Abstract
Background:
The degree of coincidence and interaction between primary biliary cholangitis (PBC) on the one hand and metabolic syndrome (MetS) or metabolic dysfunction-associated steatotic liver disease (MASLD) on the other is unknown.
Objectives:
The aim of the study was to evaluate the associations between MASLD/MetS and PBC, with the focus on liver fibrosis (F).
Design:
Cross-sectional observational study.
Methods:
Cross-sectional observational study recruited consecutive patients treated for PBC prospectively from a single tertiary center. The control group was sampled, matched for age and sex, from the general population obtained from nation wide SIRIUS database, which aims to screen the general population for liver disease. Liver stiffness as a surrogate marker of liver fibrosis was the primary outcome, while achieving complete biochemical response (CBR) was a secondary outcome of our research.
Results:
Prevalence of MASLD was 42.3% in the PBC group and 41.9% in the control group (NS). Prevalence of MetS was 38.9% in PBC and 60.7% in the control group (p < 0.001). Advanced liver fibrosis was present significantly more often in the PBC group (21.5%), even after stratification by CBR (9.8%), than in age and sex matched general population (2%; p = 0.003). MetS (odds ratio (OR) 4.561, 95% confidence interval (CI): 1.852–11.398) and the number of fulfilled MetS criteria (1.901, 95% CI: 1.239–2.915), but not MASLD (OR 3.956, 95% CI: 0.730–21.422) were associated with advanced fibrosis. Out of individual MetS criteria hyperglycemia/diabetes mellitus had the strongest association with advanced fibrosis, followed by high-density lipoprotein cholesterol criterion. Neither MASLD nor MetS was associated with CBR.
Conclusion:
Patients with PBC appear to have a lower prevalence of MetS than age and sex matched general population, despite the same prevalence of MASLD in both groups. MetS but not MASLD was associated with the presence of advanced fibrosis in patients with PBC.
Plain language summary
This study looked at how often primary biliary cholangitis (PBC), a rare autoimmune liver disease, occurs together with two common metabolic conditions: metabolic syndrome (MetS) and metabolic dysfunction-associated steatotic liver disease (MASLD). We also examined whether these conditions affect liver scarring (fibrosis) in people with PBC. We studied patients with PBC from one hospital and compared them to people of the same age and sex from a large national health screening program. We measured liver stiffness (a marker of fibrosis) and looked at whether patients had a complete biochemical response (CBR) to treatment.
Keywords
Introduction
Primary biliary cholangitis (PBC) is a chronic autoimmune, non-suppurative inflammatory disease of the liver characterized by the destruction of the small intrahepatic bile ducts and the development of liver fibrosis (F). PBC progresses to liver cirrhosis in some patients.1,2 It is a rare disease with a relatively stable incidence and an increasing prevalence in advanced countries. The diseasepredominantly affects women. 3 Two of three criteria below must be met for a diagnosis of PBC:
- increased alkaline phosphatase (ALP) value above the upper limit of the norm (ULN) lasting for at least 6 months.
- antimitochondrial antibodies M2 (AMA M2) positivity in a titer of at least 1:40; in case of AMA M2 negativity, specific antinuclear antibodies (ANA) positivity: anti-sp100 nuclear antigen antibodies (anti-sp100) or anti-glycoprotein-210 antibodies (anti-gp210).
- histological findings consistent with PBC. 1
ANAs are present in more than half of PBC patients, mainly in AMA-negative patients; in addition to the ANA subtypes anti-gp210 and anti-sp100, anti-sp140 are also frequently found.4–7
Ursodeoxycholic acid (UDCA) is the drug of choice in PBC. About two-thirds of patients achieve a biochemical response on treatment, and one-third achieve a complete biochemical response (CBR). 8 For nonresponders, we can add-on second-line registered drugs, seladelpar or elafibranor, or an alternative is off-label treatment with fibrates. Also, obeticholic acid (OCA) has been used in the past as a second-line treatment. 9 PBC patients who achieve a biochemical response have a significantly better prognosis than nonresponders. 10
Metabolic syndrome (MetS) is a cluster of biochemical and physiological abnormalities associated with the development of cardiovascular disease and Type 2 diabetes mellitus (T2DM). Several definitions of MetS have been proposed, including the harmonized criteria introduced in 2009 11 ; in the present study, we applied the 2005 International Diabetes Federation (IDF) definition, which remains widely used in epidemiological research and emphasizes central obesity as a key metabolic component. 12 We chose to use this definition to facilitate broader comparison with published data, and since IDF 2005 identifies fewer but higher risk patients with metabolic derangement, we expected better correlation with metabolic dysfunction-associated steatotic liver disease (MASLD)/nonalcoholic fatty liver disease (NAFLD) and liver fibrosis.
