Abstract
Background:
Invasive intracranial pressure (ICP) can result in complications, pain, or even aggravate intracranial hypotension (IH) or headache in patients with IH.
Objective:
To investigate whether ultrasonographic measurements of optic nerve sheath diameter (ONSD) could serve as a noninvasive IH marker.
Methods:
Ultrasonographic ONSD was measured prior to lumbar puncture (LP) and ONSD measurements compared to LP’s opening pressure. We analyzed correlations between ONSD and ICP and determined the optimal ONSD cut-off point for IH. According to their LP on admission, patients were divided into three groups: IH group, normal ICP group, and elevated ICP group. Correlations between ONSD and ICP were analyzed using Pearson’s correlation. A receiver operating characteristic (ROC) curve was generated to determine the optimal cut-off point for IH.
Results:
In total, 136 subjects (75 men, 55.1% men) were included, and 1088 ONSDs were measured. The ONSD of the IH group (2.96 ± 0.15 mm) was significantly lower than that of the normal (3.59 ± 0.33 mm) and elevated ICP groups (4.90 ± 0.42 mm, p < 0.001). There was a significant difference in ONSD within all groups (p < 0.001), and the differences between the two groups were also statistically significant. ONSD and ICP values were strongly correlated, with an r = 0.952 (95% confidence interval [CI]: 0.924–0.969; p < 0.001). After adjusting for age, diastolic blood pressure, systolic blood pressure, body mass index, waistline, and head circumference, ONSD was positively associated with ICP. ROC curve analysis revealed an area under the curve of 0.990 (95% CI: 0.975–1.000). The ONSD cut-off point for identifying decreased opening pressure on LP was 3.15 mm, with 98.3% sensitivity and 91.7% specificity.
Conclusions:
Ultrasonographic ONSD may be a noninvasive, valuable, and easy-to-perform bedside technology for evaluating IH.
Keywords
Introduction
Intracranial hypotension (IH) or elevated intracranial pressure (ICP) mainly manifests through headaches. IH is a syndrome with various etiologies and clinical presentations, characterized by orthostatic headache and low cerebrospinal fluid (CSF) pressure. IH mainly manifests by the postural features of headaches and the presence of associated symptoms. 1 However, the clinical spectrum is surprisingly varied. Headache does not invariably occur within a short time after adopting the upright posture, as many other patterns of headache have been reported, such as nonpositional headache, exertional headache, or second half-of-the-day headache. 2 The opening pressure on lumbar puncture (LP) is typically used to diagnose this disease. Although this invasive measurement is often helpful, it is complicated by miseries; even one-third of patients might experience post-LP headache. 3 Furthermore, due to CSF outflow, LP might aggravate IH or headache in patients with IH. In addition, IH-induced headaches are caused by reduced CSF volume or pressure, and headaches may develop secondary to LP. 4 Therefore, a simple, noninvasive and repeatable diagnostic technology that can aid diagnosis and follow-up is needed.
In recent years, ultrasonographic optic nerve sheath diameter (ONSD) has been increasingly used to assess elevated invasive ICP.5 –8 However, there is currently insufficient data to determine the utility of ultrasonographic ONSD in IH. 6 Since abnormal ICP, not just high but low, causes clinical headaches, it is important to determine whether ONSD can specifically identify high and low ICP. Therefore, this study investigated whether ultrasonographic measurements of ONSD could serve as a noninvasive marker of IH.
Methods
Study setting
The study was performed in the US center in the Department of Neurology at the First Hospital of Jilin University, a general public hospital in China. The protocol was designed from September 15, 2018, to November 31, 2018, and approved by the ethics committee of the First Hospital of Jilin University on January 10, 2019 (approval number, 2019-209). All methods adhered to relevant guidelines and regulations. The institutional review board approved the enrollment of patients who needed LP. All participants were informed that the obtained ultrasonic ONSD measurements would be compared to their opening pressure on LP, to which they provided written informed consent.
