Abstract
Background:
Malnutrition is common in patients with pancreatic head cancer, which can diminish their tolerance to anti-cancer treatments. Pancreatic duct blockage is one of the contributing factors leading to malnutrition.
Objectives:
This study aimed to clarify whether pancreatic stenting offers a survival benefit in patients with unresected pancreatic head cancer.
Design:
Retrospective observational study.
Methods:
Patients with newly diagnosed pancreatic cancers who received endoscopic retrograde cholangiopancreatography (ERCP) because of biliary tract obstruction were collected from a hospital-based cancer registry between 2007 and 2021. Individuals who underwent tumor resection or bypass surgery following ERCP were excluded. The primary outcome was the 1-year survival rate in patients with and without a pancreatic stent. Secondary outcomes included overall survival, cancer-specific survival, and changes in weight and nutritional markers following ERCP.
Results:
Among 562 patients, 39 (6.9%) received pancreatic stent placement. Most of the patients (90%) did not receive tumor resection because of advanced-stage disease. The stent group had a lower 1-year survival rate than the non-stent group (18% vs 30%; p = 0.047). The median survival time appeared to be shorter in the stent group (170 vs 217 days; p = 0.077). The adjusted hazard ratio of pancreatic stent placement was 1.42 (95% confidence interval, 0.75–2.67; p = 0.282). In addition, changes in weight and nutritional markers after ERCP did not differ significantly between the groups.
Conclusion:
Pancreatic stent placement in unresected pancreatic head cancer with obstructive jaundice does not improve survival or nutritional status.
Plain language summary
Malnutrition is a common issue among pancreatic cancer patients, hindering their anti-cancer treatment and leading to poor outcomes. One hypothesized cause of malnutrition is pancreatic exocrine insufficiency (PEI) due to pancreatic duct blockage by tumors. Therefore, we aimed to explore whether pancreatic stent placement could offer a survival benefit by improving nutritional status. We enrolled patients with pancreatic cancer and obstructive jaundice who underwent endoscopic retrograde cholangiopancreatography (ERCP) for biliary stenting from a hospital-based cancer registry. We compared the survival rates between patients with and without pancreatic stent placement during ERCP. However, our results indicated that pancreatic stent placement does not improve survival or nutritional status. Accordingly, pancreatic stents should be removed after ERCP as scheduled, even in pancreatic cancer patients with imaging-confirmed pancreatic ductal obstruction.
Keywords
Introduction
Pancreatic cancer is a highly lethal disease, with a 5-year overall survival rate of less than 5%. 1 The global incidence of pancreatic cancer is on the rise, making it the seventh leading cause of cancer-related deaths worldwide. 2 Approximately three-fourths of newly diagnosed pancreatic cancer cases are unresectable due to proximity to major blood vessels or the presence of distant metastases, leaving chemotherapy as the primary treatment modality. 1 However, the efficacy and tolerability of chemotherapy are often compromised by malnutrition, which is highly prevalent among pancreatic cancer patients and is strongly associated with poor prognosis.3–8 Pancreatic exocrine insufficiency (PEI) caused by pancreatic duct obstruction is recognized as a major contributor to malnutrition, particularly in patients with tumors located in the pancreatic head.9–11 In this context, we aim to evaluate whether relieving pancreatic duct obstruction through pancreatic stent placement can improve both survival outcomes and nutritional status in patients with unresected pancreatic head cancer.
