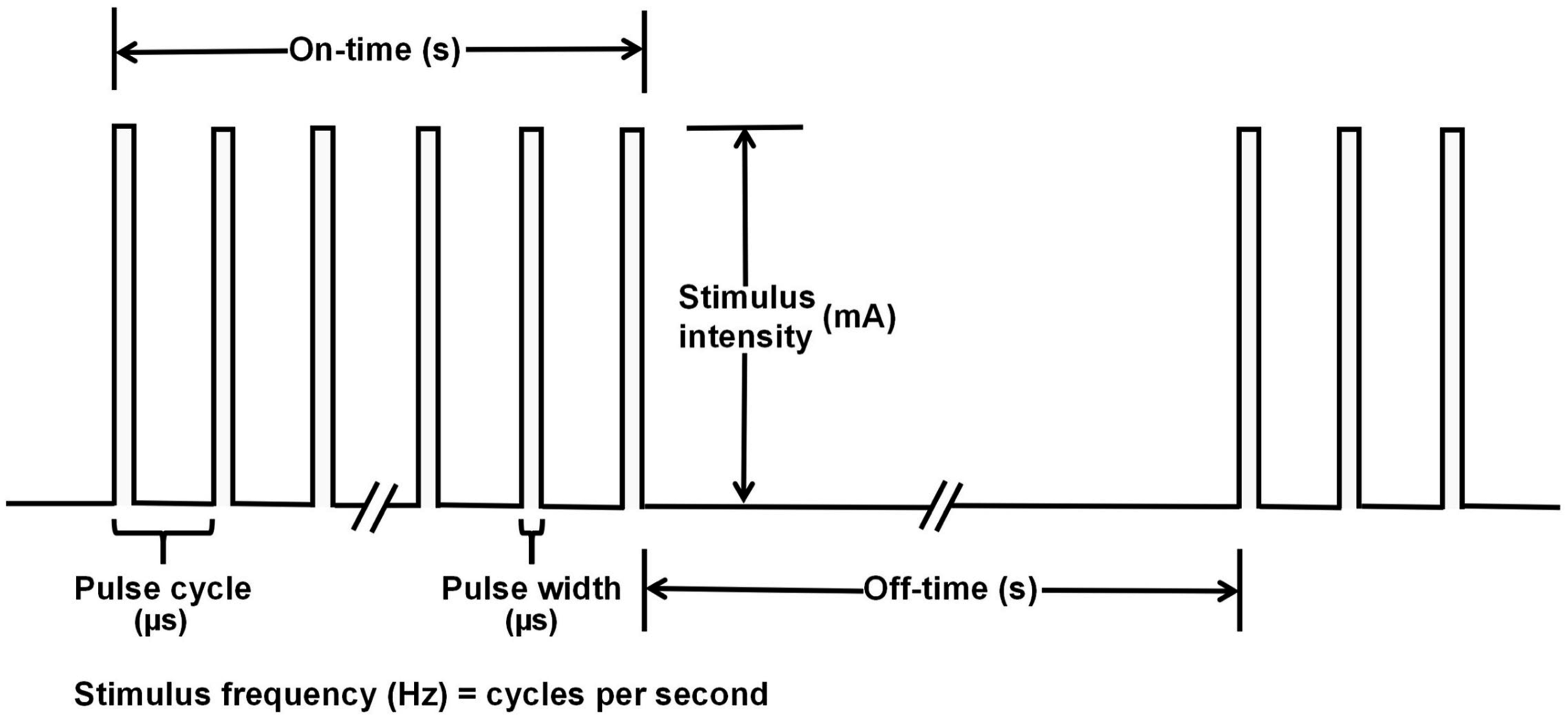
While most of these parameters have standard definitions, there are still some, such as “duration”, that have different meanings in different papers, and even some papers denote a difference in the frequency of VNS stimulation, such as once a day or twice a day.
Application of VNS
Although the mechanisms by which VNS affects stroke are not fully understood to date, its efficacy and safety have been continuously confirmed by preclinical and clinical stroke studies. The following section will discuss and summarize various VNS parameters and therapeutic effects based on findings from preclinical and clinical stroke studies.
Preclinical studies of VNS in stroke treatment
Main preclinical studies of iVNS in rodent models of stroke.