Patients
We prospectively recruited patients admitted to our neurology department who needed to undergo LP between February 2019 and December 2020. ONSD was measured using ultrasound in each patient prior to LP. ICP ⩽ 60 mmH2O was considered as IH, 9 whereas ICP > 200 mmH2O was considered elevated. 10 According to their LP on admission, patients were divided into three groups as follows: group 1 (⩽60 mmH2O), group 2 (60 < LP ⩽ 200 mmH2O), and group 3 (LP > 200 mmH2O). The exclusion criteria were as follows: age < 18 years, ophthalmic diseases, such as optic atrophy, history of glaucoma, tumors or traumas, and administration of medications that may have affected the ICP. In addition, the following patient data were recorded: sex, age, diastolic blood pressure (DBP), systolic blood pressure (SBP), and body mass index (BMI).
Measurements
Ultrasound ONSD examinations were performed in B-mode on a Philips iU22 ultrasound system (Andover, Massachusetts, USA), using a 9.3 MHz linear array transducer. The acoustic output of the ultrasound system needed to be adjusted to the requirements of orbital sonography according to the ALARA (‘as low as reasonable achievable’) principle to avoid damage to the retina and lens. 11 Patients were examined in the supine position. The investigation of both ONSDs for all patients was independently performed by two experienced practitioners, blind to each other’s study results or the patients’ disease state. Consistent with previous protocols, the probe was placed lightly over the closed upper eyelid with a thick ultrasound gel to prevent pressure on the eye. The probe position was adjusted to clearly display the entry of the optic nerve into the eyeball. The first measurement was performed in the sagittal plane (with the probe in a vertical orientation), and the second in the transverse plane (with the probe in a horizontal orientation).12,13 ONSD was assessed bilaterally, 3 mm behind the globe.14,15 Each eye was examined twice, and the mean value of the eight total measurements for both eyes was recorded as one observer’s measurement. The final ONSD measurement of each patient was derived from the average of the 16 values from the two observers to minimize variability. After ONSD measurements, LP was immediately performed by an experienced neurological resident blinded to the ultrasonographic ONSD results. The CSF opening pressure by LP was recorded in millimeters of water pressure (mm H2O). Patients were awake and placed in the left lateral position with their hips and knees flexed, and their heads as close to their knees as comfortably possible. The area around the lower back was prepared using an aseptic technique, and patients were asked to relax. After entering the subarachnoid space, patients were asked to straighten their legs, after which the opening pressure on the LP was recorded and fluid samples obtained.
Statistical analysis
For sample size estimation, the PASS software (NCSS) was used. Based on the assumption that the correlation between ONSD and ICP was 0.758, 8 the required sample size was calculated as 13 participants, with an α error of 0.05 and a statistical power of 91%. All statistical analyses were performed using SPSS (version 24.0; IBM SPSS, IBM Corp, Armonk, NY, USA) and R version 3.4.3 (University of Auckland, Oakland, New Zealand). Continuous variables are presented as median and interquartile range and compared using the Wilcoxon rank sum test. Categorical variables are represented as frequency and percentage and compared using the chi-square test (for unordered categorical variables) or the Wilcoxon rank sum test (ordinal categorical variable). We established scatterplot and generalized additive models (GAMs) using the ‘mgcv’ package to explore the associations between ONSD and ICP. For ONSD and IH, binary logistic regression analysis was performed to identify the factors that affected this association. A receiver operating characteristic (ROC) curve was generated to determine the optimal cut-off point for IH. Statistical significance was set at p < 0.05.
Results
A total of 136 subjects (75, 55.1% males) were included in the study, and 1088 ONSDs were measured. The mean patient age was 40.7 ± 14.04 years (range: 18–80 years). ONSD and ICP ranges were 2.72–6.22 mm and 30–400 mm H2O, respectively. IH, normal ICP, and elevated ICP were diagnosed in 24, 58, and 54 patients, respectively. A comparison of characteristic data between the elevated opening pressure and normal opening pressure groups is shown in Table 1. Statistically significant differences were found for sex, age, ONSD, DBP, waistline, and head circumference. The ONSD of the IH group (2.96 ± 0.15 mm) was significantly lower than that of the normal (3.59 ± 0.33 mm) and elevated ICP groups (4.90 ± 0.42 mm, p < 0.001; Table 1).
