Abstract
Background:
Alemtuzumab (ALEM) is a humanised monoclonal antibody that depletes circulating lymphocytes by selectively targeting CD52, which is expressed in high levels on T- and B-lymphocytes. This depletion is followed by lymphocyte repopulation and a cytokine expression shift towards a lesser inflammatory profile, both of which may contribute to prolonged efficacy. National recommendations for enrolling and treating multiple sclerosis (MS) patients with ALEM have been established. However, there are no recommendations in place for the treatment of MS reactivation after the ALEM treatment.
Objectives:
To evaluate the effectiveness and safety of the use of ALEM and to analyse subsequent disease-modifying treatments (DMTs). A multidimensional prediction model was developed to make a patient-specific prognosis regarding the response to ALEM.
Design:
A multicentre, prospective, non-controlled, non-interventional, observational cohort study.
Methods:
Relapsing multiple sclerosis patients (RMSp) who received ⩾1 dose of ALEM were enrolled. In each treatment year, the following baseline and prospective data were collected: age, MS history, number, type and duration of previous disease-modifying treatment (PDMT), relapse rate (REL), expanded disability status scale (EDSS), magnetic resonance imaging and serious adverse events (AE). In cases of reactivation of MS, all data about the subsequent DMT were collected.
Results:
A total of 142 RMSp from 10 MS Slovak Centres fulfilled the inclusion criteria. The average age was 35 years (standard error 8.56). The overall average EDSS was 3.87 (1.46) when ALEM was started. The average duration of PDMT was 6.0 (4.04) years, and the median number of PDMTs was 3 (0–5), while the patients were mostly treated with 2 or 3 DMTs (>65.00%). Post-ALEM treatment was needed in 39 cases (27.46%). The most frequent post-ALEM treatment indicated was ocrelizumab, followed by natalizumab (NAT), siponimod and cladribine. The ocrelizumab and NAT treatment bring little benefit to patients. Siponimod showed less EDSS increase in contrast to ocrelizumab and NAT. Another repopulation therapy, cladribine, may also be an effective option. Statistically significant predictors for the expected EDSS are age (p-value <0.0001), number of ALEM cycles (0.0066), high number of PDMT (0.0459) and the occurrence of relapses (<0.0001). There was no statistically significant effect on the patient’s gender (0.6038), duration of disease-modifying treatment before alemtuzumab (0.4466), or the occurrence of AE (0.6668).
Conclusion:
The study confirms the positive effect of ALEM on clinical and radiological outcomes. We need more data from long-term sequencing studies.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic and progressive neurological disease of the central nervous system (CNS) characterised by heterogeneous clinical manifestations and disease course.1–3 Pathologically, MS is characterised by inflammation, demyelination and neurodegenerative phenomena that occur from the earliest phases of the disease.1–3 The treatment landscape for MS has substantially changed, with the approval of more than 10 new drugs in the last decade. Based on their efficacy, currently available disease-modifying treatments (DMTs) are commonly distinguished as moderate-efficacy DMTs (glatiramer acetate, interferon-beta, teriflunomide and dimethyl fumarate (DMF)) and high-efficacy (HE) DMTs (natalizumab (NAT), fingolimod, ozanimod, siponimod, alemtuzumab (ALEM), cladribine, ocrelizumab and ofatumumab).1,3–6
Two general treatment paradigms can be applied: either a maintenance-escalation approach, in which a medication is given continuously and patients are switched to a higher efficacy drug upon disease activity, or a pulsed immune reconstitution therapy, which involves few treatment pulses with long intermittent treatment-free phases.6,7
HE treatments appear to improve the long-term outcomes of MS patients. Recent growing pieces of evidence suggest that early initiation of HE-DMTs may have a beneficial long-term impact on disease progression in MS patients, thus underlining the need to offer early treatment with an HE-DMT to MS patients.1,3,6,7
ALEM (Lemtrada®; Sanofi Belgium, EU/1/13/869/001), one of the HE-DMT, is given as a pulsed immune reconstitution therapy. 8
ALEM is a humanised monoclonal antibody that depletes circulating lymphocytes by selectively targeting CD52, expressed at high levels on T- and B-lymphocytes. This depletion is followed by lymphocyte repopulation and a cytokine expression shift towards a lesser inflammatory profile, both of which may contribute to prolonged efficacy.9,10
In the European Union, ALEM has been approved for adults with active relapsing multiple sclerosis (RMS) by clinical or imaging features since 2013. In the United States, the drug has been approved for RMS and progressive–relapsing MS treatment, but only for patients who did not have a satisfying response to two or more other immunomodulatory treatments (i.e. for the third-line therapy). 11
In November 2019, after reports of rare but severe adverse reactions to ALEM, a risk-benefit analysis of its authorised indications was conducted in accordance with Article 20 of Regulation (EC) No. 726/2004 of the European Parliament and of the Council. As a result, ALEM is now only used to treat RMS if the disease is highly active despite treatment or if the disease is worsening rapidly but ALEM is no longer used in patients with certain heart, circulation, or bleeding disorders, or patients with an additional autoimmune disorder.12,13
Study objectives
The European Medicines Agency’s summary of product characteristics does not provide a strict definition of the ideal candidate or the level of clinical or radiological disease activity for indication of the drug, allowing clinicians some degree of flexibility. Moreover, criteria for high disease activity differ among countries.8,14,15 There are national recommendations for enrolling and treating MS patients with ALEM. However, there are no recommendations for treatment of MS reactivation after the ALEM treatment.
