Abstract
Background:
Although the emergence of multiple therapeutic agents has benefited patients with human epidermal growth factor receptor 2 (HER2)-positive advanced gastric cancer (AGC), chronological improvements in treatment outcomes for real-world patients remain unclear.
Objectives:
The present study aimed to evaluate the treatment features of various agents according to their approval periods and investigate outcomes in daily practice.
Design:
Retrospective observational study.
Methods:
We retrospectively evaluated the clinical outcomes of patients with HER2-positive AGC who received first-line fluoropyrimidine-containing chemotherapy between 2011 and 2023 according to the approval period of each agent in Japan: group A (pre-immunotherapy approval), 2011–2016; group B (nivolumab approval for third-line or later treatment), 2017–2019; and group C (trastuzumab deruxtecan approval for third-line treatment), 2020–2023.
Results:
In total, 309 patients were enrolled (n = 159, 69, and 81 in groups A, B, and C, respectively). Significant differences were observed among the three groups in terms of the proportion of patients with Eastern Cooperative Oncology Group performance status of 0 or ⩾1 (p = 0.02), HER2 status of 3+ or 2+/FISH+ (p = 0.03), prior gastrectomy (p = 0.008), diffuse-type histology (p = 0.016), peritoneal metastases (p < 0.0001), and lymph node metastases (p = 0.007). The median overall survival (OS) rates were 19.5, 20.6, and 23.5 months in groups A, B, and C, respectively, with no significant difference among the groups. Progression-free survival in the first-line setting did not significantly differ among the three groups. Multivariate analyses revealed primary tumor location, prior gastrectomy, peritoneal metastases, and liver metastases as independent prognostic factors for OS. Inverse probability of treatment weighting analysis showed no significant difference in OS between groups A and B (hazard ratio (HR), 0.86; 95% confidence interval (CI): 0.70–1.05) but superior OS in group C over group A (HR, 0.54; 95% CI: 0.43–0.68) and group B (HR, 0.72; 95% CI: 0.55–0.95). Moreover, 53.3%, 61.4%, and 66.1% of patients in groups A, B, and C, respectively, received third-line treatment. The most commonly administered third-line treatments were irinotecan (63%) in group A, immunotherapy (43%) in group B, and trastuzumab deruxtecan (70%) in group C. The proportion of patients receiving trastuzumab deruxtecan at any line gradually increased across the three groups (7.5%, 30.4%, 44.4%; p < 0.0001).
Conclusion:
The emergence of novel agents and treatment modalities may have contributed to improvements in the survival of patients with HER2-positive AGC. This highlights the benefits of effective treatment strategies, including efforts to identify biomarkers and develop new agents.
Keywords
Introduction
Although the ToGA trial established trastuzumab plus chemotherapy as the standard first-line treatment for human epidermal growth factor receptor 2 (HER2)-positive advanced gastric cancer (AGC), 1 improving upon this benchmark has proven difficult. Unfortunately, the prognosis for patients with HER2-positive AGC remains limited due to various factors, including acquired resistance. 2 Moreover, the LOGiC and GATSBY trials, which evaluated other HER2-targeted agents—lapatinib and trastuzumab emtansine, respectively—as second-line treatments, failed to improve overall survival (OS).3,4 Similarly, the T-ACT study showed no benefit in continuing trastuzumab beyond progression. 5 Therefore, developing more effective treatment strategies for this distinct cancer subtype is an urgent clinical need. 6
Nonetheless, the stagnation in therapeutic advancements was disrupted by the emergence of two classes of drugs that reshaped later-line strategies for AGC. The advent of nivolumab offered a viable third-line treatment.7,8 A subgroup analysis in the ATTRACTION-2 trial indicated that nivolumab was effective, regardless of prior trastuzumab use. This suggests a potential role for the immune checkpoint inhibitor in the HER2-positive patients.7,8 More striking results were obtained for trastuzumab deruxtecan in the DESTINY-Gastric01 trial, which showed significantly improved objective response rates (51% vs 14%) and median OS (12.5 vs 8.4 months) compared with standard chemotherapy. 9 Although some real-world data suggest improvement in outcomes over the last decade, 10 the specific contribution of these sequentially approved agents to this trend has not been formally evaluated. Hence, a critical gap in understanding exists in the chronological evolution of treatment patterns and survival outcomes with progressive changes in the standard of care.