Patient characteristics.

BMI, body mass index; DBP, diastolic blood pressure; ICP, intracranial pressure; ONSD, optic nerve sheath diameter; SBP, systolic blood pressure.
Pearson correlation coefficient between the two eyes was 0.989 and that between the two observers was 0.961. A Bland–Altman analysis yielded a mean (SD) difference of −0.0089 (0.048) mm for ONSD. To understand the relationship between ONSD and ICP, we drew a scatter plot to observe the trend distribution (Figure 1). Among the total 136 subjects, ONSD and ICP values were strongly correlated, with an r = 0.952 (95% CI: 0.924–0.969; p < 0.001). There was a significant correlation between ONSD and ICP, with an r of 0.677 (95% CI: 0.376–0.873; p < 0.001), 0.606 (95% CI: 0.441–0.731; p < 0.001), and 0.871 (95% CI: 0.757–0.934; p < 0.001) in IH, normal ICP, and elevated ICP groups, respectively.

Scatterplot between optic nerve sheath diameter (ONSD) and intracranial pressure (ICP). X-axis represents ONSD, Y-axis ICP.
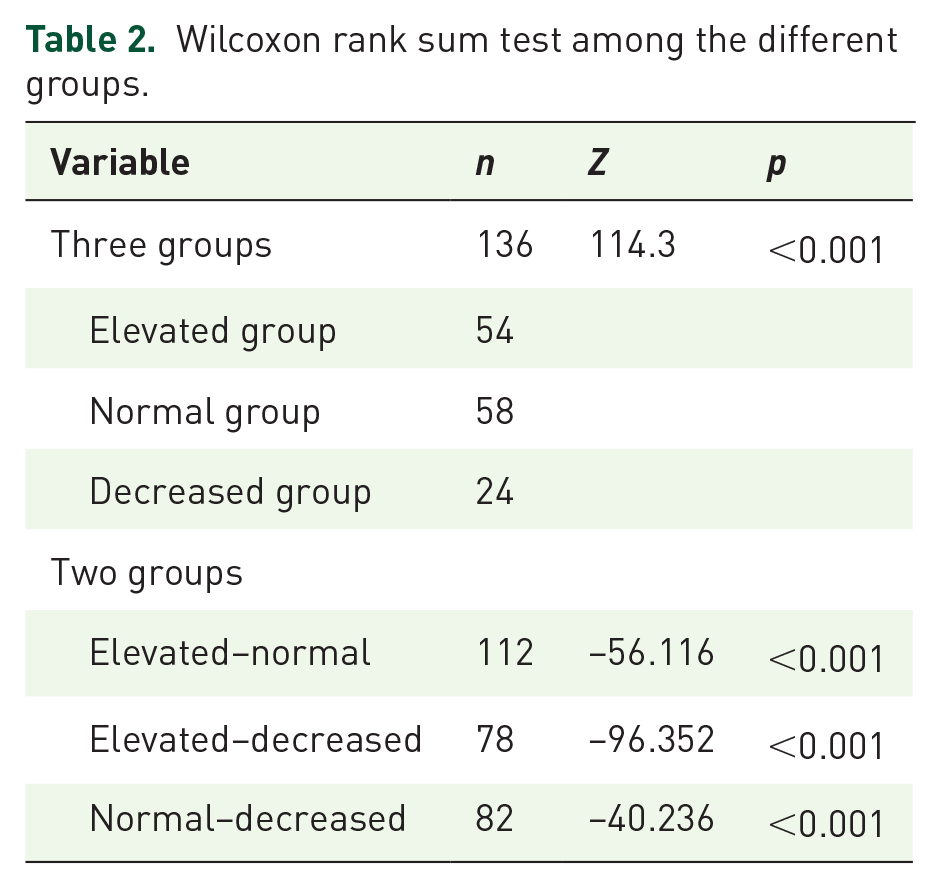
The Wilcoxon rank sum test showed a significant difference in ONSD between the three groups (p < 0.001) and that differences between the following two groups were also statistically significant: Elevated-Normal ICP groups (Z = –56.116, p < 0.001); IH-Normal ICP groups (Z = –40.236, p < 0.001), and elevated ICP-IH groups (Z = –96.352, p < 0.001; Table 2).
