Abstract
Objective
Circular RNAs belong to a category of noncoding RNAs that feature a unique continuous, covalently bonded ring configuration. Recent research indicates that circular RNAs are essential for the development of metabolic dysfunction–associated steatotic liver disease. This study sought to examine the functional role and molecular mechanism of circDock6 in metabolic dysfunction–associated steatotic liver disease.
Methods
Quantitative reverse transcription polymerase chain reaction was performed to determine circDock6 expression patterns in liver tissues from high-fat diet- and standard diet-fed mice in vivo. Triglyceride detection, western blot analysis, and oil red O staining were performed to evaluate the regulatory effect of circDock6 on metabolic dysfunction–associated steatotic liver disease in vitro.
Results
CircDock6 was found to be markedly overexpressed in high-fat diet liver tissues compared with that in standard diet tissues. Moreover, knockdown of circDock6 expression lowered triglyceride content and lipid droplet formation. Mechanistically, circDock6 acted as a molecular sponge for mmu-let-7g-5p, which regulated insulin-like growth factor 1 receptor expression and contributed to the progression of metabolic dysfunction–associated steatotic liver disease. CircDock6 knockdown suppressed metabolic dysfunction–associated steatotic liver disease progression by modulating insulin-like growth factor 1 receptor via mmu-let-7g-5p targeting.
Conclusion
Our study identified circDock6 as a novel regulator of metabolic dysfunction–associated steatotic liver disease pathogenesis through the mmu-let-7g-5p/insulin-like growth factor 1 receptor axis, indicating its potential as a therapeutic target for metabolic dysfunction–associated steatotic liver disease intervention.
Keywords
Introduction
Metabolic dysfunction–associated steatotic liver disease (MASLD) is a clinical condition marked by excessive lipid accumulation within the liver, which is influenced by several conditions including insulin resistance, metabolic disorders, genetics, and the environment. 1 The prevalence of MASLD among Chinese adults has steadily increased in recent years due to changes in dietary patterns and growing living standards. 2 Currently, over one-quarter of people worldwide suffer from MASLD. 3 MASLD encompasses several types of chronic liver diseases, including nonalcoholic steatohepatitis (NASH) and simple hepatic steatosis. NASH may advance to severe hepatic diseases, including hepatocellular carcinoma (HCC) and cirrhosis, if not treated. 4 According to research, up to 10% of those diagnosed with MASLD may eventually develop liver cancer. 5 In addition, MASLD poses a major concern to public health, as it markedly aggravates the risk of extrahepatic complications, especially diabetes and coronary artery disease.1,6
A type of noncoding RNAs known as circular RNAs (circRNAs) is distinguished by its closed-loop structure and lack of a 3′-poly(A) tail and 5′-cap. In particular, circRNAs are primarily found in the cytoplasm or stored in exosomes, where they are more stable owing to their resistance to RNA exonuclease degradation. Research has verified their extensive presence in various eukaryotic species. 7 CircRNAs are expressed in certain cells and tissues and perform several regulatory functions. According to recent studies, circRNAs can act as miRNA sponges as they contain a large number of microRNA (miRNA)-binding sites. They increase the expression of target genes by reducing the repression of target genes caused by miRNAs. CircRNAs have a significant regulatory effect in the development of diseases through these interactions.
MASLD development is mostly influenced by interactions between circRNAs, miRNAs, and their target genes, which may be biomarkers for future diagnosis and therapy, as demonstrated by bioinformatics analysis. 8 According to research, circRRM2 alleviates MASLD progression by acting as a miR-142-5p sponge to upregulate NRG1, thereby reducing hepatic triglyceride accumulation in both cellular and murine models. 9 Targeting mmu_circ_0009303 can attenuate MASLD by restoring miR-182-5p/Foxo3 signaling and normalizing PPARα- and SREBP1-mediated lipid homeostasis. 10 Hepatocyte-specific circular RCRIN overexpression inhibits MASLD progression by destabilizing RPL8-containing ribosomes, offering a novel circRNA-based therapeutic strategy. 11 These findings demonstrate that circRNA dysregulation may affect liver steatosis, which in turn may contribute to the development of MASLD.