ALEM treatment. We also collected data about subsequent DMTs and their timing and compared their efficacy. A multidimensional prediction model was developed to make a patient-specific prognosis regarding the response to ALEM.
Methods
Study design
This is a nationwide, multicentre, prospective, non-controlled, non-interventional, observational cohort study conducted in Slovakia that is close to completion. The aim is to evaluate the long-term efficacy and safety of ALEM. All patients provided informed consent before the therapy, agreeing to the use of their data for research purposes by healthcare professionals.
Patients and setting
Two cycles of ALEM were administered 12 months apart; further cycles were offered if a relapse occurred. ALEM was given by intravenous infusion on five consecutive days at baseline and three consecutive days for subsequent cycles. The dose was 12 mg/day. Another DMT was indicated in the case of non-effectiveness of ALEM or re-activation of MS and was administered in different time periods. The inclusion criteria were in accordance with the Slovak national criteria for ALEM treatment: active relapsing MS with a history of treatment failure of three other DMT or in treatment-naïve patients with highly active relapsing MS (activity defined as ⩾2 relapses and ⩾1 new/enhancing magnetic resonance imaging (MRI) lesions per year; www.health.gov).
The patients were seen quarterly for 2 years following the administration of each cycle of ALEM, at a frequency of 6-month intervals over the next 2 years, at least annually thereafter, and within a week of reporting new symptoms. Patient data were reported longitudinally on a yearly basis for a minimum of 2 and a maximum of 8 years (between 2014 and 2022), with an average of 5.5 years.
Criteria for evaluation and variables selection
Relapsing multiple sclerosis patients (RMSp) who received ⩾1 dose of ALEM were enrolled. In each treatment year, the following baseline and prospective data were collected: number of relapses (relapse rate (REL)), expanded disability status scale (EDSS), MRI results and adverse events (AEs). We also collected the following data: age, MS history, number, type and duration (duration of disease-modifying treatment before alemtuzumab (DDMT)) of previous disease-modifying treatment (PDMT) and REL. In the case of reactivation of MS, we collected all data about the subsequent DMT.
A clinical relapse was defined as the occurrence of new symptoms or exacerbation of existing symptoms that lasted for 24 h or longer in the absence of concurrent illness or fever, and occurring 30 days or more as a previous relapse. The definition of relapse used in this study did not require confirmation by a change in the EDSS.
Disability was assessed annually using the EDSS by the same rater, who was neurostatus certified.
Sustained accumulation of clinical disability was defined as an increase in the EDSS (coded as ‘1’), sustained for at least 12 months of ⩾0.5 EDSS if the baseline EDSS was 1.0–6.0 and of ⩾1.0 step if the baseline EDSS was 0.
Unchanged or decreased EDSS was coded ‘0’. The presence of relapses (REL ⩾ 1) was coded ‘1’, and the absence of them ‘0’.
MRI was performed annually by a radiologist blinded to the patient’s EDSS score. Radiological disease activity was defined in the presence of ⩾2 new/enlarging T2-lesion or of any gadolinium-enhancing lesion on post-contrast T1-scans. If the MRI scan was regarded positive, it was coded as ‘1’. The opposite was coded as ‘0’. Finally, No Evidence of Disease Activity 3 (NEDA 3) was recorded in each treatment year; each was coded as ‘1’ if the patient fulfilled all three parameters measured (stabile EDSS, no relapses, no new/enlarging or Gd+ MRI lesions) and ‘0’ if the outcome was opposite.
Information on infections and symptoms of relevant organ-specific autoimmunity was assessed during each outpatient clinic review. In addition, the patients were encouraged to contact the neurologist from the Centre to report any new symptoms. They were counselled about urgent reporting of the signs and symptoms of serious AEs. Monthly tests for blood, liver and kidney functions were performed for a period of 4 years after each ALEM cycle. Thyroid-stimulating hormone was tested quarterly. Secondary autoimmunity was defined as a new symptomatic autoimmune disease diagnosed with the positivity of organ-specific autoantibodies and was further managed by a specialist.
