Abstract
Background:
The CLARINET-MS study assessed the long-term effectiveness of cladribine tablets by following patients with multiple sclerosis (MS) in Italy, using data from the Italian MS Registry.
Methods:
Real-world data (RWD) from Italian MS patients who participated in cladribine tablets randomised clinical trials (RCTs; CLARITY, CLARITY Extension, ONWARD or ORACLE-MS) across 17 MS centres were obtained from the Italian MS Registry. RWD were collected during a set observation period, spanning from the last dose of cladribine tablets during the RCT (defined as baseline) to the last visit date in the registry, treatment switch to other disease-modifying drugs, date of last Expanded Disability Status Scale recording or date of the last relapse (whichever occurred last). Time-to-event analysis was completed using the Kaplan–Meier (KM) method. Median duration and associated 95% confidence intervals (CI) were estimated from the model.
Results:
Time span under observation in the Italian MS Registry was 1–137 (median 80.3) months. In the total Italian patient population (n = 80), the KM estimates for the probability of being relapse-free at 12, 36 and 60 months after the last dose of cladribine tablets were 84.8%, 66.2% and 57.2%, respectively. The corresponding probability of being progression-free at 60 months after the last dose was 63.7%. The KM estimate for the probability of not initiating another disease-modifying treatment at 60 months after the last dose of cladribine tablets was 28.1%, and the median time-to-treatment change was 32.1 (95% CI 15.5–39.5) months.
Conclusion:
CLARINET-MS provides an indirect measure of the long-term effectiveness of cladribine tablets. Over half of MS patients analysed did not relapse or experience disability progression during 60 months of follow-up from the last dose, suggesting that cladribine tablets remain effective in years 3 and 4 after short courses at the beginning of years 1 and 2.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated disorder affecting the central nervous system. Cladribine tablets 10 mg [3.5 mg/kg cumulative dose over 2 years (CT3.5)] are indicated for the treatment of patients with MS in the European Union (highly active relapsing MS) and the United States (relapsing forms of MS, including relapsing-remitting disease and active secondary progressive disease, in adults), as well as other countries around the world.1,2 Unlike most other disease-modifying drugs (DMDs) for MS that require continual treatment, cladribine tablets are administered in short courses at the beginning of years 1 and 2. The effect of the drug appears to last for many months or years after the administration of the last dose in year 2. 3 Indeed, cladribine tablets are hypothesised to act as an immune reconstitution therapy, exhibiting prolonged treatment effects after a short treatment dose through transient reduction and recovery of lymphocytes. 4
The efficacy and safety of cladribine tablets were investigated in the CLARITY Phase III study of patients with relapsing-remitting MS (RRMS), 5 followed by the double-blind, randomised CLARITY Extension study. 3 The Phase III study ORACLE-MS involved patients with clinically isolated syndrome (CIS) who received cladribine tablets,6,7 and the ONWARD Phase II add-on study added cladribine tablets to interferon (IFN) β in patients with both RRMS and secondary progressive MS (SPMS). 8 At the time of CLARITY (conducted 2005–2008), the discontinuous short-course treatment strategy with cladribine tablets was a novel treatment approach, leading to uncertainty from physicians regarding treatment strategy after administration of cladribine tablets. An approach to addressing such uncertainty is the collection of real-world data (RWD) from large patient registries, which allow for the investigation of long-term safety and efficacy of treatment in chronic diseases like MS.9,10 Analysis of RWD alongside clinical data would therefore provide evidence for the sustained effectiveness of cladribine tablets; indeed, long-term safety data have already been described from the observational PREMIERE registry. 11
Italy is considered a high-risk area for MS with an incidence of approximately 3000 new cases in 2018 and an estimated prevalence of 193 cases per 100,000 (360 cases per 100,000 in Sardinia),12,13 and was also one of the largest recruiters of patients into the cladribine tablets clinical development programme. In 2014, the Italian MS Association (AISM) and its research foundation (FISM) promoted and supported, through a FISM-Bari University Research Agreement, the creation of the Italian MS Registry. The objective was to merge data from all the current Italian MS centres and, thereby, provide benefits to all Italian patients affected by MS. 14 Currently the Italian MS Registry merges data of approximately 55,000 Italian MS patients from regional registries and local databases, connecting 134 Italian MS centres in total. 15
The CLARINET-MS study aimed to assess the long-term effectiveness of cladribine tablets by following patients with MS in Italy enrolled in cladribine tablets randomised clinical trials (RCTs), using data available from the Italian MS Registry.