With the rapid evolution of the therapeutic landscape and shifting of treatment options toward earlier lines of therapy, understanding the foundational impact of each major agent becomes increasingly critical for optimizing patient care. This complexity highlights the need for real-world evidence to guide clinical decision-making. Therefore, the present study sought to provide the necessary evidence by evaluating the chronological improvements in survival among patients with HER2-positive AGC who received treatment at our institution from 2011 to 2023. Moreover, to analyze the progression of treatment patterns and outcomes, the study period was divided into three eras based on the successive approval of nivolumab and trastuzumab deruxtecan.
Materials and methods
Patients
This retrospective study was conducted at the Cancer Institute Hospital of the Japanese Foundation for Cancer Research (JFCR), Tokyo, Japan. Patients included in this study were selected based on the following inclusion criteria: (1) unresectable or metastatic gastric or gastroesophageal junction cancer, (2) histologically or cytologically confirmed HER2-positive adenocarcinoma, and (3) receiving palliative fluoropyrimidine-based chemotherapy as first-line treatment between January 2011 and December 2023. The exclusion criteria were as follows: (1) initiation of palliative chemotherapy at another hospital, (2) adjuvant chemotherapy after R0 metastasectomy, and (3) presence of other advanced cancers. For analysis, the calendar period was divided into three categories (group A: 2011–2016; group B, 2017–2019; and group C, 2020–2023) based on the year in which nivolumab (September 2017) and trastuzumab deruxtecan (November 2020) had been approved for insurance coverage in Japan.
Data collection
Each patient’s baseline demographic and clinicopathological variables, including age, sex, Eastern Cooperative Oncology Group (ECOG) performance status (PS), HER2 status, primary tumor location, history of gastrectomy, disease status, histological type, metastatic sites, and details of chemotherapy for systemic disease, were collected from the institution’s electronic medical records.
Statistical analyses
OS was defined as the duration from first-line chemotherapy initiation to death from any cause. Progression-free survival (PFS) was defined as the duration from first-line chemotherapy initiation to disease progression or death from any cause. Time to treatment failure (TTF) was defined as the duration from chemotherapy initiation in each treatment line to disease progression, treatment discontinuation, or death from any cause. Post-progression survival (PPS) was defined as the duration for which patients survived following progressive disease during first-line treatment. OS, PFS, TTF, and PPS were calculated using the Kaplan–Meier method. Univariate and multivariate analyses were performed using the Cox proportional regression model to identify factors associated with OS and PFS. The proportional hazards assumption was assessed and confirmed prior to interpretation of the model results. For sensitivity analysis, the propensity score was calculated using prognostic factors for OS identified in the cohort, after which the adjusted hazard ratio (HR) and 95% confidence interval (CI) were estimated using the inverse probability of treatment weighting (IPTW) method based on propensity scores calculated via logistic regression analysis. Comparisons between groups were conducted using analysis of variance and Pearson’s Chi-square test for continuous and categorical variables, respectively. All p values were based on a two-sided hypothesis, with values <0.05 indicating statistical significance. All statistical analyses were performed using JMP version 18.0.0 (SAS Institute, Cary, NC, USA). This study conforms to the Strengthening the Ris reported in concordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (see Supplemental Materials). 11
Results
Patient characteristics
In total, 309 patients with HER2-positive AGC were analyzed and stratified into three chronological groups: group A (n = 159; 2011–2016), group B (n = 69; 2017–2020), and group C (n = 81; 2021–2023; Figure 1). Among the included patients, 216 (70%) were males, 107 (35%) received gastrectomy prior to chemotherapy, and 189 (61%) and 120 (39%) had an ECOG PS of 0 and ⩾1, respectively. Moreover, 105 (34%) patients had ⩾2 metastatic sites, 115 (37%) had a diffuse-type Lauren classification, and 245 (79%) had a HER2 IHC 3+ status. Details regarding the patients’ characteristics are described in Table 1. In general, several factors differed among the three groups, including the proportion of patients with ECOG PS of ⩾1 (A: 21%, B: 46%, C: 49%; p = 0.02), proportion of patients with HER2 IHC 3+ (A: 82%, B: 68%, C: 83%; p = 0.03), proportion of patients who received gastrectomy prior to chemotherapy for systemic disease (A: 42%, B: 32%, C: 22%; p = 0.008), presence of peritoneal metastases (A: 18%, B: 39%, C: 46%; p = 0.0005), presence of lymph node involvements (A: 35%, B: 43%, C: 56%; p = 0.007), and proportion of patients with ⩾2 metastatic organs (A: 26%, B: 41%, C: 43%; p = 0.01).

