Abstract
Pancreatic divisum (PD) is caused by the lack of fusion of the pancreatic duct during the embryonic period. Considering the incidence rate of PD, clinicians lack an understanding of the disease, which is usually asymptomatic. Some patients with PD may experience recurrent pancreatitis and progress to chronic pancreatitis. Recently, a 13-year-old boy presented with pancreatic pseudocyst, recurrent pancreatitis, and incomplete PD, and we report this patient’s clinical data regarding the diagnosis, medical imagining, and treatment. The patient had a history of recurrent pancreatitis and abdominal pain. Magnetic resonance cholangiopancreatography was chosen for diagnosis of PD, pancreatitis, and pancreatic pseudocyst, followed by endoscopic retrograde cholangiopancreatography, minor papillotomy, pancreatic pseudocyst drainage, and stent implantation. In the follow-up, the pseudocyst lesions were completely resolved, and no recurrent pancreatitis has been observed.
Keywords
Background
Pancreatic divisum (PD) is characterized by the lack of fusion in the ductal system of the dorsal and ventral anlage, leading to PD with the pancreatic drainage via the dorsal duct of Santorini through the minor papilla.1,2 Its prevalence ranges from 4% to 14% of the population, 3 and some PD patients develop serious diseases, including recurrent acute pancreatitis (RAP), chronic pancreatitis (CP), and chronic abdominal pain.
In children, pseudocyst is a common complication of pancreas-related trauma, RAP, and CP, 4 leading to the ductal disruption and secondary trauma. 5 Additionally, most pancreatic pseudocysts can be relieved with careful nursing and supportive nutrition. 6 Symptomatic pseudocysts, especially for those with a lesion diameter is more than 6 cm, require surgical intervention.6,7 However, management of pancreatic pseudocysts in children includes drainage of the pseudocysts, which can be the optimal choice, and it is accomplished by surgery or, recently, by interventional therapy such as endoscopic retrograde cholangiopancreatography (ERCP). 4
Pancreatic pseudocyst, pancreatitis, and incomplete PD in children are extremely rare. We herein report clinical data on the pancreaticobiliary system that was predicted from a rare case of pancreatic pseudocysts, pancreatitis, and incomplete PD in a 13-year-old boy.
Case presentation
A 13-year-old boy had a significant medical history of recurrent pancreatitis with one episode of acute pancreatitis, and the patient’s first pancreatitis episode occurred in October 2019 at 12 years of age. He presented to the Department of Hepatopancreatobiliary Surgery. He mentioned that he had experienced abdominal pain, abdominal distension, fatigue, and anorexia for 8 days. His family history was unremarkable. He had a negative Murphy sign and no history of previous alcohol abuse, smoking, or gallstones.
The patient’s vital signs were as follows: blood pressure, 116/68 mmHg; respiration, 14 breaths/minute; and heart rate, 85 beats per minute (bpm). Computed tomography (CT) that was conducted at our hospital revealed a pseudocyst lesion of the pancreas (Figure 1) that measured 6.84 cm × 3.17 cm and was located between the stomach and the liver. Magnetic resonance imaging (MRI) revealed an 8 cm × 4.5 cm pseudocyst lesion of the pancreas that was located at the pancreatic body and neck (Figure 2). Magnetic resonance cholangiopancreatography (MRCP) findings showed no evidence of bile duct stenosis (Figure 3). On the basis of the medical imagining, a diagnosis of incomplete PD and pancreatic pseudocyst was made.

Abdominal CT showing the pancreatic pseudocyst lesion in the transverse plane.