Methods
CLARINET-MS was a non-interventional, retrospective exploratory analysis of patients from the Italian MS Registry who had received at least one course of cladribine tablets in CLARITY, CLARITY Extension, ONWARD or ORACLE-MS. Anonymised patient data were extracted over a set observation period spanning from the last dose of cladribine tablets during the RCT (defined as baseline) to the last visit date in the registry, treatment switch to other DMDs, date of last recorded Expanded Disability Status Scale (EDSS) score, or date of the last relapse (whichever occurred last). Data were analysed to explore whether the cohort of data can provide evidence on long-term effectiveness of cladribine tablets.
Variables retrieved from the database included patient demographics, as well as clinical characteristics such as time span under observation, MS phenotype at last dose (CIS, RRMS and SPMS) and EDSS score recorded at or close to the end of treatment date. The study protocol for CLARINET-MS was approved by the Central Ethics Committee (CEC) of the University of Catania (N°: 126/2018/PO).
Study population
The study population consisted of Italian MS patients distributed across 17 Italian MS centres (participating centres are listed in the Supplementary Material). Data were obtained from the Italian MS Registry; data extraction and analyses were coordinated by the Azienda Ospedaliera Universitaria “Policlinico Vittorio Emanuele” of Catania, Italy, and the Italian MS Registry coordinator centre at the Azienda Ospedaliera – Policlinico Consorziale, Bari, Italy. Patient data were extracted by the Technical structure of the Mario Negri Institute (STO). Prior enrolment into CLARITY, CLARITY Extension, ONWARD or ORACLE-MS RCTs with at least one full course of treatment with cladribine tablets 3.5 mg/kg (approved dose) or 5.25 mg/kg (investigational dose only, not approved) in both year 1 and year 2 was required for inclusion in the CLARINET-MS analysis. Patient participation in the RCTs also must have ended, and patients were required to have given written informed consent to be included in the Italian MS Registry. There were no exclusion criteria for this non-interventional study.
Out of the 161 Italian patients enrolled in cladribine tablets RCTs, 88 patients from 17 MS centres were initially identified as meeting the inclusion criteria. The full analysis set (FAS) consisted of 80 patients, as eight patients subsequently did not qualify for the study: failure to meet the CLARINET-MS inclusion criteria (two patients), discrepancies in registration in the iMed Web database (three patients) and lack of follow-up after the last dose of cladribine tablets (three patients). The RCT that each patient was initially enrolled in was recorded (CLARITY, ONWARD or ORACLE-MS). CLARITY included patients enrolled in CLARITY Extension who received placebo in CLARITY and cladribine tablets 3.5 mg/kg in the extension study. For three patients, the enrolled RCT was not available in the database. Those patients were included in the analysis as the start and end dates of cladribine tablets intake were part of the minimum dataset collected in the iMedWeb database.
Assessments
For the observation period, time-to-first confirmed relapse was evaluated along with time-to-disability progression [defined as an increase from the baseline EDSS score of at least 1.0 point (or 0.5 points if the baseline EDSS score was ⩾5.5) that was sustained for at least 12 weeks]. Within disability progression, the time-to-first EDSS score ⩾ 6.0 (a surrogate marker for ambulatory support) 16 and time-to-first EDSS score ⩾ 7.0 (a surrogate marker for wheelchair use or being bedridden) 16 were also identified. The conversion of patients with CIS to clinically definite MS (CDMS) was analysed, with time to conversion and incidence risk investigated. Time-to-treatment change over the observation period was identified, including the number of DMDs received after the last dose of cladribine tablets and the proportion of patients free of treatment changes. Outcomes were analysed for the total patient population, as well as subgroup analysis by cladribine tablets RCT and MS phenotype.