Flow chart for patient selection.
Patient characteristics.

CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGJ, esophagogastric junction; FISH, fluorescence in situ hybridization; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; NA, not assessed; No, number; PS, performance status; Ref, reference.
p Value < 0.05.
Survival among the chronological groups
At the cutoff period for data collection (June 1, 2025), the median follow-up duration for the cohort was 72.9 months (103.1, 72.9, and 53.3 months for groups A, B, and C, respectively), with 245 (79%) patients eventually succumbing to their disease. Overall, the median OS, PFS, and the PPS after progression to first-line treatment were 20.9 months (95% CI: 17.7–23.6), 8.5 months (95% CI: 7.4–10.2), and 10.4 months (95% CI: 8.6–11.8), respectively (Figure 2(a) and (b) and Supplemental Figure 1(A)). The TTF for second- and third-line treatments were 4.3 months (95% CI: 3.7–4.8), and 3.0 months (95% CI: 2.3–3.5), respectively.

Kaplan–Meier estimates of (a) overall survival, (b) first-line PFS, and (c) overall survival and (d) first-line PFS in groups A (2011–2016), B (2017–2019), and C (2020–2023).
The median OS rates in groups A, B, and C was 19.5 months, 20.6 months (HR for group A, 0.98; 95% CI: 0.72–1.33), and 23.5 months (HR for group A, 0.80; 95% CI: 0.58–1.11; HR for group B, 0.82; 95% CI: 0.56–1.19), respectively (Figure 2(c)). The median PFS rates in groups A, B, and C was 8.5 months, 9.9 months (HR for group A, 0.92; 95% CI: 0.68–1.26), and 8.1 months (HR for group A, 0.88; 95% CI: 0.66–1.17; HR for group B, 0.94; 95% CI: 0.66–1.34), respectively (Figure 2(d)). Among those whose disease had progressed to first-line treatment, the median PPS rates in groups A, B, and C was 9.5 months, 11.7 months (HR, 0.87; 95% CI: 0.63–1.10), and 11.3 months (HR, 0.86; 95% CI: 0.61–1.20), respectively (Supplemental Figure 1(B)). Among those who received second-line treatment, the median TTF in groups A, B, and C was 4.2 months, 4.3 months (HR for group A, 0.93; 95% CI: 0.65–1.32), and 4.6 months (HR for group A, 0.92; 95% CI: 0.66–1.30), respectively (Supplemental Figure 1(C)). For those who received third-line treatment, the median TTF in groups A, B, and C was 2.8 months, 3.0 months (HR, 1.03; 95% CI: 0.68–1.55), and 3.4 months (HR, 0.93; 95% CI: 0.61–1.41), respectively (Supplemental Figure 1(D)).
Univariate and multivariate Cox regression analyses for OS and PFS
Table 2 summarizes the results of our univariate and multivariate Cox regression analyses for OS, in which baseline characteristics and laboratory test results were used as covariates. The factors found to be independently associated with prognosis in this cohort were the primary location of the esophagogastric junction (HR 0.71, 95% CI: 0.52–0.96, p = 0.03), prior gastrectomy (HR 0.46, 95% CI: 0.28–0.74, p = 0.001), peritoneal metastases (HR 1.51, 95% CI: 1.07–2.2, p = 0.018), and liver metastases (HR 1.43, 95% CI: 1.04–1.96, p = 0.026).