Methods
Patients
This retrospective study was conducted at a single referral medical center. Patients with newly diagnosed pancreatic cancer were identified through the hospital’s cancer registry system between 2007 and 2021 using ICD-9 and ICD-10 codes for pancreatic cancer (157.0, 157.1, 157.2, 157.8, C25.0, C25.1, C25.2, C25.7, C25.8, and C25.9). Patients were included in the study if they met the following criteria: (1) pathology-confirmed pancreatic adenocarcinoma; (2) tumor located at the pancreatic head causing obstructive jaundice; (3) underwent endoscopic retrograde cholangiopancreatography (ERCP) for biliary stent and/or pancreatic stent placement; and (4) had not undergone pancreatic tumor resection or double bypass surgery (gastroenterostomy and hepaticojejunostomy) after ERCP. The exclusion criteria were as follows: (1) age under 18 years; (2) prior pancreatic stent placement before the diagnosis of pancreatic cancer (e.g., for pain management in chronic pancreatitis); and (3) inability to determine the status of pancreatic stent placement due to incomplete medical records. Patients were stratified based on the placement of a pancreatic stent during the index ERCP, regardless of whether biliary drainage was ultimately achieved via ERCP or salvage techniques (such as percutaneous or endoscopic ultrasound-guided biliary drainage). This study adhered to the principles outlined in the Declaration of Helsinki. The findings were reported in accordance with the STROBE reporting guidelines for observational studies. 12
Data collection
Baseline patient characteristics, including age, sex, height, weight at diagnosis and subsequent changes, laboratory test results, and prophylactic treatments for post-ERCP pancreatitis (PEP) prevention, were retrieved through chart review. Information on cancer treatments—such as performance status, comorbidities, cancer stage according to the 8th edition of the American Joint Committee on Cancer staging system, 13 number and location of metastases, treatment modalities, treatment responses, and adverse events—was obtained from the hospital’s cancer registry system. This registry is a prospectively maintained database that includes all patients diagnosed with or treated for malignant diseases within the hospital. Survival data, including survival status, date of death, and cause of death, were retrieved from Taiwan’s National Health Insurance Research Database, a comprehensive dataset established in 2002 that covers 99.99% of Taiwan’s population. 14 The decision to place a pancreatic stent during ERCP was made at the discretion of the endoscopists. Pancreatic stents were usually placed for PEP prevention following incidental pancreatic duct cannulation or to facilitate bile duct cannulation in challenging cases. Details regarding the pancreatic stents, including caliber and length, were extracted from endoscopic reports.
Outcome measurements and definition
The primary outcome of this study was the 1-year overall survival rate following pancreatic cancer diagnosis. Secondary outcomes included 1-year cancer-specific survival, overall survival, changes in weight and nutritional markers (serum albumin levels, lymphocyte counts, and prognostic nutritional index (PNI)) at 1, 3, and 6 months after ERCP, and the incidence of PEP. The PNI was calculated using the following formula: 10 × albumin (g/dL) + 0.005 × total lymphocyte count (/μL). PEP was defined as new or worsening abdominal pain combined with amylase or lipase levels exceeding three times the upper limit of normal, persisting for more than 24 h after ERCP. 15 The severity of PEP was assessed using the BISAP score (range: 0–5), with higher scores indicating greater predicted mortality. 16
Statistical analysis
Dichotomous data were presented as counts and percentages, while continuous variables were expressed as medians with interquartile ranges (IQRs) for non-normally distributed data or as means with standard deviations for normally distributed data. Between-group differences were assessed using the χ2 test or Fisher’s exact test for dichotomous data, and the Mann–Whitney U test (for non-normally distributed data) or the unpaired Student’s t test (for normally distributed data). Potential factors associated with survival were analyzed using a univariable Cox proportional hazards model. Variables with a p-value of less than 0.2 in the univariable analysis, along with factors deemed clinically relevant to survival (e.g., cancer stage, treatment status), were included as covariates in a multivariable Cox proportional hazards model to assess the impact of pancreatic stent placement on survival. Hazard ratios (HRs) were calculated with their corresponding 95% confidence intervals (CIs). Kaplan–Meier curves were constructed to compare survival outcomes between the stent and non-stent groups, and unadjusted differences were evaluated using log-rank tests. A sensitivity analysis excluding patients with PEP was planned, as this complication could disrupt cancer treatment or contribute to mortality. In addition, patients with resectable disease would also be excluded to confirm the robustness of the conclusions. All data analyses were performed using STATA BE version 17 software (StataCorp LP, College Station, TX, USA) and Python (version 3.8.2) with the matplotlib package. A two-sided p-value of less than 0.05 was considered statistically significant for all tests.
Results
Patient characteristics
A total of 3604 patients with pancreatic cancer were identified from the cancer registry system during the study period. Of these, 3042 patients were excluded based on predefined criteria: 2653 did not undergo ERCP; 234 underwent surgery following ERCP; 126 lacked pathology-confirmed diagnoses; 26 had pancreatic malignancies other than adenocarcinoma; 2 had missing endoscopic information; and 1 had a pancreatic stent placed prior to the diagnosis of pancreatic cancer due to chronic pancreatitis. Consequently, 562 patients were eventually enrolled in the study (Figure 1). Pancreatic stents were placed in 39 (6.9%) of the 562 patients during ERCP. The most common indication was difficult bile duct access and repeated pancreatic duct cannulation (37 patients, 93%). Pancreatic stents were placed for pain control in one patient and for pancreatitis prevention during deployment of a fully covered biliary metal stent in one patient, respectively. The baseline characteristics were generally balanced between the stent and non-stent groups, except that the non-stent group had larger tumor sizes (median: 37 vs 44 mm; p = 0.020; Table 1). Overall, 90% of the patients did not undergo surgery due to locally advanced or metastatic disease. Following diagnosis, 332 patients (59%) received cancer treatment, primarily chemotherapy. Among these, gemcitabine-based first-line therapy was administered to 90% of the cohort, with no significant difference observed between the stent and non-stent groups (100% vs 89%; p = 0.381). The median follow-up duration of our cohort was 213 days (IQR, 114–403). During follow-up, 95 patients (17%) developed gastric outlet obstruction, necessitating duodenal stenting or endoscopic gastroenterostomy bypass. Pancreatic enzyme replacement therapy (PERT) was administered to approximately half of the study population, with a median dose of 3.5 tablets per day (each tablet contained 20,000 units of lipase, 75,000 units of protease, and 66,400 units of amylase). The median duration of PERT was 100 days, and treatment was initiated at a median of 43 days after the diagnosis of pancreatic cancer (Table 1).