This study investigated circDock6, a circRNA with abnormal expression, which was identified using a gene chip specific to MASLD-related circRNAs. CircDock6, which is found on chromosome 9, contains a 521-nucleotide spliced sequence. Further analysis demonstrated that tissues from high-fat diet (HFD) models displayed a marked upregulation of circRNA relative to those from standard diet (SD) models. Nevertheless, the exact function and underlying mechanisms of circDock6 dysregulation in MASLD remain uncertain. The preprint of this study 12 presents initial findings on the role of circDock6 in MASLD development.
Additional research demonstrated that knocking down circDock6 in alpha mouse liver (AML)-12 cells resulted in marked suppression of lipid metabolism and lower triglyceride concentrations. circRNA gene chip analysis identified mmu-let-7g-3p, mmu-let-7g-5p, mmu-miR-138-5p, and mmu-let-7i-5p as miRNA-binding sites of circDock6. In addition, circDock6 expression was shown to be lower in AML-12 cells in previous research, with mmu-let-7g-5p exhibiting the most pronounced overexpression. Notably, the involvement of insulin-like growth factor 1 receptor (IGF1R), a validated mmu-let-7g-5p target, has been documented in metabolic regulation and the progression of severe liver damage associated with steatosis.13,14
This research aimed to demonstrate that circDock6 binds to mmu-let-7g-5p, resulting in the indirect regulation of the mmu-let-7g-5p target gene IGF1R, thus influencing MASLD development.
Materials and methods
Animal study
The reporting of this animal study conforms to the Animal Research: Reporting of In Vivo Experiments (ARRIVE) 2.0 guidelines. 15 The sample size was based on established study protocols. Liver tissues were collected from C57BL6J male mice at 8 weeks of age, with 10 samples from mice fed with a SD and 10 from mice fed with a HFD. The Slac Laboratory Animal Center in Shanghai, China, provided all of the C57BL6J mice, which were purchased at an average weight of 20 g and then bred under specific pathogen-free (SPF) conditions (temperature of 24°C and humidity of 50%) at the Ningbo University Laboratory Animal Center. All mice were maintained in a 12-h day/night cycle at controlled room temperature. After adapting to the laboratory conditions for 1 week, the mice were randomly and unblindedly divided into two groups (10/group, 5/cage) for overt phenotypes. Potential confounders were not controlled. Subsequently, the animals were randomized to either the HFD group (experimental: 80.5% standard chow diet (SCD), 2% cholesterol, 7% lard, 10% yolk powder, and 0.5% bile salt) or SD group (control: 8% rice bran, 51% maize, 30% soybean powder, 3% bone powder, 1.3% multivitamin, and 6.7% mineral) (Research Diets, Inc., New Brunswick, NJ, USA) according to their diet. 16 Samples of liver tissue were harvested 10 weeks after feeding for the quantification of circDock6 expression. The animals were sacrificed via CO2 inhalation, and death was verified by cessation of breathing and heartbeat. No animals or data points were excluded from the analysis. All animal studies were conducted in accordance with the 8th edition of the Guidelines for the Care and Use of Laboratory Animals, 17 ensuring that the animals were treated humanely and in compliance with ethical guidelines for laboratory animal care.
Cell culture
The American Type Culture Collection (ATCC) supplied the AML-12 mouse hepatocyte cell line, which was then maintained at 37°C in an incubator containing 5% CO2 under controlled humidity with Dulbecco’s modified Eagle medium (DMEM)/nutrient mixture F-12 (F-12) (1:1) media (Gibco, USA) mixed with insulin–transferrin–selenium (ITS) Liquid Media Supplement (Sigma, Japan), dexamethasone, and 10% fetal bovine serum.
