Abstract
Background:
Nivolumab plus chemotherapy (NC) was recently approved as the first-line intervention for human epidermal growth factor receptor 2-negative advanced gastric/gastroesophageal junction cancer (GC/GEJC). Moreover, in the latest KEYNOTE-859 (NCT03675737), pembrolizumab plus chemotherapy (PC) was demonstrated to produce remarkable patient survival outcomes.
Objectives:
The clinicians and patients need to assess NC and PC preference for cancer drugs.
Design:
The cost-effective analysis.
Methods:
In an economic assessment of the United States, United Kingdom, and Chinese healthcare systems using a Markov model simulated patients with GC/GEJC, two treatment decision branches with three health states and a tracked time horizon of 15 years were developed. The overall cost and efficacy outcomes of first-line strategies PC and NC were evaluated at willingness-to-pay (WTP) thresholds of different national, including life-years (LYs), quality-adjusted life-years (QALYs), incremental cost-effectiveness ratios (ICERs), and incremental net-health benefit (INHB). Sensitivity and subgroup analyses were considered.
Results:
Given a WTP threshold of $150,000, $60,161, and $37,653 per QALY in the United States, United Kingdom, and China, respectively, both PC and NC achieved QALYs of 1.67 and 1.65 (2.51 and 2.48 LYs), 1.65 and 1.63 (2.48 and 2.45 LYs), and 1.60 and 1.58 (2.40 and 2.37 LYs), with total costs of $242,444 and $232,617, $148,367 and $127,737, and $16,693 and $24,016, respectively. Based on our sensitivity analysis, the programmed death-1 inhibitors cost produced the largest impact on the outcome. In addition, the cost-effectiveness probabilities of PC were 38.3%, 4.1%, and 100% in the three aforementioned countries, respectively.
Conclusion:
In the case of the Chinese payers’ perspective, PC appeared more dominant as first-line therapy for advanced GC/GEJC patients, whereas NC was preferred in the United States and United Kingdom.
Keywords
Introduction
Gastric cancer (GC) ranks fifth among all diagnosed cancers and is the fourth contributor to cancer-related mortality around the world. In 2020 alone, there were approximately 1.1 million new reported incidences, with over 768,000 deaths worldwide. Moreover, Asia, Europe, and the Americas accounted for close to 75%, 11%, and 9% of the total global deaths, respectively. 1 In over 50% of cases, GC diagnosis occurs after progression to an advanced stage. 2 Approximately 90–95% of GCs are adenocarcinomas, and 80% of patients with regionally advanced unresectable or metastatic GC or gastroesophageal junction cancer (GEJC) adenocarcinoma have human epidermal growth factor receptor 2-negative (HER2-negative) disease.3,4 The 5-year overall survival (OS) rate of GC patients at the advanced or metastatic stage is <5%. 5
In the past, systemic therapy for patients with unresectable advanced or recurrent metastatic HER2-negative gastric/gastroesophageal junction cancer (GC/GEJC) was dominated by cytotoxic agents, and the standard first-line therapy was fluorouracil and platinum-based chemotherapy worldwide.6–8 However, multiple extensive phase III clinical trials revealed that the aforementioned therapies yield unsatisfactory long-term OS benefits and worse outcomes in patients, with a median OS (mOS) and median progression-free survival (mPFS) of 8.8–11.0 months and 3.9–7.2 months, respectively.9–12 Given this evidence, the search for novel therapeutic options to prolong OS and manage adverse events (AEs) cannot be neglected.
