Abstract
Background:
Metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD), is an increasingly prevalent liver disorder.
Objectives:
This study investigated the effect of smoking status on various clinical outcomes in MASLD and metabolic dysfunction and alcohol-associated liver disease (MetALD).
Design:
This study is a retrospective cohort analysis utilizing data from the UK Biobank (Application ID: 117214). Participants were categorized as current, previous, or never smokers, and outcomes were analyzed using inverse probability of treatment weighting to adjust for confounders.
Methods:
The primary outcomes were all-cause mortality and liver-related mortality. Secondary outcomes included incidence of liver cirrhosis, hepatic decompensation, cardio-cerebrovascular diseases (CVD), and hepatocellular carcinoma (HCC). Multivariate Cox proportional hazards models were employed to evaluate associations.
Results:
Previous and never smokers had significantly lower hazard ratios (HRs) for mortality compared to current smokers in all cohorts (HR: 0.33, 95% confidence interval (CI): 0.31–0.35, p < 0.001 for never smokers in No SLD cohort, HR: 0.43, 95% CI: 0.41–0.44, p < 0.001 for never smokers in MASLD cohort, and HR: 0.41, 95% CI: 0.38–0.45, p < 0.001 for never smokers in MetALD cohort). Previous and never smokers showed significantly lower incidences of liver cirrhosis compared to current smokers across all cohorts, except for MetALD. Previous and never smokers showed lower incidences of CVD compared to current smokers. In the MASLD cohort, never smokers had the lowest incidence of hepatic decompensation and HCC. In the MetALD cohort, no significant differences were observed in the risk of hepatic decompensation and HCC between different smoking statuses.
Conclusion:
Smoking is related to worse survival outcomes and higher incidences of liver cirrhosis and CVD in MASLD and MetALD cohorts. Therefore, smoking cessation and prevention are crucial strategies for reducing the burden of liver disease and improving patient prognosis.
Plain language summary
Metabolic dysfunction-associated steatotic liver disease (MASLD), a common liver condition, can lead to serious health problems like liver failure, heart disease, and liver cancer. Smoking is a major health risk, but we do not fully understand how it affects people with MASLD or metabolic dysfunction and alcohol-associated liver disease (MetALD). This research was done to find out how smoking impacts survival and disease progression in people with these liver diseases so that doctors and healthcare providers can better support patients.
The researchers used information from the UK Biobank, a large health study that follows many people over time. They compared three groups of people with MASLD or MetALD: those who currently smoke, those who quit smoking, and those who have never smoked. They looked at how smoking affects survival, liver-related health problems, and other illnesses like heart disease and strokes.
The study found that people who smoke have a higher risk of dying from any cause or from liver disease than people who quit or never smoked. Smokers were also more likely to develop severe liver problems like liver cirrhosis, heart disease, and strokes. People who never smoked had the best health outcomes, with lower risks of liver and heart-related problems. However, for those with MetALD, alcohol use had a stronger effect on health than smoking.
This study shows that quitting smoking—or never starting—can improve survival and reduce the risk of severe health problems in people with MASLD. It highlights the need for healthcare programs that support smoking cessation and prevention, especially for people with liver diseases. These efforts could save lives and improve quality of care for millions of people worldwide.
Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD), is an increasingly prevalent liver disorder, which is characterized by hepatic steatosis not induced by significant alcohol consumption. 1 In the new nomenclature era, steatotic liver disease (SLD) is divided into two distinct subcategories based on the presence of cardiometabolic risk factors, and those with the risk factors are further categorized into MASLD and metabolic dysfunction and alcohol-associated liver disease (MetALD), depending on the underlying cause and alcohol consumption. 2 MASLD could develop into a spectrum of chronic liver disease, from simple steatosis to steatohepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma (HCC). 3 The rising prevalence of MASLD is closely linked to the global epidemics of obesity, type 2 diabetes, and metabolic syndrome, which are major public health problems. 4 MASLD affects approximately 30% of the global adult population with higher prevalence rates in Western countries and a growing prevalence in Asia. 5 Over the past 30 years, the prevalence of MASLD has increased from 17.6% in 1990 to 23.4% in 2019 with an increase of 1.0% annually. 6 The increasing incidence of MASLD is a significant contributor to liver-related morbidity worldwide.