Cell esterification modeling
In this study, cell esterification models were established using free fatty acids (FFAs), oleic acid (OA), and palmitic acid (PA). The preparation of the PA solution adhered to a method referenced in a previous study. 11 Then, OA was dissolved in 10 mL of a solution containing 25% bovine serum albumin (BSA) to prepare a 20 mM stock solution, whereas FFAs were kept at a 2:1 OA-to-PA ratio. The final working concentrations of 100 and 200 μM FFA mixture (OA:PA = 2:1) were applied to AML-12 cells for 24 h in complete DMEM/F12 medium containing 1% fatty acid-free BSA. Vehicle control (1% BSA without FFA) and untreated cells were included as controls. Optimal lipid accumulation was confirmed at 200 μM by oil red O staining.
qRT-PCR analysis
AML-12 cells and mouse liver tissues were subjected to RNA extraction using TRIzol reagent (Invitrogen, Karlsruhe, Germany). Subsequently, reverse transcription was performed to synthesize cDNA using a random primer-based reverse transcription kit (Promega, Madison, WI, USA). The SYBR Green master mix (Promega, Madison, WI, USA) was used in an Mx3005P QPCR System (Stratagene, La Jolla, CA, USA) to perform real-time quantitative reverse transcription-polymerase chain reaction (qRT-PCR) as per the directions stipulated by the manufacturer. The housekeeping gene for circDock6 was glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
The 2−ΔΔCt normalization method served as the analytical approach for determining circRNA expression.18,19 The following qRT-PCR primers were applied:
Forward: 5′-GGCCTAACTTTGTAGCCAAGGC-3′
Reverse: 5′-GGCCAGAGGGTCTTGGACAT-3′
Forward: 5′-TCGACAGTCAGCCGCATCTTCTTT-3′
Reverse: 5′-ACCAAATCCGTTGACTCCGACCTT-3′
Cell transfection
All small interfering RNAs (siRNAs) were designed and synthesized by Tsingke Biotechnology (Nanjing, China), which were used to knockdown the expression of circDock6. The siRNA–circDock6 sequences were as follows:
Forward: 5′-UGCCCCUGCAAGUUUCUCUTT-3′
Reverse: 5′-AGAGAAACUUGCAGGGGCATT-3′
Forward: 5′-UCUCCUGCCCCUGCAAGUUTT-3′
Reverse: 5′-AACUUGCAGGGGCAGGAGATT-3′
Forward: 5′-UCCUGCCCCUGCAAGUUUCTT-3
Reverse: 5′-GAAACUUGCAGGGGCAGGATT-3′
Forward: 5′-UUCUCCGAACGUGUCACGUTT-3′
Reverse: 5′-ACGUGACACGUUCGGAGAATT-3′
Forward: 5′-GGCUUCCUUACCCCUGUCATT-3′
Reverse: 5′-UGACAGGGGUAAGGAAGCCTT-3′
Lipofectamine 2000 (Invitrogen) was used for AML-12 cell transfection as per the directions provided by the manufacturer. A concentration of 100 nM siRNA–circDock6-2 was found to be the most effective for AML-12 cell interference.
Oil red O staining
Initially, a solution of oil red in isopropanol was formulated at a concentration of 0.5%. Then, a 3:2 mixture of the stock solution and PBS was prepared, and the mixture was allowed to stand for 30 min. A 0.45-µm filter was then used to filter the mixture. The mixture was left to sediment for 20 min before being passed through a 0.22-µm filter. Finally, the formation of lipid droplets was observed by incubating the cells for 20 min.
Triglyceride detection
The cells were subjected to a 10-min lysate lysis, 10-min heating process at 70°C, and 5-min centrifugation at 2000 rpm and ambient temperature. The triglyceride content was then determined using a glycerol-3-phosphate oxidase–phenol 4-aminoantipyrine peroxidase (GPO-PAP) reagent kit (BA1589, Applygen Technologies, Beijing, China) according to the manufacturer’s instructions. The absorbance was measured at 500 nm with a microplate reader (iMark, Bio-Rad Laboratories, Hercules, CA, USA).
Western blotting
Protein was obtained by lysing the cells in a lysis buffer and centrifuging them moderately. Next, 10% sodium dodecyl sulfate–polyacrylamide gels were used to separate approximately 20 µg of protein per lane, which was subsequently loaded onto PVDF membranes (Millipore, Billerica, MA, USA). Thereafter, the membrane was immunoblotted with primary antibodies against anti-β-actin (NB600-501, 1:1000, Bio-Techne) and anti-IGF1R (ab182408, 1:1000, Abcam) as controls. After incubating the membrane with the appropriate mouse or rabbit secondary antibody, the intensity of each protein band was quantified using an Odyssey Infrared Imaging System (LI-COR, Lincoln, NE, USA).