MRI showing the pancreatic pseudocyst lesion in the transverse plane.
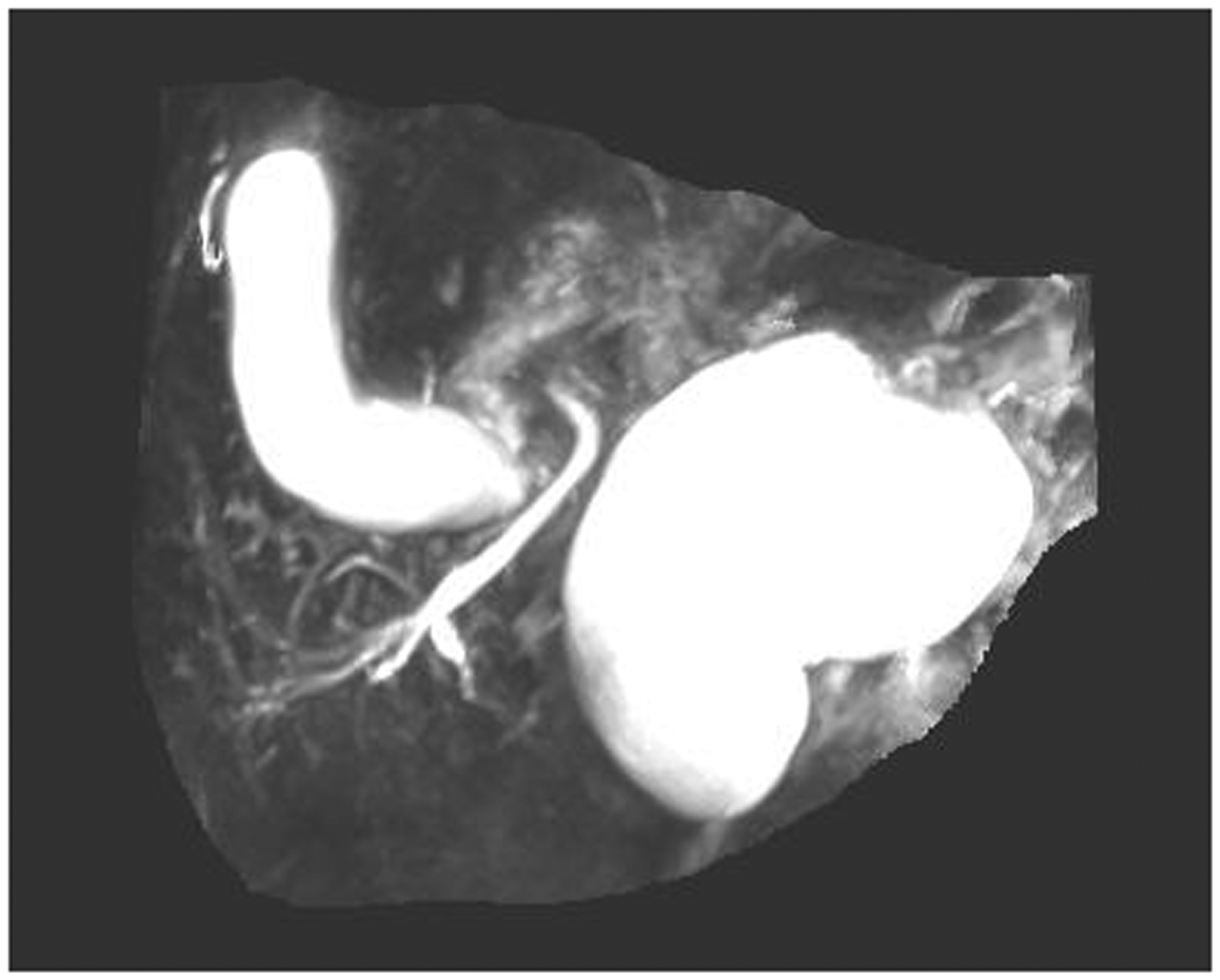
MRCP showed no evidence of bile duct stenosis.
Laboratory test results showed an increase in the levels of amylase (618 U/L, normal 0 to 130 U/L), lipase (573 U/L, normal 0 to 58 U/L), and cholestatic indexes (total bilirubin 3.2 mg/dL, normal 0.1 to 1.1 mg/dL; direct bilirubin 1.4 mg/dL, normal 0.0 to 0.4 mg/dL). A diagnosis of acute recurrent pancreatitis was then made.
After further investigation, ERCP revealed pancreatic duct stenosis (Figure 4). MRCP also showed a communication between the dorsal duct and the ventral duct. During ERCP, it is difficult to intubate the major papilla. The guide wire curls under the X-ray, and it is difficult to enter the pancreatic duct (Supplementary Figure 1), so we intubated the minor papilla (Supplementary Figure 2). After papillotomy of the minor papilla, the pancreatic duct was suspected to be connected with the pseudocyst. The pseudocyst was drained by endoscopic pancreatic duct drainage, and an aplastic stent was inserted. The pancreatic pseudocyst was successfully excised. Post-surgery, the patient remained stable and was discharged after a few days.