More recently, the immune checkpoint inhibitors (ICIs) that suppress the programmed death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) axis were shown to achieve remarkable OS benefits among GC/GEJC patients, which, in turn, revolutionized the therapy of numerous cancer. Nivolumab (Opdivo®), a fully human IgG4 monoclonal antibody, that targets PD-1 was one of the first introduced ICIs. Based on the CheckMate-649 trial (NCT02872116), nivolumab with chemotherapy (NC) as the first-line intervention for advanced GC/GEJC patients markedly enhanced patient mOS [13.8 months versus 11.6 months; hazard ratio (HR), 0.71; 98.4% confidence interval (CI): 0.59–0.86; p < 0.0001] and mPFS (7.7 months versus 6.9 months; HR, 0.68; 98% CI: 0.56–0.81; p < 0.0001) with considerably reduced grade 3–4 treatment-associated AEs [462/789 (59%)], relative to chemotherapy. Owing to these excellent clinical outcomes, in 2021, the United States Food and Drug Administration, National Medical Products Administration, and international guidelines recommended this regimen for GC/GEJC patients.5,6,13,14 Subsequently, a 3-year follow-up data were provided at the American Society of Clinical Oncology meeting in 2023, which demonstrated that NC produces marked OS (HR, 0.79; 95% CI: 0.71–0.88) and PFS (HR, 0.79; 95% CI: 0.71–0.89) benefits, relative to chemotherapy alone. 15 Similarly, at a European Society for Medical Oncology (ESMO) meeting, another extensive phase III global trial, KEYNOTE-859 (NCT03675737) revealed that pembrolizumab (KEYTRUDA®, human immunoglobulin G4 monoclonal antibody anti-PD-1) with chemotherapy (PC) also enhances patient mOS (12.9 months versus 11.5 months; HR, 0.78; 95% CI: 0.70–0.87; p < 0.0001) and mPFS (6.9 months versus 5.6 months; HR, 0.76; 95% CI: 0.67–0.85; p < 0.0001), relative to chemotherapy alone. 16 Given this evidence, PC may be the next recommended PD-1 inhibitor, based on international guidelines, and it may be widely employed in clinics.
With advancements in immunotherapy, associated clinical trials have shown great promise. However, the ultimate clinical application and guideline approval requires additional consideration of treatment-related costs to accommodate a larger proportion of eligible patients. Thus, the economic evaluation of both first-line immunotherapy regimens for advanced GC/GEJC patients is critical. The cost-effective analysis is crucial for the assessment of the balance between treatment efficacy and expenditure. Herein, our objective was to evaluate the cost-effectiveness of PC versus NC as the first-line intervention for unresected HER2-negative advanced GC/GEJC patients in China, the United States, the United Kingdom, as representative countries from Asia, America, and Europe, and to provide a reference for health insurance decisions, and rational clinical usage of drugs in varying countries.
Materials and methods
This investigation and reporting strictly followed the guidelines of the Consolidated Health Economic Evaluation Reporting Standards, established by the International Society for Pharmacoeconomics and Outcomes Research 17 (Supplemental Table S1).
Study design
The analyzed cohorts and interventions were obtained from the CheckMate-649 and KEYNOTE-859 trials.15,16,18 Despite examining distinct randomized populations, the demographics and clinical features of patients were generally similar, thus allowing a cost-effective analysis to achieve post hoc comparison (Supplemental Table S2). Based on the two trial protocols, overall, 1579 GC/GEJC patients were recruited for analysis, among which, 790 received PC (200 mg pembrolizumab intravenously) and 789 received NC (360 mg nivolumab intravenously). Moreover, both groups used XELOX (Capecitabine, 1000 mg/m2 orally twice daily on days 1–14; Oxaliplatin, 130 mg/m2 intravenously for a maximum of six cycles). The ICIs were continued for a maximum of 2 years, and all regimens were administered every 3 weeks15,16,18 (Supplemental Table S3). All patients were imaged every 6 weeks for progressive disease (PD) detection. Upon PD identification, 204 (26.4%) PC and 331 (42.0%) NC patients received second-line treatment with paclitaxel alone, based on recommendations from various clinical trials and guidelines, including the National Comprehensive Cancer Network (NCCN) and the Chinese Society of Clinical Oncology (CSCO) guidelines.6,14–16 The rest of the patients underwent optimal best supportive care (BSC) and dying patients received terminal care. The average body surface area of patients from the United States, United Kingdom, and China were 1.84, 1.72, and 1.88 m2, respectively19–21 (Table 1).
Key clinical and health preference data.

AEs, adverse events; NC, nivolumab plus chemotherapy; ND, not determined; PC, pembrolizumab plus chemotherapy.
