Abstract
The inferior vena cava (IVC) may develop abnormally because of its complex embryogenesis. An understanding of congenital variants such as duplication of the IVC is essential for clinical interventions, particularly those performed by surgeons and radiologists. We herein describe five patients who were diagnosed with duplication of the IVC by computed tomography or angiography and summarize their imaging and clinical features. All five patients were men aged 46 to 78 years. Two of the patients had pulmonary embolism and deep vein thrombosis and were treated by placement of an IVC filter and catheter-directed thrombolysis. The IVC in all patients ascended on either side of the abdominal aorta. All left IVCs terminated in the left renal vein, which crossed the aorta and joined the right IVC. The average follow-up time was 29 months (range, 14–46 months), and no patients developed venous thromboembolism or recurrence of thrombosis. Duplication of the IVC can be diagnosed by computed tomography and angiography. Its course and relationship with the renal vein must be identified for accurate planning of IVC filter placement in the setting of deep vein thrombosis and pulmonary embolism.
Keywords
Introduction
Duplication of the inferior vena cava (IVC) is a congenital anomaly with an incidence of 0.1% to 3.5%. 1 The correct diagnosis is important for retroperitoneal surgery and venous interventions. 2 Most patients have no clinical symptoms; this condition is mainly diagnosed incidentally or by imaging or autopsy. We herein report five cases of duplication of the IVC and summarize their imaging and clinical features.
Patients and methods
We searched our single-institution procedural database for all consecutive cases of duplication of the IVC treated by surgery from January 2009 to October 2020. The inclusion criterion for our study was a diagnosis of duplication of the IVC by computed tomography (CT) or angiography. Patients were excluded from the review if they had incomplete data or refused to provide written consent for inclusion of their data in the study. We recorded the clinical manifestations, CT or angiographic findings, location of the duplicated IVC, imaging characteristics, and clinical characteristics.
Results
Five patients were included in this study. All five patients were men aged 46 to 78 years. In three patients, duplication of the IVC was incidentally found on a CT scan. These patients had no vascular-related diseases. The remaining two patients had pulmonary embolism (PE) and deep vein thrombosis (DVT). One was diagnosed with duplication of the IVC by angiography. The other was found to have a slender IVC (about 12 mm) on angiography, and duplication of the IVC was subsequently diagnosed by abdominal CT. An IVC filter (Tempofilter II; B. Braun, Melsungen, Germany) was placed in the suprarenal position in these two patients. In both patients, the PE affected the main pulmonary artery and was treated by catheter-directed thrombolysis via the right subclavian vein to the main pulmonary artery (Uni-Fuse; AngioDynamics, Latham, NY, USA). For anticoagulant treatment, these two patients received low-molecular-weight heparin during hospitalization and were discharged with rivaroxaban. The patients’ demographics, main diagnosis, comorbidities, confluence of the left and right IVC, treatments, and outcomes are summarized in Table 1.
Patients’ demographics, main diagnosis, confluence of the left and right IVC, treatments, and outcomes.