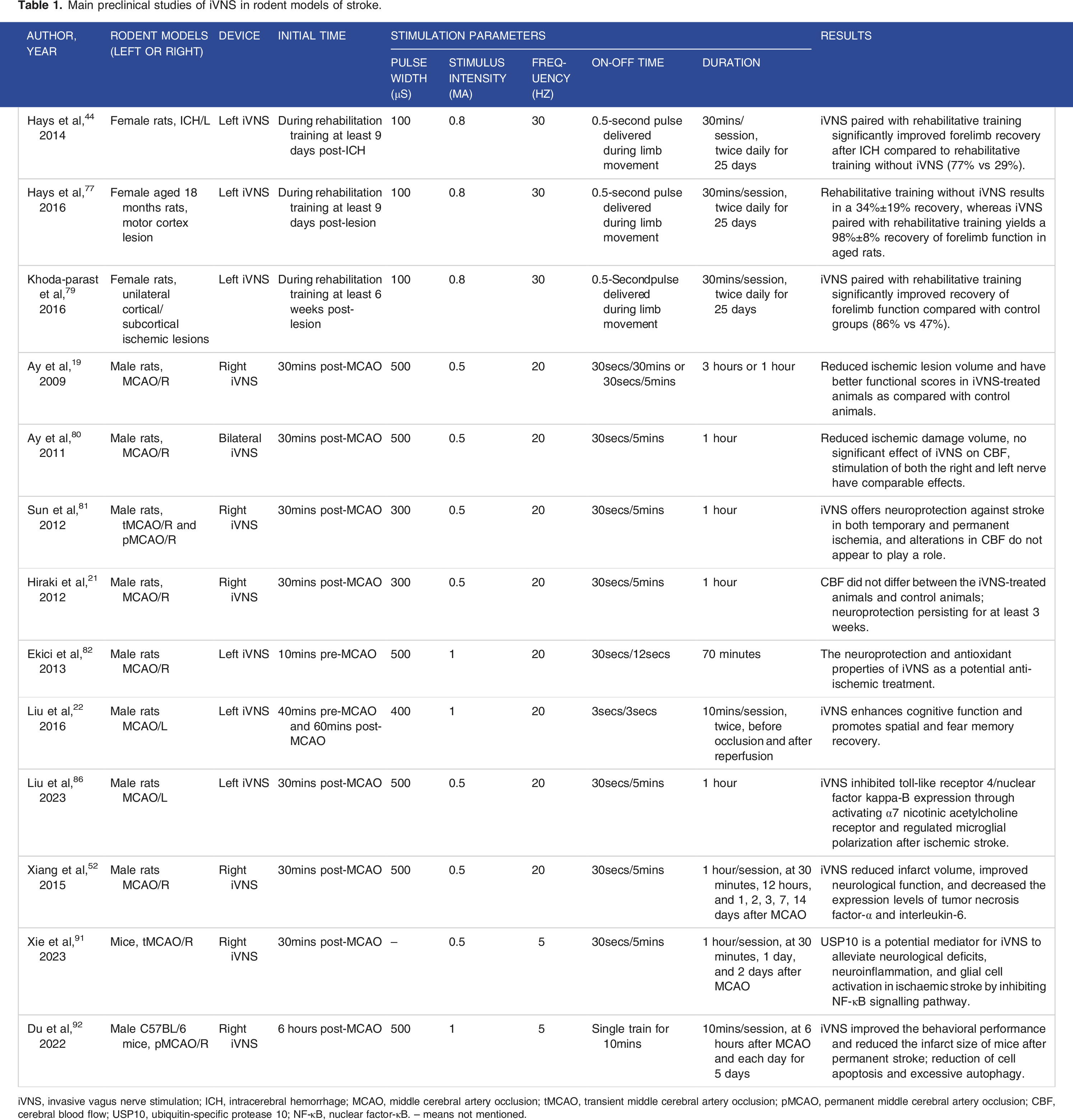
iVNS, invasive vagus nerve stimulation; ICH, intracerebral hemorrhage; MCAO, middle cerebral artery occlusion; tMCAO, transient middle cerebral artery occlusion; pMCAO, permanent middle cerebral artery occlusion; CBF, cerebral blood flow; USP10, ubiquitin-specific protease 10; NF-κB, nuclear factor-κB. -- means not mentioned.
Preclinical studies of tVNS in rodent models of stroke.

tVNS, transcutaneous vagus nerve stimulation; taVNS, transcutaneous auricular vagus nerve stimulation; tcVNS, transcutaneous cervical vagus nerve stimulation; MCAO, middle cerebral artery occlusion; tBCCAO, transient bilateral common carotid artery occlusion; BBB, blood-brain barrier; PPAR-γ, peroxisome proliferator-activated receptor-γ; α7nAchR, α7 nicotinic acetylcholine receptor; CSF, cerebrospinal fluid; VCI, vascular cognitive impairment. -- means not mentioned.