Data analysis
All experiments included a minimum of three independent biological replicates. Data were presented as mean ± standard deviation. All data met normality (Shapiro–Wilk test) and equal variance assumptions. A two-tailed Student’s t-test was applied for unpaired comparisons. All analyses were conducted using SPSS 22 (SPSS Inc., Chicago, IL), with statistical significance set at p < 0.05.
Results
OA-induced steatosis modeling in AML-12 hepatocytes
We esterified AML 12 cells using two doses of OA, PA, and FFA—100 and 200 µM—simultaneously using untreated cells as controls. After 24 h of incubation, staining with oil red O revealed that cell esterification was most effective at a concentration of 200 µM OA (Figure 1).
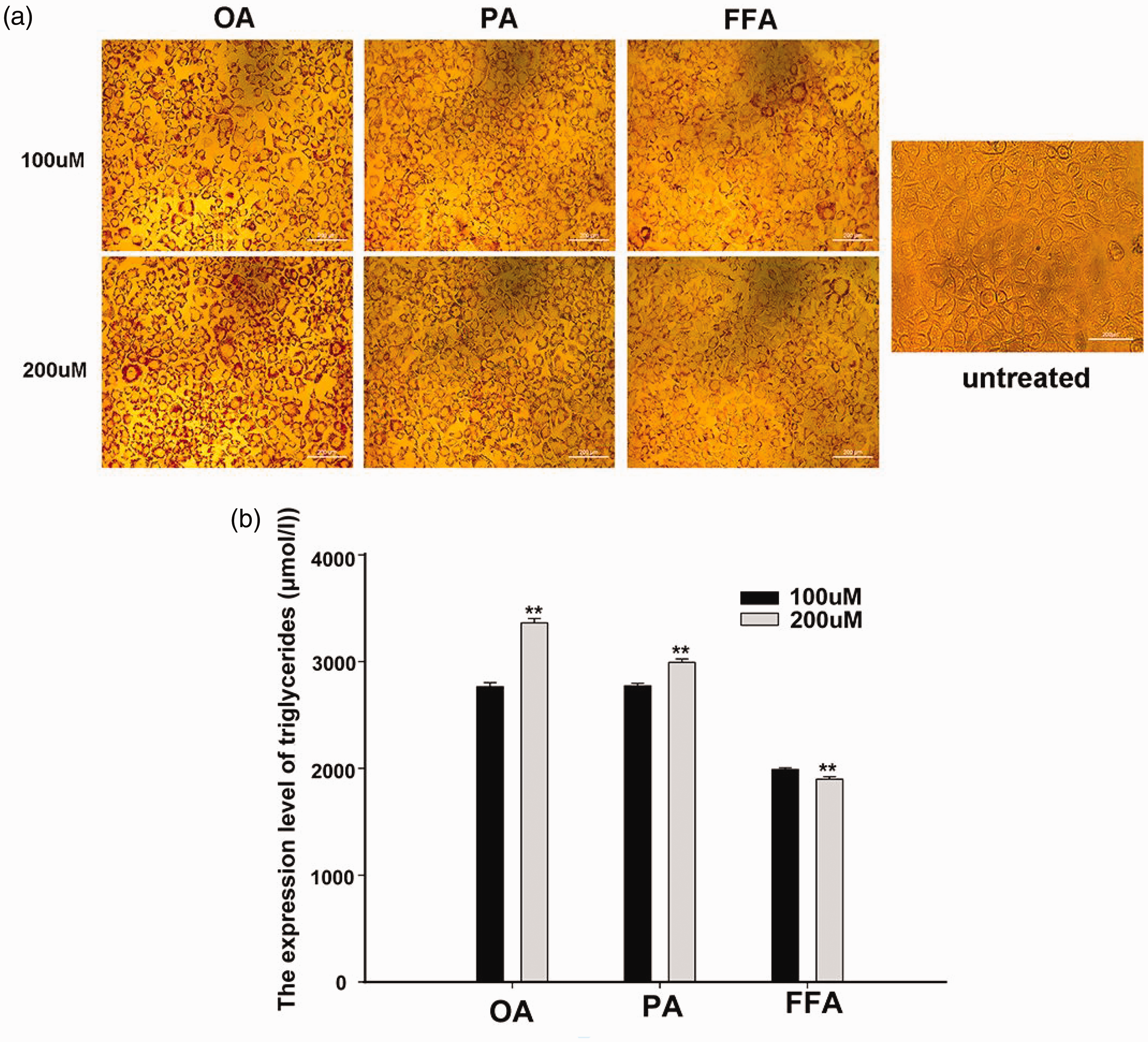
AML-12 lipidized cell model constructed using FFA, PA, and OA compared with untreated cell. (a) Esterification effects of OA, PA, and FFA at different concentrations and untreated cells and (b) concentration of triglycerides after esterification. FFA: free fatty acids; OA: oleic acid; PA: palmitic acid; AML: alpha mouse liver.
