Abstract
Background:
Predictors of recurrent stroke after endovascular treatment for symptomatic intracranial atherosclerotic stenosis (ICAS) remain uncertain.
Objectives:
Among baseline characteristics, lesion features, and cerebral perfusion changes, we try to explore which factors are associated with the risk of recurrent stroke in symptomatic ICAS after endovascular treatment.
Design:
Consecutive patients with symptomatic ICAS of 70–99% receiving endovascular treatment were enrolled. All patients underwent whole-brain computer tomography perfusion (CTP) within 3 days before and 3 days after the endovascular treatment. Baseline characteristics, lesion features, and cerebral perfusion changes were collected.
Methods:
Cerebral perfusion changes were evaluated with RAPID software and calculated as preprocedural cerebral blood flow (CBF) < 30%, time to maximum of the residue function (Tmax) > 6 s, and Tmax > 4 s volumes minus postprocedural. Cerebral perfusion changes were divided into periprocedural perfusion improvement (>0 ml) and non-improvement (⩽ 0 ml). Recurrent stroke within 180 days was collected. The Cox proportional hazards analysis analyses were performed to evaluate factors associated with recurrent stroke.
Results:
From March 2021 to December 2021, 107 patients with symptomatic ICAS were enrolled. Of the 107 enrolled patients, 30 (28.0%) patients underwent balloon angioplasty alone and 77 patients (72.0%) underwent stenting. The perioperative complications occurred in three patients. Among CBF < 30%, Tmax > 6 s, and Tmax > 4 s volumes, Tmax > 4 s volume was available to evaluate cerebral perfusion changes. Periprocedural perfusion improvement was found in 77 patients (72.0%) and non-improvement in 30 patients (28.0%). Nine patients (8.4%) suffered from recurrent stroke in 180-day follow-up. In Cox proportional hazards analysis adjusted for age and sex, perfusion non-improvement was associated with recurrent stroke [hazards ratio (HR): 4.472; 95% CI: 1.069–18.718; p = 0.040].
Conclusion:
In patients with symptomatic ICAS treated with endovascular treatment, recurrent stroke may be related to periprocedural cerebral perfusion non-improvement.
Registration:
http://www.chictr.org.cn. Unique identifier: ChiCTR2100052925.
Keywords
Introduction
Intracranial atherosclerotic stenosis (ICAS) is one of the most common causes of ischemic stroke worldwide.1–3 Patients with 70–99% symptomatic ICAS are at high risk of recurrent stroke (12.2%) at 1 year despite aggressive medical management (AMM), 4 and especially higher (37%) in those patients with impaired hemodynamics. 5 Endovascular treatment remains an alternative therapy for patients with symptomatic ICAS refractory to AMM.6–8 In two recent multiple registries focusing on endovascular treatment for symptomatic ICAS, the 1-year recurrent stroke declined to 6.3% and 8.5% compared with 20% in Stenting Versus Aggressive Medical Therapy for Intracranial Atherosclerosis (SAMMPRIS) trial.9,10
So far, the predictors of the risk of recurrent stroke after endovascular treatment for symptomatic ICAS remain uncertain. We hypothesize that some baseline characteristics, lesion features, or cerebral perfusion parameters (preprocedural, postprocedural, or periprocedural changes) might be related to the risk of recurrent stroke after endovascular treatment. As for the evaluation of cerebral perfusion parameters, RAPID software (iSchemia View) which is an automatic software for post-processing computer tomography perfusion (CTP) images can quantitatively evaluate cerebral perfusion parameters, and it had been used to identify potential beneficiary with acute large vessel occlusion through endovascular thrombectomy.11,12 Furthermore, RAPID software was used to assess impaired perfusion for patients with ICAS. 13
In this study, we collected baseline characteristics, lesion features, and preprocedural and postprocedural cerebral perfusion parameters of patients with symptomatic ICAS treated with endovascular therapy in a high-volume stroke center and evaluated which factors were associated with the 180-day outcome of recurrent stroke.