Meanwhile, smoking is one of the well-known risk factors for numerous chronic diseases, including cardiovascular disease, chronic obstructive pulmonary disease, and various malignancies.7 –9 Previous studies have expressed the crucial role of smoking in the aggravation of liver disease. A research demonstrated that smokers with NAFLD were more likely to develop advanced liver fibrosis compared to non-smokers, suggesting that smoking could exacerbate liver injury through enhanced oxidative stress and inflammatory pathways. 10 A large cohort study showed that smoking significantly increases all-cause mortality among patients with chronic liver disease such as NAFLD, and the other study found that smokers had a higher risk of liver cirrhosis and HCC, emphasizing the adverse effects of smoking on liver physiology.11,12 Furthermore, another study reported that NAFLD patients who smoked had a significantly higher risk of cardiovascular disease with carotid stenosis, which could further aggravate their overall prognosis.13,14 Further study revealed that smoking has an adverse effect on the development of NAFLD, and cessation could be an effective way to prevent the disease. 15
Despite existing studies, comprehensive research comparing the impact of smoking on various liver-related and systemic outcomes in MASLD and MetALD patients is limited. This study utilized data from the UK Biobank, a large-scale biomedical database, to analyze overall and liver-related survival, incidence of liver cirrhosis, hepatic decompensation events, and cardiovascular and cerebrovascular events in MASLD and MetALD cohorts, considering the dynamics of smoking status.
Materials and methods
Study design and data source
The study utilized data from the UK Biobank. Specifically, this study focused on participants identified without SLD (No SLD), with MASLD, or MetALD, and investigated the relationship between smoking status and various health outcomes using Data-Field number 1498. The UK Biobank Application ID for this research is 117214. The UK Biobank received ethical approval from the North West Multi-Centre Research Ethics Committee. All participants provided written informed consent. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement.
Study population
Among the UK biobank cohort, individuals without clinical information, such as demographic information, alcohol consumption, and underlying comorbidities, or with alcohol-associated liver disease, autoimmune hepatitis, chronic viral hepatitis, primary biliary cirrhosis, and cryptogenic hepatitis were excluded. The study included participants diagnosed with MASLD and MetALD based on cardiometabolic and other criteria previously suggested. 1 MASLD is defined in individuals with hepatic steatosis not induced by significant alcohol consumption, with the presence of cardiometabolic risk factors, such as obesity, type 2 diabetes, dyslipidemia, or hypertension. 16 MetALD is diagnosed as MASLD with increased alcohol intake, where alcohol consumption exceeds 30 g/day for men and 20 g/day for women. 17 SLD was defined as individuals with scores above 60, measured by fatty liver index (FLI). 18 Formula of FLI is as follows.
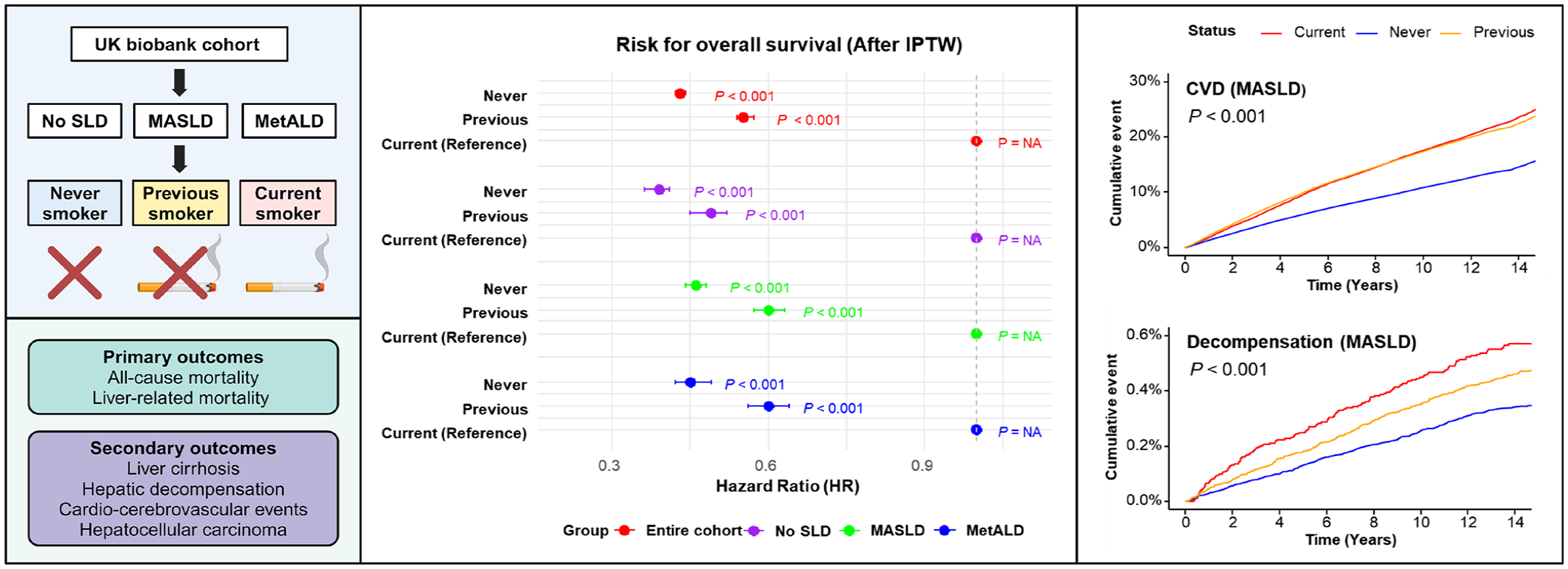
The population group was categorized into three groups based on smoking status: current smokers, previous smokers, and never smokers (Supplemental Figure 1).
The primary and secondary outcomes