MetS increases all-cause mortality, cardiovascular mortality, cancer mortality, and hepatic mortality.13–15 Steatotic liver disease is characterized by excessive hepatic fat accumulation, associated with insulin resistance, and defined by the presence of steatosis in >5% of hepatocytes. 16 MASLD increases the risk of both cardiovascular and oncological diseases. 17
The association between MetS/MASLD and PBC is poorly studied. The aim of our work is to elucidate how MetS and MASLD influence the clinical course of PBC.
Patients and methods
We conducted a cross-sectional observational study. PBC patients were consecutively recruited from the Hepatology Outpatient Clinic of the 2nd Department of Internal Medicine of L Pasteur University Hospital and PJ Safarik University, Faculty of Medicine in Kosice between January and June 2024. The study included individuals who fulfilled the diagnostic criteria for PBC according to European Association for the Study of the Liver (EASL) guidelines. 1 All patients in the PBC cohort underwent standard etiological evaluation for chronic liver diseases, including viral hepatitis B and C and other relevant causes. Exclusion criteria included only the refusal to participate, the diagnosis of idiopathic steatosis, and liver disease etiology other than PBC. PBC were consecutively examined in groups of two or three patients per day. Eligible patients were invited for follow-up visits during which laboratory testing, transient elastography, demographic and anthropometric assessments, disease-specific questionnaires, and other imaging modalities were performed. The study was approved by the Ethics Committee of the L Pasteur University Hospital in Kosice, Slovakia, on October 26, 2020, under reference number 2020/EK10076. The implementation of the study was postponed due to the COVID-19 pandemic. All patients signed an informed consent form. The general population cohort was constructed from the SIRIUS database, which is the result of an ongoing screening program in Slovakia, 18 which contained 1843 patients. Out of these patients, we excluded 522 patients with alcohol use disorder or hazardous alcohol consumption, assessed using the AUDIT-C questionnaire. Figure S1 presents a flowchart illustrating the patient selection process. There is no data on other liver diseases (i.e., mainly chronic hepatitis B and chronic hepatitis C) in the cohort, which, however, due to their low prevalence in the Slovak population, we do not consider this to be a problem. Pairing with the control group was performed in a 1:2 ratio, based on sex and age (tolerance for age ±5 years). In this way, 298 matched controls were randomly selected.
Definitions
The diagnosis of MetS was determined according to the criteria of the IDF Epidemiology Task Force Consensus Group. Participants were considered to have MetS when central obesity was present (waist circumference >94 cm for males and >80 cm for females or body mass index (BMI) >30 kg/m2) and any two of the following factors were met:
- raised triglycerides (TG) ⩾1.7 mmol/L or specific hypolipidemic treatment.
- reduced high-density lipoprotein cholesterol (HDL-C) <1.03 mmol/ in males; <1.29 mmol/L in females or specific hypolipidemic treatment.
- raised blood pressure, systolic ⩾130 or diastolic ⩾85 mm Hg; or treatment of previously diagnosed arterial hypertension (AH).
- raised fasting plasma glucose ⩾5.6 mmol/L or previously diagnosed T2DM. 12
The diagnosis of steatosis has been assessed by vibration-controlled transient elastography-controlled attenuation parameter (VCTE-CAP). CAP cut-off values for steatosis were 248 dB/m. CAP cut-off values at 248, 268, and 280 dB/m are recommended to identify steatosis (S) grading ⩾S1, ⩾S2, and =S3, respectively. MASLD has been diagnosed as the presence of hepatic steatosis in conjunction with at least one cardiometabolic risk factor and no other discernible cause. Cardiometabolic risk factors were defined according to current MASLD consensus and included overweight or obesity (BMI ⩾25 kg/m2), T2DM or prediabetes, AH or antihypertensive treatment, hypertriglyceridemia (TG ⩾1.7 mmol/L or lipid-lowering therapy), and low HDL-C (<1.03 mmol/L in men or <1.29 mmol/L in women). Minimal alcohol intake was ensured using AUDIT-C-based exclusion criteria. 17
F measured by VCTE was classified as mild to moderate (F0–F2), elasticity <10.7 kPa and advanced, elasticity ⩾10.7 kPa.19,20 CBR was defined as a normal value of total bilirubin and ALP in the laboratory examination of a PBC patient.
Outcomes
The primary outcome was to evaluate the association of liver fibrosis on the one hand with the presence of MASLD or MetS on the other hand in PBC patients.
The secondary outcomes were as follows:
- to compare the proportions of PBC patients that achieved CBR in the cohort with and without MASLD/MetS.
