Abstract
Background:
No established predictive biomarker beyond tissue HER2 status currently exists for trastuzumab deruxtecan (T-DXd) treatment response in human epidermal growth factor receptor 2 (HER2)-positive advanced gastric cancer (AGC). Serum HER2 extracellular domain (HER2 ECD) has shown potential in breast cancer; however, its clinical significance in gastric cancer remains unclear.
Objectives:
To evaluate the efficacy and safety of T-DXd in HER2-positive AGC and investigate the predictive role of serum HER2 ECD for T-DXd response.
Design:
Retrospective single-institution study.
Methods:
Data of HER2-positive AGC patients treated with T-DXd were analyzed. According to their baseline serum HER2 ECD levels, the patients were classified into high (⩾12.1 ng/mL) and low (<12.1 ng/mL) HER2 ECD groups. Tumor responses, HER2 ECD dynamics, HER2 re-biopsy status, progression-free survival (PFS), and overall survival (OS) were evaluated.
Results:
Altogether, 67 patients were enrolled. The HER2 ECD data were available only for 38 patients, with 23 and 15 patients in the high and low HER2 ECD groups, respectively. The objective response rate (ORR) was significantly higher in the high HER2 ECD group (65.2%, 15/23 vs 26.7%, 4/15; p = 0.045). Among the patients with immunohistochemistry (IHC) 2+/in situ hybridization (ISH+) tumors, responses were observed exclusively in the high HER2 ECD group (57.1%, 4/7 vs 0%, 0/8). Additionally, the HER2 ECD level changes post-treatment initiation also reflected tumor response, with no responses observed in the high HER2 ECD group. HER2 positivity in re-biopsy was associated with higher ORR (50.0%, 3/6 vs 25.0%, 1/4). Contrarily, the baseline HER2 ECD levels were not associated with PFS or OS.
Conclusion:
Baseline serum HER2 ECD may be a potential non-invasive predictor of T-DXd response in HER2-positive AGC, particularly in IHC 2+/ISH+ cases. Dynamic HER2 ECD level changes during therapy may also reflect treatment response.
Plain language summary
This study examined whether a blood marker called serum HER2 extracellular domain (HER2 ECD) may help predict response to trastuzumab deruxtecan (T-DXd) in HER2-positive advanced gastric cancer. We analyzed 38 patients treated with T-DXd and measured HER2 ECD levels before treatment and again about three weeks after starting therapy. Patients with higher baseline HER2 ECD were more likely to respond to T-DXd, and decreases after treatment were often seen in responders. These findings suggest that HER2 ECD may serve as a simple and minimally invasive marker to help identify patients who may benefit from T-DXd, although larger studies are needed to confirm this.
Introduction
Worldwide, gastric cancer (GC) ranks fifth in incidence and is the fourth leading cause of cancer-related mortality. 1 Systemic chemotherapy is the mainstay for unresectable or metastatic GC, and various combination regimens have modestly prolonged survival over the past decades.2–6 Recently, molecular targeted therapies and immunotherapies were integrated into standard care guidelines as they have opened new avenues for treatment and yielded improved outcomes. One pivotal molecular target in GC is the human epidermal growth factor receptor 2 (HER2).7,8
HER2 overexpression or amplification is observed in approximately 15%–20% of advanced GC (AGC) cases.9,10 HER2-positive tumors constitute a distinct subset characterized more often by intestinal histology and proximal (gastroesophageal junction or cardiac) location, as compared to HER2-negative tumors.6,9,11,12 Trastuzumab deruxtecan (T-DXd) is a HER2-directed antibody–drug conjugate that delivers a cytotoxic payload to HER2-expressing cells. In the phase II DESTINY-Gastric01 trial, T-DXd achieved a markedly higher objective response rate (ORR; 51.3% vs 14.3%) and longer median overall survival (OS; 12.5 vs 8.4 months) than standard chemotherapy in HER2-positive GC patients who had progressed on trastuzumab.13,14 Moreover, recent data suggest that patients with very low HER2 expression levels (so-called “HER2-low” tumors) may also benefit from T-DXd, expanding the spectrum of HER2-targeted therapy.15,16
Despite these advances, the optimal biomarkers for predicting response to HER2-targeted therapies are still being explored. In the DESTINY-Gastric01 trial, for example, patients with immunohistochemistry (IHC) 3+ tumors had an ORR of 58% on T-DXd, whereas those with IHC 2+ with in situ hybridization (ISH) positive tumors obtained an ORR of 29%. 17 This is in agreement with the findings from previous studies and indicates that strong HER2 overexpression is associated with greater sensitivity to HER2-directed therapies.6,18 However, because HER2 expression within tumors is heterogeneous and may change over time under treatment pressure, the initial diagnostic biopsy may not accurately reflect the tumor biology in subsequent treatment lines. Indeed, HER2 expression is not static; up to 29%–61% of GCs can lose HER2 positivity after progression on trastuzumab-based therapy.18,19 As a result, patients with a loss of HER2 positivity may no longer benefit from subsequent HER2-targeted treatments.18,20