Statistical methods
The statistical analysis of the data was performed in two steps. Firstly, exploratory characteristics were used to gain an insight into the underlying data structure, and second, a confirmatory part utilising a linear mixed effect model 16 was used to model the expected value of the EDSS value while controlling for significant time-dependent and subject’s specific covariates. A stepwise forward modelling procedure was used to obtain the final model (using the critical value of α = 0.05). The model contains a random intercept term to account for within patient’s and between patient’s variability sources. The statistical analysis was performed using the R software (Development Core, 2022). 17
Results
Pre-ALEM period, baseline characteristics
Altogether, there were 146 patients screened and treated with ALEM. However, four of them were excluded as they did not complete their treatment, or the data were not available. The final dataset used for the statistical analysis consisted of 142 patients (70.4% women and 29.6% men). Baseline summary statistics are reported in Table 1.
Baseline summary characteristics (sample means and the corresponding standard errors) for N = 142 eligible patients treated with ALEM.

ALEM, alemtuzumab; DDMT, duration of disease-modifying treatment before alemtuzumab; EDSS, expanded disability status scale; PDMT, previous disease-modifying treatments.
Response to ALEM treatment
The majority of the patients (116 out of 142; 81.69%) received 2 ALEM cycles (as recommended), 11 patients (7.74%) had only 1 cycle and 15 patients (10.56%) received 3 ALEM cycles. The reasons for reducing the ALEM treatment to just one cycle were AEs or noncompliance with the post-treatment monitoring. The indication for the third cycle was lower but sustained disease activity.
During the ALEM treatment within the whole follow-up period (from 1 to 8 years depending on the patient), some relapses were found in 56 out of 142 patients (39.44%), and new T2 or gadolinium-enhancing lesions in the MRI scans were found in 46.47% of all patients (66/142).
Considering the first year of the ALEM treatment, NEDA 3 was recorded for 64.08% of the patients. Relapses occurred for 39.44% of the patients, new lesions in the MRI scans for 25.35% and AEs for 35.92%. The EDSS value slightly increased when compared with the baseline (the overall average of 3.83 after the first year of treatment vs 3.79 at the baseline), but the unchanged EDSS was observed for more than 80% of the patients (see Table 2).
Summary statistics (sample means with the corresponding standard errors and scoring proportions) for the patient’s response after the first year of ALEM treatment.

ALEM, alemtuzumab; EDSS, expanded disability status scale; MRI, magnetic resonance imaging; NEDA 3, No Evidence of Disease Activity 3.
Two years after the first ALEM treatment, there were 140 patients (98 women and 42 men) being still followed. An unchanged EDSS value was observed for over 70% of the patients, and NEDA 3 was recorded for 67.86% of the patients (see Table 3 for more details).
Summary statistics (sample means with the corresponding standard errors and scoring proportions) for the patients’ response 2 years after the first ALEM treatment.

ALEM, alemtuzumab; EDSS, expanded disability status scale; MRI, magnetic resonance imaging; NEDA 3, No Evidence of Disease Activity 3.
Finally, 5 years after the first ALEM treatment (with 73 patients being still followed, 52 women and 21 men), the NEDA 3 score was assigned to 87.67% of the patients (88.46% of women and 81.71% of men). NEDA 3 rates assessed over the total period was 58.0%.
Regarding the EDSS, women had a slower progression of the disability than men (see Figure 1(a)). Older patients had an earlier increase in the neurological disability than younger ones (Figure 1(b)). A milder disease course was observed in patients who took one PDMT before starting ALEM than in those who took two or three PDMT (Figure 1(c)). Shorter disease duration before ALEM resulted in lower sustained EDSS (Figure 1(d)). Patients who received two or three ALEM treatment cycles had milder progression of disability than those treated with only one ALEM cycle (see Figure 1(e)).