In iVNS research, only one study was carried out using a rat model of ICH. 44 The results showed that compared with rehabilitation training alone, the combination of iVNS and rehabilitation training significantly improved forelimb recovery after ICH. iVNS therapy was initiated during rehabilitation training at least 9 days after ICH and was conducted 5 days a week (only weekdays) for a total of 25 days. The therapy involved two 30-minute sessions per day separated by a 2-hour break. Stimulation was administered as a 0.5-second train of 15 pulses at 30 Hz, and each biphasic pulse had an amplitude of 0.8 mA and a pulse width of 100 μs. The same stimulation parameters were used to verify the effects of iVNS combined with rehabilitation exercises on forelimb function in rat models of IS, including chronic stroke (6 weeks after stroke) and acute stroke, in adult or aged rats.64,75-79 The results demonstrated that iVNS paired with rehabilitative training significantly improved forelimb function after IS and enhanced structural plasticity in motor networks, remarkably surpassing the outcomes of rehabilitation alone.
Ay et al 19 firstly performed a study in which iVNS was delivered for 30 seconds every 30 minutes for 3 hours in experimental group 1 and every 5 minutes for 1 hour in experimental group 2 beginning 30 minutes after the induction of focal cerebral ischemia in a rat model; the constant current was set at 0.5 mA and a 30-second train of 500 μs pulses was delivered at 20 Hz. The results indicated that the ischemic lesion volume in the iVNS-treated animals was nearly less than half of that in the control animals, and the functional score improved by 50% in experimental group 1 and 44% in experimental group 2 (P < 0.05 for both groups). Subsequently, the research group proceeded to stimulate both the left and right vagus nerves with identical parameters (0.5 mA, 500 μs, 20 Hz, 30 seconds/5 minutes) for 1 hour beginning 30 minutes after the induction of right-sided ischemia. The results showed that the ischemic damage volume in the stimulated animals was significantly reduced by 41%–45% compared to that in the control group. 80 This study suggested that the protective effects of iVNS against acute ischemic brain injury are not primarily attributed to changes in CBF. Stimulation of both the right and left vagus nerves has comparable effects, and iVNS is effective after ipsilateral and contralateral focal ischemia. When the vagus nerve was stimulated 30 minutes after middle cerebral artery occlusion (MCAO) in rats via 30-second pulse trains (0.5 mA, 300 μs, 20 Hz) every 5 minutes for 1 hour, iVNS offered neuroprotection against cerebral stroke in both temporary and permanent ischemia, and alterations in CBF did not appear to play a role. 81 Moreover, this neuroprotection persisted for at least 3 weeks. 21 In another study, iVNS (1 mA, 500 μs, 20 Hz, 30 seconds/12 seconds) was initiated 10 minutes before MCAO and continued for the first 30 minutes of reperfusion in a transient MCAO and reperfusion (MCAO/R) rat model, indicating that the neuroprotective and antioxidant properties of iVNS are potential anti-ischemic treatments. 82
iVNS also enhanced cognitive function and promoted spatial and fear memory recovery in a rat model of MCAO/R. 22 The stimulation parameters included an intensity of 1 mA at a fixed frequency of 20 Hz with a 400 μs bipolar pulse width for 3 seconds with a 3-second interval, lasting for 10 minutes before occlusion and after reperfusion, respectively. Among the 26 publications reviewed, 11 articles focused on exploring the potential mechanisms of iVNS in improving IS prognosis in rat models. These mechanisms include reducing neuroinflammation and inhibiting oxidative stress, autophagy, and apoptosis pathways to decrease cerebral I/R injury.52,57,62,83-90 These studies consistently applied uniform stimulation parameters that comprised a 500 μs pulse width of 30-second square pulses (0.5 mA, 20 Hz) every 5 minutes for 1 hour. Most studies applied stimulation for 1 hour starting 30 minutes after inducing ischemia, and only Xiang et al 52 administered stimulation for 1 hour at 30 minutes, 12 hours, and 1, 2, 3, 7, and 14 days post-ischemia.
Only two studies have been conducted on mice models of MCAO. Xie et al 91 conducted iVNS at 30 minutes, 24 hours, and 48 hours after transient MCAO, with parameters of 0.5 mA and 5 Hz for 30 seconds every 5 minutes for 1 hour (pulse width not mentioned). Their findings demonstrated that iVNS can increase the expression of ubiquitin-specific protease 10 (USP10), which is a potential mediator through which iVNS alleviates neurological deficits, neuroinflammation, and glial cell activation in IS by inhibiting the nuclear factor-κB (NF-κB) signaling pathway. Du et al 92 stimulated the vagus nerve with a 500 μs pulse width and 1 mA electric pulses for 10 minutes at 5 Hz every day from 6 hours after surgery for 5 days in a permanent MCAO mouse model and verified that iVNS could effectively improve the behavioral performance of mice after permanent stroke in addition to significantly reducing the infarct size of the brain tissue.