To address this issue, serum HER2 extracellular domain (HER2 ECD), a fragment of the HER2 protein that is shed into the bloodstream in patients with HER2-overexpressing tumors, has been proposed as a minimally invasive biomarker. Serum HER2 ECD may bind trastuzumab and act as a decoy, thereby reducing its therapeutic activity.21,22 In HER2-positive breast cancer, an elevated serum HER2 ECD level is associated with higher tumor burden and poorer prognosis, and its dynamic changes after the initiation of therapy could be used to track the disease course. 23 In GC, several studies have also investigated the clinical relevance of serum HER2 ECD in patients receiving trastuzumab-based therapy. Higher baseline serum HER2 ECD levels have been reported to correlate with greater tumor burden, liver metastasis, and IHC 3+ tumors, whereas peritoneal dissemination tends to be associated with lower levels. Moreover, early decreases in serum HER2 ECD after the initiation of trastuzumab-based treatment have been associated with favorable response or disease control in some studies.24–26 Although these findings have not yielded a unified conclusion, they collectively suggest that serum HER2 ECD may reflect tumor biology and treatment sensitivity beyond tissue HER2 expression. The exploratory analyses of the DESTINY-Gastric01 trial have indicated that patients with higher baseline serum HER2 ECD levels tended to have higher ORR (36.7% vs 0%). 17 However, the utility of HER2 ECD as a biomarker in AGC is less established. Several studies in GC have reported that elevated baseline serum HER2 ECD levels were correlated with larger tumor burden, more frequent liver or peritoneal metastases, and inferior outcomes in patients treated with trastuzumab-based chemotherapy.20,22 These findings suggest that serum HER2 ECD reflects tumor biology beyond tissue HER2 status and may provide clinically relevant information for patient stratification in GC.
Given this background, the present retrospective observational study aimed to evaluate the clinical efficacy and safety of T-DXd and to investigate the predictive and monitoring utility of serum HER2 ECD levels in HER2-positive AGC patients.
Methods
Study design and patients
This single-institution, retrospective observational study was conducted at Aichi Cancer Center Hospital (Nagoya, Japan). The study protocol was approved by the Institutional Review Board (IRB) of Aichi Cancer Center (IRB ID: IR041009) and adhered to the standards and guidelines stipulated in the Declaration of Helsinki as well as the STROBE guidelines of observational retrospective studies. The requirement for obtaining patients’ written informed consent was waived due to the retrospective and non-interventional nature of the present study in accordance with national regulations and institutional policies. The participants were allowed to withdraw from the study through our institution’s website.
The study included consecutive patients with HER2-positive advanced gastric or gastroesophageal junction adenocarcinoma who were treated with T-DXd until December 2024 (data cutoff date: July 10, 2025). The primary inclusion criteria were as follows: histologically confirmed adenocarcinoma of the esophago-gastric junction (EGJ) or stomach; unresectable or recurrent disease; previously treated with trastuzumab; treatment with at least one cycle of T-DXd; Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–2; and sufficient bone marrow, hepatic, and renal functions. Patients were treated with T-DXd at either the approved dose of 6.4 mg/kg every 3 weeks or at a reduced dose (5.4 or 4.4 mg/kg) at the treating physician’s discretion (often based on PS and/or organ function) without predefined uniform criteria. We excluded patients with incomplete clinical data and concomitant malignancies that could confound the outcomes.
HER2 positivity was defined according to the established clinical guidelines as either HER2 IHC 3+ or IHC 2+ with confirmation of HER2 gene amplification by ISH. 27 The HER2 assessment was performed by expert pathologists using either a Dako Autostainer Link48 (HercepTest; Dako Corp., Carpinteria, CA, USA) or a Ventana BenchMark ULTRA (PATHWAY anti-HER2/neu (4B5); Ventana Medical Systems, Tucson, AZ, USA). Serum HER2 ECD measurement was outsourced to BML Inc. (Tokyo, Japan) and performed by chemiluminescence immunoassay using anti-HER2 monoclonal antibodies.
