Abstract
Spontaneous intracranial hypotension is an uncommon but not rare cause of new onset daily persistent headaches. A delay in diagnosis is the norm. Women are affected more commonly than men and most are in the fifth or sixth decade of life. The underlying cause is a spontaneous spinal cerebrospinal fluid (CSF) leak. Typically the headache is orthostatic in nature but other headache patterns occur as well. Associated symptoms are common and include neck pain, a change in hearing, diplopia, facial numbness, cognitive abnormalities and even coma. Typical imaging findings consist of subdural fluid collections, pachymeningeal enhancement, pituitary hyperaemia and brain sagging, but magnetic resonance imaging may be normal. Myelography is the study of choice to identify the CSF leak but is not always necessary to make the diagnosis. Treatment consists of bedrest, abdominal binder, epidural blood patching, percutaneous fibrin glue injection or surgical CSF leak repair. Outcomes have been poorly studied.
INTRODUCTION
The headache that occurs upon standing and is relieved by lying down remains commonly misdiagnosed when there is no history of a spinal tap (1, 2). Nevertheless, much has been learned about spontaneous intracranial hypotension (SIH), particularly since the early 1990s (1–19), and this review will describe the history, pathology, epidemiology, clinical presentations, diagnostic criteria, radiographic features, treatment options and outcome of these patients.
History of terminology
The spontaneous occurrence of a low cerebrospinal fluid (CSF) state was first recognized in the 1930s by the German neurologist Georg Schaltenbrand, who introduced the term aliquorrhoea (3). This reflects the absence of any CSF upon multiple attempts at lumbar puncture. Subsequently, it was realized that a small amount of CSF could be obtained—but under decreased pressure—in most patients with this syndrome, and the term hypoliquorrhoea was used (4). Since the 1950s intracranial hypotension has been the most commonly used term describing this condition. Because the intracranial pressure or opening pressure at the time of lumbar puncture is not always abnormally low in patients with SIH, the term CSF hypovolaemia was coined (20). However, loss of CSF volume also occurs with spontaneous CSF rhinorrhoea or ottorrhoea, but the typical imaging and clinical features of SIH rarely, if ever, occur under these circumstances. Moreover, CSF hypovolaemia is a misnomer, because ‘hypovolaemia’ means low blood volume. Because all or essentially all cases of SIH occur secondary to a spontaneous spinal CSF leak, the terms spontaneous spinal CSF leak is the preferred descriptive term.
EPIDEMIOLOGY
SIH has long been considered an exceedingly rare disorder. However, recent evidence suggests that it is not particularly rare and that it has to be considered an important and relatively frequent cause of new daily persistent headaches in young and middle-aged individuals. No solid community-based figures are available to calculate the incidence and prevalence of SIH in this or any other country. In one manuscript published in 1994, mention was made of a prevalence of SIH of one per 50 000 (21). This was based on the presence of two patients with known SIH in a community of about 100 000. In a more recent study based on a review of radiographic studies of patients seen at an urban emergency department, it was found that SIH was diagnosed about half as frequently as spontaneous subarachnoid haemorrhage, for an estimated annual incidence of SIH of five per 100 000 (2). In the past, SIH was probably even more frequently underdiagnosed than it is now, and it is unlikely that there has been an actual increase in its frequency, although that possibility cannot be entirely excluded.
The onset of symptoms of patients with SIH is typically in the fifth or sixth decade of life with a peak incidence around age 40 years (1–20); however, children as young as 3 years and elderly individuals as old as 86 years have also been reported. SIH affects women more frequently than men, with a female to male ratio of approximately 1.5:1.
AETIOLOGY AND PATHOGENESIS
It can be stated with confidence that SIH is caused by spontaneous spinal CSF leaks. Because spinal CSF leaks generally do not cause any local symptoms, they remain undetected unless actively looked for in a patient suspected of SIH. Also, unlike CSF rhinorrhoea or otorrhoea, there is no risk of meningitis because the CSF is directly absorbed into the sterile spinal epidural venous plexus or the perispinal soft tissues, and it is therefore not exposed to the external milieu. Patients who have clinical and radiographic features of intracranial hypotension and who also have a CSF leak at the skull base should be evaluated for the presence of a spinal CSF leak as well, because CSF otorrhoea and rhinorrhoea do not cause positional headaches.