The primary outcomes were all-cause mortality and liver-related mortality based on smoking status in each cohort. These outcomes were assessed by the time from diagnosis of MASLD or MetALD to death from any cause or liver-specific causes. The secondary outcomes were the incidence of liver cirrhosis, occurrence of hepatic decompensation events, cardio-cerebrovascular events, and occurrence of HCC. Development of liver cirrhosis during the follow-up period and decompensation events were defined as esophageal varices, portal hypertension, hepatorenal syndrome, or ascites19,20 (Supplemental Table 1).
Statistical analysis
Baseline characteristics of the study population were summarized using means and standard deviations for continuous variables, and frequencies and percentages for categorical variables. Kaplan–Meier survival curves were generated for overall and liver-related survival. Cox proportional hazard models were utilized to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between smoking status and survival outcomes, adjusted for potential confounders. The incidence rates of liver cirrhosis, hepatic decompensation events, and cardio-cerebrovascular events were calculated and compared between smoking groups. Cox proportional hazard models were used to evaluate the risk associated with different smoking statuses. A p value of less than 0.05 was considered to be statistically significant. Inverse probability of treatment weighting (IPTW) was utilized to adjust for potential confounders to balance the baseline characteristics among the smoking groups. Propensity scores for each participant were estimated using logistic regression models that included relevant variables. Participants were weighted by the inverse of their propensity score to create a pseudo-population, where the distribution of covariates was similar between smoking groups. All statistical analyses were conducted using R software (R version 4.2.3; R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics of the study population
Baseline clinical characteristics were compared based on smoking status within the entire cohort, No SLD, MASLD, and MetALD groups both before and after IPTW adjustment. IPTW adjustments effectively balanced these characteristics, as indicated by standardized mean differences (SMDs) approaching zero in the adjusted cohorts (Table 1, Supplemental Table 2, and Supplemental Figure 2). In each cohort, never smokers consistently form the largest group, followed by previous smokers and then current smokers. For instance, in the overall cohort, 55.29% are never smokers, 34.35% are previous smokers, and 10.35% are current smokers, with similar trends observed in other cohorts (Figure 1 and Supplemental Table 3).
Baseline clinical characteristics based on the liver disease status.

The table presents the baseline clinical characteristics of the study population categorized by smoking status (current, previous, and never smokers) between the entire cohort, No SLD, MASLD, and MetALD cohorts. Characteristics include sex, age at recruitment, physical activity, BMI, waist circumference, type 2 diabetes, dyslipidemia, hypertension, ALT, GGT, platelet count, and albumin levels. IPTW adjustments were applied to balance these characteristics. Data are described as mean ± standard deviation or n (%).
ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; IPTW, inverse probability of treatment weighting; MASLD, metabolic dysfunction-associated steatotic liver disease; MetALD, metabolic dysfunction and alcohol-associated liver disease; SLD, steatotic liver disease; SMD, standardized mean difference.