Wilcoxon rank sum test among the different groups.
Since abnormal ICP, both high and low, lead to headache, it is important to determine whether ONSD can specifically identify them. A probability density diagram showed no overlap between the elevated and decreased ICP groups (Figure 2). Furthermore, to confirm this result, we calculated the P95’s (3.186) 95% CI as 3.1213–3.1980 for the decreased ICP group and the elevated ICP group P2’s (4.375) 95% CI as 4.3424–4.4724. A GAM was performed to confirm the association between them after adjusting for the effects of age, DBP, SBP, BMI, waistline, and head circumference. Figure 3 shows that ONSD was positively associated with ICP. We selected age, sex, BMI, SBP, DBP, waistline, head circumference, and ONSD as the independent variables, and the dependent variable was the ICP group. Only ONSD was included in the logistic regression analysis. Using the opening pressure on LP as the standard criterion, we generated an ROC curve (Figure 4) to determine the optimal cut-off point for sensitivity and specificity. ROC curve analysis revealed an area under the curve (AUC) of 0.990 (95%CI: 0.975–1.000). The ONSD cut-off point for identifying decreased opening pressure on LP was 3.15 mm, with a sensitivity of 98.3% and a specificity of 91.7%.

Probability density diagram of increased and decreased intracranial pressure (ICP) groups. X-axis represents optic nerve sheath diameter (ONSD) values, y-axis the probability density. Red represents decreased ICP and blue increased ICP group.

Association between optic nerve sheath diameter (ONSD) and intracranial pressure (ICP) after adjusting for other factors. Y-axis shows the smoothness function value, and the numbers in brackets the estimated degrees of freedom (EDF). The solid line represents the estimated relationship and dotted lines the 95% confidence interval.
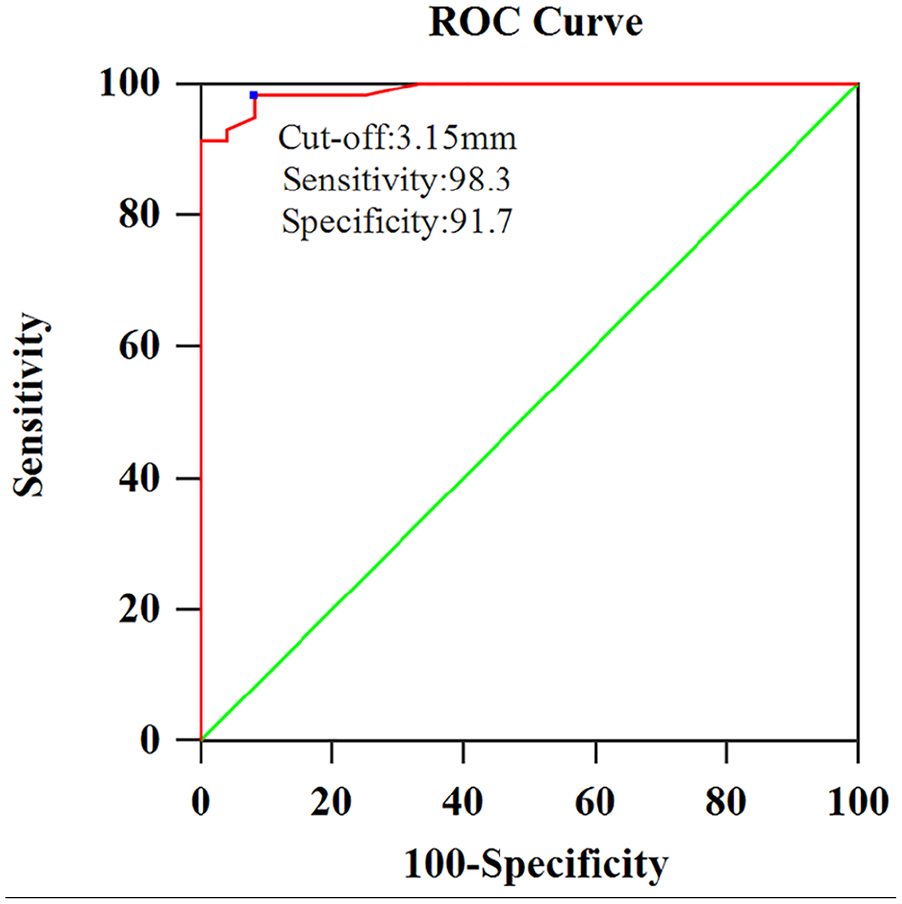
Receiver operating characteristic (ROC) curve for the optic nerve sheath diameter (ONSD). AUC = 0.990. X-axis represents 100-specificit and y-axis sensitivity.
Discussion
This study examined the ONSD and ICP of 136 patients and found that ONSD was positively associated with ICP. In addition, we determined that ultrasonographic ONSD measurement is potentially useful for identifying patients with IH.