Univariate and multivariate analyses for overall survival.

CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGJ, esophagogastric junction; FISH, fluorescence in situ hybridization; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; PS, performance status; Ref, reference.
p Value < 0.05.
Supplemental Table 1 summarizes the results of our univariate and multivariate Cox regression analyses for PFS. We identified prior gastrectomy (HR 0.61, 95% CI: 0.39–0.95, p = 0.020), peritoneal metastases (HR 1.54, 95% CI: 1.11–2.16, p = 0.009), and liver metastases (HR 1.75, 95% CI: 1.29–2.238, p < 0.001) as prognostic variables for first-line PFS.
Propensity score-weighted survival analysis according to chronological period
We conducted an exploratory analysis using IPTW to adjust for the aforementioned prognostic variables. Notably, this analysis revealed significant improvements in OS in group C. The adjusted HR for death in groups A and C was 0.54 (95% CI: 0.43–0.68, p < 0.0001; Figure 3(a)), while that for groups B and C was 0.72 (95% CI: 0.55–0.95, p = 0.02; Figure 3(b)). We found no significant difference in the adjusted OS between groups A and B (HR 0.86, 95% CI: 0.70–1.05, p = 0.15; Figure 3(c)). We also calculated the adjusted HRs using these factors as covariates. This analysis showed similar trends to those observed in the IPTW (HR in groups A and C 0.59, 95% CI: 0.42–0.84, p = 0.0031; HR in groups B and C 0.75, 95% CI: 0.51–1.11, p = 0.15; HR in groups A and B 0.78, 95% CI: 0.57–1.08, p = 0.15).
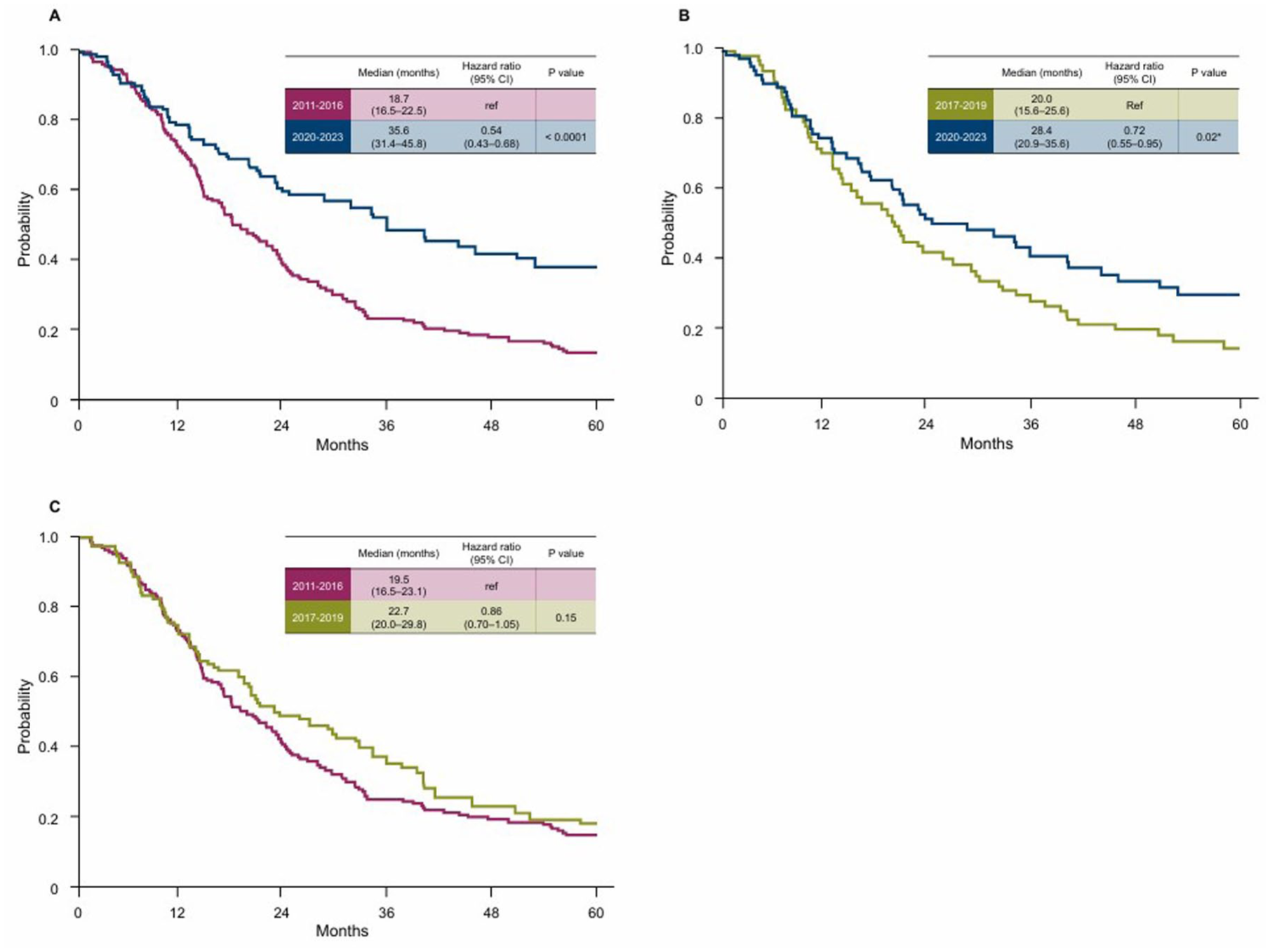
Kaplan–Meier estimates of overall survival adjusted using inverse probability of treatment weighting in (a) group A versus group C, (b) group B versus group C, and (c) group A versus group B.
Patterns of subsequent treatment