Distribution of smoking statuses across study cohorts. Proportion of current smokers, previous smokers, and never smokers. Data are presented as percentages within each cohort.
Analysis of survival across smoking statuses
The study revealed differences in overall survival (OS) with all-cause mortality and liver-related mortality among current, previous, and never smokers across various cohorts both before and after IPTW adjustment. In the entire cohort, current smokers had significantly higher all-cause mortality and liver-related mortality compared to previous and never smokers, with results consistent before and after IPTW adjustment (Supplemental Figure 3(A)–(E)). In the No SLD cohort, current smokers had significantly higher all-cause and liver-related mortality, compared to previous and never smokers (Supplemental Figure 4(A)–(E)). Within the MASLD cohort, current smokers had significantly higher all-cause and liver-related mortality compared to previous and never smokers, with consistent findings before and after IPTW adjustment (p < 0.001; Figures 2(a) and (b) and 3(a) and (b)). In the MetALD cohort, current smokers had significantly higher all-cause and liver-related mortality compared to previous and never smokers (p < 0.001; Figures 2(d) and (e) and 3(d) and (e)). Collectively, current smokers exhibited significantly higher all-cause and liver-related mortality across all cohorts of SLD, with findings remaining consistent before and after IPTW adjustment.

Overall and liver-related survival in MASLD and MetALD cohorts before IPTW adjustment. (a) OS in MASLD. (b) Liver-related survival in MASLD. (c) Incidence of liver cirrhosis in MASLD. (d) OS in MetALD. (e) Liver-related survival in MetALD. (f) Incidence of liver cirrhosis in MetALD. Statistical significance was determined using the log-rank test.

Overall and liver-related survival in MASLD and MetALD cohorts after IPTW adjustment. (a) OS in MASLD. (b) Liver-related survival in MASLD. (c) Incidence of liver cirrhosis in MASLD. (d) OS in MetALD. (e) Liver-related survival in MetALD. (f) Incidence of liver cirrhosis in MetALD. Statistical significance was determined using the log-rank test.
Analysis of liver cirrhosis incidence across smoking statuses
The study also investigated alterations of liver cirrhosis incidence among the smoking group, across the cohorts of liver disease. The entire cohort also showed a significantly higher incidence of liver cirrhosis in current smokers (Supplemental Figure 3(C) and (F)). A higher incidence of liver cirrhosis was observed in current smokers within the No SLD cohort, compared to previous and never smokers, with these trends remaining significant after IPTW adjustment (Supplemental Figure 4(C) and (F)). In addition, the incidence of liver cirrhosis was significantly higher in current smokers in the MASLD cohort (p < 0.001; Figures 2(c) and 3(c)). However, there was no significant difference in the incidence of liver cirrhosis between smoking statuses (Figures 2(f) and 3(f)). These data showed that current smokers have a significantly higher incidence of liver cirrhosis across the entire, No SLD, and MASLD cohort, while no significant difference was observed in the MetALD cohort.
Multivariate analysis of survival
We investigated the impacts of smoking status on survival across various cohorts with or without liver disease using multivariate analysis. In the No SLD cohort, previous smokers had a lower risk of all-cause and liver-related mortality compared to current smokers, and never smokers even had a lower risk, with remained significant trends after IPTW adjustment. Similarly, in the MASLD cohort, previous smokers exhibited lower all-cause and liver-related mortality compared to current smokers. In the MetALD cohort, both previous and never smokers showed significantly lower risks of all-cause and liver-related mortality compared to current smokers, both pre- and post-IPTW adjustment (Table 2, Supplemental Table 4, and Supplemental Figures 5 and 6). Overall, previous and never smokers had significantly lower risks of all-cause and liver-related mortality compared to current smokers across all cohorts, with trends remaining significant before and after IPTW adjustment.
Univariate and multivariate analyses for overall survival.