Study flowchart.
Baseline characteristics and prognostic factors between pancreatic cancer patients with and without pancreatic stenting.

Values are n (%) or median (interquartile range).
The normal range for CA 19-9 was 0–37 IU/mL.
Data available in 425 patients.
Data available in 195 patients.
Each tablet contains lipase 20,000 units, protease 75,000 units, and amylase 66,400 units.
AJCC, American Joint Committee on Cancer; BMI, body mass index; CA 19-9, carbohydrate antigen 19-9; ECOG, Eastern Cooperative Oncology Group; PERT, pancreatic enzyme replacement therapy.
Survival outcomes
The 1-year overall survival rate was lower in patients with a pancreatic stent compared to those without a stent (18% vs 30%; p = 0.047; Figure 2). The median survival in the pancreatic stent group was 170 days (IQR, 95–306), shorter than the 217 days (IQR, 118–414) observed in the non-stent group, although this difference was not statistically significant (p = 0.077; Figure 3). The unadjusted HR for pancreatic stent placement was 1.44 (95% CI, 1.00–2.07; p = 0.048). After adjusting for other prognostic factors identified in the univariable analysis—including PERT, age, Eastern Cooperative Oncology Group performance status, number of metastatic organs, chemotherapy, radiotherapy, body mass index, and carbohydrate antigen 19-9 levels—using a multivariable Cox proportional hazards model, the adjusted HR for pancreatic stent placement was 1.42 (95% CI, 0.75–2.67; p = 0.282; Table 2). Similarly, the 1-year cancer-specific survival was lower in the pancreatic stent group (18% vs 30%; p = 0.047; Supplemental Figure 1). The impact of pancreatic stents on survival remained unchanged after excluding patients with PEP or those with early-stage disease (Supplemental Figures 2 and 3). By contrast, PERT demonstrated a significant survival benefit in our study population (adjusted HR, 0.39; 95% CI, 0.26–0.59; p < 0.001; Table 2). In subgroup analysis, when patients were stratified based on PERT and pancreatic stenting, the potential adverse impact of pancreatic stent placement appeared to be mitigated by PERT. Within the PERT cohort, 1-year survival was comparable between patients with and without pancreatic stents. Although this comparison may be underpowered due to the small sample size, survival in both PERT subgroups remained significantly higher than in the non-PERT group. Notably, the subgroup consisting of patients with a pancreatic stent who did not receive PERT exhibited the poorest survival outcomes (Figure 4).
