Abstract
Alemtuzumab, one of the very effective immunomodulatory forms of treatment for patients with highly active multiple sclerosis, can also have unexpected side effects. We present a case of cystic echinococcosis causing thrombosis of the inferior vena cava in a female patient after two cycles of alemtuzumab.
Introduction
Alemtuzumab (ALEM) is a humanized monoclonal antibody indicated for the treatment of patients with active relapsing-remitting multiple sclerosis (RRMS). In clinical trials, ALEM demonstrated efficacy in both treatment-naïve patients and those relapsing after prior disease-modifying treatments (DMTs). So far, several serious adverse events have been reported. The most frequent are infusion reactions, secondary autoimmune diseases and opportunistic infections. 1
Here, we present a rare case of cystic echinococcosis (CE) in a 42-year-old White woman with active RRMS, which was revealed after ALEM treatment, causing serious venous thrombosis.
Case report
In 2009, the patient was diagnosed with RRMS and treated with interferon beta-1a and later with natalizumab (2012–2015). After a positive anti-JC virus antibody status she stopped taking natalizumab and was switched to dimethyl fumarate (2015–2016). During the 12-month treatment, she experienced two disabling relapses. Magnetic resonance imaging (MRI) showed an increased number of T2 hyperintensive demyelinating brain lesions, two of which were active. As a patient with active RRMS, she was given ALEM.
In the patient’s history, an isolated echinococcal cyst (10 mm) was detected in the liver in 2009. The diagnosis was supported by laboratory findings of elevated ELISA IgG, IgM antibodies against Echinococcus multilocularis. She received anthelmintic treatment for 6 months, until the antibodies declined. Between 2010 and 2018, she did not have any clinical symptoms of echinococcosis. She required immunomodulatory treatment due to active RRMS. The risk of the reactivation of echinococcosis was not assessed as serious, so ALEM treatment was not contraindicated.
After the first ALEM cycle (October 2016), she experienced repeated episodes of acute iridocyclitis in both eyes that stabilised after corticosteroid treatment. Recurrent acute iridocyclitis was judged as a secondary autoimmune disease associated with ALEM treatment, and this postponed the second cycle of ALEM to September 2018. Two weeks after completing the second cycle of ALEM, she developed dyspeptic syndrome lasting several days (September 2018). An ultrasound examination of her abdomen and CT-phlebography (Figure 1) showed compression of the inferior vena cava (IVC) with cystic lesions. Laboratory tests showed IgE elevated to ⩾1000 kU/L (0–100), hypochromic anaemia (HGB 90 g/L) and lymphopaenia (0.65 × 109/L). Additional laboratory testing revealed an elevation of serum antibodies against Echinococcus multilocularis, so the patient started antiparasitic treatment with 400 mg of albendazole daily. However, the patient decided to discontinue about 2 weeks into the treatment.

Coronal (a) and axial (b) CT-phlebography of abdominal veins detected compression of the IVC (white arrow) by multicystic heterogeneous masses (red arrow) and stenosis of the suprarenal IVC lumen.
In March 2019, the patient experienced an intermittent increase in body temperature to over 38°C, shivering and abdominal pain, mostly in the evenings. Although no infection was proved, the infectious disease specialist indicated antibiotics (piperacillin/tazobactam i.v.) to treat the symptoms. Additional examinations (ultrasonography of the abdomen, CT scans of the head, chest and abdomen, MRI examination of the abdomen) showed a massive reactivation of echinococcosis in the abdominal area (right retroperineum, the suprarenal part of the IVC) (Figure 2). The IVC and common iliac veins bilaterally were nearly completely obturated by hyperintense material on native T1 fat saturated sequence (Figure 3) and after contrast medium administration without an enhancement (Figures 4 and 5), possibly CE, as suggested by the radiologist.
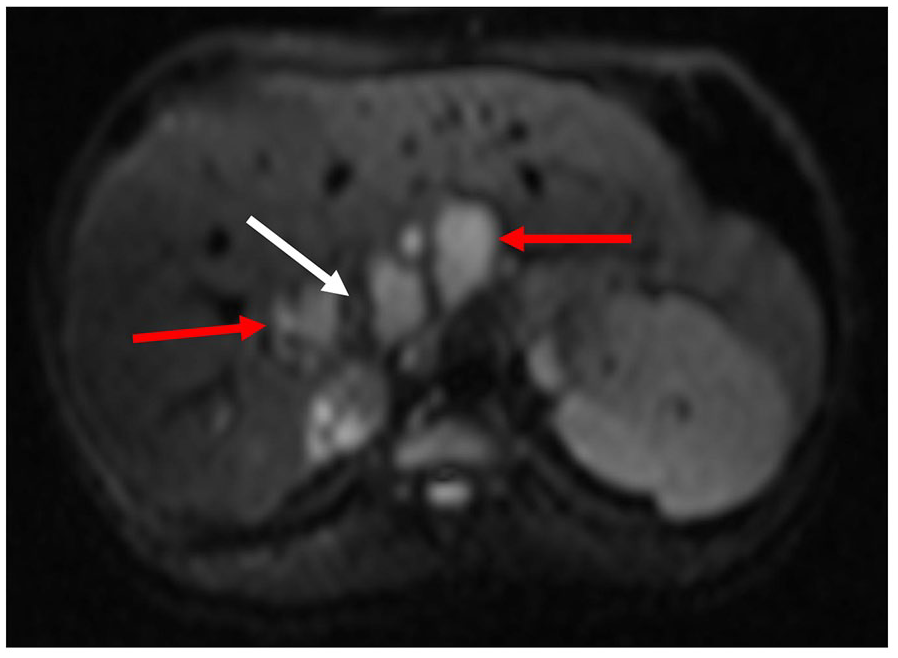
Axial abdominal MRI showed on diffusion-weighted image a hyperintense signal of multicystic masses (red arrow) localized around the stenotic IVC lumen (white arrow). Restricted diffusion of multicystic masses represented an acute stage of echinococcosis.