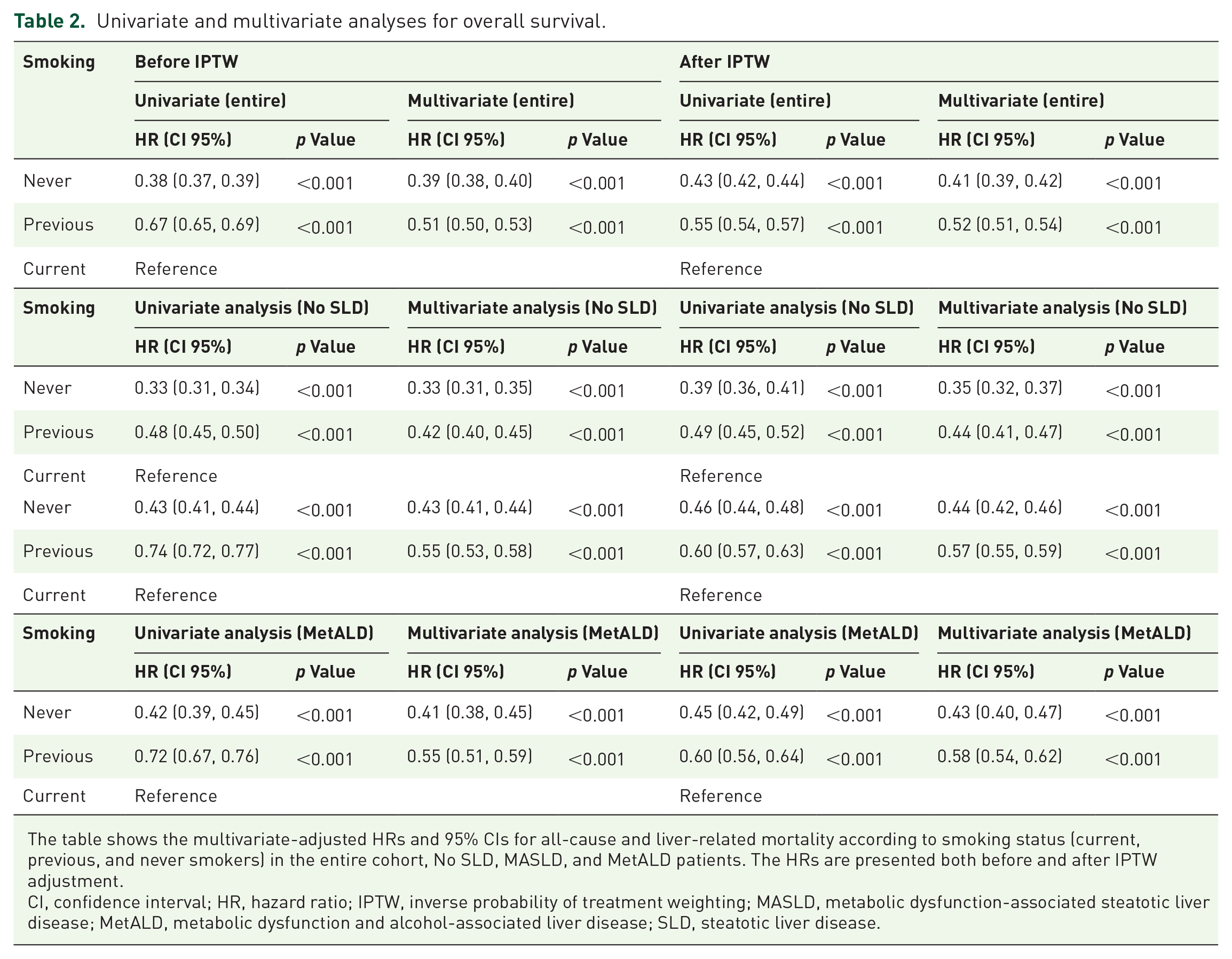
The table shows the multivariate-adjusted HRs and 95% CIs for all-cause and liver-related mortality according to smoking status (current, previous, and never smokers) in the entire cohort, No SLD, MASLD, and MetALD patients. The HRs are presented both before and after IPTW adjustment.
CI, confidence interval; HR, hazard ratio; IPTW, inverse probability of treatment weighting; MASLD, metabolic dysfunction-associated steatotic liver disease; MetALD, metabolic dysfunction and alcohol-associated liver disease; SLD, steatotic liver disease.