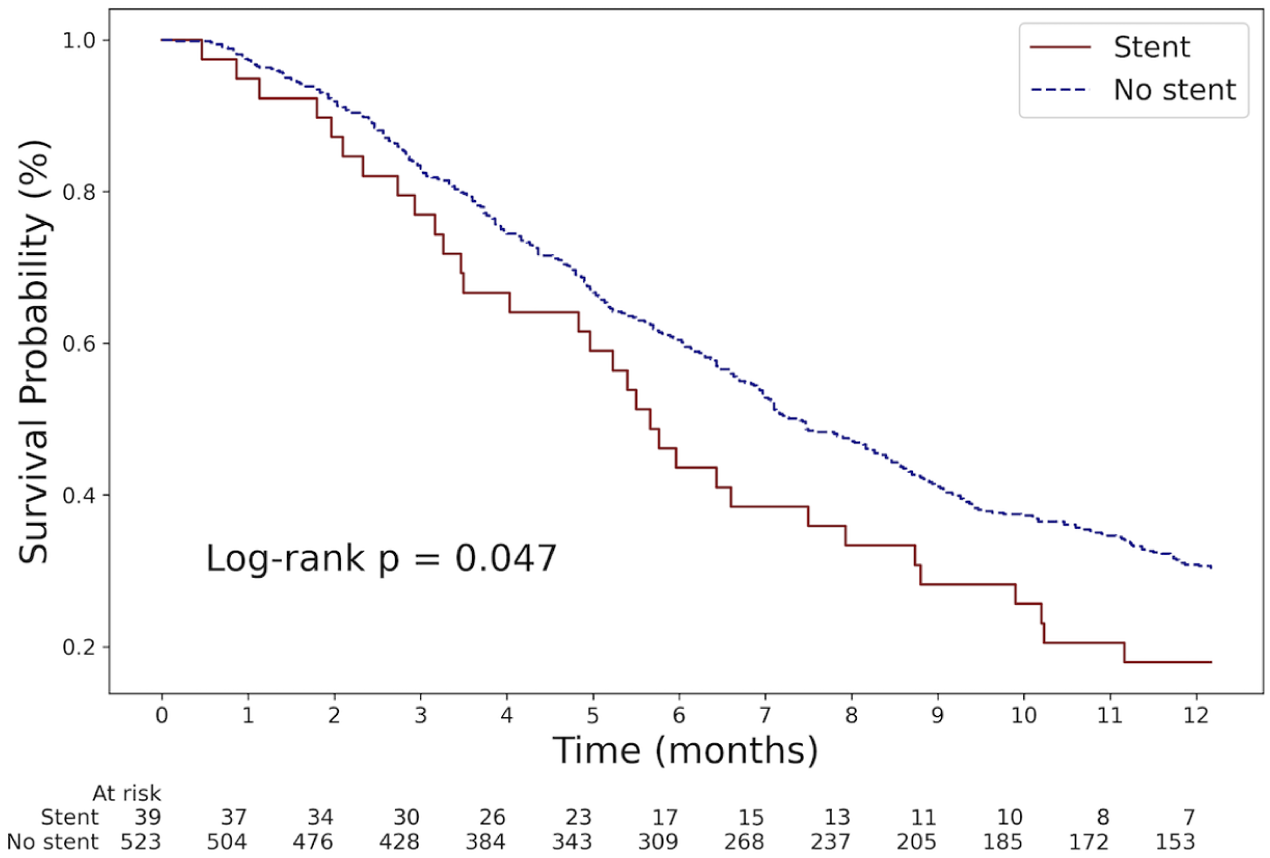
Kaplan–Meier curve for 1-year overall survival in pancreatic cancer patients with and without a pancreatic stent.

Kaplan–Meier curve for overall survival in pancreatic cancer patients with and without a pancreatic stent.
Baseline characteristics and prognostic factors for 1-year overall survival in univariable and multivariable Cox regression analyses.
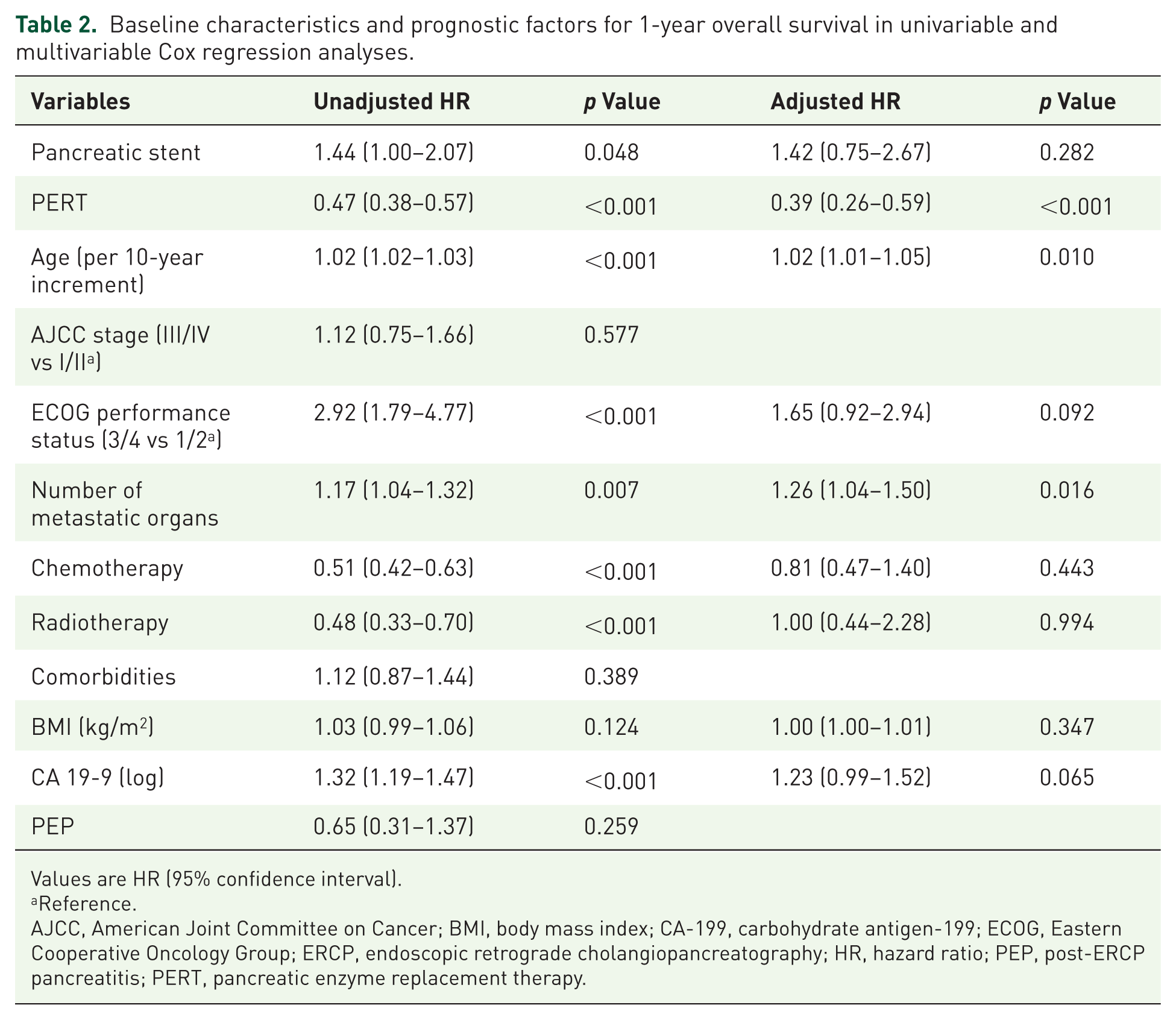
Values are HR (95% confidence interval).
Reference.
AJCC, American Joint Committee on Cancer; BMI, body mass index; CA-199, carbohydrate antigen-199; ECOG, Eastern Cooperative Oncology Group; ERCP, endoscopic retrograde cholangiopancreatography; HR, hazard ratio; PEP, post-ERCP pancreatitis; PERT, pancreatic enzyme replacement therapy.
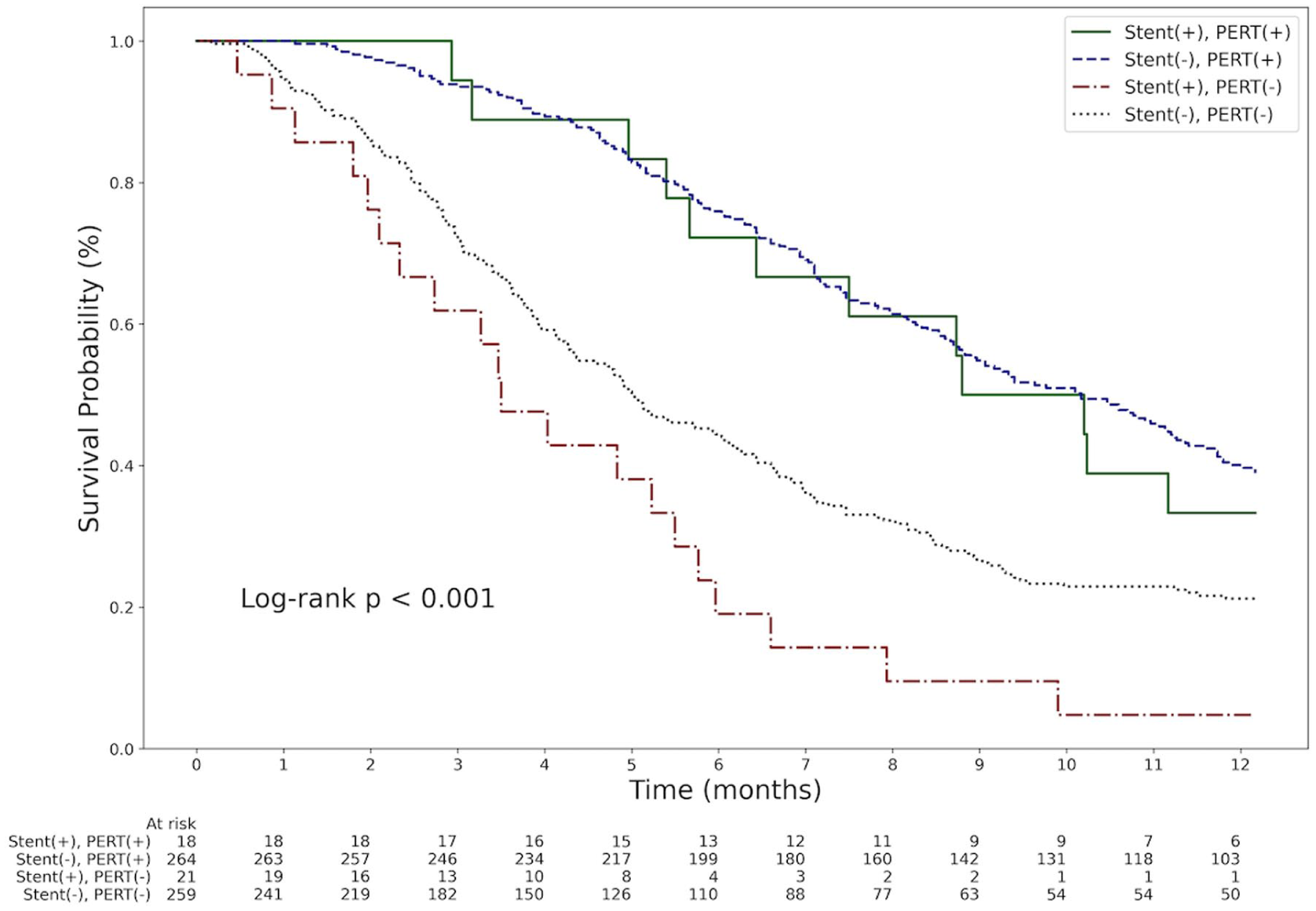
Kaplan–Meier curve for the interaction between pancreatic stent placement and PERT in 1-year overall survival.
Changes of weight and nutritional markers
One month after ERCP, patients with a pancreatic stent experienced no significant weight loss (median: 0 kg; IQR, −1.0 to 0.3), whereas patients without a pancreatic stent lost a median of 0.6 kg (IQR, −2.9 to 0.0; p = 0.048; Table 