Abstract
Objective
We aimed to analyze the pathological changes of the occlusive area of the inferior vena cava (IVC) in patients with Budd–Chiari syndrome (BCS) and to explore its possible etiology and pathogenesis.
Methods
Color Doppler ultrasound and IVC angiography were used to evaluate causes of occlusion. Clamp biopsies were performed in the occlusive area of the IVC in 31 patients with BCS, and the specimens were examined pathologically. Clinical data were collected retrospectively.
Results
The success rate of clamp biopsy was 100% (31/31) and the sensitivity of pathology was 96.8% (30/31). By microscopic analysis, we found 27 cases of fibrous connective tissue, including 8 cases of thrombosis and 19 cases of inflammatory cell infiltration, vitreous degeneration, or mucinous degeneration of fibrous connective tissue; 3 cases showed thrombus organized with fibrosis.
Conclusion
The occluding lesion in the IVC of BCS may be caused by local thrombosis, followed gradually by organization and fibrosis.
Introduction
Budd–Chiari syndrome (BCS) is an uncommon heterogeneous group of diseases characterized by the obstruction of the hepatic vein (HV), the inferior vena cava (IVC), or both, leading to liver tissue damage caused by retrohepatic portal hypertension and significant morbidity and mortality.1–4 Patil et al. 5 have described the pathological findings of BCS. Pure HV obstruction is predominant in Western countries, 3 whereas IVC obstruction is reported more often in Asia. Clinical manifestations might differ between patients with IVC obstruction and those with HV branch obstruction.6,7 Until now, the most common explanation for the pathogenesis of BCS was thrombosis (with a variety of causes, commonly myelodysplastic syndrome) or membrane formation within the IVC, among others. However, the cause of IVC constriction without thrombosis is unknown, and the pathogenesis of IVC constriction is of interest to many researchers. To determine the pathological changes associated with IVC constriction in BCS, especially that of idiopathic origin, we performed clamp biopsies to obtain tissue for a histopathologic study of IVC.
Materials and methods
Clinical data
This retrospective study was approved by the Ethics Committee of The First Affiliated Hospital of Zhengzhou University. Written informed consent was signed by all participants before interventional procedures. All methods were carried out in accordance with local guidelines and regulations for a clinical study. Approximately 810 patients with BCS were treated in the Department of Interventional Radiology in the First Affiliated Hospital of Zhengzhou University from March 2011 to January 2018. Thirty-one patients with complicated BCS, 18 men and 13 women, were selected for biopsy to investigate pathology. The age range was from 34 to 66 years, with a median of 49.0 years. The median time of medical history was 3.0 years (range 0.5–30.0 years). Clinical manifestations, including hepatosplenomegaly, abdominal distension, edema, or pigmentation of lower extremities, were recorded. Color Doppler ultrasound and IVC angiography were used to evaluate the causes of IVC occlusion (Figure 1).

CT and color Doppler ultrasound before and after interventional treatments. (a) CT cross section showed that there was no contrast agent observed in IVC. (b) CT coronal section showed the morphology of IVC, and (c) color Doppler ultrasound showed that the echo in IVC was uneven. (d) After a recyclable stent was placed, CT showed blood flow in IVC. (e) CT coronal imaging showed that the recovered stent had the correct shape and position, and (f) color Doppler ultrasound showed that the IVC was unobstructed without thrombosis after stent placement.
Clamp biopsy procedure
Angiography of IVC was established through access to the femoral vein. For patients with IVC stenosis, a reinforcing guidewire and a 5F catheter were introduced to the superior vena cava; for patients with IVC occlusion, the occluded segment of the IVC was punctured and a reinforcing guidewire introduced to the superior vena cava after confirming puncture by angiography. Along the reinforcing guidewire, a 10F to 14F long sheath tube was introduced below the obstructed section of the IVC. The head end of the long sheath was located below the section of IVC stenosis, and endoscopic biopsy forceps (Olympus, Tokyo, Japan) were inserted through the sheath tube into the site of the lesion. The area of IVC obstruction was biopsied using the biopsy forceps. IVC balloon dilatation or stent placement (or both) was performed following the biopsy (Figure 2). 8 Balloon angioplasty was performed in 19 patients, and stents were placed in 4 patients (two recyclable and two permanent stents). Three patients underwent catheter-directed thrombolysis, and the remaining patients underwent oral anticoagulant therapy with rivaroxaban.