The precise cause of the spontaneous spinal CSF leaks remain unknown in the vast majority of patients, but underlying fragility of the spinal meninges generally is suspected. The aetiology and pathogenesis of spinal CSF leaks is multifactorial and, for example, a history of a more or less trivial traumatic event preceding the onset of symptoms can be elicited in approximately one-third of patients, suggesting an important role for mechanical factors as well (22–25). At the time of surgery, a wide variety of dural abnormalities may be observed, including dural holes or rents, complex fragile meninge diverticula, or even complete absence of the dura that normally covers the spinal nerve root (21, 26, 27).
There are several lines of evidence to suggest that a heritable underlying connective tissue disorder plays an important role in the development of spontaneous spinal CSF leaks (26, 28–33). Familial SIH has been described (32). Based on physical examination alone, evidence for underlying generalized connective tissue disorder can be found in up to two-thirds of patients (31). A named generalized connective tissue disorder is found in < 5% of patients (31). Of the named heritable connective tissue disorders associated with spontaneous spinal CSF leaks, Marfan syndrome, Ehlers–Danlos syndrome type II and autosomal dominant polycystic kidney disease are the most important (34–42). In addition, about 20% of patients with spontaneous spinal CSF leaks have isolated skeletal manifestations of Marfan syndrome such as tall stature, arachnodactyly, highly arched palate and joint hypermobility (29, 31). Even upon extensive evaluation, such as slit lamp examination of the eye and echocardiography, no other stigmata of the syndrome can be detected. It has now been shown that these patients do not harbour mutations in the Marfan syndrome gene FBN1 encoding fibrillin-1 or in the Marfan type II syndrome gene TGFBR2 encoding transforming growth factor-beta receptor proteins (29, 33). In addition to dural fragility related to an underlying connective tissue disorder, some patients with spontaneous spinal CSF leak are found to have osseous spinal pathology (43–48). Most commonly, this is an acquired degenerative disk disorder piercing the dura, but occasionally congenital bony spurs can be found. Before precise imaging was able to detect the underlying spinal CSF leak, it was speculated that SIH resulted from decreased secretion of CSF or a generalized increase in CSF absorption. However, there are no data to support such alternative mechanisms.
Although it is a widely held belief that the symptoms related to SIH are the result of low CSF volume, it has also been postulated that the orthostatic headaches seen in SIH may result from an altered distribution of craniospinal elasticity (49). This is due to spinal loss of CSF resulting in spinal dural sac collapse and increased compliance of the lower spinal CSF space. Through this mechanism, decreasing the compliance of the lower spinal CSF space can be restored by increasing dural stiffness.
DIAGNOSTIC CRITERIA
The most widely used diagnostic criteria for SIH are those published in 2004 in the International Classification of Headache Disorders, 2nd edn (ICHD-II) (Table 1) (50). However, it has become well established that the spectrum of clinical as well as radiographic manifestations of SIH is unusually broad, and this is not reflected by the ICHD-II criteria. Most notably, not all patients have the typical orthostatic headache that occurs within 15 min after sitting or standing, nor do all patients have meningeal enhancement on magnetic resonance imaging (MRI) or complete resolution of headache within 72 h of epidural blood patching. Therefore, new diagnostic criteria for spinal spontaneous CSF leaks and intracranial hypotension were developed that rely on both well-established radiographic and clinical criteria (Table 2) (51). However, these criteria need to be refined.
Diagnostic criteria for headache due to spontaneous spinal CSF leak and intracranial hypotension according to the International Classification of Headache Disorders, 2nd edn, 2004 (42)

CSF, cerebrospinal fluid; CT, computed tomography; MRI, magnetic resonance imaging.
Diagnostic criteria for spontaneous spinal CSF leak and intracranial hypotension

Patients with onset of symptoms following dural puncture or other penetrating spinal trauma are excluded.