Decision models
Three decision analysis Markov model examining three mutually exclusive health states (PFS, PD, and death) was established to completely simulate the 15-year horizon of the disease developmental process of advanced GC/GEJC patients, at which time more than 99% of patients transition to a state of death, with a cycle of 6 weeks, according to the dosed regimen and follow-up regimen (Supplemental Figure S1). We retrieved OS curve data from the PC and NC cohorts from both trials to reconstruct individual OS data and compute time-dependent transition probability (Tp).15,16 The reconstructed data were then fitted and extrapolated with exponential, log-logistic, log-normal, Gompertz, and Weibull functions. Subsequently, using the Akaike information criteria and Bayesian information criteria, in combination with a visual test (Supplemental Figure S2 and Table S4), we selected the survival function with the best Weibull fitting, and two main parameters, namely, scale (λ) and shape (γ) were calculated.
22
The employed Tp (PD and death) calculation formula was as follows:
Owing to a lack of a ‘head-to-head’ comparison between PC and NC, we performed an indirect comparison using fixed-effects meta-regression models for frequency network meta-analysis to yield HRs of OS and PFS of PC and NC. 23 Considering that NC was the control and PC was the experimental cohort, we converted the corresponding parameters based on the Hoyle et al. method, and the employed formula is as follows: γpembrolizumab group = γnivolumab group and λpembrolizumab group = λnivolumab group * HR. 24 Subsequently, model building, data point extraction, and parameter computation employed TreeAge software (version TreeAge Pro 2022, available at: https://www.treeage.com), GetData Graph Digitizer (version 2.26, available at: http://www.getdata-graph-digitizer.com/index.php), and RStudio (version 1.2.5042, available at: https://www.rstudio.com) with the package ‘netmeta’, respectively 25 (Table 1).
Effectiveness and cost
We calculated and summarized the total cost between treatments, lics (LYs), quality-adjusted life years (QALYs), incremental cost-effectiveness ratio (ICERs), and incremental net-health benefit (INHB) at different willingness-to-pay (WTP) threshold of $150,000/QALY, $60,161/QALY (£50,000/QALY), and $37,653/QALY (three times the Chinese GDP per capita in 2021) of the United States, United Kingdom, and China, respectively.19,26 Based on our observations, the United States, United Kingdom, and China annual discount rates were 3%, 3.5%, and 5% for key outcomes, respectively. 27
The utility was used to describe the patient’s quality of life (QoL) throughout a natural disease, and the values ranged from 0 (death) to 1 (perfect health status). Regrettably, patient utility values were not reported in the CheckMate-649 and KEYNOTE-859 trials. Hence, we extracted utility values from a cost-effective investigation of ICI regimen in GC/GEJC patients in this model, and the utility values of PFS and PD state were 0.797 and 0.577, respectively. 20 The AE-related disutility values were acquired from published literature 28 (Table 1).
We incorporated direct costs, such as that of drugs, imaging, laboratory tests, administration, severe AEs (grade 3 or higher with incidence rates ⩾5%)-related treatments, BSC, and terminal care (Table 2). In the meantime, the post-progressive and other routine care (office visits, microsatellite instability-high [MSI-H], PD-L1 tests, and follow-up) costs were comparable between the two groups and were not considered. Drug prices were derived from local related websites within the United States, United Kingdom, and Xiangya Hospital of Central South University.29–31 Other expenses were extracted from previously published articles.19,20,32–37 Subsequently, using the consumer price indices for the United States and United Kingdom, we adjusted the relevant medical service-related costs for inflation in January 2023. 38 Chinese healthcare was not affected by inflation, but the patient assistance programs and health insurance policies were severely impacted. The model costs were converted to US dollars [$1 = ¥6.8601 and $1 = £0.8311 (February 2023)] (Table 2).
Cost estimates.

The costs of drugs were estimated based on the price of Xiangya Hospital Central South University, 2023.