ERCP revealed pancreatic duct stenosis.
During follow-up, the patient was in a stable condition. We removed the original stent and implanted a new stent at 4 months after the original stent insertion (Figures 5, 6). The pseudocyst was completely resolved, and the patient was discharged with no evidence of lesion recurrence. After 8 months of follow-up, the follow-up CT and ERCP (Figures 7, 8) revealed no evidence of pseudocyst recurrence, and the stent was removed. This child recovered uneventfully.

Abdominal CT showing no evidence of lesion recurrence at 4 months after stent insertion.

ERCP showing no evidence of lesion recurrence at 4 months after stent insertion.

Abdominal CT showed no evidence of pseudocyst recurrence after 8 months.

ERCP showed no evidence of lesion recurrence after 8 months.
Discussion
Previous studies have reported that the PD shows an increasing prevalence in pancreatitis patients.8,9 A congenital anatomical abnormality of the pancreas is often observed, which leads to dorsal and ventral pancreatic ducts that drain via the minor papilla. In patients with PD, pancreatic drainage mainly occurs via the minor papilla, leading to high duct pressure and possible pancreatitis. 10 Some patients with PD can develop a symptomatic disease including RAP, CP, and recurrent pancreatitis, and attention should be paid to this condition by clinicians. 11 Recurrent pancreatitis refers to episodes of acute pancreatitis (AP) that could lead to CP. Our patient presented with abdominal pain, abdominal distension, fatigue, and anorexia. The diagnosis of incomplete PD and was mainly based on the ERCP results, and the diagnosis of pseudocyst was mainly based on the contrast-enhanced abdominal CT scan and MRI results. Considering that this patient had one episode of pancreatitis and a diagnosis of PD, we also made a consistent diagnosis, which was recurrent pancreatitis.
MRCP is considered a non-invasive approach that can visualize the pancreatic ducts without the using of contrast-enhanced material. 12 MRCP is also a sensitive method for diagnosing PD. 13 An evidence-based review 14 showed that the sensitivity and specificity of MRCP for PD was 52% and 97%, respectively. MRCP is also an optimal choice for diagnosing other congenital pancreaticobiliary malformations. Thus, clinicians may need the details that are provided by pancreatic imaging including the ductal anatomy and the radiologist’s report on the scans. This case also highlights the importance of imaging interpretation by clinicians.
Endotherapy can be a feasible procedure for treating patients with PD with pseudocyst. Endotherapy also may be an effective treatment option for patients with symptomatic PD, especially for patients with AP. ERCP is considered the gold standard for diagnosing PD. 15 In PD patients who undergo ERCP, the incidence of post-ERCP adverse events ranges from 8.2% to 10.6%, 16 and post-ERCP pancreatitis is most frequently observed. In our case, a stent was inserted for the pseudocyst, and no severe adverse events were observed. Currently, minor papillotomy and stent implantation during ERCP are the preferred methods to treat symptomatic PD. Low adverse event levels also make it the first-line treatment for patients with PD. 11 However, draining pancreatic pseudocysts, especially in children, remains controversial because adverse events may occur more frequently than in adults. 17
Pancreatic pseudocyst, pancreatitis, and incomplete PD in children are rare in the English and Chinese literature, and to the best of our knowledge, only three well documented cases have been reported.18 –20 All of these documented cases were treated using Roux-en-Y fistulojejunostomy, so our case is the first report of a child with pancreatic pseudocyst, pancreatitis, and incomplete PD who was treated using endotherapy.