CSF, cerebrospinal fluid; MRI, magnetic resonance imaging.
CLINICAL PRESENTATION
Orthostatic headache
The prototypical and most common manifestation of SIH is an orthostatic headache that may occur within seconds to minutes of assuming the upright position. In others, this period of time may be as long as several hours. Improvement of headache after lying down is somewhat less variable, but usually occurs within 30 min. The headache is usually described as diffuse and may be throbbing or non-throbbing. Alternatively, the headache may be localized to the frontotemporal or most commonly the occipital regions. Occasionally, the headache is not symmetric, but only rarely is it uniquely unilateral. A clue to the diagnosis may be offered by the patient who may describe the headache as an anvil sitting on top of the head, an ice cube in an empty glass, a pulling sensation from the head down to the neck, or the feeling of the brain being sucked down to the toes. Additional clues include the patient's recumbent position in the physician's office, the pillow they carry along with them at all times to allow them to lie down comfortably, or the fact that the patient volunteers that very tight-fitting clothes help with their symptoms. The vast majority of patients are able to recall the day the headache started, if not the exact time. The initial onset of headache in most patients is gradual or subacute, reaching maximal intensity in several minutes to hours, but in others the onset of headache is essentially instantaneous. Patients with such a thunderclap-type headache often will be suspected of having a subarachnoid haemorrhage and may undergo numerous invasive procedures such as cerebral angiography (52). The severity of headache varies widely. It is likely that many mild cases remain undiagnosed, whereas other patients are incapacitated for years or decades and are unable to engage in any useful activity while being upright. The exact cause of the headache is not known but may be related to the downward displacement of the brain due to loss of CSF buoyancy causing traction of pain-sensitive structures, particularly the intracranial or upper cervical dura. Alternatively, compensatory dilation of pain-sensitive intracranial venous structures may have a role in causing the headache. It should be noted that not all orthostatic headaches are caused by spontaneous spinal CSF leaks, and other diagnoses should be taken into consideration (53, 54).
Other headache patterns
An orthostatic headache is the clinical hallmark of SIH, as reflected by all diagnostic criteria, but it is well known that this posture-related component often becomes less prominent or even disappears over time when the underlying spinal CSF leak remains untreated. Rarely, the reverse time course is seen, with a non-positional headache preceding the onset of a typical orthostatic headache. In addition, patients with absolutely no postural related component to their headache from the onset have been described (55–57). Other headache patterns include exertional headaches, headaches that occur at the end of the day, or even paradoxical headaches that worsen when lying down (58–60). Intermittent headaches, possibly related to the presence of intermittent spinal CSF leaks, may occur at intervals of weeks or months or even years. Rarely, a patient denies having any headache, and this is usually seen when other symptoms of SIH predominate the clinical picture.
MISCELLANEOUS SYMPTOMS
In addition to headache, an ever expanding variety of additional symptoms have been reported in patients suffering from SIH. The most common, occurring in > 50% of patients with SIH, include posterior neck pain or stiffness, nausea and vomiting. The next most common symptoms, seen in approximately 10–50% of patients with SIH, include a change of hearing, which may be described as echoing or as being under water, tinnitus, and a disturbed sense of balance (61). Less common manifestations seen in < 10% of patients include visual blurring, visual field defects, diplopia, facial numbness or facial pain, facial weakness or spasm, dysgeusia, Parkinsonism, ataxia, cerebellar haemorrhage, dementia, interscapular pain, quadriplegia, upper extremity radicular symptoms, and galactorrhoea (62–75).
DIAGNOSIS
Brain MRI
MRI has revolutionized not only the recognition of SIH but also our understanding of the disease itself. MRI has greatly facilitated the ability to arrive at the diagnosis with confidence without having to resort to invasive procedures such as intracranial pressure monitoring or spinal puncture. Recognition of the MRI features is probably the most important factor responsible for the ever increasing number of patients diagnosed with SIH since the early 1990s. On the other hand, incomplete understanding of the variability of the MRI findings has resulted in the diagnosis of SIH being erroneously excluded in patients with normal brain MRI findings. There are five characteristic imaging features of SIH detectable on brain MRI for which the mnemonic SEEPS has been proposed (17). These imaging features include subdural fluid collections, enhancement of the pachymeninges, engorgement of venous structures, pituitary hyperaemia and sagging of the brain (Table 3) (Fig. 1).