Data analysis
The data are descriptive, with no formal comparison with other treatments. Only aggregated, anonymised data were obtained from the database. Quantitative variables were summarised as mean [standard deviation (SD)] and median [interquartile range (IQR)]. Categorical variables were summarised as a number (percentage). The analysis of all objectives was based on a time-to-event analysis based on the Kaplan–Meier (KM) method. Median duration and associated 95% confidence intervals (CI) were estimated from the model.
Results
The time span under observation in the registry ranged between 1 and 137 [median (interquartile range): 80.3 (60–87)] months. Over 75% of the study population had follow-up data greater than 60 months.
Among 80 patients evaluated, the mean age at baseline (start of observation period) was 39 ± 10.2 years, 57.5% were female and the majority of patients had an EDSS score between 1 and 5. There were 10 patients with CIS phenotype, 60 RRMS patients and 10 SPMS patients (Table 1).
Patient demographics.

MS phenotype of patients included in this analysis were determined at baseline, defined as the last dose of cladribine tablets during the preceding randomised clinical trials.
patients with ⩾1 EDSS score available.
CIS, clinically isolated syndrome; EDSS, Expanded Disability Status Scale; MS, multiple sclerosis; RRMS, relapsing-remitting MS; SD, standard deviation; SPMS, secondary progressive MS.
Confirmed relapses
In the overall patient population, the KM estimates for the probability of being relapse-free at 12, 36 and 60 months after the last dose of cladribine tablets were 84.8%, 66.2% and 57.2%, respectively. By study, at 36 months after the last dose, the KM estimates for the probability of being relapse-free were 66.2% for CLARITY, 57.1% for ONWARD and 70.8% for ORACLE-MS (Figure 1). At 60 months, the corresponding KM estimates were 58.9%, 57.1% and 54.2%, respectively. According to MS phenotype, the KM estimates for the probability of being relapse-free at 60 months after the last dose of cladribine tablets were 44.4% for CIS patients, 57.8% for RRMS patients and 68.6% for SPMS patients (Supplementary Figure 1).

Time-to-first confirmed relapse, by randomised clinical trials.
The mean annual relapse rate for the total patient population was 0.19 (95% CI 0.13–0.27); however, the median time-to-first confirmed relapse could not be estimated.
Disability progression
Of the 80 patients in the total patient population, 5 were excluded from the FAS due to lack of EDSS scores (one patient) or only one EDSS score available (four patients). At 12, 36 and 60 months after the last dose of cladribine tablets, KM estimates for the probability of being free of disability progression in the overall patient population were 97.3%, 73.3% and 63.7%, respectively. The median patient time-to-disability progression could not be estimated. The KM estimates for the probability of being free of disability progression at 36 months after the last dose were 63.7% for CLARITY patients, 67.1% for ONWARD patients and 84.7% for ORACLE-MS patients (Figure 2). At 60 months, the corresponding by-study KM estimates were 51.9%, 50.3% and 80.7%, respectively. By MS phenotype, the KM estimates for the probability of being free of disability progression at 60 months after the last dose were 100% for CIS patients, 65.6% for RRMS patients and 13.0% for SPMS patients (Supplementary Figure 2).

Time-to-disability progression, by randomised clinical trials.