Methods
Study design and subjects
This is a prospective, single-arm, exploratory study of consecutive patients with symptomatic ICAS from an endovascular treatment team in a national stroke center. This study was approved by the Institutional Review Board of Beijing Tiantan Hospital, Capital Medical University (KY 2021-075-02). Written informed consent was obtained from the patients or their legal guardians. Inclusion criteria were (1) age ⩾ 18 years; (2) ischemic stroke (modified Rankin Scale ⩽ 3) or transient ischemic attack due to ⩾ 70% intracranial stenosis (in cases with tandem stenoses, the most severe stenosis was considered as the responsible lesion); (3) refractory to medical treatment (stroke recurrence under aggressive medical treatment including dual antiplatelet therapy and intensive management of risk factors); (4) more than one atherosclerotic risk factors including hypertension, hyperlipidemia, diabetes mellitus, and cigarette smoking. Exclusion criteria were (1) non-atherosclerotic stenosis, including Moyamoya disease, vasculitis, and dissection; (2) cardiac embolism; and (3) contrast agent allergy.
For enrolled patients, baseline characteristics were collected. Risk factors were defined as follows: hypertension was defined as a history of hypertension or taking any hypotensive drugs; diabetes mellitus was defined as a history of diabetes mellitus, taking any hypoglycemic agents or glycosylated hemoglobin ⩾ 7%; hyperlipidemia was defined as low-density lipoprotein cholesterol ⩾ 2.6 mmol/liter at the time of admission, a history of hyperlipidemia, or receiving lipid-lowering treatment; coronary artery disease was defined as a history of myocardial infarction or angina pectoris; smoking was defined as current smokers or previous smoking history. 14 The degree of stenosis was measured according to the Warfarin-Aspirin for Symptomatic Intracranial Disease (WASID) trial. 15 The most severe stenotic and the reference sites were decided by two physicians with consensus. If they did not reach an agreement, the third physician participated. Non-responsible artery (including extracranial and intracranial segments) stenosis > 50% was recorded. Multiple stenoses were defined as patients having more than one non-responsible extracranial or intracranial stenosis.
CTP scanning parameters
All patients underwent whole-brain CTP within 3 days before and 3 days after the endovascular treatment. CTP images were acquired using a 256-slice axial CT scanner (GE Revolution CT). Contrast agent, 50 ml (Omnipaque, 350 mg I/ml; GE Healthcare, Shanghai, CN) was injected into the antecubital vein at a rate of 5 ml/s using an automated injector (Ulrich Injection System; Ulrich GMBH & Co. KG, Germany). After a 5-s delay, CTP was performed with the following acquisition parameters: 80 kV tube voltage, 150 mA, 5 mm slice thickness, 256 × 0.625 mm collimation, 0.5 s rotation time, 2.0 s cycle time, 25-cm field of view (FOV), 512 × 512 image matrix size, and 32 slices. A total of 512 slices were obtained with a 160 mm scan length and scan time of about 40 s.
Cerebral perfusion parameters
CTP data were post-processed with the automated RAPID software. Cerebral perfusion parameters were described as voxels with CBF < 30% volume, time to maximum of the residue function (Tmax) > 6 s volume and Tmax > 4 s volume. Periprocedural perfusion changes were defined as preprocedural cerebral perfusion parameters minus postprocedural. If the change was > 0 ml, perfusion improvement was defined, and ⩽ 0 ml, perfusion non-improvement was defined.
Endovascular procedure
Endovascular treatment was performed by an experienced team. Under general anesthesia, a bolus dose of 75 U/kg of intravenous heparin was administered followed by half the dose after an hour after placement of the vascular access.
The endovascular treatment included balloon angioplasty alone and stenting.