3). However, the difference in weight reduction between the groups was not significant at 3 and 6 months after ERCP. Similarly, changes in serum albumin level and lymphocyte count were comparable between the two groups (Table 3). Moreover, the median PNI of the study cohort before ERCP was 43 (IQR, 37–48), with no significant difference between the stent and non-stent groups (median: 42 (IQR, 39–48) vs 43 (IQR, 37–48); p = 0.726). Changes in PNI after ERCP were also comparable between the two groups at each time point (Table 3).
Changes in body weight and nutritional markers after ERCP.

Values are median (interquartile range).
ERCP, endoscopic retrograde cholangiopancreatography.
Detail of ERCP
Overall, the patients received ERCP on a median of 14 days (IQR, 3–67) after their cancer diagnosis. The median pancreatic duct diameter before stenting was 6.3 mm (IQR, 4.8–8.0), with no significant difference between the stent and non-stent groups (median: 5.7 vs 6.3 mm; p = 0.462). The most commonly used pancreatic stent size was 5 French in caliber and 7 cm in length. The overall incidence of PEP was 2.3%, which was higher in the stent group compared to the non-stent group, though the difference was not statistically significant (5.1% vs 2.1%; p = 0.227; Table 4). However, patients with a pancreatic stent tended to experience more severe PEP, as indicated by a significantly higher median BISAP score (2.5 vs 1; p = 0.045). Nevertheless, the interval between cancer diagnosis and the initiation of the first cancer treatment was not influenced by the incidence or severity of PEP (Table 4). Among the 32 patients who underwent follow-up imaging after pancreatic stent deployment, pancreatic stent dysfunction was observed in 12 patients (31%), and spontaneous stent dislodgement occurred in 9 patients (23%). Four pancreatic stents were removed: three during biliary stent revision because the pancreatic stents were visibly occluded, and one due to upper gastrointestinal bleeding that occurred 28 days after stent deployment. In the latter case, a 7-Fr, 10-cm straight pancreatic stent had been placed. Esophagogastroduodenoscopy revealed bleeding originating from the pancreatic stent, and stent-induced pancreatic duct injury with suspected hemosuccus pancreaticus was diagnosed. After stent removal, two patients subsequently underwent placement of new pancreatic stents.