- to compare the prevalence of MetS, MASLD, and advanced fibrosis between PBC patients and the general population.
Statistical analysis
Interval variables are reported as mean ± standard deviation, log-transformed interval variables as median (interquartile range), categorical variables are reported as absolute and relative counts. Variables with typically skewed distribution (long right tail) were log-transformed (Liver stiffness, bilirubin-total, conjugated bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), ALP, gamma-glutamyl transferase (GGT; all four expressed as percentage of norm), c-reactive protein (CRP), ferritin, Lipoprotein a, homeostatic model assessment for insulin resistance (HOMA-IR)). Nonparametric test (Mann–Whitney) was used to compare variables with atypical nonparametric distribution (glucose), normally distributed variables (all other variables) were tested by T-test. Multivariate logistics and linear regressions were used to evaluate the associations of predictors, adjusted for confounders, with fibrosis and CBR. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 21
Results
A total of 152 patients with PBC were included in our analysis, 11 men (7.2%) and 141 women (92.8%). There were 66 (43.4%) patients with steatosis, of whom 3 had idiopathic steatosis (2%) and 63 patients (42.3%) had MASLD. MetS was present in 58 (38.9%) patients with PBC. Patients with idiopathic steatosis were excluded from further analysis. No patient fulfilled criteria for alcohol use disorder or hazardous alcohol consumption, with 94 patients (61.8%) reporting being complete abstainers, 49 patients (32.2%) admitting to consuming once a month or less, and 9 patients (5.9%) consuming 2–4 times a month. CBR was reported by 82 (55%) patients in the PBC patient cohort. All patients with PBC received UDCA as first-line therapy. In addition, 34 of 149 patients (22.8%) were treated with fibrates, either as add-on therapy for PBC or for the management of hypertriglyceridemia, according to clinical indication. No patient received seladelpar or elafibranor. An overview of the cohort of patients with PBC is given in Table 1.
An overview of the cohort of PBC patients.

Statistically significant associations (p < 0.05) are highlighted in bold.
ACS, acute coronary syndrome; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CAD, coronary artery disease; CAP, controlled attenuation parameter; CBR, complete biochemical response; cm, centimeter; CRP, c-reactive protein; dB/m, decibels per meter; F, fibrosis stage; GGT, gamma-glutamyl transferase; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment for insulin resistance; kPa, kilopascal; L, liter; LDL-C, low-density lipoprotein cholesterol; MASLD, metabolic dysfunction-associated steatotic liver disease; MetS, metabolic syndrome; mg/L, miligram per liter; mmol/L, millimol per liter; n, number; nmol/L, nanomoles per liter; PAO, peripheral arterial atherosclerotic occlusive disease; PBC, primary biliary cholangitis; Plt, platelets; TC, total cholesterol; TG, triglycerides; ULN, upper limit of the normal; VCTE, vibration-controlled transient elastography; μg/L, microgram per liter; μmol/L, micromol per liter.
As expected, patients with MASLD had higher BMI and waist circumference, and more commonly MetS. There was no difference in the activity of liver enzymes or CRP, but MASLD patients had significantly higher levels of serum ferritin. In lipoprotein metabolism, patients with MASLD had higher TG levels and lower HDL-C levels. There was no significant difference in the occurrence of CBR and advanced fibrosis between patients with and without MASLD.
The primary outcome of our study was to investigate whether patients with MASLD or MetS frequently suffer from advanced fibrosis. The results of this observation are shown in Table 2.
Parameters associated with advanced fibrosis in PBC patients.