Balloon angioplasty alone was recommended for patients with the following features: tortuous segment distal to the stenosis that is not safe for landing a stent delivery system, large branch arteries arising from the stenosis, or diameter of the parent artery < 2 mm. Balloon angioplasty alone was also used for patients who were reluctant to implant stent. The protocol of balloon angioplasty was the followings. Submaximal dilation was recommended to avoid arterial dissection, vessel rupture, and the snowplow effect of compressed plaque into perforator arteries. The balloon size was 80% of the actual luminal diameter or 60% in lesions directly adjacent to angiographically visible perforators.8,16 The balloon length should cover the lesion. The balloon was dilated slowly until it was pillar-shaped. After being inflated for at least 30 s, the balloon was deflated slowly. Rescue stenting was performed if an obvious recoil was observed or the antegrade blood flow was poor [modified Thrombolysis in Cerebral Infarction (mTICI) 0–2a]. In the stenting group, stent type (self-expanding stent or balloon-mounted stent) was determined based on access characteristics and lesion features [Mori types: type A, short (5 mm or less in length) concentric or moderately eccentric lesions less than totally occlusive; type B, tubular (5–10 mm in length), extremely eccentric or totally occluded lesions, less than 3 months old; and type C, diffuse (more than 10 mm in length), extremely angulated (>90°) lesions with excessive tortuosity of the proximal segment] and operator experiences. 17 For patients with smooth arterial access and Mori type A lesion, a balloon-mounted stent [Apollo stent (MicroPort NeuroTech, Shanghai, China)] was preferred. For patients with tortuous arterial access, extremely angulated Mori type C lesions, or lesions with a significant mismatch in the diameter between the proximal and the distal segments, balloon dilatation [Gateway balloon (Stryker, Natick, MA)] plus self-expanding stent [Wingspan stent (Stryker, Natick, MA, USA)] was preferred. It is difficult to deploy Wingspan stents for long lesions with an extremely tortuous path and curved lesions with a tiny vessel diameter, therefore the Enterprise stent (Codman Neurovascular) or Neuroform EZ stent (Boston Scientific) was used for its excellent conformability, lower radial force, and good wall apposition.18,19
Successful recanalization was defined as < 50% residual stenosis and the mTICI ⩾ 2b.20,21
In the periprocedural period, strict blood pressure management (at least 20% reduction over baseline) was performed in case of postprocedural hyperperfusion syndrome.
Clinically relevant ischemic and hemorrhagic stroke within 30 days of the procedure were recorded.
Postprocedural medical management
Dual antiplatelet therapy was performed 3 months after the procedure, then mono antiplatelet therapy continued. A weight-based dose of 0.4–0.6 ml nadroparin was given every 12 h subcutaneously for 1–2 days. Controlling risk factors included the following: systolic blood pressure less than 140 mmHg (or 130 mmHg in patients with diabetes mellitus), low-density lipoprotein less than 1.81 mmol/liter, smoking cessation, glycosylated hemoglobin less than 7%, and lifestyle modifications.
Recurrent stroke assessment during follow-up
Recurrent stroke was defined as a new neurological deficit lasting for at least 24 h due to new ischemic infarction in the territories of the responsible artery beyond 30 days after the endovascular procedure. Hemorrhagic stroke and ischemic stroke in the territories of the non-responsible artery were also recorded. Patients were evaluated at 180 days after the procedure via telephone or face-to-face follow-up. If a stroke was suspected, magnetic resonance imaging or non-contrast CT for the brain was performed. After the follow-up data were analyzed, the outcomes were determined by two clinicians in consensus (L.Y. and Z.H.).
Statistical analysis
We performed statistical analysis using SPSS software. Normal distribution measurement data were expressed as means and standard deviations (SD), while skew distribution data were expressed as the median and interquartile range (IQR). Counting data were expressed in frequencies and percentages. Patients with perfusion improvement and non-improvement were compared using the Mann–Whitney U test for continuous variables and the chi-square or Fisher’s exact test for categorical variables. Multivariable logistic regression was performed to evaluate the factors associated with perfusion non-improvement. Patients with stroke recurrence and no-recurrence were compared using the Mann–Whitney U test for continuous variables and the chi-square or Fisher’s exact test for categorical variables. The Kaplan–Meier (K-M) survival curve analysis was used to test perfusion non-improvement associated with stroke recurrence. The Cox proportional hazards analysis was performed to assess association between factors and stroke recurrence after adjusting for age and sex. A p value less than 0.05 was considered a statistically significant difference.
Results
Patient baseline characteristics
From March 2021 to December 2021, among 190 patients, 107 patients with symptomatic ICAS were enrolled (Figure 1). The mean age was 57.5 ± 10.6 years, and 79 patients (73.8%) were men. The risk factors were summarized in Table 1.