Sensitivity and subgroup analyses
Uncertainty was assessed using sensitivity analysis. We performed a one-way sensitivity analysis for over 30 key model parameters, using a parameter range of ±20% of the baseline value. 22 The first 11 parameters that influenced the order of results are displayed in the tornado diagram. We next conducted a probabilistic sensitivity analysis using 10,000 Monte Carlo simulations to represent the cost-effectiveness probability between the two treatment cohorts with WTP values of various countries, displayed by acceptability curves and scatter plots. 25
In all, we performed eight major subgroup patient analyses. First, we indirectly compared the HRs of subgroups, namely, PC or NC versus chemotherapy, to obtain OS HRs for each cohort. There was a lack of sufficient data for subgroup analysis given the consistency with the general population, particularly in the HRs of PFS (PC versus NC) in the subgroup (0.962, 95% CI: 0.817–1.134).15,16,22 The HRs of individual subgroups of OS or PFS were then incorporated into the model for cost-effective analysis from the aforementioned three national healthcare system perspectives.
Results
Base-case analysis
The estimated total costs over 15 years for the aforementioned treatment options were $242,444, $148,367, and $16,693 for PC in the United States, United Kingdom, and China; as well as $232,617, $127,737, and $24,016 for NC. In the United States, United Kingdom, and China, the QALYs (LYs) were 1.67 (2.51), 1.65 (2.48), and 1.60 (2.40) for PC; and 1.65 (2.48), 1.63 (2.45), and 1.58 (2.37) for NC. The resulting ICERs (PC versus NC) revealed that PC was more cost-effective at the prespecified WTP threshold of China (ICER, −$333,696 per QALY). Alternately, NC was more cost-effective in the United States and United Kingdom (ICER, $413,706 and $886,326 per QALY). In addition, INHB from PC was −0.04, −0.32, and 0.22, relative to NC in the United States, United Kingdom, and China, respectively (Table 3).
Results of the base-case.

Pembrolizumab plus chemotherapy showed higher effectiveness and lower cost, as compared with nivolumab plus chemotherapy.
ICER, incremental cost-effectiveness ratio; INHB, incremental net-health benefits; LYs, life-years; NA, not applicable; QALYs, quality-adjusted life-years.