Our previous study found that ultrasonographic ONSD correlated with ICP on admission and suggested that this noninvasive method could be potentially useful for identifying patients with elevated ICP. 16 The results of this study are consistent with that of ICP and ONSD values obtained on admission being strongly correlated in different samples. Furthermore, this study focused on the ONSD of patients with IH and confirmed ONSD as a predictor of IH.
To our knowledge, only a few studies have described ONSD in patients with IH. A study including three patients found that the subarachnoid space on magnetic resonance imaging (MRI) is decreased in patients with CSF hypovolemia. 17 Another study using qualitative signs in MRI of 19 patients with CSH showed decreased ONSD diameters. 18 In recent years, a retrospective study of computed tomography (CT) found that ONSD in patients with IH was significantly lower than in hypertension cases. 19 Some reports measured ONSD using MRI17,20 or ocular sonography.4,21,22 Our study also demonstrated that ONSD decreases in patients with IH. We conjecture that the ONSD depends on pressure and is associated with its peculiar elastic properties. It was reported that the subarachnoid space of the human optic nerve contains various trabeculae, septa, and pillars which are arranged between the pia mater and arachnoid. Those elastic complex structures could stretch and shrink. 23 Therefore, the presumably folded trabeculae likely stretch, leading to dilated ONSD when the ICP is elevated. Similarly, the trabeculae could refold, and the dilated optic nerve sheath shrinks when ICP is reduced. Our study confirmed that ONSD examinations could evaluate the elevated ICP and IH because of the elastic complex structure of the optic nerve sheath noninvasively and at the bedside (Figure 5).

Illustrations and ultrasonography images showing the changes in optic nerve sheath diameter (ONSD): (a) The trabeculae are refolded and the ONSD shrinks during intracranial hypotension. (b) The ultrasonic ONSD was 2.5 mm while the lumbar puncture pressure of this patient was 40 mm H2O. (c) The folded trabeculae are stretched leading to dilated ONSD while the intracranial pressure (ICP) is elevated. (d) The ultrasonic ONSD was 6.72 mm while the lumbar puncture pressure of this patient was 400 mm H2O.