EDSS score ⩾ 6.0
The number of patients eligible for analysis was 74 (one patient had a baseline EDSS of 6.0). In the overall patient population, the KM estimates for the probability of achieving EDSS score ⩾ 6.0 at 12, 36 and 60 months after the last dose of cladribine tablets were 0%, 8.7% and 16.4%, respectively. The median time-to-first EDSS score ⩾ 6.0 could not be estimated. Further analysis was carried out in terms of categorical EDSS score at baseline. The KM estimate for the probability of EDSS score ⩾ 6.0 for patients with baseline EDSS score of 0–2.5 (n = 49) was 1.3% at 60 months. For patients with baseline EDSS score of ⩾ 3.0 (n = 25) the KM estimate for the probability of EDSS score ⩾ 6.0 was 45.2% at 60 months. The KM estimates for the probability of EDSS score ⩾ 6.0 at 36 months after the last dose were 13.4% for CLARITY patients, 18.2% for ONWARD and 0% for ORACLE-MS (Figure 3). At 60 months after the last dose, the corresponding KM estimates were 24.1%, 36.4% and 0%, respectively. By MS phenotype, the KM estimates for the probability of EDSS score ⩾ 6.0 at 60 months after the last dose were 0% for CIS patients, 7.9% for RRMS patients and 87.0% for SPMS patients (Supplementary Figure 3).

Time-to-EDSS ⩾ 6.0, by randomised clinical trials.
EDSS score ⩾ 7.0
The number of patients in the total patient population eligible for analysis was 75. At 12, 36 and 60 months after last dose, the KM estimates for the probability of achieving EDSS score ⩾ 7.0 were 0%, 0% and 1.5%, respectively, in the overall patient population. Further analysis grouping patients by categorical EDSS score at baseline found that the KM estimate for the probability of EDSS score ⩾ 7.0 for patients with a baseline EDSS score of 0–2.5 (n = 49) was 0% at 60 months. The corresponding KM estimate for probability of EDSS score ⩾ 7.0 at 60 months for patients with baseline EDSS score ⩾ 3.0 (n = 26) was 4.3%. Subgroup analysis could not be performed due to the limited number of patients with EDSS ⩾ 7.0 in the study population over the observation period.
Conversion of CIS to CDMS
At the time of enrolment in the registry, there were 10 patients from the ORACLE-MS study with CIS who were therefore eligible for study of conversion to CDMS during follow-up. During the observation period, 60% of patients converted to CDMS; the incidence rate was 0.02/month, with a total time at risk of 292 months. The first conversion to CDMS occurred 1 month after the last dose of cladribine tablets, and the last conversion took place after 41 months.
Time-to-treatment change