A patient with Budd–Chiari syndrome treated with balloon dilatation, clamp biopsies, and stent placement. (a) Angiography showed a membranous occlusion in IVC such that the contrast agent could not return to the right atrium. (b) The guidewire and catheter were successfully introduced through the segment of membranous occlusion. (c) A clamp biopsy of the occlusion was performed via the long sheath of the catheter. (d) Pre-dilation was performed using a small balloon catheter. (e) A recyclable stent was inserted. (f) Angiography showed that the IVC was unobstructed after stent placement. (g) Large balloon dilation was performed after removal of the stent. (h) Angiography showed that blood flow was fluent in the IVC.
Preparation of specimens
Tissue specimens from the occluded IVC were embedded in paraffin, cut into 5-µm-thick sections, stained with hematoxylin and eosin, and studied microscopically. Pathological changes, including fundamental laminar structure and histological characteristics of thrombi, were observed by light microscopic evaluation.
Results
In this study, the hepatic portion of the IVC was studied in 31 patients with BCS, and no cause was found for occlusion of the IVC. A sufficient amount of vena cava tissue was available for evaluation in all cases (31/31). As shown in Table 1, the clinical manifestations were hepatosplenomegaly (n = 5), abdominal distension (n = 9), edema of lower extremities (n = 12), pigmentation of lower extremities (n = 5), and varicose veins of abdominal wall (n = 5). Color Doppler ultrasound showed 5 cases of severe membranous stenosis of IVC, 14 cases of membranous occlusion, 2 cases of segmental stenosis, and 10 cases of segmental occlusion. IVC angiography was used to confirm the ultrasound diagnoses. The white, dark red, or red tissues were taken out in all 31 cases, with a thickness of 2 to 5 mm; all specimens met the requirements for pathological diagnosis. The technical success rate of biopsies was 100% (31/31). All cases were successfully biopsied except for one case of myocardial tissue taken by mistake; the sensitivity of pathology was 96.8% (30/31).
Clinical characteristics of enrolled patients.
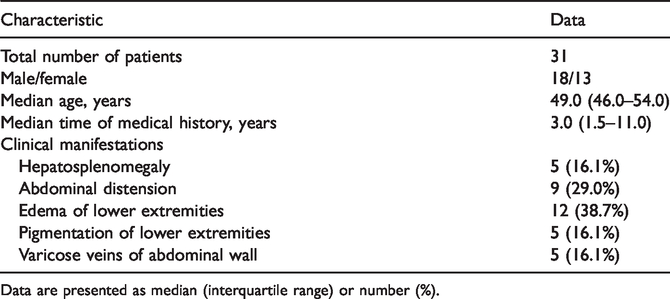
Data are presented as median (interquartile range) or number (%).
Pathology showed a normal basic structure of the venous wall in the occluding lesions. Twenty-seven biopsy samples were diagnosed as fibroconnective tissue, including 10 cases of mucinous degeneration, 2 of vitreous degeneration, 2 of red fresh thrombus, 6 of mixed thrombus (fresh thrombi, organized thrombi, fibrous tissue), and 2 of neovascularization in fibrous connective tissue, indicating that thrombus was mechanically recanalized. The remaining five cases showed only fibroconnective tissue. Three cases showed thrombus organized with fibrosis (Figure 3). In 19 cases, chronic inflammatory cell infiltration was found.

Pathological examination. (a, b) Fibrous connective tissue and vitreous degeneration; (c, d) pathological changes of thrombus.
Discussion
Thrombogenic condition is a known cause of BCS; the primary hepatic vein lesions are associated with coagulation disorders, myeloproliferative disorder, use of oral contraceptives, or paroxysmal nocturnal hemoglobinuria, among other underlying causes. Occlusion in IVC is much less common in Western countries but seems to occur frequently in developing countries. 7 There has been no systematic study of the histopathology of occlusion in IVC, 9 and the etiology and pathogenesis of BCS are not very clear; thus, a histopathologic study is warranted.
According to the congenital malformation theory, occlusion in the IVC is thought to be a congenital anomaly, although its cause is not clearly established. However, patients with congenital BCS have a clinical course frequently complicated by hepatocellular carcinoma, which is generally considered a disease different from the conventional type of BCS. This theory fails to explain why most patients with IVC occlusion are adults.
In BCS, obstruction often occurs in the hepatic portion of the IVC, and it can occur following mechanical injury or infection. The mechanical injury theory holds that constant movements of the diaphragm cause mechanical strain and possibly microscopic injuries to the intima of the IVC that could trigger thrombosis or invite bacterial infection into the IVC. Hirooka and Kimura 10 postulated that a membrane formed within the liver due to abnormal blood draining in the early embryonal liver. The membrane was described as fibrous connective tissue with some elastic and smooth muscle fibers, histology identical to that seen in our study, which we interpreted as a natural sequela to thrombosis.
The thrombosis theory holds that thrombosis and its gradual organization, caused by a variable blood hypercoagulable state, is the cause of BCS. Occlusion in the IVC is probably a sequela to thrombosis, considering that a transition can be observed from fresh thrombus to fibrous connective tissue with or without calcification in the obstructed IVC. In our biopsies, the three-layer fundamental structure of the IVC wall was clearly recognized and no obvious evidence of congenital malformation was found.
Our findings in this study indicate that BCS is likely to be an acquired disease. Among the specimens obtained, three cases showed mixed thrombus and the remainder showed fibrous connective tissue combined with thrombosis. These findings suggest that the disease is initially caused by thrombosis induced by various causes. With development of the disease, thrombus organization and fibrosis lead to formation of fibrous connective tissue, resulting in membranous or segmental obstruction.
There are some limitations to this study. This was a retrospective study and the sample size was relatively small. A prospective study with a large patient population is needed. Because of the lack of a control group, we conducted no statistical analyses.
Fibrous connective tissue in the area of IVC obstruction is often accompanied by vitreous or mucinous degeneration. Chronic inflammatory cell infiltrations involving a number of different cell types was seen in 19 patients, suggesting that the inflammatory reaction is closely related to formation and development of IVC obstruction. Kage et al. 9 found that stenosis and occlusion of the IVC is due to thrombosis and its sequelae, not to abnormal congenital development.
Conclusions
Occluding lesions in the IVC of BCS may be caused by local thrombosis, followed gradually by organization and fibrosis.
Footnotes
Authors’ contributions
Yonghua Bi and Xiaoyan Zhu researched the literature and conceived the study. Mengfei Yi was involved in data analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by National Natural Science Foundation of China (Grant No. 81501569).