Data collection
Clinical data were extracted from the medical records. The serum HER2 ECD level was measured if a pre-treatment blood sample was available, using a chemiluminescence immunoassay using anti-HER2 monoclonal antibodies.
In a subset of patients, repeat HER2 assessment by tumor biopsy was performed prior to starting T-DXd. The results of these re-assessments were categorized as persistence or loss of HER2 positivity. Tumor responses were evaluated by imaging studies and assessed according to RECIST version 1.1 criteria in patients with measurable lesions. 28 The disease control rate (DCR) refers to the proportion of patients who achieved stable disease (SD) or complete response (CR)/partial response (PR), whereas the ORR refers to the proportion of patients who achieved either a PR or CR, as their best response. Progression-free survival (PFS) refers to the time period from the date of starting T-DXd therapy to the time of death from any cause or disease progression. RECIST version 1.1 and/or clinical assessment were used by the treating physician to evaluate the disease progression. OS refers to the time period from the date of starting T-DXd therapy to the date of death or last follow-up visit.
The post-treatment serum HER2 ECD level was defined as the earliest available HER2 ECD measurement obtained within 4 weeks after the initiation of T-DXd. We also captured the following T-DXd treatment intensity metrics: number of cycles administered, treatment duration, relative dose intensity (RDI, the actual dose delivered per unit time divided by standard dose intensity), and any dose reductions or interruptions. The treatment-related adverse events were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0. 29
Statistical analysis
R software version 4.1.0 (R Project for Statistical Computing, Vienna, Austria) was used to carry out the statistical analyses. All tests were two-sided, and p-values <0.05 denoted statistically significant differences. The baseline clinicopathological features were compared using Chi-squared or Fisher’s exact tests. A cut-off value of ⩾12.1 ng/mL was used to define elevated HER2 ECD levels. It was determined based on the maximum Youden index on the receiver-operating characteristic (ROC) curve for the ORR. The Kaplan–Meier (KM) method of analysis was employed to estimate the PFS and OS. Survival in different patient groups was analyzed using Cox proportional hazards regression analysis. Univariate analyses were used to assess the predictive ability of the following baseline clinicopathological features for outcomes: sex (male vs female); age (⩾65 vs <65 or ⩾70 vs <70 years); ECOG PS (⩾1 vs 0); histology (intestinal vs diffuse); location of the primary tumor (EGJ/cardia vs stomach); history of gastrectomy (yes vs no); disease status (metastatic vs recurrent); HER2 status (IHC3+ vs IHC2+/ISH+); number of metastasis (⩾2 vs <2); liver metastasis (yes vs no); peritoneal metastasis (yes vs no); lung metastasis (yes vs no); lymph node metastasis (yes vs no); bone metastasis (yes vs no); presence of ascites (moderate/severe vs mild/none); serum lactate dehydrogenase (LDH; ⩾upper limit of normal (ULN) vs <ULN); serum alkaline phosphatase (ALP; ⩾ULN vs <ULN); serum carbohydrate antigen 19-9 (CA19-9; ⩾ULN vs <ULN); serum carcinoembryonic antigen (⩾ULN vs <ULN); modified Glasgow prognostic score (GPS; 0 vs 1–2), neutrophil-to-lymphocyte ratio (NLR; ⩾3.2 vs <3.2); duration of trastuzumab treatment before T-DXd therapy (⩾9.2 vs <9.2 months); prior immune checkpoint inhibitor treatment (yes vs no); and initial T-DXd dose (6.4 vs 5.4 or 4.4 mg/kg). The modified Glasgow Prognostic Score (mGPS) was defined based on a previous report. 30 The cutoff value for the NLR was set at 3.2, which was the median value for the patients included in this study. The clinicopathological factors with p-values of <0.05 in the univariate analysis were included in the multivariate Cox proportional model. Owing to the smaller subset with available HER2 ECD data (n = 38), the analysis of HER2 ECD as a prognostic factor was done in a univariate analysis only, because the limited sample size precluded multivariate analyses. No formal sample size calculation was performed because of the retrospective nature of the study; the number of patients analyzed reflects all available cases during the specified period.