Pretreatment (upper row) and post-treatment (lower row) axial fluid-attenuated inversion recovery magnetic resonance imaging (MRI) (A) showing resolution of bilateral subdural haematomas (arrowheads) and engorgement of the superior sagittal sinus (arrow) in a 35-year-old man; axial T1-weighted MRIs after intravenous administration of gadolinium (B) showing resolution of enhancement of the pachymeninges (arrowheads) in a 39-year-old woman; and sagittal T1-weighted MRIs showing resolution of sagging of the brain [flattening of the pons with obliteration of the prepontine cistern (arrowheads) and downward displacement of the cerebellar tonsils mimicking a Chiari malformation (arrowhead)] and hyperaemia/enlargement of the pituitary gland (arrow) in a 41-year-old woman. Also note restoration of ventricular size post treatment in all panels.
Magnetic resonance imaging findings

There has been recognition of an association between subdural fluid collections and intracranial hypotension since at least the 1950s (19, 76–82). Subdural fluid collections are common in SIH, occurring in about 50% of patients. Mostly, these subdural fluid collections represent hygromas and they are thin, bilateral, located over the cerebral convexities, and they do not cause any appreciable mass effect. They also may be seen in the posterior fossa, particularly over the cerebellar convexities or in the retroclival region. However, subdural haematomas with varying degrees of mass effect also are not uncommon in SIH and they have been described in about 20% of patients recognized as suffering from SIH. Fortunately, most of these subdural haematomas can be managed with treatment directed at the underlying spinal CSF leak (82). Thus, only a rare patient needs a craniotomy or burr hole evacuation of subdural haematomas. If a patient with an underlying spinal CSF leak undergoes treatment directed at the subdural haematoma without treatment of the CSF leak, then the risk of a recurrence of a subdural haematoma is quite high.
Enhancement of the pachymeninges has become the most well-known imaging abnormality of SIH (83, 84). It was first described in 1991 by Mokri and colleagues (83). The enhancement is diffuse, linear, spares the leptomeninges, and involves both the supratentorial and intratentorial compartments. It has now become well established that the pathological substrate for the enhancement is small thin-walled dilated blood vessels in the subdural zone. The relationship between this meningeal enhancement and SIH has been believed to be so close that the term ‘syndrome of orthostatic headache and diffused pachymeningeal gadolinium enhancement’ was coined (13). However, it is important to realize that up to 20% of patients recognized as having SIH never develop any meningeal enhancement (85, 86). Engorgement of venous structures may be difficult to detect on the initial MRI scan. However, in those cases where it is detected it usually affects the dural venous sinuses or large cerebral veins (87–89). Mostly it can be appreciated only when pretreatment and post-treatment images are compared.
Pituitary hyperaemia has recently been recognized as an imaging feature of SIH (90–93). The pituitary hyperaemia may become quite striking and may mimic pituitary tumour or pituitary hyperplasia.
Sagging or downward displacement of the brain is a very specific imaging finding of SIH. It is often accompanied by ventricular collapse (94). Sagging of the brain was first described in 1975 by Billings on the basis of a pneumo-encephalogram in a patient with SIH (9). The sagging of the brain is associated with several features that can be identified most readily on sagittal MRI imaging. These features include effacement of perichiasmatic cisterns with bowing of the optic chiasm over the pituitary fossa, effacement of the prepontine cistern with flattening of pons against the clivus, and downward displacement of cerebellar tonsils which may mimic a Chiari Type I malformation. Sagging of the brain in SIH is occasionally attributed to coexisting subdural fluid collections, but the degree of sagging generally is out of proportion to any mass effect from the subdural fluid collections. It should be realized that occasionally it is difficult to distinguish sagging of the brain from isolated downward displacement of the cerebellar tonsils. Although downward displacement of the cerebellar tonsils can be seen as the only imaging feature of spontaneous spinal CSF leaks, it is almost always accompanied by other imaging features of brain sagging.