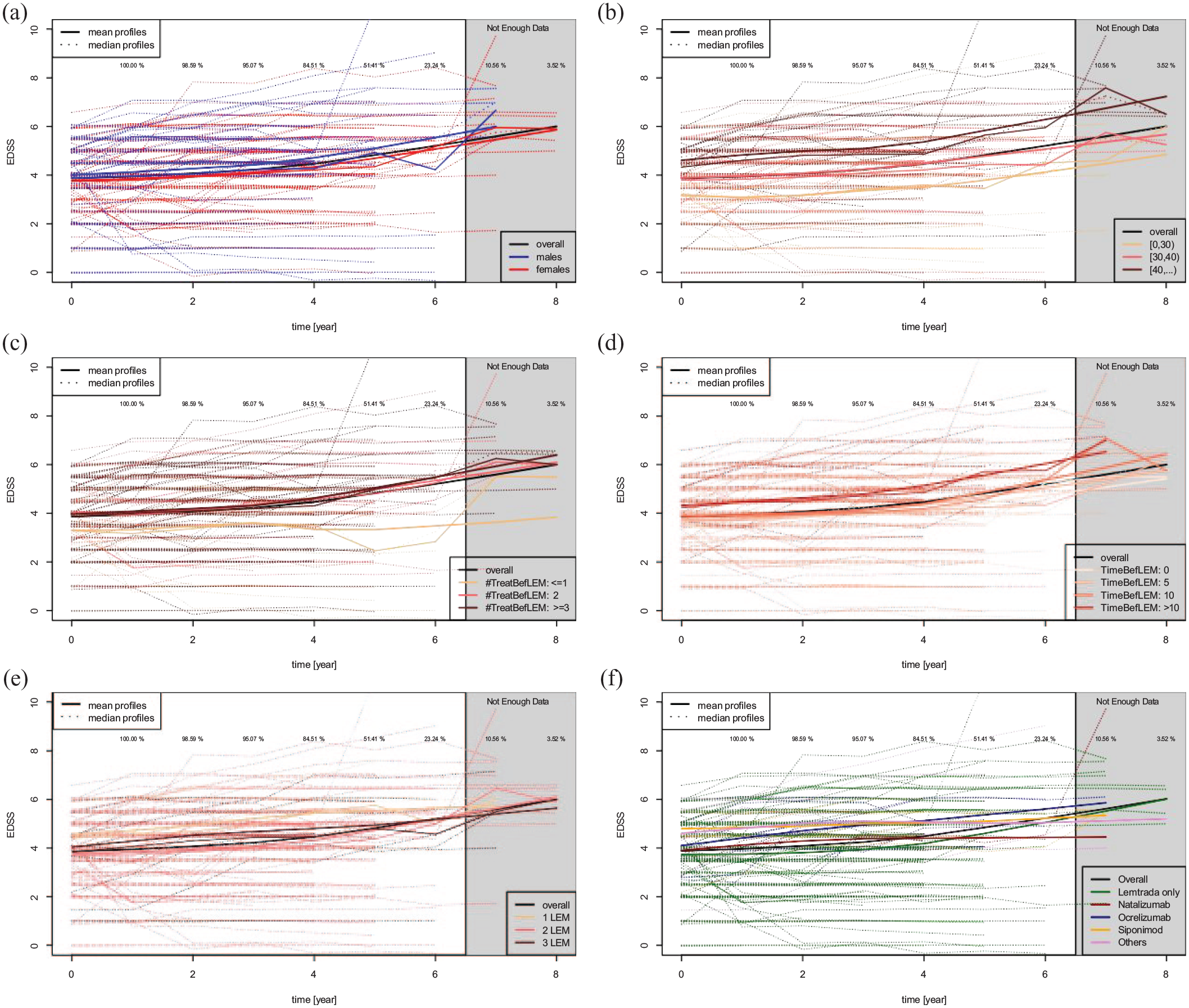
Subject-specific EDSS profiles (dotted lines) within the whole considered follow-up period with the overall average/median profiles (black solid lines) and group-specific profiles (coloured solid lines) while distinguishing for different patients characteristics in different subplots. (a) Women (red) versus men (blue). (b) Age in years: <30 (orange), [30, 40) (red), ⩾40 (brown). (c) PDMT: ⩽1 (orange), 2 (red), ⩾3 (brown). (d) PDMT duration: 0, 5, 10 and more than 10 years. (e) ALEM cycles: 1 (orange), 2 (red) and ⩾3 (brown). (f) Post-ALEM DMTs.
Post-ALEM treatment
Out of the 142 patients enrolled at the beginning, the post-ALEM treatment was needed for 39 patients (27.46%) and no further treatment was required in 103 cases (72.54%). The most frequent post-ALEM treatment indicated was ocrelizumab (12 cases), followed by siponimod (10), NAT (6), cladribine (4) and dimetyl fumarate (3). Other DMT variants (glatiramer acetate, interferon beta 1a s.c., fingolimod and autologous transplantation of haematopoietic stem cells) were only used in one case each. The shortest interval between the last ALEM infusion and the post-treatment was 12 months.
At the time of the treatment change, the patients who started with ocrelizumab or NAT had lower EDSS than patients receiving siponimod. However, compared with the siponimod-treated patients, the ocrelizumab-treated patients progressed faster and achieved higher disability scores during the observation period. The difference was not statistically significant. NAT was superior in efficacy, but in terms of safety, it has not been found to be a better option when compared to other DMTs (see Figure 1(f)).