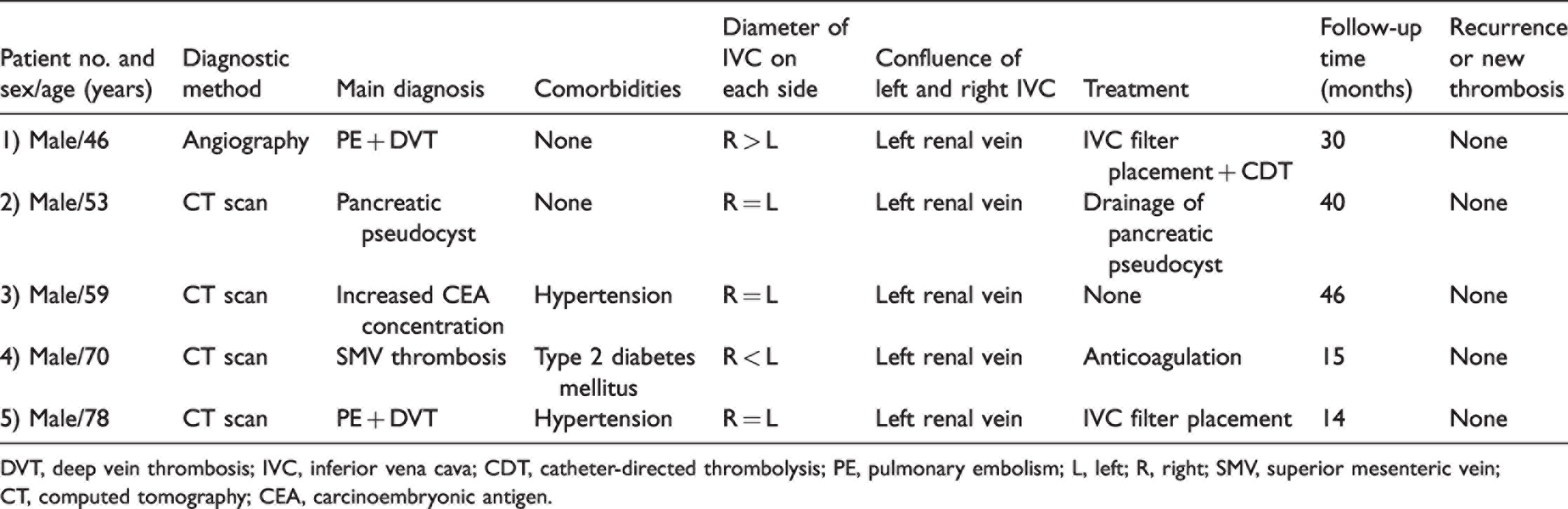
DVT, deep vein thrombosis; IVC, inferior vena cava; CDT, catheter-directed thrombolysis; PE, pulmonary embolism; L, left; R, right; SMV, superior mesenteric vein; CT, computed tomography; CEA, carcinoembryonic antigen.
In all patients, both IVCs ascended on either side of the abdominal aorta (Figure 1). The left IVC joined the left renal vein, crossed the aorta as the left renal vein, and finally drained into the right IVC.

Angiography and computed tomography findings. (a) Angiography shows a normal-caliber right-sided IVC. The left common iliac vein inflow is not seen. (b) Angiography shows a small-caliber left-sided IVC and (c) Angiography shows a left IVC terminating in the left renal vein and then communicating with the common suprarenal IVC. 2, 3, 4, 5: Computed tomography images showing a duplicated IVC. The arrows point to the duplicated IVC on either side of the aorta.
No patients exhibited a connection between the iliac vein and contralateral IVC. The diameters of the duplicated IVCs were similar in three patients, the left-sided IVC was wider in one patient, and the right-sided IVC was wider in one patient.
The average follow-up time was 29 months (range, 14–46 months), and no patients developed venous thromboembolism or recurrence of thrombosis.
Discussion
The IVC is formed between weeks 6 and 10 of gestation. Duplication occurs in an estimated 0.2% to 3.0% of the general population. 3 Embryogenesis of the IVC is a complex process involving anastomosis, asymmetric degeneration, and vessel growth of three pairs of embryonic veins (posterior cardinal, subcardinal, and supracardinal veins). 4 Formation of the IVC depends on the presence of this irregular network of vessels, which is constantly altered during the embryonic stage. This has been suggested as an alternative theory to explain the IVC anomaly. 3
There are two main types of duplicated IVC. 4 In the most common variety, as in our group of patients, both IVCs ascend on either side of the abdominal aorta and the left IVC joins the left renal vein, which then crosses the aorta and drains into the right-sided IVC. Each IVC usually receives its corresponding suprarenal, renal, and lumbar veins, although the left-sided IVC may drain into the left renal vein rather than directly into the right-sided IVC. A CT scan usually shows an IVC on each side of the abdominal aorta. The diameters of the two IVCs can be similar or different. The right IVC is obviously enlarged above the renal vein. One or more cross-communicating veins may be seen between the left- and right-sided IVCs. In the less common variety of duplicated IVC, both vessels are ipsilateral and most often on the right side. 5 Double helical IVC duplication has also been reported; in such cases, the bilateral common iliac veins cross each other and spiral upward as duplicated IVCs before joining the renal veins. 6
Duplication of the IVC is one of various congenital anomalies, some of which have potential clinical consequences. However, IVC duplication is usually asymptomatic and often diagnosed during routine imaging studies. DVT of the lower extremities and PE are possible clinical manifestations, and the prevalence of IVC thrombosis ranges from 60% to 80% among patients with congenital IVC anomalies. 7 Virchow’s triad describes the three main factors contributing to thrombosis: hypercoagulability, vessel injury, and venous stasis. Duplication of the IVC may predispose to venous thromboembolism due to resultant venous stasis because the blood return may be inadequate. This inadequate blood return may increase the blood pressure in the veins of the lower extremities, resulting in venous stasis and subsequent DVT. 8 For young patients with DVT of the lower extremities, we must be alert to IVC anomalies including duplication of the IVC. Among our five patients, three had no thrombosis event and were incidentally diagnosed by imaging. The literature also contains many cases in which duplication of the IVC was diagnosed in other situations, such as during autopsy and retroperitoneal surgery (particularly for abdominal aortic aneurysm repair and live donor nephrectomy). 