At the cutoff point for data collection, 271 (88%) patients had discontinued first-line treatment. The Sankey diagram shows an overview of the treatment journey for patients with HE2-positive AGC (Supplemental Figure 2). Among them, 115 (80.4%), 48 (78.7%), and 60 (89.6%) belonging to groups A, B, and C received second-line treatment, respectively (Supplemental Figure 3(A)). After discontinuing second-line treatment, 72 (53.3%), 35 (61.4%), and 37 (66.1%) patients in groups A, B, and C received a third-line treatment. Regarding fourth-line treatment, 29 (21.5%), 26 (45.6%), and 25 (45.5%), patients in groups A, B, and C, respectively, received antitumor agents. Supplemental Figure 3(B) details subsequent treatment agents in each line according to the groups. Most of the patients received taxane-based chemotherapy as second-line treatment, whereas irinotecan (63%), immunotherapy (43%), and trastuzumab deruxtecan (70%) were the most frequently provided third-line treatments in groups A, B, and C, respectively. The proportion of patients receiving immunotherapy with or without other agents at any treatment line was significantly higher in group B than in the other two groups (A: 17.6%, B: 50.7%, C: 43.2%; p < 0.0001), whereas the proportion of patients receiving trastuzumab deruxtecan as any-line treatment gradually increased across the three groups (7.5%, 30.4%, and 44.4% for groups A, B, and C, respectively; p < 0.0001; Supplemental Figure 4). The use of irinotecan had gradually decreased for the three groups (37.1%, 17.4%, and 3.7% for groups A, B, and C; p < 0.0001).
Curative intent surgery during chemotherapy for systemic disease was performed in 43 (13.9%) patients (A: n = 26, 16.4%; B: n = 7, 10.1%; C: n = 10, 12.3%). Among them, the median OS was 91.8 (95% CI: 32.0–not reached (NR)) in group A, 58.0 (95% CI: 15.4–NR) in group B, and NR months (95% CI: 22.7–NR) in group C (Supplemental Figure 5(A)). By contrast, among those who did not undergo surgery for curative intent, the median OS was 16.8 (95% CI: 14.1–21.7) in group A, 19.7 (95% CI: 13.5–26.7) in group B, and 20.5 (95% CI: 15.8–31.4) months in group C (Supplemental Figure 5(B)).