Statistical model
Statistically significant effects that play an important role when estimating the subject-specific EDSS value are: age (p-value <0.0001), the indicator whether the patient was assigned to just one or more ALEM treatments (0.0066), PDMT (0.0459) and, finally, the occurrence of relapses (<0.0001). There was no statistically significant effect of the patient’s gender (0.6038), DDMT before ALEM (0.4466), the type of the post-ALEM treatment (0.0523) and the occurrence of AEs (0.6668).
The expected EDSS score for a patient with an average age of 35.4 years, after one ALEM treatment, and no relapses, is 3.95 (with the 95% confidence interval (3.02, 4.89)). For a patient with the same age, however, after two ALEM treatments, the expected EDSS score is 2.73 (2.16, 3.29). After the first year of the treatment, the expected EDSS score increases roughly by 0.04 (−0.01, 0.09), which is not a statistically significant difference. After 2 years, however, the expected EDSS increases (compared to the baseline) by 0.11 (0.02, 0.19), and the EDSS increase accelerates in time achieving an expected increase (with respect to the baseline EDSS) of 0.5 (0.37, 0.59) after 5 years after the first ALEM treatment (see Table 4 for more details).
Linear model (with a random intercept term) for the expected EDSS value given statistically significant (with α = 0.05) effects.
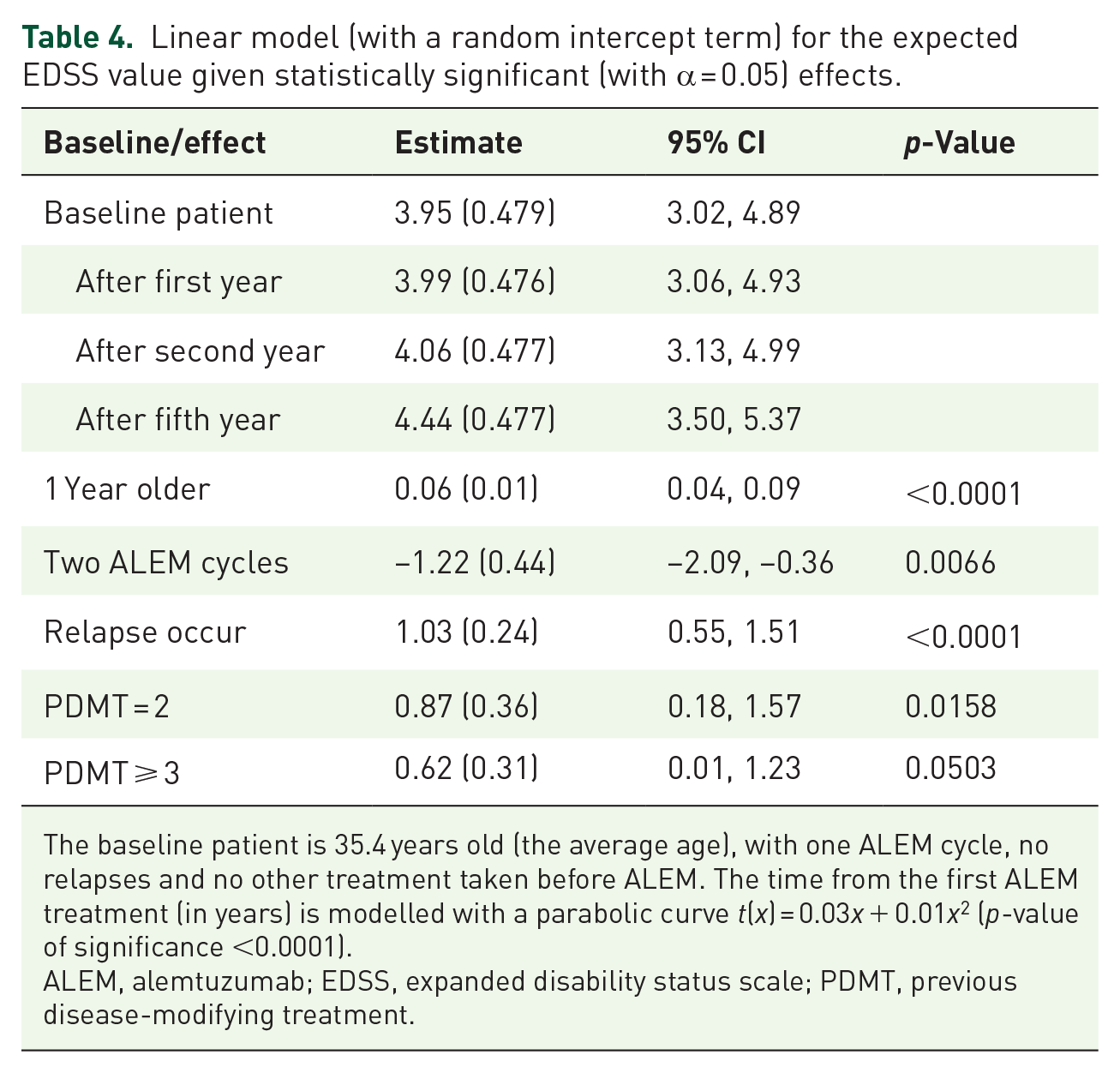
The baseline patient is 35.4 years old (the average age), with one ALEM cycle, no relapses and no other treatment taken before ALEM. The time from the first ALEM treatment (in years) is modelled with a parabolic curve t(x) = 0.03x + 0.01x2 (p-value of significance <0.0001).