CircDock6 upregulation in HFD murine liver tissues
The animal experimental design is shown in Figure 1(a). To validate the chip-predicted circDock6 expression in MASLD, 11 PCR analysis was conducted on liver tissues from 10 SD-fed and 10 HFD-fed mice. Consistent with the chip results, the data showed significant overexpression of circDock6 in HFD liver tissues (Figure 2(b)).

CircDock6 expression levels were detected in SD and HFD tissues. (a) A schematic diagram illustrating the experimental design of the animal study. (b) The expression of circDock6 was considerably higher in HFD tissues than in SD tissues. (c) HFD samples showed significant circDock6 expression, according to standardized SD data and (d) SD samples showed low circDock6 expression based on standardized HFD data. *p < 0.05, **p < 0.01, ***p < 0.001. SD: standard diet; HFD: high-fat diet.
As the liver tissues from the SD and HFD groups were not paired, the circDock6 expression levels of each HFD liver tissue were visualized by standardizing them against the liver tissues from 10 SD-fed mice. The analysis demonstrated a substantial overexpression in HFD liver tissues relative to the standardized SD samples (Figure 2(c)). Similarly, the expression levels in SD liver tissues were evaluated following the standardization of data from 10 HFD liver tissues, showing the opposite trend (Figure 2(d)).
CircDock6 silencing ameliorates lipid accumulation in cultured hepatocytes
Three siRNAs targeting circDock6 were designed and synthesized based on the distinctive structure of circRNA. The inhibitory effects of these siRNAs on circDock6 expression were evaluated by transfecting them into lipid-enriched AML-12 cells at doses of 80, 100, and 120 nM. According to experimental data, the most effective suppression was achieved by siRNA2–circDock6 at 100 nM (Figure 3(a)). After determining that 100 nM siRNA was the optimal interference concentration, to avoid off-target effects, negative control (NC) and scrambled siRNAs were used as negative controls. The results confirmed that the observed interference was not due to off-target effects, allowing us to proceed with conventional NC as the negative control in subsequent experiments (Figure 3(b)).

Steatosis in MASLD can be inhibited by silencing the expression of circDock6. (a) The most significant interference effect in AML-12 cells was exhibited by screened siRNA2. (b) Verification of the interference effect of siRNA2 on two negative controls, NC and scramble siRNA2. (c) Lipid droplet formation may be inhibited by low circDock6 expression and (d) knockdown of circDock6 expression resulted in a reduction in triglyceride levels. *p < 0.05, **p < 0.01, ***p < 0.001. MASLD: metabolic dysfunction–associated steatotic liver disease; AML: alpha mouse liver.
The regulatory function of circDock6 in lipid-enriched AML-12 cells associated with MASLD was investigated through oil red O staining analysis and triglyceride quantification after circDock6 knockdown, with NC and scramble siRNA as the negative control. Silencing of circDock6 dramatically suppressed the lipid droplet formation (Figure 3(c)) and led to a marked decrease in triglyceride levels (Figure 3(d)).