During the observation period, 54 patients (67.5%) switched to another DMD (1–2 DMDs, n = 47; ⩾3 DMDs, n = 7). A wide variety of immunomodulatory and immunosuppressive therapies were utilised during treatment switching (Table 2), with IFN β-1a or 1b as the most frequent treatment used.
List of MS-related treatments after last cladribine tablets course.

IFN, interferon; MS, multiple sclerosis.
In the total patient population, the KM estimates for the probability of not initiating another treatment at 12, 36 and 60 months after the last dose of cladribine tablets were 62.2%, 46.7% and 28.1%, respectively (Figure 4). The median time-to-treatment change from the last dose was 32.1 (95% CI 15.5–39.5) months.
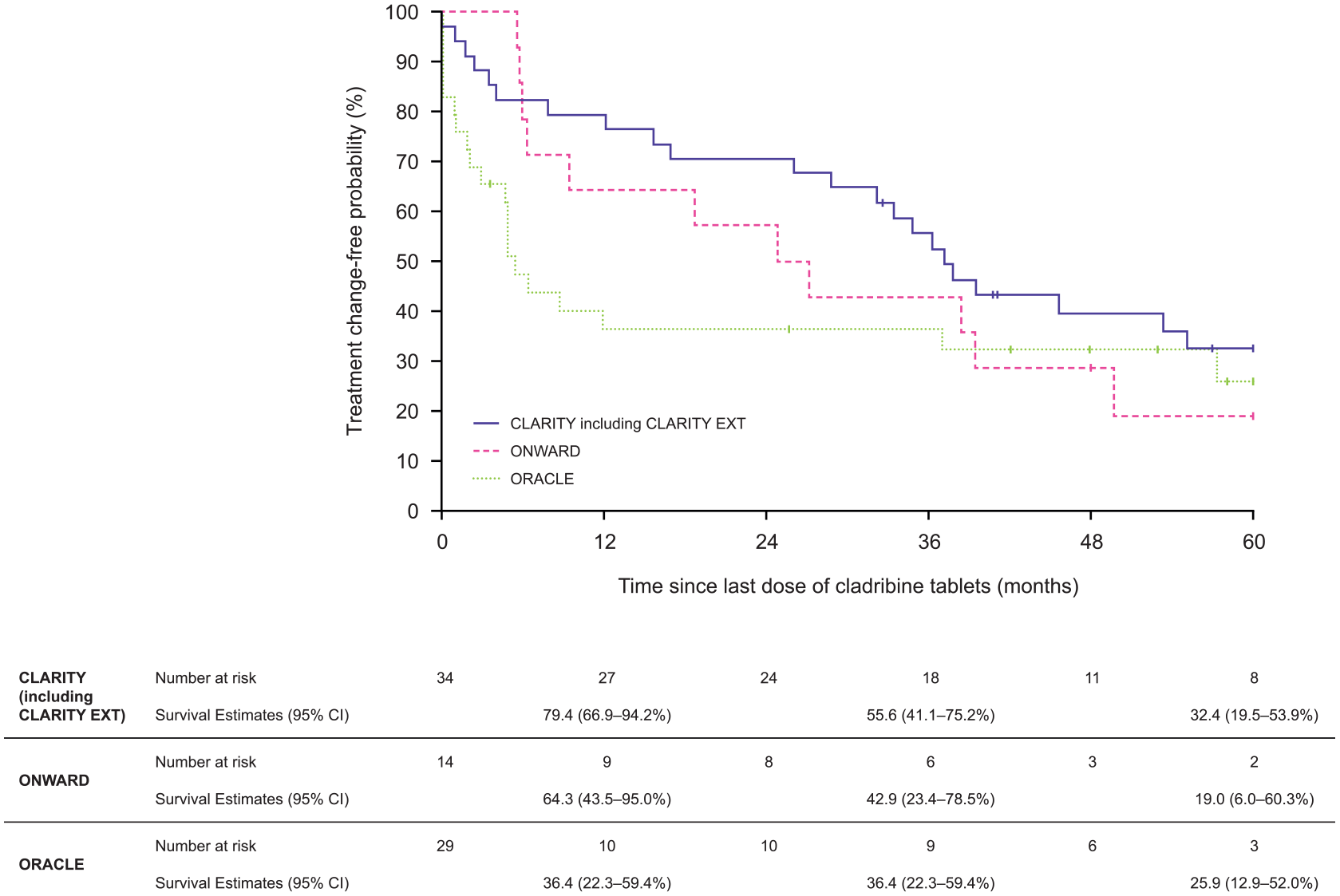
Time-to-treatment change, by randomised clinical trials.
For CLARITY, at 12, 36 and 60 months after last dose the KM estimates were 79.4%, 55.6% and 32.4%, respectively. At 36 and 60 months after last dose the KM estimates were 42.9% and 19.0%, respectively, for ONWARD patients. For ORACLE-MS patients, 36 and 60 months after last dose the KM estimates were 36.4% and 25.9%, respectively. By study, the median time-to-treatment change from last dose in CLARITY patients was 37.1 [95% CI 32.1–not estimable (NE)] months, 26.0 (95% CI 9.3–NE) months for ONWARD and 5.4 (95% CI 4.6–NE) for ORACLE-MS.
At 60 months, according to MS phenotype, the probability of not initiating another treat-ment was 15.0% for CIS patients, 30.1% for RRMS patients and 29.6% for SPMS patients (Supplementary Figure 4).
Discussion
CLARINET-MS aimed to explore whether RWD available from the Italian MS Registry could provide evidence for the sustained effectiveness of cladribine tablets. Overall, study results confirmed the effectiveness of cladribine tablets demonstrated in previous clinical studies, but over a longer observable period and in a real-world setting. Indeed, over 50% of analysed patients in CLARINET-MS were free from relapse or disability progression at 60 months from the last dose of cladribine tablets, found both in the overall population and by RCT type. More favourable trends were observed in ORACLE-MS patients, possibly due to diagnostic criteria at the time of study entry including patients with a mild phenotype in terms of inflammatory activity and disability. Of note, the risk of conversion from CIS to CDMS in ORACLE-MS was reduced by 67% over 2 years versus placebo;6,7 the findings from CLARINET-MS extend this window to a reduced risk of conversion to CDMS over 5 years. Of the 10 patients who were positively identified as CIS, for example, 40% had not converted to CDMS at 5 years after the last dose of cladribine tablets, with the last conversion at 41 months after the last dose.