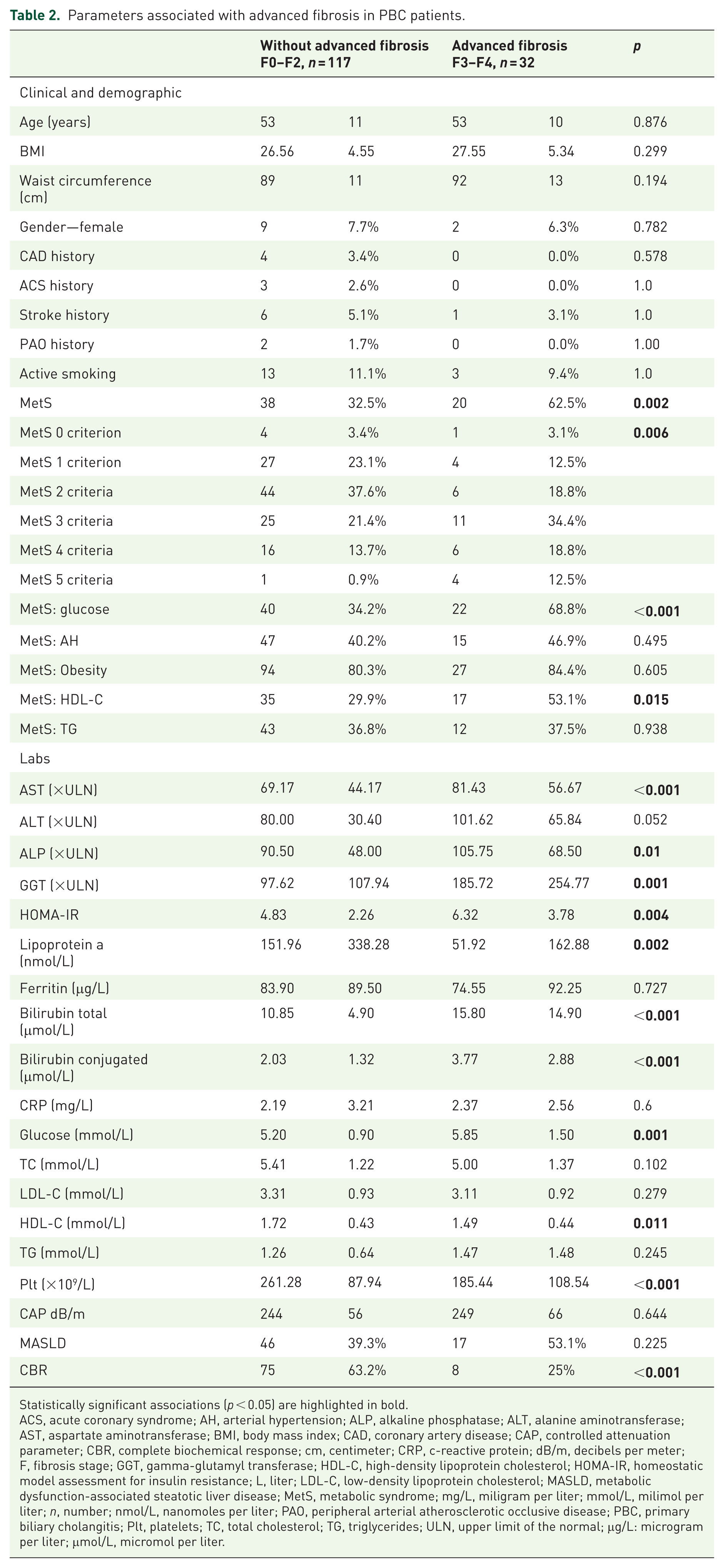
Statistically significant associations (p < 0.05) are highlighted in bold.
ACS, acute coronary syndrome; AH, arterial hypertension; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CAD, coronary artery disease; CAP, controlled attenuation parameter; CBR, complete biochemical response; cm, centimeter; CRP, c-reactive protein; dB/m, decibels per meter; F, fibrosis stage; GGT, gamma-glutamyl transferase; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment for insulin resistance; L, liter; LDL-C, low-density lipoprotein cholesterol; MASLD, metabolic dysfunction-associated steatotic liver disease; MetS, metabolic syndrome; mg/L, miligram per liter; mmol/L, milimol per liter; n, number; nmol/L, nanomoles per liter; PAO, peripheral arterial atherosclerotic occlusive disease; PBC, primary biliary cholangitis; Plt, platelets; TC, total cholesterol; TG, triglycerides; ULN, upper limit of the normal; μg/L: microgram per liter; μmol/L, micromol per liter.
The comparison showed that patients with more severe fibrosis suffered more often from MetS, fulfilled more MetS criteria, and, of course, had higher levels of hepatic enzymes and bilirubin. PBC patients with advanced fibrosis had significantly lower lipoprotein a, HDL-C levels, and platelets, and significantly higher glycemia and HOMA-IR. There was no significant difference between these two groups in the presence of steatosis assessed by CAP and the presence of MASLD. PBC patients without advanced fibrosis more frequently achieved CBR.
When adjusting for age, sex, and CBR achievement using a series of multivariate regression models, the association between fibrosis on the one hand and lipoprotein a, HOMA-IR, HDL-C, and the presence of MetS on the other hand remained significant. Patients with MetS were approximately 4.5 times more likely to have advanced fibrosis than patients without MetS. Each additional MetS criterion was associated with a 1.9-fold increase in the risk of advanced fibrosis in PBC patients. The results are summarized in Table 3. All variables, except for the presence of MASLD were adjusted for steatosis. MASLD includes steatosis in its definition.
Series of regression models adjusted for age, gender, CBR, and fatty liver presence.

Outcome is the presence of advanced fibrosis (F3–F4). Statistically significant associations (p < 0.05) are highlighted in bold.