MRI and CT can also be of diagnostic value in these disorders and have many advantages, such as high image accuracy and measurements. However, their use is limited by cost, radiation exposure, and transportation. Conversely, ultrasound is a safe, bedside, inexpensive, rapid, well-tolerated modality with the potential to evaluate ICP. Interestingly, in some cases where the MRI showed collapsed subarachnoid space, the optic nerve sheath could not be measured.17,20 However, sonographic ONSD can be detected and measured in patients with IH. This is significant for the initial IH diagnosis, and the dynamic observation of changes in IH and assessment of treatment approaches. In addition, other measurement issues of MRI or CT are eye motion artifacts. However, advising the patient to look at a single point in the gantry of the MR or CT scanner may minimize this artifact. 24 Furthermore, the duration of MR or CT scans is longer than that of ultrasonography, which can also quickly save images at any time.
Moreover, it is difficult to ensure a specified measurement slice in MRI because the eyes cannot move according to the scan imagery. In addition, MRI slices were not consistently located with respect to eyeball position. 20 In contrast, the patient can adjust eye movements in real-time during ultrasound examination to save the best ultrasonographic images and measured sites.
For IH, patients typically complain of orthostatic headaches accompanied by various symptoms, such as photophobia, nausea, neck stiffness, tinnitus, and hearing impairment. In some patients, however, headaches are not orthostatic and may mimic other types of headaches. Elevated ICP can also cause headaches. Therefore, it is important to evaluate ICP in patients with headaches using a noninvasive, easy-to-perform, and cheap technology. Based on this study results, this technology was useful to identify patients with abnormal ICP and efficiently avoid lower ICP in patients with IH undergoing LP (Figure 5). Although LP was regarded as a diagnostic criterion standard, it can cause lower ICP and/or increased headaches; therefore, it is not an ideal diagnostic procedure and should be avoided in these patients. In addition, ultrasound ONSD can avoid invasive monitoring. Infections, hemorrhage, and decalibration in long-term observations are known complications of invasive ICP monitoring. The basic objectives of this study were to explore the benefits of time-saving, effective and complication reduction in noninvasive ICP monitoring.
Furthermore, in recent years, pediatric studies have usually performed ultrasonographic ONSD measurements in patients with increased ICP.25,26 The prevention and treatment of IH is always neglected because atypical symptoms and expression are limited in children. It is noninvasive convenient, painless, fast, and could facilitate repeat inspection. Therefore the advantages of ultrasonographic ONSD may be investigated for IH in future pediatric studies. An ONSD protocol posed that the diagnostic accuracy of ONSD ultrasonography varies according to patient characteristics (e.g. age, weight, etc.). 25 However, few studies have confirmed whether interindividual factors affect the use of ultrasonographic ONSD measurements as a screening tool for IH. Age, sex, BMI, SBP, DBP, waistline, and head circumference were found to have no significant influence on the relationship between ONSD and ICP in our study. In recent years, some studies have found a decreased ONSD in patients with IH. However, few studies attempted to investigate the cut-off point for IH. In this study, we determined a preliminary cut-off point for IH as a basis for future investigations on the diagnostic value of sonographic ONSD in IH. Our previous study found that ethnic differences or genetic distinctions in ONSD might affect the cut-off point for elevated ICP. Thus, we propose that ethnic differences should be noted and maybe appropriately applied to the corresponding ultrasonographic ONSD criteria for IH in future studies.
Limitations
First, this was a single-center study, and as such, it should be considered a basis for future investigations of sonographic ONSD in IH. Second, the diagnostic value of ONSD measurements may be limited because of individual variations; therefore, our results are considered preliminary. In the future, the most accurate standard should be confirmed by studies with larger samples to validate the implementation of the technique in the general population. Third, this study included a relatively heterogeneous patient group with various etiologies for IH.
Conclusion
Ultrasonographic ONSD may be a noninvasive, valuable, and easily performable bedside technology for evaluating IH.
Footnotes
Acknowledgements
The authors thank the patients who participated in this study and their relatives.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fund Project of Jilin Provincial Finance Department (JLSWSRCZX2021-066).