Flowchart.
Comparison of periprocedural perfusion improvement and non-improvement.

BA, basilar artery; CTP, computed tomography perfusion; ICA, internal carotid artery; MCA, middle cerebral artery; SD, standard deviation; Tmax, time to maximum of the residue function; VA, vertebral artery.
Lesion characteristics
The responsible artery included intracranial internal carotid artery in 17 patients, middle cerebral artery in 42 patients, intracranial vertebral artery in 22 patients, and basilar artery in 26 patients. As for Mori types, type A was found in 18 patients (16.8%), type B in 55 patients (51.4%), and type C in 34 patients (31.8%). The median degree of preprocedural stenosis was 76.4% (IQR: 72.2–88.4%). The median lesion length was 8.4 mm (IQR: 6.3–10.4 mm). Multiple stenosis was found in 36.4% of patients.
Endovascular treatment
Of the 107 enrolled patients, 30 (28.0%) patients underwent balloon angioplasty alone and 77 patients (72.0%) underwent stenting. The perioperative complications occurred in three patients. The three periprocedural complications displayed as non-disabling ischemic stroke due to perforator stroke in one case and embolic stroke in the other two cases.
Preprocedural and postprocedural cerebral perfusion parameters, and periprocedural perfusion changes
Preprocedural CBF < 30% volume, Tmax > 4 s volume, and Tmax > 6 s volume was 0 ml (IQR: 0–0 ml), 74 ml (IQR: 28–178 ml), and 0 ml (IQR: 0–15 ml). Postprocedural CBF < 30% volume, Tmax > 4 s volume, and Tmax > 6 s volume was 0 ml (IQR: 0–0 ml), 22 ml (IQR: 0–101 ml), and 0 ml (IQR: 0–0 ml). Periprocedural CBF < 30% volume, Tmax > 4 s volume, and Tmax > 6 s volume changes were 0 ml (IQR: 0–0 ml), 35 ml (IQR: 0–98 ml), and 0 ml (IQR: 0–14 ml). Among the three parameters, Tmax > 4 s volume was suitable to evaluate periprocedural perfusion changes. Among 107 patients, postprocedural perfusion improvement was found in 77 patients (Figure 2) and non-improvement in 30 patients (Figure 3).

An adult patient presented with slurred speech and right limbs weakness for 3 years. (a) Digital subtraction angiography showed 99.0% of stenosis of basilar artery (arrowhead). The Tmax > 4 s volume was 74 ml before procedure. (b) After balloon dilation (Gateway, 1.5 mm × 9 mm), the degree of postprocedural residual stenosis was 36.2% (arrow) and the Tmax > 4 s volume dropped to 29 ml. No recurrent stroke occurred in the following 180 days.

An adult patient presented with paroxysmal dizziness and blurred vision for 5 months. (a) Digital subtraction angiography showed 74.2% of stenosis of intracranial vertebral artery (arrowhead). The Tmax > 4 s volume was 7 ml before procedure. (b) After stent implantation (Apollo, 2.5 mm × 8 mm), the degree of postprocedural residual stenosis was 16.2% (arrow) and the Tmax > 4 s volume was 8 ml. Recurrent stroke occurred on the 178th day after procedure.
Comparison of patients with periprocedural perfusion improvement and non-improvement
Patients with periprocedural perfusion improvement were younger (56.0 versus 61.3, p = 0.017), longer length of lesion (8.9 versus 6.6 mm, p = 0.001), larger preprocedural Tmax > 4 s volume (98.0 versus 39.5 ml, p = 0.001), and severe degree of preprocedural stenosis (79.0% versus 74.1%, p = 0.008) compared with patients with periprocedural perfusion non-improvement (Table 1). After adjusted for age and sex, length of lesion [odds ratio (OR): 0.824; 95% CI: 0.689–0.984; p = 0.033], degree of preprocedural stenosis (OR: 0.943; 95% CI: 0.896–0.993; p = 0.025), and preprocedural Tmax > 4 s volume (OR: 0.994; 95% CI: 0.988–0.999; p = 0.023) were associated with periprocedural perfusion non-improvement (Table 2).