CBR, complete biochemical response; CI, confidence interval; F, fibrosis stage; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment for insulin resistance; log, logarithm; MASLD, metabolic dysfunction-associated steatotic liver disease; MetS, metabolic syndrome; OR, odds ratio.
The greatest difference between the groups with and without advanced fibrosis in PBC patients was in the hyperglycemia/T2DM criterion and the low HDL-C criterion. In a multivariate regression model adjusted for age, sex, and presence of CBR, fulfilling the glycemia/T2DM criterion was the most significant predictor of advanced fibrosis (odds ratio (OR) 3.888, 95% confidence interval (CI): 1.518–9.963) followed by the HDL-C criterion (OR 2.622, 95% CI: 1.017–6.761). The other MetS criteria were not associated with the risk of advanced fibrosis; see Table 4.
Multivariate regression model with F3–F4 fibrosis as the outcome.

Statistically significant associations (p < 0.05) are highlighted in bold.
AH, arterial hypertension; CBR, complete biochemical response; CI, confidence interval; EXP(B), exponentiation of the B coefficient; F, fibrosis stage; HDL-C, high-density lipoprotein cholesterol; MASLD, metabolic dysfunction-associated steatotic liver disease; MetS, metabolic syndrome; OR, odds ratio; T2DM, type2 diabetes mellitus; TG, triglycerides.
Linear association of liver stiffness and MASLD/MetS
The liver stiffness was numerically higher in the group with MASLD, but the difference did not reach statistical significance (Table 1). The median stiffness (interquartile range) was significantly higher in patients with MetS compared to non-MetS patients; 10.72 (7.2) versus 7.77 (3.8) kPa, p = 0.021. Linear regression adjusted for age, sex, and the presence of CBR showed that the presence of MetS (Beta 0.176, p = 0.037) but not MASLD (Beta 0.122, p = 0.140) was associated with increasing liver stiffness.
Eighty-two PBC patients (55%) achieved CBR. A secondary outcome of our study was to determine whether the presence of MASLD or MetS predicts the achievement of CBR. The factors associated with CBR are presented in Table 5.
Factors associated with complete biochemical response.

Statistically significant associations (p < 0.05) are highlighted in bold.
ACS, acute coronary syndrome; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; CAD, coronary artery disease; CAP, controlled attenuation parameter; CBR, complete biochemical response; cm, centimeter; CRP, c-reactive protein; dB/m, decibels per meter; GGT, gamma-glutamyl transferase; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment for insulin resistance; kPa, kilopascal; L, liter; LDL-C, low-density lipoprotein cholesterol; MASLD, metabolic dysfunction-associated steatotic liver disease; MetS, metabolic syndrome; mg/L, miligram per liter; mmol/L, milimol per liter; n, number; nmol/L, nanomoles per liter; PAO, peripheral arterial atherosclerotic occlusive disease; Plt, platelets; TC, total cholesterol; TG, triglycerides; ULN, upper limit of the normal; VCTE, vibration-controlled transient elastography; μg/L, microgram per liter; μmol/L, micromol per liter.
Neither the presence of MetS nor the presence of MASLD differed between patients who did and who did not achieve CBR in the treatment of PBC. Surprisingly, patients with CBR on PBC treatment had higher CAP values. Achieving CBR on PBC treatment was associated with some hepatic parameters and, surprisingly, with higher lipoprotein a level. Patients with advanced fibrosis were less likely to achieve CBR.
A multivariate regression model of the relationship between liver steatosis and CBR was used to control for some variables. The model included age, sex, liver stiffness, degree of hepatic steatosis, and lipoprotein a level (see Table 6). The overall model fit (strength of association with biochemical response was not high, R2 0.181).
Multivariate regression model with complete biochemical response as an outcome.

Statistically significant associations (p < 0.05) are highlighted in bold.
CAP, controlled attenuation parameter; CI, confidence interval; EXP(B), exponentiation of the B coefficient; log, logarithm; TE, transient elastography.
After controlling the above variables, lower stage fibrosis remained associated with CBR (a 1-log rise of stiffness in kPa reduced the response rate by 90%). Also, surprisingly, higher CAP remained associated with CBR (a 10 dB/m rise in CAP increases the CBR rate by almost 10%). When controlling for the above variables, lipoprotein a was not significantly associated with CBR.
Comparison of the prevalence of MetS and MASLD between patients with PBC and patients from the general population was another secondary outcome of our research. The prevalence of individual metabolic abnormalities and the significance of differences between the PBC patient cohort and the general population cohort are summarized in Table 7.
Metabolic abnormalities between the PBC cohort and the general population cohort.