Sensitivity analysis
Using the tornado diagram (Figure 1), we revealed that the model parameter with the largest impact on outcome was the cost of ICIs (range $1045–$25,921, ICER range −$810,991/QALY to $1,638,405/QALY). The PFS utility and AEs cost also had considerable influence on the model. The HR of OS (PC versus NC), as well as the administration, imaging, and laboratory costs had less impact on the paired results. No matter how the input model parameters varied in their range, the ICER value for each country did not change.
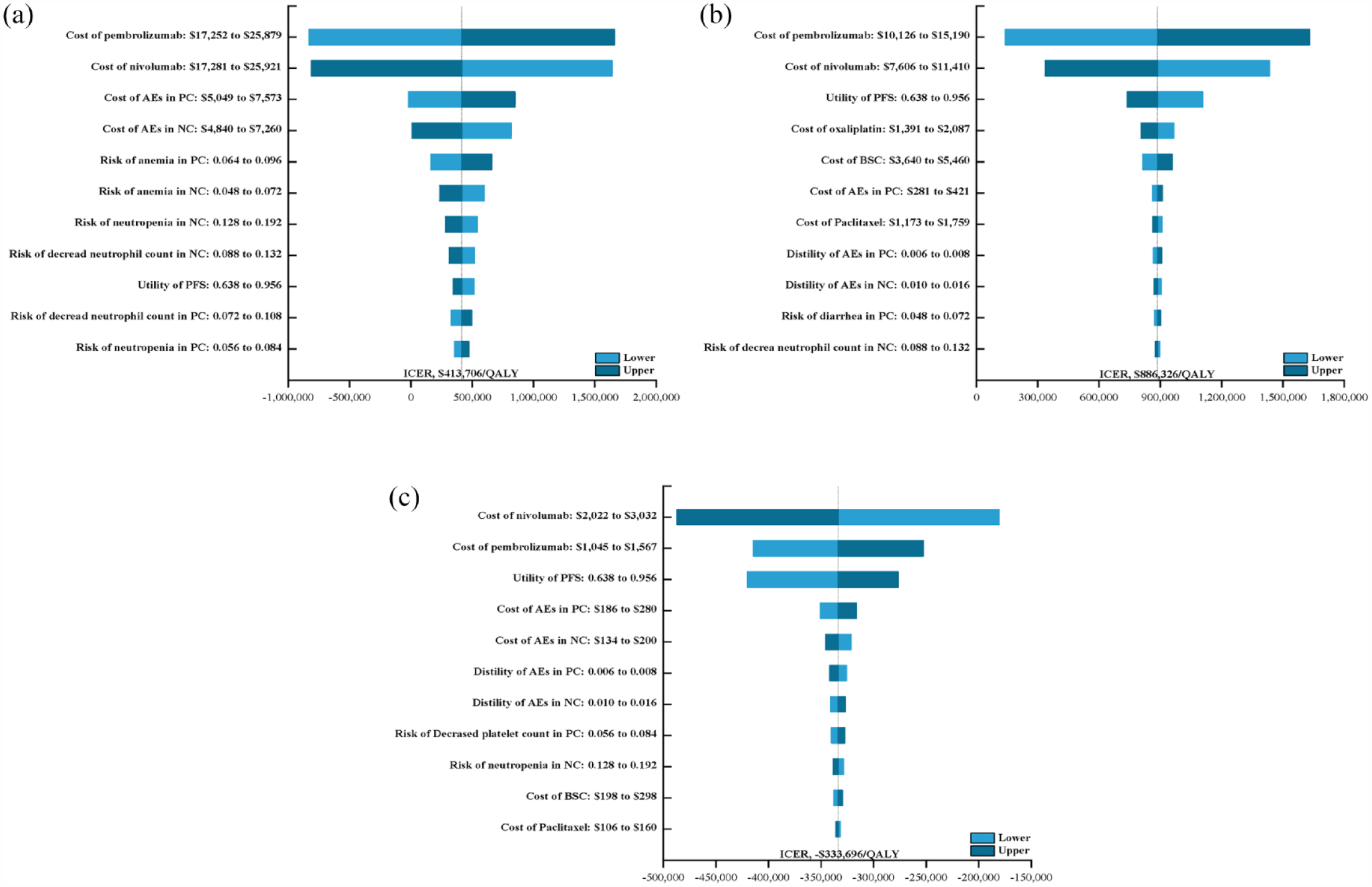
The one-way sensitivity analyses for pembrolizumab plus chemotherapy strategy compared to nivolumab plus chemotherapy strategy in the United States (a), the United Kingdom (b), and China (c).
Based on our probability sensitivity analysis, for the total patient population, the probabilities of NC cost-effectiveness, concerning PC cost-effectiveness, were 61.2% and 95.9% at the WTP threshold of $15,000 and $60,161 per QALY in the United States and United Kingdom, respectively. However, the odds of PC being cost-effective about NC was 100% at the WTP threshold of $37,653 per QALY in China (Figure 2 and Supplemental Figure S3). Moreover, the probability of cost-effectiveness of PC was greater than 50% at the WTP threshold of 40,000 and 850,000 per QALY in the United States and United Kingdom, respectively (Figure 2).

The cost-effectiveness acceptability curves for pembrolizumab plus chemotherapy strategy compared to nivolumab plus chemotherapy strategy in the United States (a), the United Kingdom (b), and China (c).
Subgroup analysis
In a majority of Chinese subgroups, PC was superior in reducing mortality risk, and the ICER of PC versus NC ranged between −$1,557,940 and $591,768 per QALY with a 63.1–99.9% probability of cost-effectiveness. Solely for the subgroup of patients who had PD-L1 expression, NC was superior to pembrolizumab plus chemotherapy (PC) in improving OS. However, NC was more cost-effective in all United Kingdom patient subgroups. In the United States, the cost-effectiveness of PC and NC patient subgroups was comparable (Supplemental Table S5).