Results
Patients
Sixty-nine consecutive patients with HER2-positive AGC received T-DXd therapy. Two patients were excluded due to ECOG PS of 3 (Supplemental Figure 1), leaving 67 patients for the present analysis. The patients’ baseline characteristics are summarized in Table 1. The patients’ median age was 68 years (range, 33–88 years), and 71.6% of the patients were male. The ECOG PS was 0 and 1–2 in 47.8% and 52.2% of patients, respectively. Based on histology, 67.2% and 32.8% had intestinal- and diffuse-type tumors, respectively. The tissue HER2 status was IHC 3+ and IHC 2+/ISH+ in 64.2% and 35.8% of the patients, respectively. All patients had undergone at least two prior lines of treatment: 52.2% had two, 14.9% had three, and 32.8% had four or more prior regimens.
Baseline characteristics.

ALP, alkaline phosphatase; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; ECD, extracellular domain; ECOG, Eastern Cooperative Oncology Group; EGJ, esophago-gastric junction; HER2, human epidermal growth factor receptor 2; ICI, immune checkpoint inhibitor; IHC, immunohistochemistry; ISH, in situ hybridization; LDH, lactate dehydrogenase; mGPS, modified Glasgow Prognostic Score; NLR, neutrophil-to-lymphocyte ratio; PS, performance status; T-DXd, trastuzumab deruxtecan; ULN, upper limit of normal.
Before initiating the T-DXd treatment, the baseline serum HER2 ECD level was available only for 38 patients (57% of the cohort), as the serum samples were not obtained in the rest of the patients. The median HER2 ECD level was 11.5 ng/mL (range, 2.3–134.5 ng/mL). ROC curve analysis using ORR as the endpoint identified 12.1 ng/mL as the optimal threshold by the Youden index (AUC 0.675 (95% confidence interval (CI), 0.495–0.854); sensitivity, 0.789; specificity, 0.579; Youden index, 0.368; Supplemental Figure 2). Based on this, 23 and 15 patients were classified as having high (⩾12.1 ng/mL) and low HER2 ECD (<12.1 ng/mL) levels, respectively. Patients with high HER2 ECD levels tended to have a more advanced disease, for example, higher rates of liver (65.2% vs 31.3%) and peritoneal metastases (60.9% vs 37.5%) than the low HER2 ECD group, although these differences were not statistically significant (Supplemental Table 1).
Regarding T-DXd dosing, 46 patients received the standard dose (6.4 mg/kg), whereas 21 started at a reduced dose (5.4 mg/kg in 15 patients; 4.4 mg/kg in 6 patients). Lower doses were prescribed due to poor clinical conditions (n = 16), advanced age (n = 4), or prior lung disease (n = 1). Patients receiving lower doses were significantly older (median 70 vs 66 years; p = 0.033) and had higher mGPS (1–2 vs 0, 81.0% vs 52.2%; p = 0.032; Supplemental Table 2). The median number of T-DXd cycles was 5 (range, 1–55), with a treatment duration of 2.8 (range, 0.1–39.3 months). The median RDI was 84.6%, which was numerically higher in the standard-dose group (92.4%, interquartile range (IQR) 79.2–98.6) than in the reduced group (70.2%, IQR 60.3–81.1); RDI of ⩾80% was achieved in 73.9% and 28.6% of the patients in the standard- and reduced-dose groups, respectively (Supplemental Table 3).
Efficacy outcomes
The median follow-up duration after initiating T-DXd treatment was 11.3 months (IQR, 8.2–19.0 months). The ORR was 40.9% (27 of 66 patients with measurable lesions), with 3 patients (4.5%) achieving CR and 24 patients (36.3%) achieving PR (Figure 1). The DCR was 71.2%, with the SD and progressive disease rates being 30.3% and 24.2%, respectively.

Tumor response in the overall evaluable population (n = 66). Waterfall plot illustrating the maximum percentage change in the sum of target lesion diameters from baseline in all evaluable patients treated with T-DXd. Each bar represents an individual patient.