Multivariable analysis of factors associated with periprocedural perfusion non-improvement.
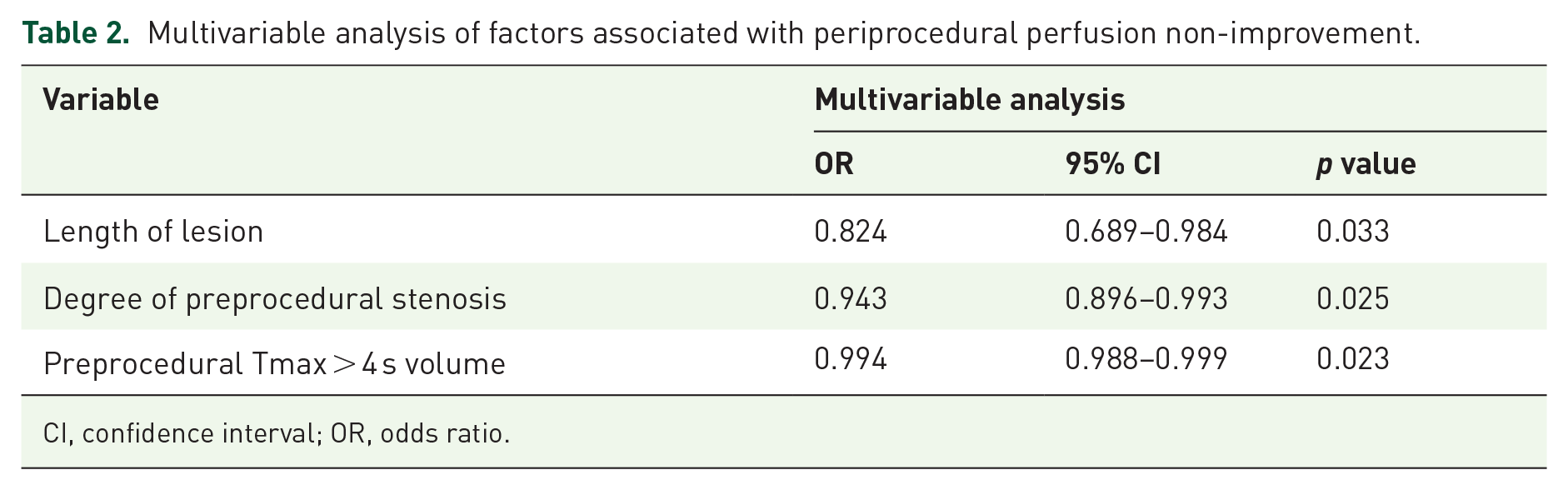
CI, confidence interval; OR, odds ratio.
Clinical follow-up outcome
During the 180-day follow-up, nine patients with recurrent stroke were recorded. Compared with patients without recurrent stroke, patients with recurrent stroke had a higher prevalence of perfusion non-improvement (66.7% versus 24.5%, p = 0.014) (Table 3). The K-M survival curve analysis revealed that perfusion non-improvement was associated with recurrent stroke (p = 0.007) (Figure 4). In Cox proportional hazards analysis adjusted for age and sex, patients with perfusion non-improvement had high risk of recurrent stroke at 180 days than those without [hazards ratio (HR): 4.472; 95% CI: 1.069–18.718; p = 0.040].
Comparison of patients with recurrent stroke and without.

BA, basilar artery; CTP, computed tomography perfusion; ICA, internal carotid artery; MCA, middle cerebral artery; SD, standard deviation; Tmax, time to maximum of the residue function; VA, vertebral artery.