ALEM, alemtuzumab; EDSS, expanded disability status scale; PDMT, previous disease-modifying treatment.
If the patient was already assigned to one other treatment before ALEM (i.e. PDMT = 2), then the expected EDSS value is roughly 4.82 (with the corresponding 95% confidence interval (3.91, 5.74); again for a patient with the average age of 35.4 years, with one ALEM treatment and no side effects). If the patient undertook three or more treatments before the ALEM treatment (i.e. PDMT = 3), then the model estimated expected EDSS is 4.57 (3.70, 5.44).
Finally, the age of the patient has a positive effect in a sense that two patients with the age difference of 10 years will have an estimated difference in their expected EDSS values equal to 1.26 (0.61, 1.91; with the older patient having higher EDSS).
There are some post-ALEM treatment effects observed in the data, but the differences were not found to be significant in the final model. However, there was a trend towards significance in ocrelizumab (p-value 0.0523).
Discussion
The presented data from the real clinical practice could help with making ALEM-treatment decisions. More importantly, the presented work provides information about sequencing post-ALEM treatment and offers a valid statistical prediction model. For the current analysis, the data from 142 patients from 10 study sites with the average observation period of 5.5 years (range 2–8) were analysed.
In our research, the observation period was longer than in other real-world studies,14,15,18,19 but the demographic data and basic variables studied are comparable.
The results demonstrate that the reduction of disease activity with ALEM continues over 5 years. During this period, NEDA 3 showed its persistency in 58.0%; 60.6% of RMSp were relapse-free, and most patients had stable or improved EDSS scores (the EDSS was maintained, in average, for 2.7 years), while 85.7% remained free of MRI activity.
We found a similar outcome of the long-term efficacy of ALEM when comparing results with registration studies. In the CARE-MS II study, the proportions of patients achieving NEDA 3 were 52.9%, 54.2% and 58.2% in years 3, 4 and 5, respectively. 20 In the 5-year follow-up of CARE-MS I, the percentages were 61.7%, 60.2% and 62.4% for years 3, 4 and 5, respectively. 21 The follow-up results of the safety profile were also similar to those of prior studies CARE-MS I and II, indicating consistency.
The long-term efficacy (6-year follow-up) of ALEM was also presented in patients fulfilling criteria for high disease activity (high relapse rate in 1–2 years prior to ALEM, high mean baseline EDSS score).22,23 The authors presented a substantially declined relapse rate and progression of disability. Moreover, 69% of patients in CARE-MS I and 68% in CARE-MS II remained free of MRI disease activity. 22 We did not evaluate highly active RMSp separately, nevertheless, 85.72% of RMSp remained free of MRI activity during a 5-year follow-up.
The Central European board of Multiple Sclerosis Experts expressed the opinion that there is no doubt about the usefulness of additional data constituting more rational and personalised treatment choices in MS patients. 24
This inspired us to investigate which patients respond well to ALEM. Our model identified the following predictors impacting a worse outcome when EDSS was endpoint: ⩾40 years of age, ⩾2 PDMT, only one cycle of ALEM. Gender, duration of PDMT, further DMT and occurrence of AEs did not show statistical significance.
Results of Tuohy et al. 25 identified similar predictors of disability outcomes. They concluded that, in patients with early active relapsing-remitting disease (median EDSS 3.5, median disease duration 3 years), two cycles of ALEM, with up to three further cycles due to a relapse, lead to a stabilisation of disability, in most patients treated over an average 7-year follow-up.
Results of another study by López-Real et al. presented data that was partly contradictory to ours. They found a lower probability of achieving NEDA 3 in younger patients and women. 15 This was not the case in our study. ALEM was highly effective between 19 and 40 years of age when EDSS was a primary endpoint. Other data of López-Real et al. were comparable to ours: a lower probability of achieving NEDA 3 in patients with a high annualised relapse rate and an elevated number of previous treatments. Another recent study also supports the efficacy of ALEM in the older rather than the younger population. They revealed an increased risk of relapse in paediatric – than in adult – onset MS, cut-off was the age of 22.75 years. However, survival analysis did not disclose any difference between the groups in terms of clinical disability, including progression independent of relapse activity. 26
Van Wijmeersch et al. 22 summarise, the two-course regiment maximised clinical and MRI benefits, as was also supported by our results.