Information of ERCP and PEP.

Values are N (%) or median (interquartile range).
ERCP, endoscopic retrograde cholangiopancreatography; NA, not applicable; NSAIDs, non-steroidal anti-inflammatory drugs; PEP, post-ERCP pancreatitis.
Discussion
Malnutrition is a common issue in patients with pancreatic cancer, particularly those with advanced-stage disease. More than 80% of patients experience significant weight loss prior to diagnosis.3,4 Poor nutritional status adversely affects their tolerability to surgery, chemotherapy, quality of life, and overall survival.5–8 Several mechanisms contribute to malnutrition in pancreatic cancer, including the Warburg effect, tumor-specific factors such as islet amyloid polypeptide that suppress appetite, and PEI. 9 A meta-analysis reported that the pooled prevalence of PEI in advanced pancreatic cancer is as high as 72% (95% CI, 55–86), despite variations in the diagnostic criteria across studies. 17 Tumors located at the pancreatic head are more likely to cause PEI compared to those in the body or tail (RR 3.36; 95% CI, 1.07–10.54).10,17 Pancreatic duct obstruction plays a critical role in the development of PEI, with additional potential contributors including fibrosis of the pancreatic parenchyma and loss of pancreatic exocrine tissue.9,18 PERT has demonstrated benefits for pancreatic cancer patients by improving nutritional status, stabilizing body weight, increasing tolerability to cancer treatment, and prolonging survival.19–21 However, the use of pancreatic enzymes is often cumbersome, as they need to be taken with each meal. Therefore, this study aims to evaluate whether relieving pancreatic duct obstruction with pancreatic stent placement can achieve similar improvements in survival and nutritional status.
In our current study, we demonstrated that pancreatic stent placement does not improve 1-year survival, overall survival, or nutritional status. On the contrary, it may have a potentially negative impact on 1-year survival. This result remained consistent even after excluding patients with PEP to eliminate the potential increase in morbidity and mortality associated with pancreatitis. We hypothesize that this adverse outcome is driven by stent-induced pancreatic ductal obstruction following occlusion, which subsequently aggravates PEI in pancreatic cancer patients. Pancreatic stent placement is a commonly employed technique during ERCP to facilitate biliary tract cannulation and prevent PEP.22,23 Small-caliber pancreatic stents (3–5 French) are recommended to avoid obstructing the bile duct orifice.22,23 In our daily practice, pancreatic stents were not removed on a scheduled basis. The median stent indwelling time in our study population was 87 days (IQR, 25–170). Consequently, small-caliber stents may become occluded over time, potentially blocking the pancreatic duct and exacerbating PEI. Follow-up imaging of our patients frequently revealed recurrent pancreatic duct dilatation after stent placement, indirectly supporting this hypothesis. In subgroup analysis, the potential adverse impact of pancreatic stents was mitigated by PERT, likely due to its ability to alleviate the aggravating impact of PEI caused by stent occlusion. Regarding nutritional outcomes, patients with a pancreatic stent exhibited a statistically significant, although clinically modest, less weight loss at 1 month after cancer diagnosis, possibly reflecting the short-term benefit of a patent stent. However, weight changes and nutritional markers did not differ significantly at 3 and 6 months after diagnosis, which may be attributed to stent occlusion occurring over time.
Previous studies have demonstrated the short-term safety and efficacy of pancreatic stents in pain control for pancreatic cancer patients, with technical success rates reported to range from 68% to 100%. 24 A 10 French plastic stent is generally preferred; however, 7 or 8.5 French stents may be used as alternatives when the pancreatic duct is too narrow for a 10 French stent. 25 In patients with an obstructive pain pattern (defined by post-prandial abdominal pain), more than two-thirds experienced significant pain relief following stent placement.24–26 The mean duration of response varies widely, ranging from 30 to 270 days. 24 Despite these findings, a robust relationship between pancreatic stent placement and pain control remains unproven due to the poor quality and limited sample sizes of these studies. The recently published guideline by the American Society for Gastrointestinal Endoscopy recommends celiac plexus neurolysis as a complementary modality for pain control rather than pancreatic stents, highlighting the limited evidence available to justify the use of stents for this indication. 27 Moreover, the long-term outcomes of pancreatic stent placement in pancreatic cancer patients have been largely overlooked. To the best of our knowledge, this is the first study to address this issue comprehensively. As discussed previously, pancreatic stent placement did not confer survival or nutritional benefits. Consistent with ESGE guidelines, 28 our study suggests that pancreatic stents should be considered for removal 5–10 days following ERCP to mitigate the risk of complications associated with prolonged stenting.