Among the 12 animal studies on tVNS, 8 articles utilized taVNS and 4 articles utilized tcVNS, 10 studies were carried out in ischemic rats, while 2 studies were conducted in ischemic mice. Ay et al 93 were the first to demonstrate that the delivery of taVNS (0.5 mA, 500 μs, 20 Hz) via a 30-second train at 5-minute intervals for 1 hour beginning 30 minutes after contralateral transient MCAO resulted in a 28% reduction in infarct volume in rats and led to sustained improvements in neurological function up to 24 hours post-stimulation. A follow-up study by the same group showed that tcVNS beginning 4 hours post-MCAO also reduced infarct volume. 20 Electrical stimulation (1000 μs pulse width, 12 V sine waves repeated at 25 Hz) was delivered in the form of 2-minute trains every 10 minutes for 1 hour. Similar tcVNS parameters have also been utilized in rat and mouse models of MCAO. These studies have shown that tcVNS can potentially decrease ischemic brain damage by safeguarding the integrity of the BBB, suppressing CSD, promoting microglial M2 polarization, and boosting anti-inflammatory activity.23,69,94
Studies have also shown that taVNS can protect against cerebral I/R injury by promoting angiogenesis and enhancing axonal plasticity73,74,95,96 using the same stimulation parameters as those used in previous studies, 93 differing only in duration. Vascular cognitive impairment (VCI) was induced by transient bilateral common carotid artery occlusion (tBCCAO) surgery in mice, and both long-term (20 minutes per stimulation for 6 days) and short-term (20 minutes per stimulation for 2 days) taVNS (1 mA, 330 μs, 20 Hz) stimulations were shown to facilitate CSF circulation and ameliorate VCI symptoms. 97 taVNS at 1 mA and 10 Hz for 30 minutes per day over 7 days (pulse width not mentioned) following cerebral I/R injury in rats was found to reduce pro-inflammatory factor levels and enhance motor function recovery. 98 Additionally, daily treatment with taVNS at 20 Hz and 2 mA with a 500 μs pulse width for 30 minutes for 3 weeks improved white matter repair and alleviated dysphagia symptoms in a rat model of MCAO. 99
VNS in patients with stroke
Clinical studies of iVNS in patients with stroke.
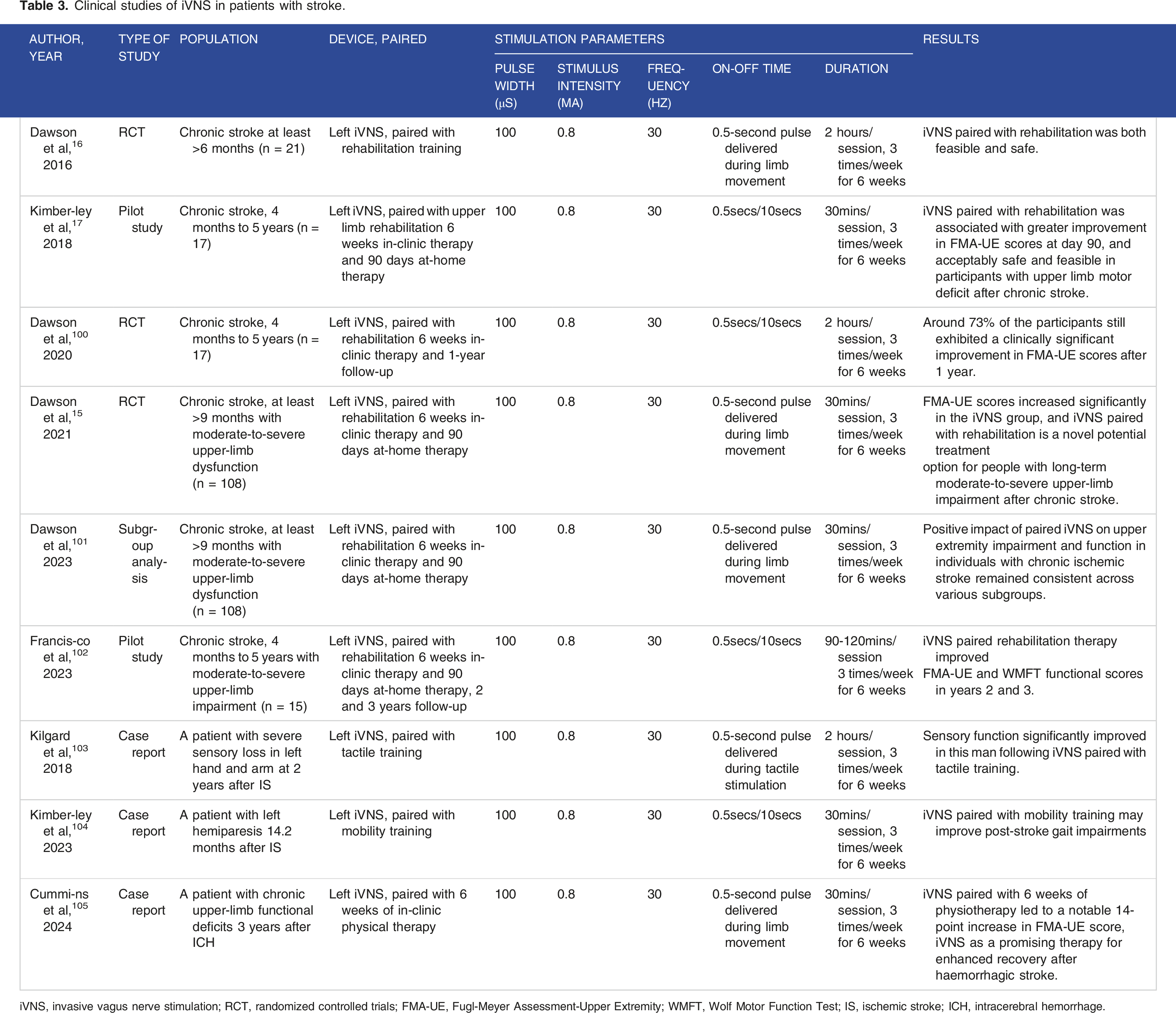
iVNS, invasive vagus nerve stimulation; RCT, randomized controlled trials; FMA-UE, Fugl-Meyer Assessment-Upper Extremity; WMFT, Wolf Motor Function Test; IS, ischemic stroke; ICH, intracerebral hemorrhage.