Discussion
This large-scale, single-center retrospective study provides a detailed chronological analysis of the real-world outcomes of HER2-positive AGC across three time periods. Although the differences between randomized trials and real-world data must be recognized, our cohort outperformed the efficacious outcomes observed in the ToGA trial. 1 This was likely due to improvements in chemotherapy management practices and treatment developments. Although our initial unadjusted analyses found no significant improvements in OS among the groups, the apparent lack of progress could have been attributed to significant imbalances in baseline prognostic factors. Notably, patients in more recent eras presented with worse prognostic profiles, such as a higher incidence of peritoneal metastases and poorer PS, than did those in earlier eras. After accounting for these confounding variables, a significant improvement in OS was observed for patients in group C. This core finding demonstrates that despite the escalating difficulty in treating this patient population over time, the introduction of novel therapies successfully translated into a tangible survival benefit in real-world settings. Although other studies have reported chronological improvements in the management of gastric cancer, 12 our risk-adjusted analysis provides robust evidence that the survival benefits truly reflect therapeutic progress and not merely a consequence of shifting patient selection.
The observed improvements in survival in our most recent cohort were achieved in a patient population with more adverse prognostic factors, underscoring the potency of modern therapeutic agents. These advances can be primarily attributed to the successful integration of trastuzumab deruxtecan into clinical practice. Its superior efficacy, which was first established in the pivotal DESTINY-Gastric01 trial, 9 was confirmed by our real-world data presented herein. Our findings show that the use of trastuzumab deruxtecan increased dramatically from 7.5% in group A to 44.4% in group C, coinciding with the observed improvements in OS. The efficacious outcomes of trastuzumab deruxtecan in our study are consistent with those of the EN-DEAVOR study. They affirm the effectiveness and manageable safety risks of trastuzumab deruxtecan in Japanese routine practice.13–15 Hence, opportunities to introduce trastuzumab deruxtecan for the management of HER2-positive AGC should not be overlooked.
Our study failed to reveal the impact of the emergence of immunotherapy for this population. The appearance of immune checkpoint inhibitors following the ATTRACTION-2 trial might have contributed to the treatment continuum, further enriching later-line options aside from trastuzumab deruxtecan. 8 Although the use of immunotherapy as third- or later-line treatment increased dramatically from group A to group B, OS remained comparable between the two groups even after adjusting for certain prognostic factors. This finding suggests that the population-level benefit derived from the integration of immunotherapy was modest in real-world settings. The reasons for this might be complicated, but could include the common use of irinotecan as later-line treatment in group A, as well as the different proportions of patients who underwent curative intent surgery between groups A and B. The benefits of immunotherapy for HER2-positive AGC have been limited to certain patients, and the unselected nature of a real-world cohort might have diluted the overall therapeutic effect of immunotherapy. These findings encourage efforts to identify patients with HER2-positive AGC who might benefit from immunotherapy, although predictive biomarkers, including PD-L1 expression status, were rarely evaluated in the present study.
Although the successful integration of these effective later-line agents has transformed the treatment landscape, it has also created new clinical challenges regarding optimal sequencing. 13 This complexity is rapidly increasing as potent therapies shift into earlier lines of treatment. For instance, the KEYNOTE-811 trial demonstrated that adding pembrolizumab to trastuzumab plus chemotherapy significantly improved survival among patients with HER2-positive and PD-L1-positive AGC, thereby establishing a new option for first-line treatment. 16 A meta-analysis investigating the efficacy of immune checkpoint inhibitors combined with trastuzumab and chemotherapy found anti-PD-1 antibodies to be consistently efficacious in this population. Outcomes were particularly promising in PD-L1-positive patients and first-line treatments. 17 Similarly, trastuzumab deruxtecan is poised to move into the second-line setting after the DESTINY-Gastric04 trial showed that it produced better OS than did standard second-line paclitaxel plus ramucirumab in HER2 positivity-maintaining AGC after progression to first-line trastuzumab. 18 Looking ahead, we believe that the treatment landscape is set to become even more complex, with pivotal first-line trials of agents including zanidatamab 19 (HERIZON-GEA-01; NCT05152147) and new combination strategies with trastuzumab deruxtecan (DESTINY-Gastric05 20 ; NCT06731478, ARTEMIDE-Gastric01 21 ; NCT06764875) underway. This front-line shift in both immune checkpoint inhibitors and potent therapeutic agents fundamentally alters the treatment paradigm from a linear approach to a more dynamic, multifaceted strategy. However, despite this clear progress, our analysis also revealed limited improvements in the prognosis of patients with poor baseline characteristics, highlighting a critical unmet need for novel therapies or strategies specifically for this high-risk subgroup.