In vitro mechanistic dissection of the circDock6/mmu-let-7g-5p/IGF1R axis in hepatocyte models
According to the abovementioned results, circDock6 suppression successfully lowered triglyceride levels and prevented lipid droplet accumulation. However, the specific regulatory molecules in question have not yet been identified. We assessed the expression of mmu-let-7g-3p, mmu-let-7g-5p, mmu-miR-138-5p, and mmu-let-7i-5p, which were identified through gene chip predictions as potential circDock6-binding miRNAs.
PCR analysis indicated that among the gene chip-predicted miRNAs, mmu-let-7g-5p demonstrated the most substantial overexpression following circDock6 silencing (Figure 4(a)). In addition, the binding sites between circDock6 and mmu-let-7g-5p were identified (Figure 4(b)). Moreover, the mmu-let-7g-5p target gene IGF1R showed a corresponding decrease in both mRNA and protein expression levels (Figure 4(c) and (d)).
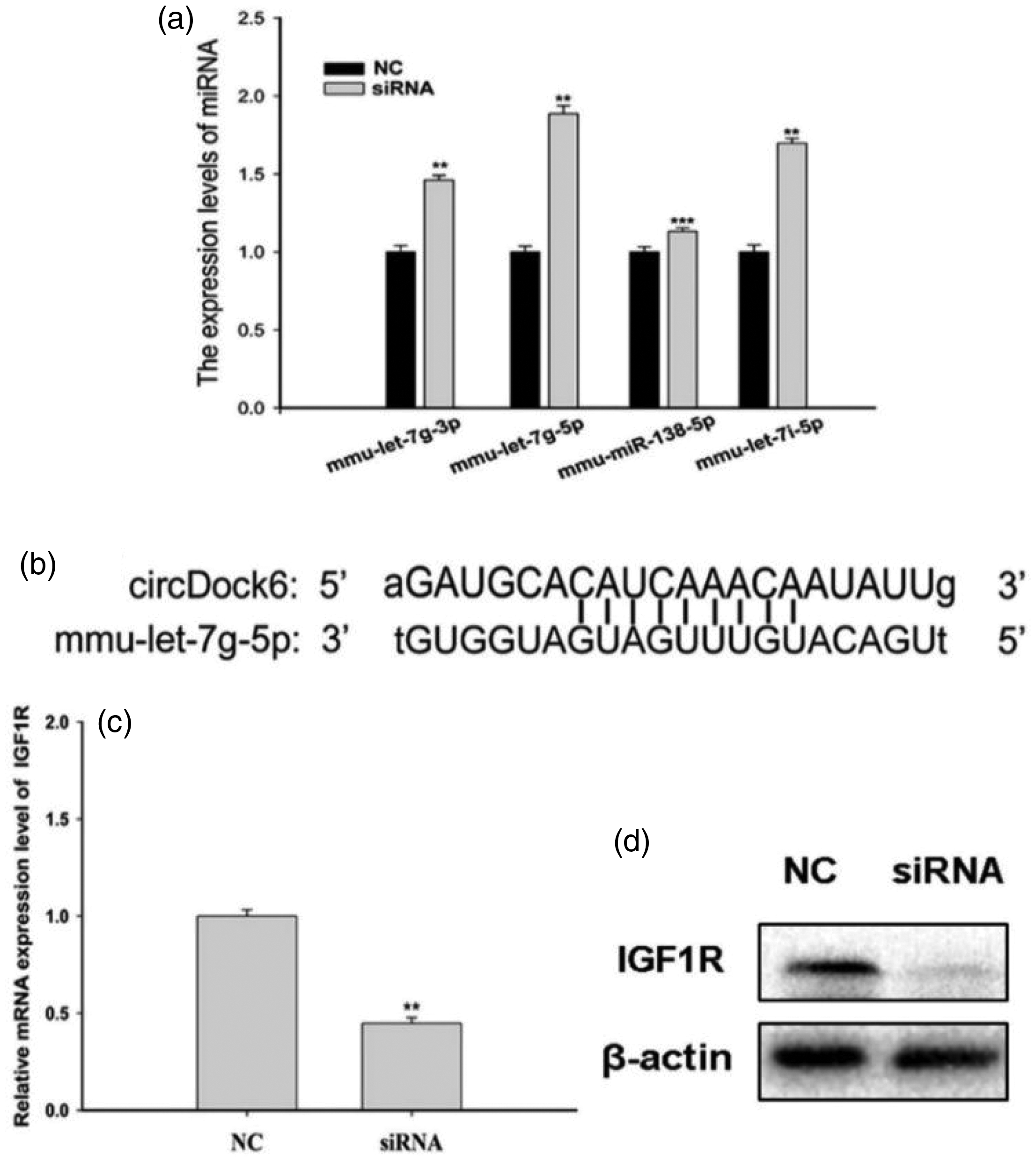
The change in mmu-let-7g-5p and its target gene IGF1R expression levels upon circDock6 knockdown. (a) miRNA expression levels when circDock6 expression is knocked down. (b) CircDock6 functions as a sponge for mmu-let-7g-5p and (c) the expression levels of circDock6 and IGF1R were downregulated. *p < 0.05, **p < 0.01, ***p < 0.001. IGF1R: insulin-like growth factor 1 receptor.