Clinical studies of tVNS in patients with stroke.

tVNS, transcutaneous vagus nerve stimulation; taVNS, transcutaneous auricular vagus nerve stimulation; tcVNS, transcutaneous cervical vagus nerve stimulation; RCT, randomized controlled trials; FMA-UE, Fugl-Meyer Assessment-Upper Extremity; MAAVNS, motor-activated auricular vagus nerve stimulation; AIS, acute ischemic stroke; ICH, intracerebral hemorrhage; -- means not mentioned.
Pairing iVNS with rehabilitation has been shown to enhance post-stroke neuroplasticity and facilitate the recovery of forelimb motor function in preclinical animal models. Dawson et al 16 conducted the first clinical study in which they randomly assigned 21 participants with IS occurring more than 6 months prior and moderate to severe upper-limb impairment into two groups. One group received 0.5 seconds of iVNS (0.8 mA, 100 μs, 30 Hz) paired with rehabilitation (3 times a week for 2 hours each session over 6 weeks), while the other group underwent individual rehabilitation alone. The study demonstrated that iVNS was feasible and safe, with a significantly greater improvement in FMA-UE scores in participants treated with iVNS than in the control group. Consistent stimulation parameters were applied in a randomized, multicenter, double-blind, sham-controlled pilot study. 17 All participants received VNS implants randomized to active or control stimulation and underwent 6 weeks of clinical rehabilitation, followed by a 90-day home exercise program. This study also suggested that pairing rehabilitation therapy with iVNS is an acceptably safe, feasible, and effective intervention for treating upper-limb weakness following IS. A subsequent study by the same group extended the follow-up period to 1 year to investigate the long-term safety, feasibility, and adherence to home self-management for patients with chronic stroke, showing that approximately 73% of the participants still exhibited a clinically significant improvement in FMA-UE scores after 1 year. 100
The VNS-REHAB study strongly supported the use of iVNS combined with rehabilitation for treating upper-limb motor dysfunction in patients after chronic IS, which served as a cornerstone of the FDA’s approval of VNS for treating moderate to severe upper-limb motor dysfunction in chronic stroke patients. 15 Subgroup analysis of the VNS-REHAB trial showed that the positive impact of paired iVNS on upper-limb impairment and function in individuals with chronic IS remained consistent across various subgroups, including age, sex, severity of impairment, time since stroke, stroke location, and affected side. 101 Moreover, an analogous experimental design was utilized in a long-term follow-up study, which revealed that the marked effects of paired iVNS treatment over the course of 1 year were maintained in years 2 and 3, with further improvements in both impairment and function observed. 102
Two specific cases were reported from the clinical trials of Dawson et al 16 and Kimberley et al, 17 in which patients with left upper-limb sensory loss and gait impairments more than 1 year after chronic IS, respectively, improved their sensory function and gait impairments to varying degrees after undergoing iVNS combined with tactile stimulation and mobility training.103,104 Recently, a case report described the initial use of VNS implantation in a patient who experienced persistent upper-limb dysfunction 3 years following ICH. 105 The stimulation parameters were the same as those used for IS, and iVNS paired with 6 weeks of physiotherapy led to a notable 14-point increase in the FMA-UE score, suggesting that iVNS is a promising therapy for enhanced recovery after hemorrhagic stroke.