An elevated baseline serum HER2 ECD level was the only factor identified as a predictor of ORR, with the high HER2 ECD group showing significantly higher ORR than the low HER2 ECD group (65.2%, 15/23 vs 26.7%, 4/15; p = 0.045; Figure 2). Although the ORR was numerically higher in IHC 3+ than in IHC 2+/ISH+ tumors (48.8%, 21/43 vs 26.1%, 6/23), the difference was not significant (p = 0.126). The ORR was also comparable between the standard- and reduced-dose groups (39.1%, 18/46 vs 45.0%, 9/20; p = 0.862; Supplemental Table 4). Notably, among the IHC 2+/ISH+ patients, those with high HER2 ECD levels demonstrated a remarkably higher ORR (57.1%, 4/7) than those with low HER2 ECD levels, who showed no response (0%, 0/8). Among the patients with paired pre- and post-treatment HER2 ECD measurements (n = 25), the post-treatment HER2 ECD measurement was obtained at a median of 21 days (range 17–28 days) after the start of T-DXd therapy. The ORRs were 62.5% in patients with HER2 ECD decline (high to low), 83.3% in those with sustained high HER2 ECD (high to high), 33.3% in those with sustained low HER2 ECD (low to low), and 0% in those with increasing HER2 ECD (low to high), without statistical significance (p = 0.099) between groups (Supplemental Figure 3). Among 10 patients who underwent repeated tumor HER2 testing, those with a loss of HER2 positivity had a lower ORR (25.0%, 1/4) than those who maintained their HER2 positivity (ORR, 50.0%, 3/6; Figure 3).

The objective response rates according to the HER2 status and serum HER2 ECD level. Bar plots showing the objective response rates to T-DXd across four subgroups are illustrated: (1) tumor HER2 status (IHC 3+ vs IHC 2+/ISH+); (2) baseline serum HER2 ECD level (high, ⩾12.1 ng/mL vs low, <12.1 ng/mL); (3) HER2 ECD level within the IHC 3+ subgroup; and (4) HER2 ECD level within the IHC 2+/ISH+ subgroup.

The objective response rates according to the HER2 status on re-biopsy. Bar plot showing the objective response rates among patients who underwent repeated tumor HER2 testing.
KM analysis revealed a median PFS of 4.1 months (95% CI, 2.8–5.4 months) and median OS of 10.7 months (95% CI, 6.5–12.3 months) in the whole population (Supplemental Figure 4). Among the baseline clinical variables, peritoneal metastasis, ascites, serum LDH, CA19-9, and mGPS showed significant associations with either PFS or OS in the univariate analysis (Tables 2 and 3). In the multivariate analysis, no baseline clinical variable was independently associated with