Most of the MRI features of SIH can be explained as compensatory changes related to the loss of CSF volume. According to the Monroe–Kelly hypothesis, the sum of the volumes of intracranial blood CSF and cerebral tissue must remain constant in an intact cranium. Thus, the loss of CSF from the spine can be compensated for by increasing the vascular component, accounting for pachymeningeal enhancement, engorgement of venous structures, and pituitary hyperaemia. It can also be compensated for by an increase in intracranial CSF component accounting for subdural hygromas. Subdural haematomas may be caused by tearing of bridging veins or rupture of the dilated thin-walled blood vessels in the subdural zones. Sagging of the brain is caused by loss of CSF buoyancy. The application of the Monroe–Kelly hypothesis to explain the features seen in intracranial hypotension was first described by Billings and colleagues in 1979 (9) and was later expanded on by Fishman and Dillon (83) and Mokri (20).
Improvement of MRI abnormalities can be seen within hours to weeks of successful treatment of the CSF leak. Clinical improvement generally precedes that demonstrated on MRI, and in some patients, particularly those who have not receive specific treatment for their CSF leak, considerable clinical improvement is shown over time, whereas the MRI abnormalities persist. Large subdural haematomas may require up to 3 months to resolve after treatment of the spinal CSF leak.
Brain computed tomography
Although not as conclusive as MRI, computed tomography (CT) scanning can demonstrate subdural fluid collections, cerebellar tonsillar herniation, ventricular collapse as well as obliteration of the subarachnoid cisterns. Obliteration of the subarachnoid cisterns can give the picture of pseudo-subarachnoid haemorrhage (95). CT is of important diagnostic value particularly in the emergency department setting.
Myelography
Myelography with iodinated contrast followed by thin cut CT of the entire spine has been shown to be the study of choice to define accurately the location and extent of a CSF leak (12) (Fig. 2). Myelography with intrathecal administration of gadolinium followed by MRI is an excellent alternative (96, 97). The CSF leak may vary from a small amount of CSF tracking along a single nerve root to extensive bilateral collections of CSF within the paraspinal soft tissues. Single or multiple meningeal diverticula may be demonstrated. However, the diverticula may be below the level of detection of myelography and may not be uncovered until the time of surgery. Most spinal CSF leaks can be found at the cervicothoracic junction or along the thoracic spine. However, CT cuts not always cover the sacral spine following myelography, and sacral CSF leaks, although uncommon, may be missed. Frequently, multiple simultaneous CSF leaks are demonstrated. Retrospinal collections of contrast at C1-C2 are seen in about 5–10% of patients and they should not be mistaken for the actual site of the CSF leak (98). Delayed imaging may be required to visualize slow or intermittent leaks, and ultra early CT may be required to identify the site of a rapid high-volume leaks. Occasionally, repeating the myelogram following a course of several days of intrathecal infusion of mock CSF is required to visualize a CSF leak.