The surgical implantation required for iVNS may make it unsuitable as a rehabilitation treatment for many stroke patients with comorbidities. As a result, there have been 11 clinical studies on tVNS in cerebral stroke patients, 9 of which utilized taVNS. Capone et al 106 were the first to demonstrate the safety and feasibility of taVNS in patients with chronic ischemic or hemorrhagic stroke. taVNS (2.8-7.2 mA, 300 μs, 20 Hz) was administered in 30-second training sessions at 5-minute intervals for 60 minutes per day for 10 days before robotic rehabilitation. taVNS combined with robot-assisted rehabilitation led to a slight but significant improvement in arm functionality. However, the duration of taVNS in this study was shorter than that in studies on iVNS, which typically lasted at least 6 weeks. Subsequently, Redgrave et al 107 confirmed the positive effects of taVNS when combined with upper-limb rehabilitation for the first time. In their study, patients with residual upper-limb dysfunction at more than 3 months after IS underwent 60 minutes of taVNS (1-3.2 mA, 100 μs, 25 Hz) therapy 3 times per week for 6 weeks. They engaged in motor rehabilitation through repetitive task practice and received a taVNS pulse at the beginning of each movement. The intervention was found to be safe and well tolerated, resulting in a significant change in the FMA-UE score, with a mean increase per participant of 17.1 points. Similarly, Chang et al 108 showed that taVNS (4.5 mA, 300 μs, 30 Hz) delivered prior to extension movements significantly reduced spasticity in the affected arm, and significantly changed bicep peak surface electromyography (sEMG) amplitudes during extension in chronic stroke patients. A novel neurostimulation system, known as motor-activated auricular vagus nerve stimulation (MAAVNS), has been designed to deliver taVNS simultaneously with repetitive movements during post-stroke motor rehabilitation. A prospective, randomized, double-blind trial involving individuals with unilateral hemiparesis from chronic stroke suggested that pairing stimulation with movement may improve the timing of stimulation, potentially leading to more efficient treatment. Although the MAAVNS group received significantly less stimulation overall, the effect size of the MAAVNS group was double that of the unpaired taVNS group. 109
In addition to upper-limb recovery, early studies of taVNS have shown promise in addressing other post-stroke neurological deficits, such as sensory loss, 110 dysphagia, 111 and insomnia. 112 In studies of subacute cerebral stroke, Wu et al 18 and Li et al 113 both reported improvements in upper-limb motor function with taVNS (300 μs, 20 Hz, 30 seconds/5 minutes) delivered before rehabilitation at 0.5 to 3 months post-stroke and <1 month post-stroke, respectively.
The NOVIS trial (NOn-invasive Vagus nerve stimulation in acute Ischemic Stroke) was conducted in the Netherlands and was the earliest registration study to investigate the use of tVNS in acute stroke. 114 This single-center, prospective, randomized controlled trial with blinded outcome assessment involved 150 patients with acute anterior circulation IS within 12 hours of symptom onset. Participants were randomized to active tcVNS or sham stimulation, with two stimulations of 2 minutes each applied every 15 minutes in the first 3 hours, followed by two 2-minute stimulations every 8 hours over the next 5 days or until discharge. The primary outcome measured was the final infarct volume on day 5, according to MRI, to determine the potential benefits of tcVNS in the acute stroke setting without interfering with revascularization therapy. The TRanscutaneous cervical Vagus nErve stimulatioN as a treatment for acUte Stroke (TR-VENUS) trial first showed the safety and feasibility of tVNS in patients with AIS and ICH. 14 Patients were randomly assigned to standard-dose tcVNS (2 minutes/10 minutes for 1 hour), high-dose tcVNS (2 minutes/10 minutes for two 1-hour sessions separated by a 3-hour interval), or sham stimulation within 6 hours of symptom onset, suggesting that high-dose tcVNS could significantly reduce infarct growth in patients with diffusion mismatch.
Current challenges and future directions
Although various mechanisms and effects of VNS on cerebral stroke from the perspective of animal experiments and clinical trials have been reported in the existing retrieved relevant literature, the specific therapeutic mechanisms need to be studied in depth. The large-scale clinical application of VNS still faces many difficulties and challenges owing to the individual variability of clinical patients and the diversity of comorbidities.
Adjusting stimulation parameters for VNS
The ideal stimulation parameters for VNS are directly related to the clinical outcome. Therefore, determining the optimal stimulation conditions for VNS has become an essential issue in the clinical application of VNS. The above studies of VNS in animals and humans demonstrated that iVNS can effectively restore forelimb function in animals and improve human upper-limb function, sensation, and gait disorders after stroke. iVNS was administered at a pulse width of 100 μs, stimulation intensity of 0.8 mA, and frequency of 30 Hz with a pulse train of 0.5 seconds paired with rehabilitation training. However, the duration of stimulation varied. Other animal studies used iVNS with a pulse width of 300-500 μs, a stimulation intensity of 0.5 or 1 mA, and a frequency of 20 or 30 Hz, with a 30-second stimulation cycle every 5 minutes for 1 hour. A lower stimulation frequency, such as 5 Hz, has also been employed in mice.