Statistically significant associations (p < 0.05) are highlighted in bold.
BMI, body mass index; CAP, controlled attenuation parameter; cm, centimeter; dB/m, decibels per meter; kPa, kilopascal; n, number; PBC, primary biliary cholangitis; TE, transient elastography.
There was no difference in the prevalence of steatosis or MASLD between the PBC group and the control group. Patients with PBC, of course, had a higher prevalence of advanced fibrosis, even after excluding PBC patients without CBR (9.8% vs 2%, p = 0.003). Patients with PBC had a lower prevalence of MetS by more than 20% (38.9% vs 60.7%). Subjects in the control group had more frequent AH and hypertriglyceridemia. Surprisingly, the overlap between MASLD and MetS was rather low. Only 55.2% of patients with MetS had MASLD, and only 51% of patients with MASLD had MetS in the PBC group. Similar observations are in the control group, where 53% of patients with MetS had MASLD and 76.8% of patients with MASLD had MetS in the control group, p for complete comparison = 0.035.
Discussion
Several papers have been published evaluating the incidence of steatosis diagnosed by CAP or histology in patients with PBC,22–27 however, literature data on the prevalence of MASLD in patients with PBC are poor. A major reason for this is the fact that the concept of MASLD was adopted relatively recently—in 2023. The key factor for the diagnosis of MASLD is the assessment of the diagnosis of liver steatosis, which can be determined histologically, using CAP or abdominal ultrasonography. The limitation of using CAP is the fact that it has only been validated for isolated hepatic steatosis, thus their cut-off points need specific evaluation for overlap PBC/MASLD. 28 Hepatic steatosis was evaluated in PBC patients both histologically and using transient elastography in a German study. Of the 51 PBC patients examined, only 16 had steatosis, with 13 classified as stage S1. The authors determined a cut-off for the diagnosis of hepatic steatosis in PBC patients of 276 dB/m; the total area under the receiver operating characteristic curve 0.81, sensitivity and specificity were relatively low: 0.71 and 0.81, respectively. 26 Despite this finding, cut-offs recommended by EASL for the diagnosis of hepatic steatosis and its different stages in PBC patients and in general population were used in our research. 17 Although this approach ensures consistency with current guidelines and comparability with existing MASLD literature, we acknowledge that the use of standard CAP cut-offs in PBC may be associated with some degree of steatosis misclassification.
Prevalence of MASLD in PBC patients in our study was approximately 40%, which was like the prevalence of MASLD in the Slovak general population. A retrospective study from the University of Alberta in Canada, which included 115 patients with PBC followed between 1984 and 2023, found 33% MASLD prevalence. Fatty liver in this study was assessed using transient elastography; the cut-off for the diagnosis of steatosis was slightly higher than in our study—288 dB/m. 24 In another study from two Spanish centers with 129 patients with histologically confirmed PBC, 27.9% of patients had concomitant MASLD. 23 More precise cut-off specifications for hepatic steatosis in PBC patients using transient elastography will probably be very difficult, because histological examination in PBC patients is not currently routinely recommended. 1
None of the above papers evaluates the incidence of MetS in PBC patients. MetS prevalence in our study was 38.9% in PBC compared to 60.7% in the control group (p < 0.001); the difference between the two groups was more than half. The high prevalence of MetS in both groups was probably associated with a relatively higher mean age in both groups (approximately 53 years). The overall MetS prevalence in our country is highly age-dependent and varies from 2.8% in the 10–17 years age group to 52.0% in people older than 65 years. 29 PBC patients had less frequent AH and, surprisingly, less frequently met the hypertriglyceridemia/fibrates criterion compared to the general population. PBC patients usually exhibit lipid abnormalities due to impaired bile acid secretion and altered lipid metabolism. PBC patients often have increased total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and HDL-C levels. 30 HDL-C levels are typically elevated in early PBC stages, but they decrease with disease progression. 31 TG levels in PBC patients are generally normal or slightly elevated, which is associated with lower lipoprotein lipase activity. 32 PBC treatment affects lipogram. UDCA lowers TC, LDL-C with no effect on HDL-C or TG. 33 OCA in PBC patients reduces TC levels due to HDL-C reduction, effect on LDL-C remains unclear. 34 The effect of hypolipidemic treatment in the context of MetS incidence is not indicative, because statin and fibrate use is included in the MetS criteria. The prevalence of MetS in our study was comparable to the MetS prevalence in other clinical studies. The Serbian authors found a lower prevalence of MetS in PBC patients compared to the control cohort: 31% versus 43%. 35 The results from Italy were similar, 32% PBC patients fulfilled the criteria for MetS. 36 A retrospective cohort study involving 789 PBC patients from China reported the following prevalences of metabolic risk factors: hyperlipidemia: 34.3%, AH: 16.0%, T2DM: 11.9%. 37 We also found a high prevalence of AH and prediabetes/T2DM in our PBC cohort. The Asian population has a lower obesity prevalence and different visceral fat composition. Genetic predisposition and different lifestyles also contributed to the different prevalence of individual MetS factors between our and Chinese cohorts.