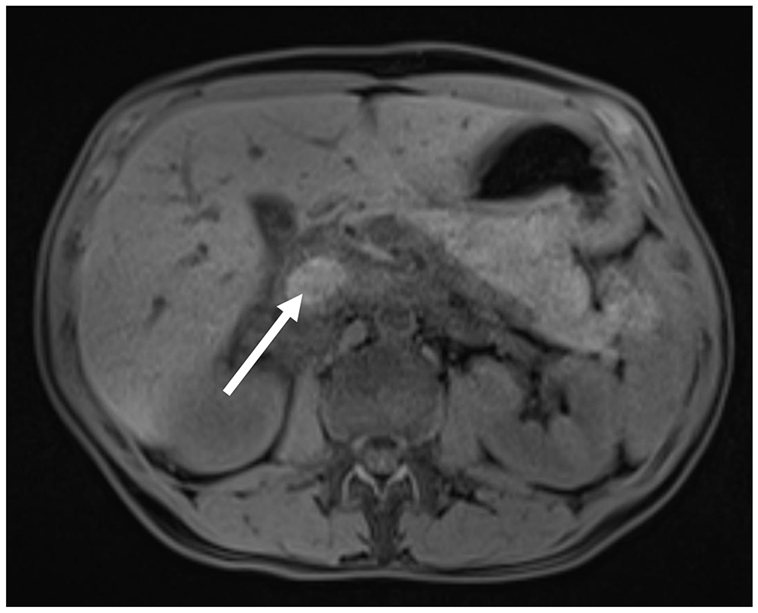
Axial abdominal MRI showed on native T1 fat saturated sequence an abnormal hyperintense signal of the IVC lumen (white arrow) that indicated a suspicious thrombosis of the IVC.
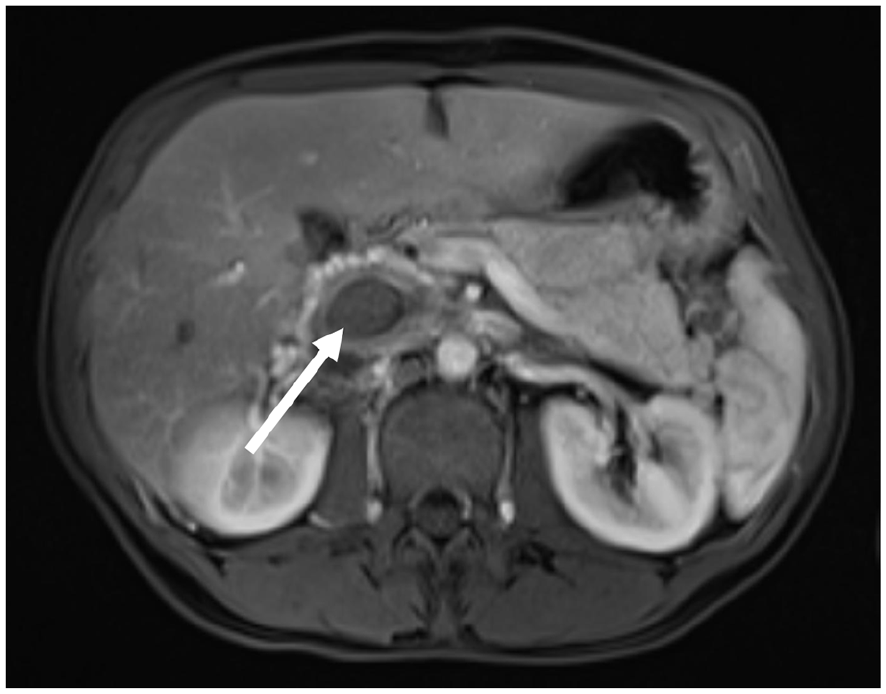
Axial abdominal MRI showed on postcontrast T1 fat saturated sequence a hypointense signal of the IVC lumen (white arrow) that confirmed a thrombosis of the IVC.