The optimal surgical choice can be discussed, and duct stent insertion via the minor papilla, Roux-en-Y fistulojejunostomy, or other feasible treatments can be accepted, but there is currently no consensus. Surgical procedures of the Roux-en-Y fistulojejunostomy are complex, and thus, Roux-en-Y fistulojejunostomy seems to be an inferior procedure compared with endotherapy in terms of simplicity and reversibility. On the basis of our experience, the operation time for the Roux-en-Y fistulojejunostomy is longer than endotherapy because of the digestive tract reconstruction. The occurrence of postoperative complications are high because of the loss of organ and tissue, so we suggest that Roux-en-Y fistulojejunostomy is not the optimal surgical choice for this kind of patient.21,22 Jokic et al. 19 reported that external drainage as a therapeutic method was successful in treating these patients, which caused us to hypothesize that a less invasive treatment such as endotherapy and external drainage can be considered for unique cases. On the basis of our experience with this case, MRCP may be the first choice for diagnosing PD and pancreatic pseudocyst, while ERCP and endotherapy can be reserved for uncertain cases and to treat symptomatic patients. Additionally, our procedures provide less invasive alternatives, and sufficient pseudocyst drainage may reduce the pressure that is caused by the pseudocyst lesions and decrease the incidence of recurrent pancreatitis. Stent insertion plays another important role in pancreatic decompression. However, clinicians should avoid prolonged stenting because it may lead to sepsis and/or cause damage to the pancreas because a metal stent needs to be removed within 2 to 4 weeks or it will cause pancreatic injury and other complications. 23 Because plastic stents can last longer in the pancreatic duct than metal stents, in our case, a plastic stent was inserted to avoid complications such as pancreatic injury that is caused by metal stent implantation. Thus, we suggest that it is important for patients with symptomatic PD to control the pressure inside and outside the pancreas to prevent the recurrent pancreatitis and other accompanying adverse events.
In conclusion, our paper reported a rare case of pancreatic pseudocyst, pancreatitis, and incomplete PD. This case can be useful for interpreting the diagnosis and for treating patients with PD.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211014395 - Supplemental material for Pancreatic pseudocyst, pancreatitis, and incomplete pancreas divisum in a child treated with endotherapy: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605211014395 for Pancreatic pseudocyst, pancreatitis, and incomplete pancreas divisum in a child treated with endotherapy: a case report by Jingxin Yan, Zheheng Zhang, Zhixin Wang, Wenhao Yu, Xiaolei Xu, Yaxuan Wang and Haining Fan in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605211014395 - Supplemental material for Pancreatic pseudocyst, pancreatitis, and incomplete pancreas divisum in a child treated with endotherapy: a case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605211014395 for Pancreatic pseudocyst, pancreatitis, and incomplete pancreas divisum in a child treated with endotherapy: a case report by Jingxin Yan, Zheheng Zhang, Zhixin Wang, Wenhao Yu, Xiaolei Xu, Yaxuan Wang and Haining Fan in Journal of International Medical Research
Footnotes
Acknowledgments
Special thanks to Prof. Lushun Zhang and Li Cheng, who are teachers at Chengdu Medical College, for their comments on the initial manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval
This article was approved by the Ethics Committee of the Affiliated Hospital of Qinghai University, which is located at No. 29 Tongren Road, Xining City, Qinghai Province, China. The patient and his parents agreed to the use of his imaging and clinical data for publication and academic research, and they provided written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Natural Science Foundation of Qinghai (grant number: 2017-ZJ-936Q), CAS “Light of West China” Program (2018), and Thousand Talents Program of Qinghai Province (2017).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