We have not observed a presumed large overlap between MASLD and MetS. Only about half of the patients in our cohort who had MetS had also liver steatosis, which is true for PBC as well as control group. However, in PBC group, we observed a significantly lower rate of MetS in patients with MASLD compared to the control group (53% vs 77%), which means that the overlap between steatosis and MetS is lower in the PBC group, compared to the control group (p = 0.035). This limited overlap is largely explained by differences in diagnostic criteria: MASLD identifies patients with hepatic steatosis and even mild metabolic abnormalities, whereas MetS reflects a more advanced, cumulative metabolic burden requiring multiple concurrent risk factors. Our results are reflecting the published data, for example, in a landmark paper by Younossi et al., 38 the prevalence of MetS in patients with liver steatosis was 42.54% (95% CI: 30.06–56.05). Vice versa, the pooled overall prevalence of NAFLD (now termed MASLD) in patients with MetS was 54.1% (95% CI: 44.1%–63.9%) in a meta-analysis of studies from the South Asian region. 39
A key question is how MASLD will affect the course of PBC. The stage of fibrosis is considered the most significant prognostic factor in chronic liver disease. It reflects the severity of liver damage and allows prediction of the clinical consequences of the disease—such as the development of cirrhosis, portal hypertension, hepatocellular carcinoma, or the need for liver transplantation. In our study, MASLD was not statistically associated with advanced fibrosis in patients with PBC (OR 3.956, 95% CI: 0.730–21.422), but this may be due to the small number of patients with MASLD in our cohort. Although MASLD was not significantly associated with advanced fibrosis in our cohort, the corresponding CI was wide, reflecting limited statistical power. Therefore, the absence of a statistically significant association should be interpreted with caution, and a type II error cannot be excluded.
However, the presence of MetS was associated with 4.5-fold higher odds of advanced fibrosis compared to patients without MetS (OR 4.561, 95% CI: 1.825–11.398). Thus, we can hypothesize that these findings suggest that metabolic risk factors may be involved in fibrogenesis in patients with PBC. Recent CaReMe-oriented concepts 40 emphasize MASLD as a hepatic manifestation of systemic cardiometabolic risk rather than an isolated liver condition. In this context, our findings further support the notion that cumulative metabolic dysfunction, captured by MetS, may be more relevant for fibrosis severity in PBC than the presence of steatosis alone.
The difference in the impact on fibrosis progression between MASLD and MetS is mainly due to the different diagnostic criteria of the two entities. MASLD is defined by the presence of steatosis and at least one cardiometabolic risk factor. This definition also allows the inclusion of patients with slightly expressed or isolated metabolic abnormalities in whom the overall metabolic burden may not be strong enough to significantly activate profibrotic pathways. In contrast, MetS, defined according to the IDF criteria, requires concomitant fulfillment of at least three of the five major risk factors. This cumulative burden represents a more advanced form of metabolic dysregulation that leads to increased fibrogenesis. 41 Our study showed that severe metabolic dysfunction is probably more important than hepatic steatosis in the pathogenesis of advanced fibrosis. This theory is supported by the fact that each MetS criterion in our cohort was additionally associated with PBC patients, with an average 1.9-fold increase in the odds of advanced fibrosis in PBC patients. Fulfillment of the glycemia/T2DM criterion showed the strongest association with advanced fibrosis (OR 3.888, 95% CI: 1.518–9.963) followed by the HDL-C criterion (OR 2.622, 95% CI: 1.017–6.761). Other studies have also confirmed a strong association of T2DM with an increased risk of liver fibrosis, assessed by non-invasive methods or histology.42–44 A recent meta-analysis found approximately 20% prevalence of advanced fibrosis in patients with T2DM. 45 The association between concomitant MASLD in PBC patients and adverse liver outcomes has been evaluated in two papers cited above. Their conclusions are controversial. In the Canadian study, MASLD did not worsen the PBC course. MASLD in PBC patients did not increase the prevalence of liver cirrhosis, decompensation, liver transplantation, or death. PBC patients with MASLD have comparable event-free and overall survival compared to those without MASLD. 24 In the Spanish PBC study, PBC patients with MASLD had higher mortality, higher liver-related mortality, and higher need for transplantation compared to the PBC patients without MASLD. Liver steatosis, dyslipidemia, and advanced fibrosis were independently associated with worse outcomes in this study. 23 Several studies have presented different results of the association between NAFLD and fibrosis in PBC patients. PBC activity, severity, and progression were not adversely affected by concomitant NAFLD in a retrospective study by Minuk et al. 46 In contrast, in two other studies, steatosis was associated with advanced fibrosis.22,25