PFS, whereas peritoneal metastasis (hazard ratio (HR), 3.07; 95% CI, 1.62–5.81; p = 0.001) and elevated LDH (HR, 2.04; 95% CI, 1.09–3.83; p = 0.026) remained significant prognostic factors for poor OS. There were no significant differences in the median PFS (5.2 vs 4.4 months; HR, 0.92; 95% CI, 0.47–1.83; p = 0.820) or OS (9.9 vs 10.7 months; HR, 0.93; 95% CI, 0.47–1.88; p = 0.853) between the high and low HER2 ECD groups (Supplemental Figure 5).
Univariate and multivariate analyses for progression-free survival.
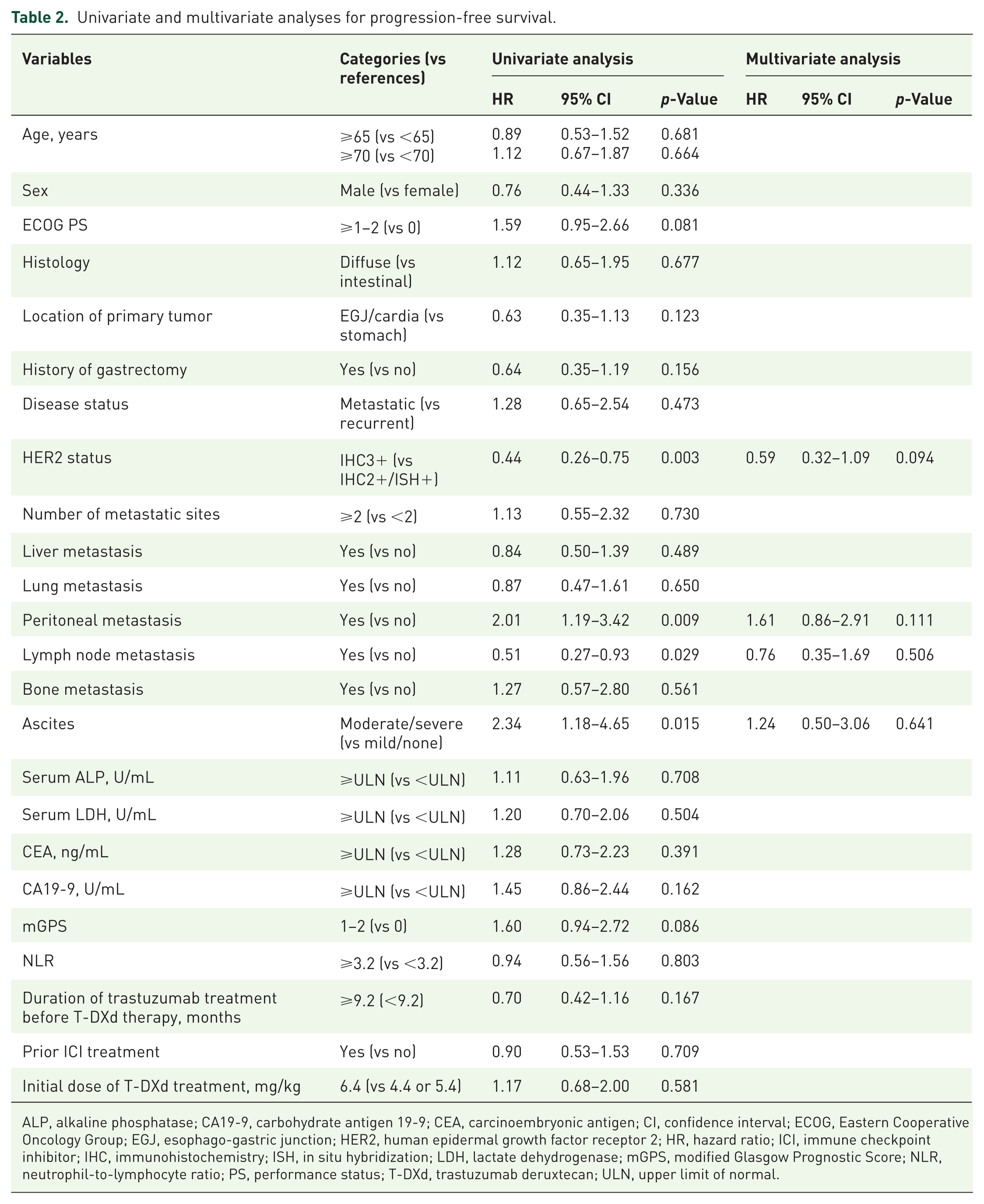
ALP, alkaline phosphatase; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGJ, esophago-gastric junction; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; ICI, immune checkpoint inhibitor; IHC, immunohistochemistry; ISH, in situ hybridization; LDH, lactate dehydrogenase; mGPS, modified Glasgow Prognostic Score; NLR, neutrophil-to-lymphocyte ratio; PS, performance status; T-DXd, trastuzumab deruxtecan; ULN, upper limit of normal.
Univariate and multivariate analyses for overall survival.

ALP, alkaline phosphatase; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EGJ, esophago-gastric junction; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; ICI, immune checkpoint inhibitor; IHC, immunohistochemistry; ISH, in situ hybridization; LDH, lactate dehydrogenase; mGPS, modified Glasgow Prognostic Score; NLR, neutrophil-to-lymphocyte ratio; PS, performance status; T-DXd, trastuzumab deruxtecan; ULN, upper limit of normal.
Safety
Disease progression was the most common reason for discontinuing T-DXd therapy (83.5%), and 10.4% discontinued T-DXd therapy because of adverse events, including interstitial lung disease (ILD) in three patients and fatigue in four patients. After discontinuation, the proportions of patients receiving subsequent chemotherapy and best supportive care were 56.9% and 43.1%, respectively.
Supplemental Table 5 lists the adverse events during the T-DXd therapy. The most common adverse events were neutropenia (73.1%, 49/67), anemia (67.2%, 45/67), and nausea (49.3%, 33/67). Notably, anemia and fatigue were significantly more frequent in the 6.4-mg/kg dose group compared to the 5.4- or 4.4-mg/kg dose group (p = 0.002 and 0.001, respectively). Drug-related ILD occurred in nine patients (13.4%, 9/67), with all of these patients receiving a dose of 6.4 mg/kg (p = 0.048); all cases were grade 1 or 2 and resolved with corticosteroid treatment and therapy interruption. There were no treatment-related deaths in our series.