Our findings should be interpreted with several limitations in mind. The primary limitation of this study was its retrospective design, which carries an inherent risk of selection bias and unmeasured confounders that could influence outcomes. Although our propensity score-weighted analysis was designed to control for known variables, residual confounders may still have existed. Second, the proportion of patients who underwent surgery with curative intent differed between the three groups, which may also have influenced survival outcomes in this cohort. Furthermore, our data were obtained from a single Japanese institution. This limits the generalizability of our results, especially internationally, as treatment patterns and drug approval timings can differ between countries. In addition, the decreased inclusion of patients with serious underlying diseases or multiple comorbidities over the study period may have introduced selection bias, as the characteristics of our patients may have differed from those of patients in community hospitals. Furthermore, dividing the study period based on the times at which novel agents were approved in Japan may not be an appropriate means of evaluating chronological improvements in survival among this patient population. Further observations are needed, including the exploration of other methods to address this. Lastly, attributing the observed survival benefit solely to novel pharmacological agents is challenging, as gradual improvements in overall supportive care over the 12-year period may also have contributed to the improved outcomes.
Not all the approved agents, including trastuzumab, nivolumab, and trastuzumab deruxtecan, were available to all patients with HER2-positive AGC in clinical practice. Among our cohort, patients with more advanced disease, poor PS, or comorbidities were unsuitable for some treatments. However, such patients are usually excluded from clinical trials. In addition, many factors can change over time and affect treatment outcomes, including patient characteristics, the applicability of chemotherapy, the late-line treatment options, and the criteria for surgery with curative intent. Despite these limitations, we believe that the current study offers a valuable evaluation of the chronological improvements in survival among this patient population and may help clinicians to better understand the treatment of HER2-positive AGC in clinical practice.
Conclusion
The current study provides compelling real-world evidence that the sequential introduction of novel agents, particularly trastuzumab deruxtecan and, to some extent, immunotherapy, might have contributed to improvements in OS in patients with HER2-positive AGC, despite the concurrent worsening of patient risk profiles. Our findings suggest the benefits of introducing modern therapies into routine practice and highlight the challenges of optimizing treatment sequences in an increasingly complex therapeutic landscape.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261441026 – Supplemental material for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023
Supplemental material, sj-docx-1-tag-10.1177_17562848261441026 for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023 by Keitaro Shimozaki, Akira Ooki, Kaoru Yoshikawa, Koichiro Yoshino, Shohei Udagawa, Hiroki Osumi, Shota Fukuoka, Takeru Wakatsuki, Mariko Ogura, Daisuke Takahari, Eiji Shinozaki, Keisho Chin and Kensei Yamaguchi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848261441026 – Supplemental material for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023
Supplemental material, sj-docx-2-tag-10.1177_17562848261441026 for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023 by Keitaro Shimozaki, Akira Ooki, Kaoru Yoshikawa, Koichiro Yoshino, Shohei Udagawa, Hiroki Osumi, Shota Fukuoka, Takeru Wakatsuki, Mariko Ogura, Daisuke Takahari, Eiji Shinozaki, Keisho Chin and Kensei Yamaguchi in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848261441026 – Supplemental material for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023
Supplemental material, sj-docx-3-tag-10.1177_17562848261441026 for Increased treatment options and improved survival in real-world patients with HER2-positive advanced gastric cancer from 2011 to 2023 by Keitaro Shimozaki, Akira Ooki, Kaoru Yoshikawa, Koichiro Yoshino, Shohei Udagawa, Hiroki Osumi, Shota Fukuoka, Takeru Wakatsuki, Mariko Ogura, Daisuke Takahari, Eiji Shinozaki, Keisho Chin and Kensei Yamaguchi in Therapeutic Advances in Gastroenterology