Multivariate analysis of liver cirrhosis incidence
We also revealed the effect of smoking status on the incidence of liver cirrhosis across the liver disease cohorts using multivariate analysis. In the No SLD cohort, previous smokers showed lower incidences of liver cirrhosis than current smokers, and never smokers showed the lowest incidence. In the MASLD cohort, only never smokers continued to show a significantly lower incidence compared to current smokers after IPTW adjustment. In the MetALD cohort, the difference of liver cirrhosis was not significantly different between smoking status (Supplemental Table 5 and Supplemental Figure 7). These data found that previous and never smokers had significantly lowered incidences of liver cirrhosis compared to current smokers, except for the MetALD cohort.
Effect of smoking on cardiovascular and cerebrovascular disease
We examined the effect of smoking status on the incidence of cardio-cerebrovascular disease (CVD) across different SLD cohorts. In the No SLD cohort, current smokers had a significantly higher incidence of CVD, cardiovascular events, and cerebrovascular events compared to previous and never smokers (p < 0.001; Figure 4(a), Supplemental Figure 8(A) and (D)). In the MASLD cohort, current smokers also had a significantly higher incidence of CVD, with current and previous smokers having similar rates, but both higher than never smokers. This was especially pronounced in cardiovascular events (p < 0.001; Figure 4(b), Supplemental Figure 8(B) and (E)). In the MetALD cohort, the trend was similar, with current and previous smokers having higher CVD incidence than never smokers, particularly for cardiovascular events (p < 0.001; Figure 4(c), Supplemental Figure 8(C) and (F)).

Incidence of cardio-cerebrovascular disease, and hepatic decompensation in No SLD, MASLD, and MetALD cohorts. (a) CVD in No SLD. (b) CVD in MASLD. (c) CVD in MetALD. (d) Hepatic decompensation in No SLD. (e) Hepatic decompensation in MASLD. (f) Hepatic decompensation in MetALD. Statistical significance was determined using the log-rank test.
We also analyzed the effect of smoking status on CVD and its subcategories across different SLD cohorts, with HRs and CIs. In the entire cohort, previous smokers had a significantly lower incidence of CVD, with never smokers having an even lower incidence compared to current smokers (Table 3). For cardiovascular and cerebrovascular events, previous and never smokers had progressively lower risks compared to current smokers (Supplemental Tables 6 and 7). In the No SLD cohort, similar trends were observed with previous and never smokers having lower incidences of CVD (Table 3, Supplemental Tables 6 and 7). In the MASLD cohort, previous smokers had a significantly lower incidence of CVD, but for cardiovascular events, only never smokers showed a significantly lower risk (Supplemental Table 6). In the MetALD cohort, although previous smokers did not show a significant difference in CVD incidence, never smokers were revealed to show a significant reduction in incidence (Table 3). For cardiovascular events, only never smokers had a significantly lower risk, while both previous and never smokers had lower risks for cerebrovascular events (Supplemental Tables 6 and 7).
Risk of cardio-cerebrovascular events according to the liver disease and smoking status.

The table displays the multivariate-adjusted HRs and 95% CIs for cardio-cerebrovascular events, according to smoking status (current, previous, and never smokers) in the entire cohort, No SLD, MASLD, and MetALD participants.
CI, confidence interval; HR, hazard ratio; MASLD, metabolic dysfunction-associated steatotic liver disease; MetALD, metabolic dysfunction and alcohol-associated liver disease; SLD, steatotic liver disease.
Collectively, current and previous smokers had a significantly higher incidence of CVD across all cohorts, with never smokers showing the lowest risk, and these trends remained consistent in both overall incidence and HR analyses.
Hepatic decompensation and smoking in various SLDs
We further investigated the effect of smoking status on the incidence of hepatic decompensation. In the entire cohort, previous smokers had a significantly lower incidence of hepatic decompensation, with never smokers having an even lower incidence compared to current smokers (Table 4). The most common decompensation event was ascites, followed by variceal bleeding (Supplemental Table 8). In the No SLD cohort, Kaplan–Meier analysis showed marginal differences between never and previous smokers, with never smokers having a slightly better outcome (p = 0.045; Figure 4(d)). In the MASLD cohort, the data revealed that never smokers had a significantly lower incidence of hepatic decompensation compared to previous and current smokers (p < 0.001; Figure 4(e)). The most common decompensation event was ascites, followed by esophageal varices (Supplemental Table 8). In the MetALD cohort, there was no significant difference in hepatic decompensation incidence between smoking statuses (p = 0.580; Figure 4(f)). Previous and never smokers did not show significantly lower incidences compared to current smokers (Table 4). Overall, previous and never smokers had significantly lower incidences of hepatic decompensation compared to current smokers in the No SLD and MASLD cohorts, with never smokers showing the lowest risk, whereas no significant difference was observed in the MetALD cohort.