The type and timing of escalation play an enormous role in clinical practice. Investigation of sequential drug use and long-term safety outcomes is of utmost importance. 24
Data documenting outcomes in patients who switched to other DMTs from ALEM are limited. Currently, there is no substantial evidence to guide the sequencing of HE DMTs in patients failing on second-line therapies. Data from randomised controlled and observational studies comparing the efficacy and safety of HE DMTs are lacking. There is no standard definition of treatment failure, and the lack of consensus on different outcomes that may predict the future course of disease adds to the complexity of decision-making for treatment sequencing.24,25
We aimed to identify the most effective further DMT to stabilise the disease. Out of the further DMTs used (ocrelizumab, NAT, cladribine, siponimod, DMF, glatiramer acetate, interferon beta 1a, fingolimod), and autologous transplantation of bone marrow, we only found a trend towards significance in ocrelizumab.
ALEM is a HE treatment with a long-term effect but many potential side effects, which makes treatment decisions difficult as it is not easy to make a linear switch and choose the appropriate treatment.20,28
The available information on the effectivity of ocrelizumab after ALEM is not sufficient yet. Vališ et al. 29 presented one isolated case of successful ocrelizumab (OCR) treatment. They pinpointed the importance of keeping in mind the mechanism of action of both DMTs to avoid overlapping effects on the immune system. In case of failure of ALEM, they recommended testing B-lymphocytes and considering an early switch to OCR. 29 Adamec and Habek 30 also confirmed the effectivity of OCR after ALEM. Lapucci et al. 31 presented good efficacy and safety of OCR treatment of MS patients who switched to OCR due to the persistence of disease activity after two courses of ALEM. Studies with long-term evaluation of switching ALEM to OCR are rare.
In our study, OCR was the most frequent treatment choice after ALEM, indicated in 12 post-ALEM patients. OCR treatment started on average 29 months after ALEM. Reactivation of disease activity would have been the factor that favours OCR. However, the ocrelizumab-treated patients progressed early and achieved the highest EDSS score of all patients tested. We did not prove its effectivity although we hypothesised early repopulation of B-lymphocytes, as was found typical in patients after ALEM with more aggressive disease.29–31
Siponimod, the second most frequently used DMT, was indicated in 10 patients that converted into secondary progression. Compared to ocrelizumab- and natalizumab-treated RMSp, the patients receiving siponimod had higher EDSS, which remained stable over the observation period. To our knowledge, no other data have been published about this type of treatment sequencing. Insight into conversion to secondary progressive phase can be gained from CARE I and II, where ALEM was also tested in MS patients with higher EDSS than 5.0,20,21 where active secondary progression needs to be considered. Siponimod, aimed at secondary progressive MS, is an obvious choice. In our study, there was a longer period (36 months) between ALEM and siponimod compared to ALEM and OCR, and we can confirm its efficacy.
Horáková et al. 32 followed on CARE-MS I, CARE-MS II and CAMMS03409 and evaluated conversion to secondary progressive MS after ALEM. They used Lorscheider’s definition (EDSS score 4, pyramidal score 2 and disability progression by one EDSS point if EDSS score = 5.5 or 0.5 EDSS points if EDSS score 6.0, without relapse, with confirmed progression over 3 months including confirmation within the functional system leading to the progression event). 33 Using the definition, Horáková et al. 32 found approximately 18% of the patients converted to secondary progressive MS over a median of 5.8 years. However, they did not evaluate further DMT.
Natalizumab (NAT), indicated in 6 of our patients, was initiated after a median of 16 months, in patients that only partially responded to ALEM. NAT is a very effective drug used to treat patients with an aggressive disease. The positive impact of the drug is apparent on many parameters: annualised relapse rate, EDSS, number of new lesions or enhancing lesions and quality of life. 34 Its limitations are the risk of progressive multifocal encephalopathy (PML). One of our patients later experienced PML after switching to NAT from ALEM and unfortunately died. Here, the risk of PML might have been potentiated by ALEM treatment. Another death was from multiple myeloma. Following ALEM treatment, B cells repopulate in the relative absence of CD4 T regulatory and CD8 T suppressor cells. T cells control the affinity maturation of antibodies within lymphoid tissue. 35 A low CD4/CD8 was independently associated with increased cancer risk in patients with human immunodeficiency virus. Immunodeficiency was also associated with a high prevalence of oncogenic virus coinfection. 36 However, since NAT reduces lymphocyte extravasation not only to the CNS, immune surveillance and responses in peripheral tissues may be insufficient.36,37 We hypothesise that the complex pathological mechanisms could explain the harmful cumulative effect of ALEM and NAT on the immune system in these cases.