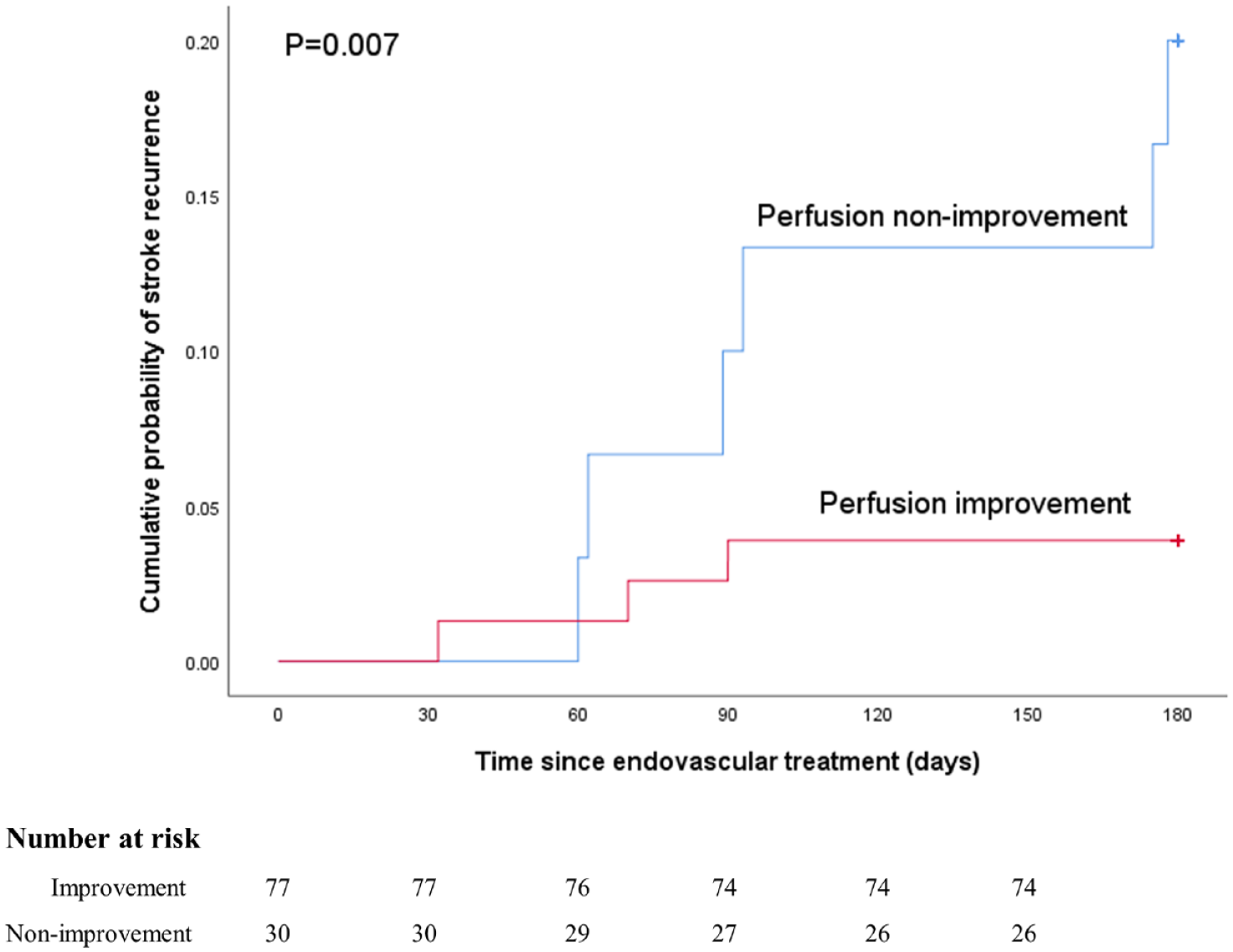
K-M survival analysis showing an association between periprocedural perfusion change and recurrent stroke.
Discussion
In this study, in a group of patients with symptomatic ICAS refractory to medical treatment, the majority (72.0%) of patients achieved periprocedural perfusion improvement after endovascular treatment, and 180-day recurrent stroke may be associated with periprocedural perfusion non-improvement after endovascular treatment.