Achieving a biochemical response predicts a good course of PBC, reduces the prevalence of hepatic events, and improves survival.10,47 The goal in the treatment of PBC is to achieve CBR. These patients have better survival than patients with an incomplete biochemical response when treated with UDCA.48,49
More than half of the PBC patients in our cohort achieved CBR with first-line UDCA treatment or second-line therapy with OCA or fenofibrate. Logically, patients with CBR had a lower stage of fibrosis but, surprisingly, a higher degree of steatosis compared to PBC patients without CBR. MetS in PBC patients did not affect the achievement of CBR. Although neither MASLD nor MetS was directly associated with CBR, advanced fibrosis was strongly linked to non-achievement of CBR. This suggests that metabolic risk factors may influence prognosis in PBC primarily through their association with fibrosis severity rather than through a direct effect on biochemical response. The Italian authors studied the biochemical response to UDCA treatment in PBC patients with MetS and found no difference in achieving biochemical response in PBC patients with and without MetS, which is consistent with our results. 36
The main contribution of our work is that it is the first to assess the simultaneous occurrence of MASLD and MetS in PBC patients and to evaluate their impact on PBC progression. It highlights the need for active monitoring of metabolic factors in patients with PBC, as these influence both hepatic and non-hepatic prognosis.
Our study has several limitations: the observational character of the study, the monocentric design, the relatively small number of patients, and the assessment of the stage of fibrosis and steatosis by transient elastography. Another limitation of our study relates to the definition of MetS. Using the IDF 2005 definition, which requires central obesity, may have underestimated MetS in lean but metabolically unhealthy PBC patients and could have influenced the observed associations with fibrosis. Another limitation of this study concerns the control cohort derived from the SIRIUS database, a nationwide liver disease screening program in Slovakia. While subjects with significant alcohol intake were excluded, detailed etiological data on other chronic liver diseases were not systematically available. Therefore, a small proportion of unrecognized chronic liver disease in controls cannot be fully excluded and may have influenced liver stiffness or steatosis measurements. Furthermore, the impact of the type of treatment could not be fully explored. Given the observational design, the heterogeneity of indications for fibrate use, and the cohort size that precluded us from exploring too many predictors, the type of medication was not included in multivariable models. Also, the exploration of predictors of CBR achievement was not the primary aim of the study.
We will evaluate over time the impact of MetS and MASLD on the progression of hepatic findings and liver fibrosis, on the incidence of major hepatic events, and on all causes, hepatic and non-hepatic mortality. As PBC is a rare disease, the impact of MetS and MASLD on its course may be elucidated by a clinical trial with a multicentric design.
Conclusion
Patients with PBC appear to have a lower prevalence of MetS than age and sex matched general population, despite the same prevalence of MASLD in both groups. In this cross-sectional study, MetS was associated with advanced fibrosis in patients with PBC, whereas no statistically significant association was observed for MASLD. These findings highlight the importance of monitoring and managing metabolic risk factors in PBC patients to potentially improve the prognosis.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261428810 – Supplemental material for Metabolic syndrome and its individual components but not MASLD are associated with advanced fibrosis in primary biliary cholangitis
Supplemental material, sj-docx-1-tag-10.1177_17562848261428810 for Metabolic syndrome and its individual components but not MASLD are associated with advanced fibrosis in primary biliary cholangitis by Sylvia Drazilova, Tomas Koky, Martin Janicko, Marek Brenisin, Maria Marekova, Ivana Spakova, Miroslava Rabajdova, Maria Rasiova, Marianna Barbierik Vachalcova, Dominik Pella, Marian Macej, Slavomira Komarova and Peter Jarcuska in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848261428810 – Supplemental material for Metabolic syndrome and its individual components but not MASLD are associated with advanced fibrosis in primary biliary cholangitis
Supplemental material, sj-jpg-2-tag-10.1177_17562848261428810 for Metabolic syndrome and its individual components but not MASLD are associated with advanced fibrosis in primary biliary cholangitis by Sylvia Drazilova, Tomas Koky, Martin Janicko, Marek Brenisin, Maria Marekova, Ivana Spakova, Miroslava Rabajdova, Maria Rasiova, Marianna Barbierik Vachalcova, Dominik Pella, Marian Macej, Slavomira Komarova and Peter Jarcuska in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