Analysis of Italian MS Registry data ran from the last dose of cladribine tablets over the established observation period in the registry. Exploring this period of time allowed for the long-term effectiveness of cladribine tablets to be determined, assessing how long before relapse or disease progression occurred and further treatment was required. This study showed that many patients remained free of clinical activity for many years after the last dose. As the last dose of cladribine tablets was the starting point of the observation period, patients had a different last dose date and as such the observation period was not uniform. However, the related impact on the study objectives was minimal, as the majority of patients had an average observation of 60 months from the last dose of cladribine tablets. The date for EDSS score at baseline also varied among patients, since EDSS score date captured did not exactly match with the last dose date captured in the registry.
Most DMDs currently in use require continuous and frequent dosing, whereas cladribine tablets require only two short periods of dosing over 2 years; a full course of cladribine tablets is defined as receiving treatment in both year 1 and year 2. When the cladribine RCTs were conducted, the long-term effect of cladribine was unknown and the delay of the marketing authorisation application (2011–2014) meant that further treatment with cladribine tablets was not an option for physicians who may have felt that patients required an additional treatment course. We therefore evaluated treatment switching as part of the analysis. The median time to treatment switching of 32.1 months suggests that cladribine tablets have long-term efficacy of at least 3 years after the last dose. The reasons for treatment switching are not recorded in the registry, but do not appear to be necessarily related to relapse or disability progression. This is reflected in the results, where there is an apparent disparity between the number of patients who had progressed and the number of patients who switched during the time span under observation. In the total patient population at 60 months after the last dose, for example, the KM estimate for the probability of a patient experiencing disability progression was 63.7% yet the corresponding estimate for the probability of receiving a treatment change was 28.1%. CLARITY patients with more than one relapse could be offered an alternative DMD, and those in ORACLE-MS who converted to CDMS had a protocol-mandated switch to subcutaneous (sc) IFN β. Furthermore, all ONWARD patients received sc IFN β as a combination therapy. Patients receiving cladribine tablets 5.25 mg/kg were switched to sc IFN β only following a protocol amendment addressing an unexpectedly high level of patients (88.2%) experiencing grade 3/4 lymphopenia during the study. 8
Treatment switching may have occurred due to a lack of information available to physicians regarding the mechanism of action/long-term effect of cladribine tablets and/or physician unfamiliarity with a discontinuous short course treatment strategy (whereby patients receive the first course of cladribine tablets in year 1 followed by a long period before receiving the second course in year 2). For example, the mechanism of action of cladribine tablets was initially thought to be related to the reduction in lymphocyte counts. 17 Patients may therefore have been switched from cladribine tablets due to physician concerns that the return of total lymphocyte counts to normal range would lead to treatment reactivation. Indeed, in a recent analysis of patients who received one course of cladribine in the International MSBase registry, almost three quarters of patients who switched from cladribine tablets to another DMD did so before a relapse. 18 However, from CLARITY Extension it is now known that the clinical efficacy of cladribine tablets lasts beyond the initial reduction and recovery of total lymphocyte counts to the normal range. 3 Despite the lack of guidance on situations when patients might need additional DMD treatment following cladribine tablets, it is notable that over 30% of patients did not receive another DMD after receiving cladribine tablets in the clinical trials.
As CLARITY Extension was a randomised study, cladribine tablets were not given on the basis of clinical need. On average, the effect of treatment on clinical outcomes was similar in patients randomised to placebo or cladribine tablets over the duration of CLARITY Extension study. 3 CLARITY Extension was not designed to test whether cladribine tablets were an appropriate treatment option for patients experiencing disease activity after the first two courses.