The strength of our study is that we used a national database to track patients’ survival status, thereby minimizing attrition bias. However, several limitations should be acknowledged. First, due to the retrospective design, unaddressed confounding factors might have influenced the decision to place a pancreatic stent. While pancreatic stents are typically reserved for difficult biliary cannulation, our analysis did not adjust for technical factors such as pancreatic duct anatomy, endoscopist experience, or ampullary morphology. Consequently, residual confounding related to procedural difficulty and survival may persist. Nonetheless, the prognostic factors were fairly balanced between the groups, except that the non-stent group had a larger tumor size compared to the stent group, which was a disadvantage for patients without a stent. Second, the study population was highly selected, focusing on patients with unresected pancreatic cancer and obstructive jaundice who underwent ERCP, which may limit the generalizability of the findings to a broader population. For example, patients who did not survive long enough to develop obstructive jaundice would not have been included in the study because ERCP was not performed. However, this selection process was applied to both the pancreatic stent and non-pancreatic stent groups. Therefore, although selection bias may exist, it is unlikely that our results were affected by immortal time bias. Third, we did not replace pancreatic stents after spontaneous dislodgement, leading to variability in stent indwelling times among patients. Fourth, due to the retrospective nature of this study, patient adherence to PERT could not be accurately assessed. Therefore, the presumed dosage was calculated based on prescription records. Fifth, fecal elastase testing was not routinely performed in our clinical practice to directly assess PEI. As a result, nutritional parameters were used as surrogate indicators of PEI, given that malnutrition may be associated with PEI due to impaired digestion. However, nutritional status in cancer patients is multifactorial, often confounded by chemotherapy-induced emesis, mucositis-limiting oral intake, or anorexia. Therefore, while useful, nutritional status and weight remain imperfect surrogates for the definitive evaluation of PEI. This limitation may restrict causal inference regarding the relationship between pancreatic stent dysfunction and PEI. Sixth, our study encompasses an extensive study period. Although the majority of patients initially received gemcitabine-based chemotherapy, subsequent treatment selections following disease progression may have varied as therapeutic strategies evolved over time. This evolution in the standard of care could potentially confound the survival analysis of our pancreatic cancer cohort. Finally, there was a notable imbalance in the number of patients between the groups, as only a small proportion (6.9%) underwent pancreatic stent placement. This reflects the relatively low frequency of pancreatic stent placement in clinical practice. According to the literature, approximately 11% of ERCP procedures in patients with normal anatomy involve difficult biliary cannulation in real-world settings. 29 As such, the proportion of pancreatic stent placement observed in our study is consistent with these expectations. Nevertheless, this imbalance may have reduced the statistical power to detect differences between groups. Moreover, the interaction analysis between pancreatic stenting and PERT was likely underpowered due to the limited subgroup sample size. Further prospective studies with adequate statistical power are warranted to validate our results.
Conclusion
Pancreatic stent placement in patients with unresected pancreatic head cancer and obstructive jaundice does not improve 1-year survival, overall survival, or nutritional status. Based on these findings, we recommend the scheduled early removal of pancreatic stents after ERCP to avoid potential complications.
Supplemental Material
sj-pdf-4-tag-10.1177_17562848261437173 – Supplemental material for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study
Supplemental material, sj-pdf-4-tag-10.1177_17562848261437173 for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study by Weng-Fai Wong, Yu-Ting Kuo, Ming-Lun Han, Chieh-Chang Chen and Hsiu-Po Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-1-tag-10.1177_17562848261437173 – Supplemental material for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study
Supplemental material, sj-tiff-1-tag-10.1177_17562848261437173 for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study by Weng-Fai Wong, Yu-Ting Kuo, Ming-Lun Han, Chieh-Chang Chen and Hsiu-Po Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-2-tag-10.1177_17562848261437173 – Supplemental material for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study
Supplemental material, sj-tiff-2-tag-10.1177_17562848261437173 for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study by Weng-Fai Wong, Yu-Ting Kuo, Ming-Lun Han, Chieh-Chang Chen and Hsiu-Po Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-3-tag-10.1177_17562848261437173 – Supplemental material for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study
Supplemental material, sj-tiff-3-tag-10.1177_17562848261437173 for Survival impact of pancreatic stenting in unresected pancreatic head cancer with obstructive jaundice: a retrospective cohort study by Weng-Fai Wong, Yu-Ting Kuo, Ming-Lun Han, Chieh-Chang Chen and Hsiu-Po Wang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