Discussion
This study was a retrospective, single-institution analysis, and therefore subject to multiple potential biases, including heterogeneity in the number of treatment cycles, discretionary dose reductions, and lack of standardized reduction criteria. These limitations may have affected the interpretation of efficacy and safety outcomes. The present investigation is the first real-world study to demonstrate that baseline serum HER2 ECD levels may predict the objective response to T-DXd treatment in HER2-positive AGC patients. A high baseline HER2 ECD level was associated with more advanced disease, including higher rates of liver and peritoneal metastases. Notably, among tumors with IHC 2+/ISH+ HER2 expression, the responses to T-DXd were observed only in cases with high HER2 ECD levels. Furthermore, the changes in HER2 ECD levels following treatment initiation correlated with the response patterns, and patients who retained their HER2 positivity on re-biopsy showed higher response rates. Contrarily, the baseline HER2 ECD level was not associated with PFS or OS, whereas peritoneal metastasis and elevated LDH level independently predicted poorer survival.
Importantly, our findings highlight serum HER2 ECD as a more robust predictor of response to T-DXd compared to conventional tissue-based HER2 assessment. The pronounced predictive value of HER2 ECD, particularly among IHC 2+/ISH+ tumors, which are typically considered less responsive to HER2-targeted therapies, underscores its potential clinical utility in refining patient selection beyond the limitations of the current HER2 scoring systems by tissue.6,13 Unlike the DESTINY-Gastric01 trial, which reported a modest association between serum HER2 ECD and T-DXd efficacy, our study provides the first real-world validation that HER2 ECD clearly stratifies the responders, especially in IHC 2+/ISH+ cases. 17 This may be because serum HER2 ECD reflects the total burden of HER2-expressing clones, thereby complementing tissue assessment that may underestimate intratumoral heterogeneity. Although the threshold (⩾12.1 ng/mL) used in our study showed promising discriminatory ability, its generalizability should be interpreted with caution until validated in independent cohorts. Furthermore, although statistical significance was observed in the ORR difference between high and low HER2 ECD groups, the p-value was close to the conventional threshold, which warrants cautious interpretation. The DESTINY-Gastric01 trial did not adjust for baseline disease severity and used an exploratory subgroup analysis, our analysis incorporated clinical context descriptively, although only univariate analyses could be performed due to the limited sample size, and demonstrated that tissue HER2 status alone may be insufficient for accurate prediction. In addition, other biological determinants beyond HER2, such as programmed death-ligand 1 expression, microsatellite instability, claudin 18.2, and fibroblast growth factor receptor 2b, may also influence treatment response and disease progression. Future studies integrating these biomarkers with serum HER2 ECD are warranted to provide a more comprehensive understanding of therapeutic outcomes in GC.
Additionally, this is the first report to show that early post-treatment HER2 ECD dynamics may reflect tumor response, suggesting its potential for real-time treatment monitoring. The patients whose HER2 ECD declined or remained high after treatment initiation had higher response rates, whereas those with increasing HER2 ECD levels did not show any response. In this analysis, “remained high/low” denotes values that stayed on the same side of the 12.1 ng/mL threshold despite minor within-range fluctuations, and the four categories were used to jointly convey baseline position relative to the threshold and early direction of change. A prior study on HER2-positive breast cancer also reported that increasing serum HER2 ECD levels at recurrence were associated with worse survival, supporting the prognostic value of HER2 ECD kinetics in treatment monitoring. 31 Although this evidence comes from a different disease setting, it underscores the potential biological relevance of dynamic HER2 ECD changes across HER2-driven cancers. These data highlight the potential utility of HER2 ECD, not only at baseline, as a dynamic, non-invasive biomarker for early efficacy assessment in HER2-positive AGC cases. A plausible biological explanation is that effective tumor killing by T-DXd reduces the shedding of the HER2 ECD from tumor cells, leading to a decline in circulating HER2 ECD levels, whereas sustained or increasing levels may reflect ongoing tumor activity.21–23 These findings should be regarded as hypothesis-generating, and prospective studies are warranted to validate the clinical utility of dynamic HER2 ECD monitoring.
Our findings are also in agreement with the results from previous reports demonstrating that HER2 status re-evaluation prior to T-DXd treatment may aid in predicting the treatment response.17,18 In our cohort, patients who retained HER2 positivity on repeat biopsy showed higher response rates than those who lost HER2 expression. This supports the notion that serum HER2 ECD may function as a surrogate biomarker in cases where tissue reassessment is not feasible. However, because only a very limited number of patients had both serum HER2 ECD and re-biopsy data available, we were unable to perform a reliable statistical comparison of ECD levels between persistence and HER2 loss groups.