The number of MS patients followed by the Italian MS centres and the availability of their complete clinical dataset in the iMedWeb database were the main reasons supporting a study designed on the Italian MS Registry. For other centres that may be considering analysing their data, the results from CLARINET-MS highlight the usefulness of this type of study for determining long-term effectiveness. RWD from 80 Italian MS patients enrolled by 17 Italian centres in the cladribine tablets pivotal clinical trials were analysed. We needed then to rely on what was present in the Italian MS Registry database at the time of data extraction, conscious that not all the sites involved in the pivotal trials of cladribine tablets were participating in the Italian MS Registry at that time. All patients that were enrolled in the studies and therefore available in the Registry were included in the CLARINET-MS study.
In terms of limitations, data obtained from the Italian MS Registry did not allow for safety outcomes to be reported. However, safety outcomes for cladribine tablets have previously been reported from an integrated analysis of data from the clinical trial programme and the completed PREMIERE registry. 11 Patients in the different cladribine tablets RCTs may have been randomised to a cumulative dose of 5.25 mg/kg instead of the now recommended 3.5 mg/kg dose. The difference in long-term efficacy in patients receiving higher cumulative doses of cladribine tablets compared with the recommended 3.5 mg/kg dose has not been investigated, as there were an insufficient number of patients to determine this in the current study. Finally, only 10 patients were actively identified as having CIS. Although the low patient numbers limit the analysis of conversion from CIS to CDMS, it is notable that 40% of these patients remained free of any event that would define MS.
Conclusion
The results from CLARINET-MS provide an indirect measure of the long-term effectiveness of cladribine tablets. Over half of patients analysed did not relapse or experience disability progression, demonstrating that further treatment was not required for most patients to prevent disease progression. While some patients switched to another treatment, this was not necessarily related to relapse or EDSS progression. CLARINET-MS therefore suggests that cladribine tablets remain effective in years 3 and 4 after short oral courses at the beginning of years 1 and 2, and in some patients with RRMS and relapsing SPMS up to 60 months after the last dose.
Supplemental Material
Supplementary_Material – Supplemental material for Long-term effectiveness in patients previously treated with cladribine tablets: a real-world analysis of the Italian multiple sclerosis registry (CLARINET-MS)
Supplemental material, Supplementary_Material for Long-term effectiveness in patients previously treated with cladribine tablets: a real-world analysis of the Italian multiple sclerosis registry (CLARINET-MS) by Francesco Patti, Andrea Visconti, Antonio Capacchione, Sanjeev Roy and Maria Trojano in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors would like to thank Dominic Jack of Global Medical Affairs, Merck KGaA, for assisting with the interpretation of the study data. The authors would also like to thank patients and their families, the Italian MS Registry, Mosconi and Lepore of STO, investigators, co-investigators and the study teams at each of the 17 participating Italian MS centres and at Merck KGaA, Darmstadt, Germany. Medical writing assistance was provided by Joseph Ward of in Science Communications, Springer Healthcare Ltd, UK, and was funded by Merck KGaA, Darmstadt, Germany.
Conflict of interest
FP has served on scientific Advisory Boards for Almirall, Bayer, Biogen, Celgene, Merck, Novartis, Roche, Sanofi and TEVA; he also received speaker honoraria from the same companies and research grants for his department by Biogen and Merck.
AV and AC are employees of Merck Serono S.p.A., Rome, Italy, an affiliate of Merck KGaA, Darmstadt, Germany.
SR is an employee of Merck, Aubonne, Switzerland, a division of Merck KGaA, Darmstadt, Germany.
MT has served on scientific Advisory Boards for Biogen, Novartis, Roche, Merck and Genzyme; has received speaker honoraria from Biogen Idec, Merck, Roche, Teva, Sanofi-Genzyme and Novartis; and has received research grants for her Institution from Biogen Idec, Merck, Roche and Novartis.
Funding
This study was sponsored by Merck Serono S.p.A. Italy, an affiliate of Merck KGaA, Darmstadt, Germany.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