In animal studies of tVNS, the pulse width typically ranged from 300 to 1000 μs, the current intensity ranged from 0.5 to 2 mA, the frequency ranged from 10 to 25 Hz, and the on-off time varied between 30 seconds/5 minutes or 2 minutes/10 minutes, lasting 1 hour. On the other hand, human studies commonly involved increasing the stimulation intensity by 0.1 mA per stimulation until the highest reported value by participants was reached. The intensity of stimulation ranged between 1 and 7.2 mA, the pulse width ranged from 100 to 300 μs, and the frequency ranged from 20 to 30 Hz. Notably, there were significant differences in on-off time and duration. Additionally, tcVNS was used in constant voltage mode in both animal and human studies.
Activation of neurons in the LC is believed to mediate many of the effects of VNS in the CNS. 115 Stimulation parameters are the primary determinants of neuromodulator network activation in response to VNS. Typically, an individual train of VNS consists of electrical pulses characterized by specific intensity and width delivered at a particular rate for a designated duration. Modifying any of these stimulation parameters has a straightforward impact on the activation of neuromodulatory networks. Increased current intensities and longer pulse widths can enhance the release of neurotransmitters and promote greater spiking activity in the noradrenergic LC, indicating heightened neuromodulatory engagement.116-118 Likewise, faster frequencies elicit a higher rate of neural activity in the LC, while longer trains result in an extended duration of spiking. 115 Most studies used a VNS frequency of 20-30 Hz in both iVNS and tVNS.
Additionally, studies have demonstrated that varying stimulation intensities differentially activate neuroplasticity. 119 Low-intensity VNS produces insufficient activation and fails to promote neuroplasticity. In contrast, moderate-intensity VNS results in adequate activation, thereby enhancing neuroplasticity in conjunction with paired training. Paradoxically, high-intensity stimulation also fails to promote neuroplasticity, leading to an inverted U-shaped relationship between stimulation intensity and VNS-dependent neuroplasticity. Pruitt et al 120 sought to determine the VNS intensity that yields the greatest recovery of forelimb function in a stroke model. Moderate-intensity VNS at 0.8 mA significantly enhanced recovery of function compared to all other groups. Neither 0.4 mA nor 1.6 mA VNS was sufficient to improve functional recovery compared to equivalent rehabilitation without VNS. This study suggests that different stimulation intensities exert varying effects on neuroplasticity and may influence the impact of VNS on neurological function. The safest and most effective combination of VNS parameters has yet to be identified, underscoring the importance of continuously optimizing and adjusting VNS stimulation parameters.
Time window for VNS
In acute stroke, preclinical studies have primarily focused on utilizing VNS approximately 30 minutes after stroke. Ay et al 20 reported that tVNS administered 4 hours after a stroke still reduced the infarct size, but this protective effect was not observed when tVNS was administered 5 hours after a stroke. The evaluation of the effect of VNS treatment at different time points after stroke is particularly necessary in clinical trials owing to the delay in hospitalization and diagnosis of stroke patients, which makes it difficult for patients to undergo emergency measures similar to those used in animal studies. Research on VNS and stroke rehabilitation indicates that task-specific plasticity can be enhanced by pairing specific tasks with VNS. While most clinical studies focus on patients with chronic stroke to minimize confounding factors, this approach may overlook an optimal recovery window.
Optimizing animal models
The neuroprotective and neuroplasticity effects of iVNS and tVNS have been demonstrated in preclinical animal models of cerebral stroke. However, preclinical animal models have limitations that need to be addressed to align more closely with clinical needs. First, many studies utilize a transient MCAO/R model, which may not fully capture the reality of stroke treatment in patients who do not always receive urgent revascularization. To better understand the impact of VNS on reperfusion injury, it is crucial to develop models of permanent vessel occlusion that mirror common stroke characteristics. Second, existing animal models focus on proximal middle cerebral artery occlusion; therefore, the effects of VNS on small vessel occlusion and posterior circulation infarction are unclear. Third, most studies have been conducted on male animals despite known sex differences in stroke mechanisms, 121 emphasizing the need for research using female animal models. Finally, the complexity of stroke in humans, influenced by factors such as hypertension, aging, diabetes, heart disease, and medications, warrants preclinical studies on animals with common comorbidities to improve the translation of the findings to clinical practice.