Discussion
With increasing GC/GEJC incidences, new diagnostic techniques and therapeutic options are frequently proposed, which leads to a considerable rise in GC/GEJC-related medical expenditures. 39 The annual medical cost for GC/GEJC patients is predicted to be around $3672 million, $7498 million, and $9099 million in North America, Europe, and Asia, respectively. 39 Considering the augmented economic burden of the cancer healthcare system in various countries, a straightforward and theory-based economic assessment is both urgent and necessary. NC was recently approved for first-line therapy of advanced GC/GEJC patients. However, a 3-year follow-up revealed that PC also achieves remarkable positive OS outcomes. Given these options, physicians and patients at present face a dilemma in determining the optimal approach for personalized care.15,16,18
To our knowledge, there are no studies that indirectly compared the cost-effectiveness of ICIs from multiple healthcare systems around the world. The available studies have all examined immunotherapy versus chemotherapy. In two studies by Jiang and Shu, they evaluated NC with chemotherapy in treating Chinese HER2-negative advanced GC/GEJC patients. They revealed ICERs of $191,266 and $278,658.71 per QALY, respectively.20,37 Second, Kashiwa reported that NC or nivolumab alone contrasts chemotherapy produced a cost-effective or non-cost-effective comparison with chemotherapy in the Japanese healthcare system. 40 In addition, the pharmacoeconomic evaluation by Giuliani based on the CheckMate 649 and KEYMAT-062 trials found that pembrolizumab monotherapy for GC/GEJC first-line treatment in patients with PD-L1 CPS of 10 or higher could be considered cost-effective in Italy. 41 The differences between their results and ours are explained as follows: First, their study compared a single ICI baseline treatment with standard treatment. Second, based on the short-term data from the CheckMate-649, ATTRACTION-4, and ATTRACTION-2 trials, their model cannot fully represent real-world patients. Third, the serious AEs (SAEs)-related adverse utility was not described, and the aforementioned utility is known to have a strong correlation with QoL. Fourth, there is no known patient stratification, based on economic assessments. Therefore, economic information based on patient subgroups may be beneficial in forging treatment decisions. Together, these deficiencies prompted us to conduct the first cost-effective analysis of NC and PC as first-line therapy for advanced GC/GEJC patients from the United States, United Kingdom, and Chinese healthcare sectors.
Herein, we estimated an average survival improvement of 0.03 LYs and 0.02 QALYs per patient, together with average additional costs of $9826 and $20,630 when comparing PC to NC in the United States and United Kingdom. The resulting ICER was $413,706 and $886,326 per QALY gained, suggesting that NC was far more cost-effective in America at a cutoff of $150,000 and $60,161 per QALY. The additional costs related to PC primarily originated from AEs management and BSC, which were related to the extension of OS duration and improvement of QoL. However, PC provided the same benefit and a $7327 reduction, thereby becoming a dominant strategy at a WTP cutoff of $37,653/QALY. This is potentially due to the differences in pricing of the same treatment in multiple national settings, as well as healthcare decisions. Sensitivity analysis revealed that the costs of nivolumab and pembrolizumab were the most significant influencers on patient outcomes. If the cost of pembrolizumab were to be reduced to $50 or $24 per mg, and the cost of nivolumab was to be increased to $33 or $18 per mg, then PC would be more cost-effective in the United States and the United Kingdom at WTP threshold of $150,000/QALY and $60,161/QALY, respectively. Based on our observation, NC was cost-effective for the Chinese population at a WTP cutoff of $37,653/QALY, when the cost of pembrolizumab was increased to over $6 per mg or nivolumab dropped to $7 per mg. Given this evidence, if the therapeutic strategies with high total cost were reduced by 5%, 14%, and 31% in the United States, United Kingdom, and China, respectively, then the ICER would approach zero, suggesting that the total NC and PC costs would be almost the same. This finding suggests that even though immunotherapy holds great promise in clinical practice, the selection of ICIs in different countries, based on economic conditions, remains a matter of significant consideration. Consequently, price adjustment is an appropriate and realistic approach to enhance the ICIs-based treatment cost-effectiveness, as well as to balance the affordability of patients and ease the financial burden on healthcare systems in various countries.