Genetic rescue validates circDock6-dependent regulation in lipid-loaded hepatocytes
A rescue experiment was conducted to confirm that circDock6 modulates MASLD by interacting with mmu-let-7g-5p and the subsequent indirect regulation of its target gene IGF1R. Both circDock6 and mmu-let-7g-5p were silenced concurrently in this experiment to evaluate their impact on triglyceride levels and lipid droplet formation.
The findings showed no appreciable changes in triglyceride content or lipid droplet formation when circDock6 and mmu-let-7g-5p were co-suppressed (Figure 5(a) and (b)). Similarly, no change was observed in the levels of IGF1R mRNA and protein expression (Figure 5(c) and (d)).

Steatosis in MASLD and IGF1R expression were investigated in a rescue experiment. (a, b) There was no significant change in the triglyceride levels or lipid droplets and (c, d) no significant changes were observed in IGF1R mRNA or protein levels. MASLD: metabolic dysfunction–associated steatotic liver disease; IGF1R: insulin-like growth factor 1 receptor.
Discussion
MASLD ranks among the most widespread liver conditions globally as a metabolic condition hallmarked by pathological triglyceride accumulation in hepatocytes.20–22 As MASLD can advance to serious complications such as liver failure, HCC, and cirrhosis, its increasing incidence poses serious health concerns.1,4,6
Research on MASLD increasingly indicates circRNAs as critical factors in its pathogenesis and progression. CircRNA SCAR, found in mitochondria, has been linked to postinflammation in recent years, underscoring the essential function of circRNAs in early-stage detection. 23 Notably, previous studies on nonalcoholic fatty liver disease (NAFLD) revealed that circ_0057558 regulates disease progression through the ROCK1/AMPK signaling pathway by functioning as a miR-206 sponge. 24 Accumulating evidence from systematic reviews further suggests that circRNAs serve as promising therapeutic targets across the spectrum of fatty liver diseases, including both NAFLD and its newly defined successor MASLD. 25
This study investigated the function of circDock6 and its underlying mechanism in MASLD. Initial microarray analysis indicated aberrant MASLD expression. In line with the microarray results, tissue-based studies confirmed the significant upregulation of circDock6 expression in HFD liver tissues compared with that in SD liver tissues. Further cellular research showed that circDock6 expression suppression dramatically decreased MASLD-related steatosis in lipid-loaded AML-12 cells.
The mechanistic pathway of the effects of circDock6 on MASLD was verified by examining and confirming the expression of mmu-let-7g-5p, which was determined to be a circDock6-interacting molecule based on chip predictions. Our results demonstrated the upregulation of mmu-let-7g-5p when circDock6 was silenced and the concurrent downregulation of its target gene, IGF1R. Furthermore, cell rescue studies showed that neither the IGF1R protein expression levels nor the steatosis associated with MASLD were markedly affected by the simultaneous knockdown of circDock6 and mmu-let-7g-5p.