Limitations of clinical studies
Most clinical studies currently have primarily focused on chronic IS, with fewer studies on AIS and hemorrhagic stroke. Therefore, it is crucial to carry out randomized, large-scale, blinded clinical studies on AIS or ICH patients. Additionally, the majority of clinical studies have utilized VNS to enhance upper-limb motor function following chronic stroke. It would be valuable to assess whether VNS can also improve other cortical-based neurological deficits after stroke, such as lower-limb weakness, sensory dysfunction, speech disorders, dysphagia, cognitive impairment, and sleep disorders.
Biomarkers of VNS
The biomarkers of VNS can be categorized into those reflecting the degree of vagal nerve activation and those associated with clinical outcome improvements. Specifically, these includes biomarkers of vagal nerve activity, such as heart rate variability (HRV) and vagal somatosensory evoked potentials, as well as LC-norepinephrine markers, including pupil dilation, P300 event-related potentials, and salivary alpha-amylase (SAA). 122 Identifying biomarkers for vagal nerve activation and response in VNS is essential. Firstly, it can guide clinical application studies to identify responsive and non-responsive patients and help allocate scarce resources. Secondly, it can provide researchers with optimal stimulation sites and parameters, ultimately allowing for the personalization of VNS parameters at the patient level and further enhancing treatment efficacy. Finally, this endeavor may contribute to a better understanding of the underlying mechanisms of VNS in acute and chronic stroke, facilitating the development of drug targets for neuroprotection and neuroplasticity.
VNS programming
VNS programming, akin to deep brain stimulation (DBS) programming for Parkinson’s disease, involves adjusting specific parameters to optimize therapeutic effects and minimize side effects. Regular follow-ups and patient feedback are essential for evaluating the efficacy of the stimulation and identifying any adverse effects. VNS programming can modify stimulation parameters in response to the patient’s treatment outcomes and side effects, thereby achieving an optimal balance between effective stimulation and manageable adverse effects. Over time, patients may acclimate to VNS treatment or may require its integration with other therapies, such as medication and psychotherapy. This programming process facilitates ongoing reassessment and adjustment of stimulation parameters in accordance with the overall treatment strategy. VNS programming is a dynamic and patient-centered approach that necessitates careful consideration of stimulation parameters, patient-specific factors, and continuous monitoring. While there are principles that guide programming, the ultimate goal is to tailor the therapy to each individual to maximize benefits and minimize side effects.
Closed-loop VNS
Most neurostimulation systems currently available operate in an open-loop configuration, wherein stimulation is administered based on preprogrammed settings, independent of changes in the patient’s clinical symptoms or underlying disease. However, the physiology of the nervous system and regulated organs can be dynamic, leading to variations in the effects of the same stimulus depending on the individual patient’s state. Consequently, open-loop stimulation may not effectively restore the desired function or may even induce side effects. In such cases, a neuromodulatory intervention in a closed-loop mode may be more appropriate.
Closed-loop VNS is a technology that employs closed-loop control mechanisms based on specific physiological feedback signals to stimulate the vagus nerve and regulate physiological functions. This approach typically utilizes sensors to monitor specific physiological parameters, such as heart rate, respiratory rate, and brain electrical activity. When these parameters exceed predetermined thresholds, the stimulator automatically generates electrical stimulation signals, which are transmitted to the vagus nerve via electrodes, thereby adjusting physiological functions and restoring them to the normal range. Moreover, the stimulation parameters can be dynamically adjusted in real time to optimize the therapeutic effect, thereby enhancing the overall effectiveness of the treatment. 123 These closed-loop VNS systems have been applied to conditions such as epilepsy, chronic pain, and depression.124-126 In the field of stroke rehabilitation, electromyography-gated action pairing taVNS has been reported as a potential neuromodulation approach for post-stroke motor rehabilitation. 109
Conclusion
This article provides a comprehensive overview of recent animal experiments and clinical studies on using VNS to improve cerebral stroke prognosis. It discusses the current types and stimulation parameters of VNS, explores potential mechanisms of action, and elucidates the neuroprotective and neuroplasticity effects of VNS in clinical settings. Although VNS has shown promise in improving neurological function in preclinical stroke models and early clinical trials for stroke rehabilitation, there are still unresolved questions regarding its optimal use and therapeutic mechanisms. There is currently no consensus on the optimal parameters for VNS. There may be no perfect combination of current intensity, pulse width, frequency, on-off time, and duration. Instead, there may be a wide range of parameters that are biologically active and can lead to promising behavioral effects. Achieving safe and effective clinical transformation of VNS requires further animal and clinical studies to thoroughly document and address the efficacy and potential side effects of VNS in clinical practice, refine animal models and stimulation parameters, and develop innovative stimulation devices to broaden the application of VNS in clinical settings.
Footnotes
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82072133, 82371467), Guangdong Basic and Applied Basic Research Foundation (2021A1515010922), and Guangzhou Science and Technology Plan Project (202206010032).