Despite its predictive value for tumor response, the baseline HER2 ECD level was not associated with PFS or OS in the current study. This apparent discrepancy may reflect the dual nature of high HER2 ECD levels, which may indicate both HER2-driven biology and more aggressive disease features, including the development of liver or peritoneal metastases. Indeed, prior breast cancer research has shown similar associations between high serum HER2 ECD levels and greater tumor burden. 32 The DESTINY-Gastric01 trial also reported numerically longer OS in patients with higher HER2 ECD, but did not adjust for baseline disease severity. 17 In our study, peritoneal metastasis and elevated LDH levels, not HER2 ECD, were independently associated with poor OS, highlighting the multifactorial nature of the prognosis. Additional factors, including treatment resistance, post-progression therapies, heterogeneous patient backgrounds such as PS, histology, and prior treatment history may also obscure the prognostic relevance of HER2 ECD. In particular, because only 38 patients had available HER2 ECD data, multivariate adjustment could not be performed, which further limits the strength of this analysis.
The safety outcomes in our cohort were generally consistent with those of prior reports.13,14 Although most adverse events were manageable, anemia, fatigue, and ILD were more frequently observed in the standard-dose group, whereas ILD events were numerically less frequent among patients who initiated T-DXd at reduced doses. In the DESTINY-Gastric02 trial, conducted in Western patients, the incidence of adjudicated drug-related ILD or pneumonitis was 5.1%, compared to 9.6% in the Asia-based DESTINY-Gastric01 trial.13,33 This disparity may reflect ethnic differences, as pooled analyses have suggested a higher susceptibility to T-DXd-associated ILD among Asian patients. 34 Taken together, these exploratory observations raise the possibility that initiating treatment at a reduced dose may help mitigate toxicity in selected patients, although confirmation in larger studies is required. Consistent with previous reports such as Jubashi et al., 35 we treated patients with different starting doses as a single cohort. Although RDI and adverse events were presented, the limited sample size precluded further analyses stratified by initial dose or dose reductions, and therefore, all patients were analyzed together. This should be recognized as a limitation of our study.
Several limitations of the present study should be acknowledged. This was a retrospective, single-institution study with a limited sample size, particularly in subgroups analyzed for HER2 ECD dynamics and re-biopsy status. Although the HER2 ECD cutoff value (⩾12.1 ng/mL) stratified the response effectively in this cohort, it was derived retrospectively and lacks external validation. Thus, while potentially clinically useful, our study data should be interpreted as exploratory. The DESTINY-Gastric04 trial demonstrated the efficacy of T-DXd in patients who maintained their HER2 positivity. 36 However, as re-biopsy is not always feasible in clinical practice, measuring the serum HER2 ECD level may offer a practical, non-invasive alternative for treatment selection. Further prospective studies, including the ongoing biomarker EN-MARK trial (jRCTs031240055), are needed to confirm the clinical utility of this approach.
Conclusion
Collectively, our study findings underscore the potential clinical utility of serum HER2 ECD as a non-invasive, dynamic biomarker for identifying patients most likely to benefit from T-DXd. Further prospective studies are warranted in the future to validate the role of serum HER2 ECD in response prediction, treatment monitoring, and integration into routine clinical decision-making.
Supplemental Material
sj-doc-1-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-doc-1-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-10-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-docx-10-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-11-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-docx-11-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-7-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-docx-7-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-8-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-docx-8-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-9-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-docx-9-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pptx-2-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-pptx-2-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pptx-3-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-pptx-3-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pptx-4-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-pptx-4-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pptx-5-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-pptx-5-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pptx-6-tag-10.1177_17562848251410771 – Supplemental material for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study
Supplemental material, sj-pptx-6-tag-10.1177_17562848251410771 for Serum HER2 extracellular domain as a predictive biomarker for trastuzumab deruxtecan treatment response in HER2-positive gastric cancer: a real-world study by Yukiya Narita, Ryosuke Kumanishi, Koshi Takahashi, Toshiki Masuishi, Hiroya Taniguchi, Shigenori Kadowaki, Kazunori Honda, Masashi Ando, Tadahisa Fukui, Masahiro Tajika and Kei Muro in Therapeutic Advances in Gastroenterology
Footnotes
Appendix
Acknowledgements
Authors’ note
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