Coronal abdominal MRI showed on postcontrast T1 fat saturated sequence a hypointense signal of the IVC (white arrow), bilateral iliac veins (red arrow) and renal veins (yellow arrow) that confirmed their thrombosis.
In the liver, the isolated echinococcal cyst transformed into CE and was enlarged to 39 mm.
In March 2019, a peripheral blood count showed normal neutrophils (7.07 × 109/L) and increased monocytes (0.98 × 109/L), but lymphocytes remained decreased (0.86 × 109/L). Tests of kidneys, liver and coagulation parameters were normal. Microbiological samples from blood, urine, tonsils and the nose were all negative. To exclude intracardiac dissemination, echocardiography was performed with normal results.
After the initiation of antiparasitic therapy (albendazole 400 mg/day) and intravenous antibiotic treatment, the patient improved. We supposed that the febrile reaction was caused by the re-activation of chronic echinococcosis with dissemination of CE to other abdominal regions, affecting the veins.
In April 2019, once the treatment of the acute symptoms of CE had been completed, the patient’s case was discussed with surgeons in the University Hospital in Martin, but the surgical removal of CE was not advised due to a high peri-operative risk of secondary complications. They confirmed chronic but asymptomatic thrombosis of the IVC and common iliac veins, with developed collaterals.
During September and October 2019, the patient experienced another episode of acute iridocyclitis as well as an episode of acute thyroiditis. At present (November/December 2019), the patient is stabilised. She is on anticoagulation medication (apixaban 10 mg/day) and continuing the antiparasitic treatment (albendazole 400 mg/day). She also needed local ophthalmological therapy. The therapy for acute thyroiditis (thiamazole) has now stopped.
Discussion
The Echinococcus organisms, the cause of CE, are parasitic helminths with life cycles involving humans as intermediate hosts. In humans, CE disease reflects the development and growth of fluid-filled hydatid cysts mainly in the liver, lungs and occasionally other organs. It seems that the immune system plays a crucial role in limiting cystic development. 2
In long-term immune-compromised patients, CE disease was described as an opportunistic infection in patients with HIV. 3 To our knowledge, we are presenting the first documented case of ALEM causing aggressive CE disease.
In our patient, chronic thrombosis of the IVC was the most dangerous consequence of aggressive CE disease. The vessels were not only compressed by CE, but echinococcosis might have been the cause of IVC thrombosis by transmitting CE into the venous lumen. The disproportion of cytokines released after the administration of ALEM, 1 probably coworking with over-activated helminths, could explain the thrombotic pathology of the vascular compartment.
However, in patients with RRMS, pulmonary embolism and other thrombotic events have been reported as adverse effects of ALEM. 4 Moreover, a recently published meta-analysis estimates MS to be associated with an increased risk of venous thromboembolism (VTE). 5 As has been noted by the authors, it is unclear whether an increased VTE risk in patients with RRMS lies in the disease itself, in its complications or in the treatment. 5 In this case, we consider there to be a strong link between VTE and CE.
ALEM is known as a repopulation treatment. Although a new population of lymphocytes is expected 6 weeks after infusions, 1 this was not so in our case. The decline in all subpopulations of T lymphocytes and B lymphocytes was prolonged to over 12 months. We also found a decline in NK cells, which may activate CE disease. 2 Currently flow-cytometry results are not included in the indication criteria. We suppose that the testing of both cellular and humoral immunity could be beneficial in evaluating the risks of potential complications. Our case also raises the question of whether a prolonged decline in lymphocytes in peripheral blood would have signalled both the prolonged effect of ALEM to stop MS and an increased risk of further complications. In the presented case, we detected as CE and subsequent deep venous thrombosis so the re-activation of secondary autoimmune diseases (acute uveitis, acute thyrotoxicosis).
Before ALEM, our patient received three other immunomodulatory treatments with different mechanisms of action that could also have impacted the activity of the immune system. It seems safer to use long-lasting ALEM as one of the first DMTs in patients with active RRMS, since using it after three or more DMTs with cumulated effects on the immune system presents a high risk of unpredictable complications.
Although the development of monoclonal antibodies for therapeutic use such as ALEM allows the targeting of specific immune mechanisms that can have neuroprotective effects, we should also discuss critically the pitfalls and potential hazards of these new therapeutic strategies. 6
Conclusion
Our case pinpoints the importance of pharmacological vigilance during long-term immunomodulatory interventions. Future studies may expand our knowledge about the impacts of sequencing immunomodulatory medications on both innate and adaptive immunity with regards to secondary immunodeficiency and its potential side effects. In ALEM-treated patients, more information from real clinical practice is needed. Experiences with secondary complications and their evolution should be shared.
Footnotes
Authors contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
The patient signed a written agreement to publish her case. The adverse events were reported via study OBS13434.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Grant VEGA 1/0301/19.