Among the three perfusion parameters from RAPID software, Tmax > 4 s volume may be most suitable to evaluate cerebral perfusion in patients with symptomatic ICAS. In studies focusing on acute ischemic stroke, RAPID software has been used to evaluate whether patients with a longer time window can benefit from endovascular thrombectomy. In this setting, patients with a larger penumbra (Tmax > 6 s volume minus CBF < 30% volume) may benefit from endovascular thrombectomy.11,12 Unlike patients with acute stroke due to large vessel occlusion, patients with symptomatic ICAS may have better cerebral perfusion due to a minor neurologic deficit. So far, several perfusion image modalities have been used to evaluate cerebral perfusion, including positron emission computed tomography, magnetic resonance imaging (using arterial spin labeling), and CT perfusion.22–24 However, they have rarely been used to evaluate the cerebral perfusion injury secondary to symptomatic ICAS. In this study, Tmax > 4 s volume in symptomatic ICAS is larger than Tmax > 6 s volume and CBF < 30% volume. It suggested that Tmax > 4 s volume may be the most suitable parameter to reflect the cerebral perfusion injury in the setting of symptomatic ICAS.
Our study showed that most patients (72.0%) with symptomatic ICAS may have cerebral perfusion improvement due to endovascular treatment. Unexpectedly, there are some patients (28.0%) having cerebral perfusion non-improvement. This study demonstrated that length of lesion, degree of preprocedural stenosis, and preprocedural Tmax > 4 s volume were associated with perfusion improvement. Strictly selected patients may benefit from endovascular therapy. Another important reason was strict blood pressure management after procedure (preprocedural 141.7 ± 19.9 versus postprocedural 122.6 ± 10.6 mmHg). Considering the RAPID perfusion parameters reflect the whole brain perfusion, decrease in cerebral perfusion of other vascular beds overwhelmed cerebral perfusion increase during the periprocedural period.
Previous studies demonstrated that hypoperfusion was one of the biomarkers of recurrent stroke for patients with ICAS.25,26 Improved perfusion after endovascular treatment may be an effective way to reduce stroke recurrence. In this study, the 180-day recurrent stroke was related to periprocedural perfusion non-improvement. There was no difference in periprocedural complications (within 30 days), so the 180-day recurrent stroke reflects the non-procedural events. This finding suggested that after overcoming the periprocedural risk of endovascular treatment, a higher cerebral perfusion improvement achieved from the responsible recanalization may indicate a lower risk of ischemic events occurrence.
In addition, baseline characteristics and lesion features were not associated with the 180-day recurrent stroke. In the SAMMPRIS trial, old infarct in the territory of the stenosis, new stroke presentation, and absence of statin use at enrollment were independent predictors of recurrent stroke in their medical group. 27 So far, there are few studies focusing on the association of recurrent stroke with baseline features after intracranial endovascular treatment. Our results suggest that there may be difference in predictors of recurrent stroke in patients with endovascular treatment and without.
This study has several limitations. First, this was a single-center study involving a small sample size of Chinese ethnicity, which may decrease the power of our findings. Second, the duration of the follow-up was shorter, which may not be generalized to the prediction of long-term recurrent stroke after endovascular treatment. Third, the present patient cohort included patients with anterior circulation and posterior circulation, and CTP may be less reliable in the posterior circulation than in the anterior circulation. Fourth, nine patients with 0 ml of preprocedural Tmax > 4 s volume were all classified into perfusion non-improvement according to the definition of perfusion improvement or not in this study. Among them, seven patients with 0 ml of pre- and postprocedural Tmax > 4 s volume were enrolled in perfusion non-improvement group. However, potential perfusion improvement may exist for these patients due to strict blood pressure control after procedure. Further studies are needed to confirm this.
Conclusion
In patients with symptomatic ICAS treated with endovascular treatment, recurrent stroke may be related to periprocedural cerebral perfusion non-improvement.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221143178 – Supplemental material for Association of periprocedural perfusion non-improvement with recurrent stroke after endovascular treatment for Intracranial Atherosclerotic Stenosis
Supplemental material, sj-docx-1-tan-10.1177_17562864221143178 for Association of periprocedural perfusion non-improvement with recurrent stroke after endovascular treatment for Intracranial Atherosclerotic Stenosis by Long Yan, Zhikai Hou, Weilun Fu, Ying Yu, Rongrong Cui, Zhongrong Miao, Xin Lou and Ning Ma in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