IGF1R is a transmembrane tyrosine kinase that is frequently overexpressed in tumors 26 and plays important roles in systemic metabolic actions, cancer growth, proliferation, migration, and metastasis.27,28 It is also predominantly expressed in liver tissues, where it plays crucial roles in liver function and processes such as liver fibrosis. Given the involvement of IGF1R in liver fibrosis, it is particularly relevant in the context of NAFLD, wherein liver fibrosis is a key pathological feature. In addition, IGF1R is involved in DNA damage repair, cell senescence, lipid metabolism, and oxidative stress. 29 Activation of IGF1R leads to the autophosphorylation of tyrosine residues in the kinase domain, triggering multiple downstream signaling pathways, such as MAPK, PI3K/Akt, JAK/STAT, and mTOR. These pathways collectively regulate key cellular processes, including growth, transformation, differentiation, and survival, while also influencing cell metabolism and preventing apoptosis. 30 Such pathways are particularly important in NAFLD, as they may contribute to the progression from simple steatosis to more severe liver diseases, including fibrosis and HCC. Therefore, the current strategy for anticancer treatment focuses on the development of IGF1R inhibitors.
In this experimental study, we found that knocking down circDock6 in cells led to an increase in mmu-let-7g-5p expression, acting as a molecular sponge, whereas the expression of its target gene, IGF1R, was downregulated. Based on these findings, we conducted rescue experiments to downregulate mmu-let-7g-5p expression by knocking down circDock6. Surprisingly, no change was observed in IGF1R expression. These findings provide new insights into potential strategies for blocking the IGF1R effect. However, practical challenges remain in translating circDock6-based therapies to clinical settings, such as efficient delivery systems and ensuring their stability and safety in vivo. Although circDock6 can bind to multiple miRNAs, our experimental research found that its binding effect is most notable when it binds to mmu-let-7g-5p, as all molecules have intricate relationships with each other. As a novel noncoding RNA, the unique circular structure of circDock6 makes it resistant to degradation by RNase R, ensuring its stability in vivo. This stability is crucial for its potential in future research and suggests promising therapeutic applications. However, the therapeutic relevance of modulating the circDock6/let-7g-5p/IGF1R axis needs further critical evaluation, particularly in the context of NAFLD, wherein the modulation of this pathway could offer new treatment avenues. Moreover, the feasibility and safety of circDock6-based treatments still face challenges, requiring more experimental validation and human data.
In summary, the findings demonstrated the significant overexpression of circDock6 in liver tissues from HFD-fed mice and its function as a molecular sponge for mmu-let-7g-5p, consequently influencing IGF1R expression and promoting MASLD development.
This study has several limitations. First, the study focused primarily on in vitro experiments, and previous in vivo studies only verified fatty liver modeling and circDock6 expression in HFD versus SD mouse liver tissues, omitting liver histology analysis such as hematoxylin & eosin staining. These will be addressed in future in vivo studies. In addition, the in vivo knockdown of circDock6 was not performed, but we plan to conduct these experiments to strengthen the findings. We also aim to conduct circDock6 knockout studies in mice. Second, we intend to validate circDock6 expression in human liver specimens, with plans to explore its potential as an early biomarker of MASLD after collecting sufficient tissue samples.
Footnotes
Acknowledgments
None.
Author contributions
The experiments were conducted by HP L. The study was designed by XYD. PFL contributed to the concept of the study, prepared the manuscript, and performed the data analysis.
Availability of supporting data
Requests for the data supporting the findings of this study can be addressed to the corresponding author.
Consent for publication
Not applicable.
Declaration of conflicting interests
None.
Ethical approval
The animal experiments were approved by the Experimental Animal Ethics Committee of Ningbo University (Approval No. AEWC-NBU20220198; 21 March 2022) and were conducted in strict accordance with relevant guidelines and regulations, following the ARRIVE guidelines for reporting animal research.
Funding
The Natural Science Foundation of Zhejiang Provincial [No. LQ18H160015] and Ningbo Science and Technology Bureau | Natural Science Foundation of Ningbo [No. 2022J222] provided funding for this study.