9,10 This suggests that not all duplicated IVCs lead to venous thrombosis or PE. Therefore, which type of duplicated IVC is likely to cause venous thrombosis requires further study. Notably, IVC anomalies have become a recognized risk factor for DVT of the lower extremities, especially in young people. 11 In one study, potential IVC abnormalities were found in 5% of young patients with spontaneous unprovoked deep vein thrombosis. 11 An IVC anomaly is classified according to the location of the anomaly, which can be divided into three types: subrenal, renal, and suprarenal. However, which type is more likely to result in venous thrombosis is unclear. 7 Theoretically, when duplication of the IVC results in slower blood flow, venous thrombosis is more likely to occur. Therefore, when encountering patients with a duplicated IVC, attention should be paid to determining whether venous compression is present because such patients may also have chronic venous insufficiency.
Although a duplicated IVC is rare and frequently asymptomatic, it is still clinically significant. Thus, the following points should be considered.
(1) Avoid misdiagnosis or missed diagnosis. A duplicated IVC can potentially be misdiagnosed as lymphadenopathy, left pyeloureteric dilatation, retroperitoneal cysts, and loops of small bowel.2,3 This may even lead to unnecessary or erroneous treatment. (2) The presence of DVT and PE must be considered. Especially in young patients, when there are no other common risk factors for venous thrombosis, the duplicated IVC anomaly and the influence of the anticoagulation time on blood flow should be fully considered.12 When performing endovascular treatment such as IVC filter implantation, the possibility of a duplicated IVC should be taken into account, and if necessary, a bilateral IVC filter should be inserted or placed above the confluence of the left and right IVCs.13 (3) Duplication of the IVC can complicate retroperitoneal surgery.14 Summarizing the anatomical characteristics of the duplicated IVC may be necessary to determine the feasibility of retroperitoneal surgery. Duplication of the IVC and/or its association with supernumerary renal veins and anomalous tributaries complicate dissection and increase the risk of hemorrhage.4 The CT scan should be fully analyzed to effectively identify and dissociate the IVC during surgery. In transplant donors, this is sometimes difficult to accomplish before surgery. Surgeons must be familiar with the variations in the IVC to avoid damaging blood vessels or organs, especially when the duplicated IVC enters the left renal vein.3 (4) Radiologists’ experience is important because their reports have a significant effect on patient management. Radiologists must be familiar with variations of the IVC to facilitate timely identification, provide accurate reports, and guide diagnosis and treatment.
Conclusion
Duplication of the IVC can be diagnosed by CT and angiography. The imaging diagnosis is of great significance in identification of a duplicate IVC and selection of appropriate vascular interventions. Additionally, its course and relationship with the renal vein must be determined for effective planning of IVC filter placement in the setting of DVT and PE.
Footnotes
Authors’ contributions
WRL was involved in project development, data collection, and manuscript writing. HF and LJ were involved in project development, image collection, data analysis, and manuscript writing. XMC was involved in data collection and analysis. ZWZ was involved in project development and manuscript review and editing. The manuscript was approved by all authors for publication.
Declaration of conflicting interest
The authors declare no conflict of interest.
Ethics statement
This study was reviewed and approved by Institutional Ethical Review Board of Beijing Friendship Hospital (No. YYYXYJ-2021-361). Written informed consent was obtained from the patients for publication of this report and any accompanying images.
Funding information
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