The Central European Board of Multiple Sclerosis Experts agreed and advocated that early treatment and escalation therapy are necessary for patients with active disease as timely initiation of treatment or a switch to higher-efficacy agents are likely to improve long-term clinical outcomes. 24
However, certain drugs might limit the use of subsequent agents by facilitating AEs in the long term. 37 It may be a case of NAT. NAT is the DMT with a different mechanism of action, targeted mostly at autoaggressive T-lymphocytes. 38 Following ALEM treatment, B cells repopulate. 39 However, before starting NAT, the cellular immunity profile was not taken into consideration about further treatment. NAT was chosen empirically.
In our study, cladribine was used in four patients. So far, there is only one other report of six patients with MS who experienced disease activity after ALEM and were subsequently treated with cladribine. The patients were followed over 2 years. The authors reported expected efficacy and tolerable safety profile. The patients were relapse-free, and their EDSS remained stable during the follow-up period. 40 We found similar results. The very low number of cladribine-treated patients and the short observation time make it difficult to assess the efficacy of cladribine sufficiently. A prolonged observation period is necessary to assess cladribine’s long-term risks and benefits. Cladribine, as another repopulation therapy, may be of value due to its short-term use but the long-term effect on the immune system, and low AE score. A recent population-based Danish study evaluating the treatment outcome of cladribine for up to 2 years showed a high risk of experiencing relapses and disability accumulation when MS patients were switching from highly active DMT, for example, ALEM or NAT. 41 However, they did not evaluate ALEM-treated patients separately. The authors suggested that these patients belonged to a group of highly active patients, and many of them had been effectively controlled on ALEM or NAT for some time. 41 Further studies are needed to confirm the effectiveness of cladribine treatment after ALEM.
In our patients, the switch to dimetyl fumarate (DMF) was mostly due to disease reactivation and/or significant adverse effects of ALEM. DMF was indicated in three patients. One of them was a woman, diagnosed with cystic echinococcosis several weeks after finishing ALEM. The patient is now stabilised, although still at risk of progression of her disease. 42
Other treatments (glatiramer acetate, interferon beta Ia, fingolimod) were used each in one patient, so it was not possible to make a definitive assessment. Indicating Autologous Hematopoietic Stem Cell Transplantation (aHSCT) after ALEM is challenging, raising concerns about patients’ safety. Boffa et al. 43 reported short-term safety and efficacy data from three patients treated with aHSCT following ALEM treatment. They found that early AEs were consistent with expected transplant toxicities. All patients were reported to be free of disease activity. The authors suggested that aHSCT can be considered as a rescue treatment strategy for MS patients with persistent disease activity despite ALEM treatment. 43
Limitations of the study
Our main limitation is that a power analysis for sample size calculation was not done before starting the study. Moreover, our periods of observation were not the same length for all individuals, so the number of studied individuals was lower in the fifth compared to the first year. We used a 12-month period, not a 6-month period, to confirm disability progression, which may have been argued as non-standard. However, we considered it a more accurate method because of the long-term follow-up.
The limitations of this multicentre study are the absence of contemporaneous controls and the variability in patient follow-up times. However, a minimum of patients was lost to follow-up, and inter-rater variability in EDSS score assignment was minimised by having one rater perform all EDSS assessments over 5 years of follow-up. This ‘real-world’ experience provides additional information to pivotal trials. In addition, in this study, we addressed the issue of treatment sequencing, which is not a routine outcome. Indeed, we are aware of the low number of patients switched to other DMTs which makes the statistical analysis of treatment sequencing impossible.
Conclusion
ALEM provides a durable response in a high number of patients, who do not require further medication for several years. Clinicians should be careful and follow the patients, closely monitoring the disease progression. Our data suggest that the best predictors for ALEM are younger age, short disease duration and low number of PDMT.
In patients with reactivation of the disease, other DMT should be considered. The treatment sequencing after ALEM can vary, depending largely on the expected patient’s response to DMT, disease activity, rate of neurodegeneration and comorbidities. Our results show OCR and NAT to be the most frequent post-ALEM DMTs. The ocrelizumab and NAT treatment bring little benefit to patients. Siponimod showed less EDSS increase in contrast to ocrelizumab and NAT. Another repopulation therapy, cladribine, may also be an effective option due to its short-term use but the long-term effect on the immune system and low AE score. We need more data from long-term sequencing studies. Sharing experiences with post-ALEM treatment will help develop guidelines and help improve the prognosis of patients.