More importantly, based on our subgroup analysis, the cost-effectiveness of ⩾1% PD-L1 expression patients who received NC was more cost-effective than patients with PD-L1 expression <1% in the United States and United Kingdom. Alternately, the opposite was true for Chinese patients. This result was similar to a prior report, phase III ATTRACTION-2, which examined nivolumab monotherapy among an Asian population. The objective response rate (ORR) was 11%, the 12-month OS rate was increased to 27%, and the survival benefit was independent of PD-L1 expression 42 ; In the CheckMate-032 trial involving western patients, the ORR rate of PD-L1-positive tumors was markedly enhanced, compared to negative tumors (27% versus 12%). 42 It was further concluded that the PD-L1 content in tumor-infiltrating immune cells was a strong indicator of good prognosis among non-Asian patients but not among Asian patients. 43 Hence, assessing the immunotherapeutic impact on Asian and non-Asian GC/GEJC patients is an important consideration. Therefore, clinicians and program makers in different countries and regions must pay extra attention to predictive markers when considering personalized care for limited potential populations. In doing so, they will be able to better assess whether innovative treatment regimens yield clinical benefits at a reasonable cost, which, in turn, is crucial to the expansion of their use and sustainability within a given healthcare system.
Several advantages deserve to be highlighted. First, our analyses simulated the long-term data collection of clinical trials with follow-up over 3 years. This improved the robustness of OS estimates by the model. Second, we considered the incidence and cost of grade ⩾3 AEs, which were closely associated with QoL, and we employed the disutility values to correct for average utility values. As a result, our analyses more accurately represented the effectiveness of the analyzed treatments. Third, we examined the relative cost-effectiveness of the PC and NC regimens within a range of patient subgroups, thereby providing valuable clinical references. Finally, our evaluations were based on the perspective of health systems from multiple countries, including North America, Europe, and East Asia, which were represented by the United States, the United Kingdom, and China. Therefore, our conclusions will not only apply to a single country but also be generalized to multiple healthcare systems.
The study also has certain limitations. First, in the absence of a ‘head-to-head’ comparison, we relied on cross-trial comparisons between two randomized clinical trials using an indirect comparison method. We did not assess heterogeneity between trials, however, the study populations within these two trials exhibited similar characteristics. Therefore, trial heterogeneity was minimized by calculating the corresponding HR, the methods used are all from previously published articles.28,44–46 Second, we inferred long-term clinical OS beyond the follow-up time from K-M curves from the CheckMate-649 (follow-up of 36.2 months) and KEYNOTE-859 (follow-up of 46.3 months) trials. This may be slightly different from the real data. However, owing to the good fitness of the model, model uncertainty for long-term OS was small. Third, according to the guidelines, we assumed that all patients progressed to receive paclitaxel as the second-line treatment. This may not accurately reflect the national clinical practice as physicians may individualize treatment options based on patient conditions. However, upon further analysis, we observed that the cost of second-line treatment did not alter the patient outcome. Fourth, only SAEs with an incidence greater than 5% were included in the cost-effectiveness analysis. This may have underestimated the direct medical cost. However, the unitary sensitivity analysis revealed that this would not affect model stability. Finally, since the two analyzed trials did not report the PFS and PD utility values in detail, our reference to previously published articles may be somewhat different from the actual national GC/GEJC patients. Meanwhile, our sensitivity analysis revealed almost no effect.
Conclusion
In conclusion, in terms of the Chinese healthcare system, PC achieved better survival benefits and was more economical as a first-line treatment for HER2-negative advanced GC/GEJC patients at current prices. By contrast, NC was more cost-effective for patients within the US or UK healthcare system. Therefore, altering the current economic outcome may require an adjustment in the cost of both ICIs. Hence, real-world investigations are necessary to further validate these differences and provide an objective reference for the pricing of healthcare policies in various countries, as well as the updating of international guidelines.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848231207200 – Supplemental material for Immune checkpoint inhibitors plus chemotherapy for HER2-negative advanced gastric/gastroesophageal junction cancer: a cost-effectiveness analysis
Supplemental material, sj-pdf-1-tag-10.1177_17562848231207200 for Immune checkpoint inhibitors plus chemotherapy for HER2-negative advanced gastric/gastroesophageal junction cancer: a cost-effectiveness analysis by Youwen Zhu, Kun Liu, Hong Zhu and Haijun Wu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