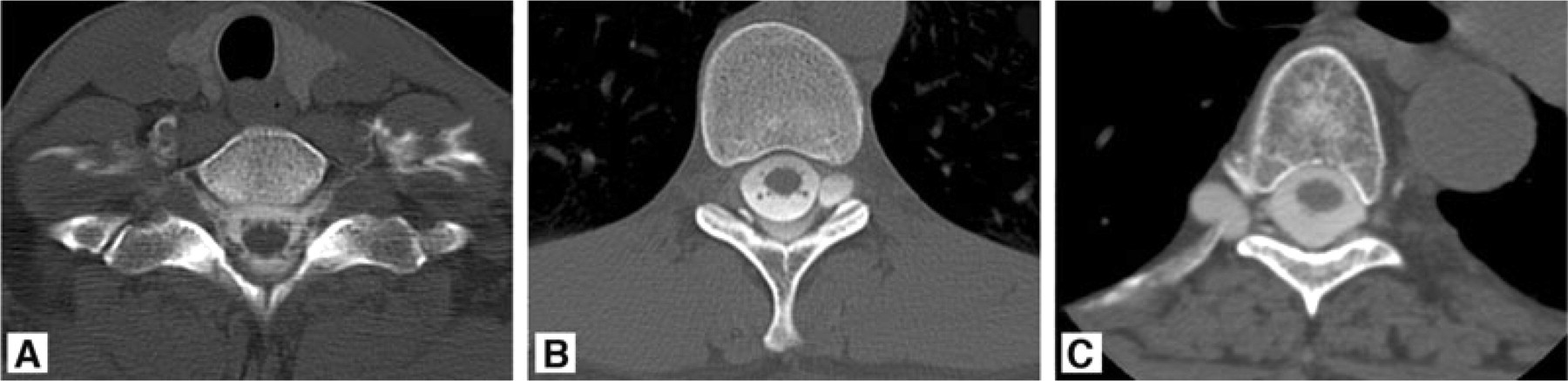
Post-myelography computed tomography showing bilateral cervicothoracic cerebrospinal fluid (CSF) leaks without an associated meningeal diverticulum (A); left thoracic meningeal diverticulum with an associated CSF leak (B); and right thoracic meningeal diverticulum without an associated CSF leak (C).
The fear of cerebral herniation caused by performing a lumbar puncture for myelography is entirely theoretical and has never been documented. Not only is the dural hole made by the lumbar puncture relatively small, but CSF pressure is already low. Aggravation of symptoms is reported by only approximately 5% of patients with SIH and it is generally mild. Of course, the occurrence of low-pressure headache is higher in those patients who do not have SIH but are evaluated for this with a myelogram.
Radionuclide cisternography
Radionuclide cisternography has been used extensively for many decades in the evaluation of SIH (99–101). However, its usefulness is relatively limited because of poor resolution. The exact site of the CSF leak remains obscure in as many as one-third of patients with proven leaks at myelography. Typical findings of radionuclide cisternography include early accumulation of tracer in the kidneys and bladder, slow accent along the spinal axis and a paucity of activity over the cerebral convexities (99–101). In our practice, radionuclide cisternography is rarely utilized. However, when the diagnosis of intracranial hypotension is in doubt and myelography is normal, then indirect findings of radionuclide cisternography may be useful.
Spinal MRI
In the past, relatively scant attention has been given to spinal MRI in the diagnosis of SIH, mainly because spinal MRI is not always effective in localizing the CSF leak. However, numerous spinal manifestations of SIH have been described, including dilated epidural or intradural veins, dural enhancement, meningeal diverticula, extrathecal CSF collections, syringomyelia and the presence of retrospinal fluid collections at C1-C2 (102–108). MR myelography may be useful in some patients to demonstrate a leak not seen on conventional MRI sequences (109).
Lumbar puncture
Typically, CSF opening pressure is < 6 cm of water and it can be immeasurable or even negative. However, it has been well documented that some patients with significant spinal CSF leaks have consistently normal CSF opening pressures (12, 13). Similarly, intracranial pressure may be normal. Examination of spinal CSF often demonstrates abnormal results and it may show a primarily lymphocytic pleocytosis and elevated protein content or xanthochromia that is probably due to increased permeability of dilated meningeal blood vessels and a decrease of CSF flow in the lumbar subarachnoid space.
TREATMENT AND OUTCOMES
Although data are completely lacking, it is often stated that most cases of SIH resolve spontaneously without any specific therapy. Fortunately, numerous options are available to treat patients with spontaneous hypotension who seek medical attention because of persistent symptoms. A purely conservative approach consists of bed rest, oral hydration, a generous caffeine intake and the use of an abdominal binder. An abdominal binder may be particularly effective, but the patient should be warned that after spending a full day upright utilizing the abdominal binder it is important to lie down while removing the binder and not get up for at least several minutes. Otherwise, if the abdominal binder is removed while the patient is upright, severe headache may occur within seconds. Other non-invasive treatments include the administration of mineralocorticoids, corticosteroids, intravenous caffeine or oral theophylline. However, the effectiveness of those treatments is limited.
The mainstay of treatment for SIH is the injection of autologous blood into the spinal epidural space, the so-called epidural blood patch (110, 111). Relief of symptoms is often instantaneous, thereby also serving a diagnostic purpose, since it is likely to be due to replacement of the loss of CSF volume with blood volume within the spinal canal. Initially, about 10–20 cm3 of blood is used and it is effective in relieving symptoms in about one-third of patients, presumably by forming a dural tamponade, thereby sealing the leak. Another mechanism of action may be restriction of CSF flow within the spinal epidural space, thereby interfering with CSF absorption. Finally, changing dural resistance/stiffness may be the final common pathway with regard to the efficacy of epidural blood patching.
If the epidural blood patch is unsuccessful it can be repeated, and consideration should be given to a large-volume epidural blood patch. Large-volume epidural blood patch is generally considered any blood patch with a volume > 20 cm3 of blood, and we have given up to 100 cm3 of blood. It should be remembered that the initial epidural blood patch, which was very successful in relieving symptoms, was done with only 0.5 cm3 of blood. Given the potentially high volume of injected blood, a minimum of 5 days between blood patches is advised. The volume of blood that can be injected is mainly limited by the development of local back pain or radiculopathy. Since most spinal CSF leaks occur in the thoracic or cervical spine, it is recommended to place the patient in a Trendelenburg position following the lumbar epidural blood patch. This will allow blood to travel over many spinal segments towards the site of the leak.
If epidural blood patching fails to provide sustained relief, then consideration should be given to the placement of a directed epidural blood patch or percutaneous placement of fibrin sealant. These therapies require that the exact site of the CSF leak be known, and placement of fibrin sealant probably provides the best chance of alleviating symptoms (112, 113). In the approximately one-quarter to one-third of patients for whom epidural blood patching has not been effective, excellent relief of symptoms can be expected with the percutaneous placement of fibrin sealant, thereby avoiding surgery. Commercially available fibrin sealant (Tisseel) contains a bovine product, and sensitization to this product has been reported if the percutaneous placement of fibrin sealant is repeated within 3–6 months of the initial treatment period (114). Therefore, we generally recommend waiting at least 3–6 months before the treatment is repeated.
The surgical treatment for spinal CSF leaks is reserved for those patients in whom other measures have failed. Surgical repair of CSF leaks is generally safe and often succeeds in providing relief for those patients in whom a structural abnormality or focal CSF leak is identified. Leaking meningeal diverticula can be ligated with metal aneurysm clips or by placement of a muscle pledget along with gel foam and fibrin sealant around the area of the leak. Rarely, the dural rent can be repaired with primary suturing. Intradural exploration may be required in some patients.
Intrathecal infusion of saline or mock CSF should not be expected to seal a CSF leak, but it may be required as an effective temporizing measure to restore CSF volume until the leak can be permanently repaired in patients who require urgent treatment, such as those with a decreased level of consciousness.
The recurrence of headache following successful treatment of SIH may indicate a recurrent CSF leak (115). However, if the pattern of headache is changed, then consideration should be given to the development of rebound intracranial hypertension or dural venous sinus thrombosis (116–118). Rebound intracranial hypertension can be treated with a course of a Diamox.
Data on long-term outcomes of patients suffering from SIH are scarce, but it has been stated that recurrence can be seen in approximately 10% of patients, regardless of treatment. Outcome studies have shown that patients with abnormal brain MRI findings and a focal spinal CSF leak have an excellent prognosis, whereas those with normal initial MRI findings and a diffuse multilevel spinal CSF leak appear to have a poor prognosis (119). Some patients have persistent symptoms following treatment, in spite of documented resolution of CSF leakage. Such patients are difficult to treat, and it is unclear whether they have residual altered CSF dynamics or perhaps small residual CSF leaks below the level of detection of current imaging techniques.