Risk of hepatic decompensation according to the liver disease and smoking status.
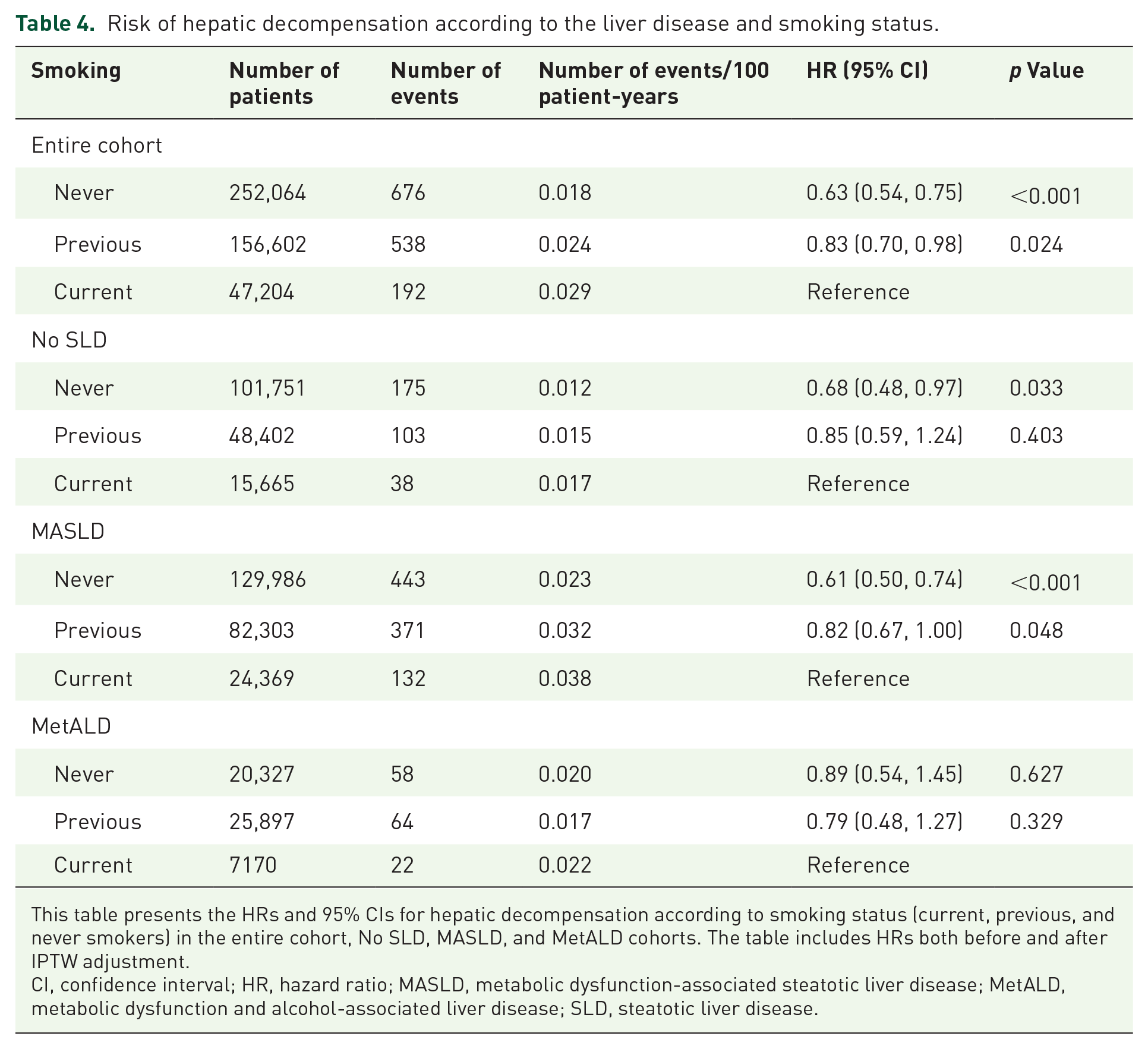
This table presents the HRs and 95% CIs for hepatic decompensation according to smoking status (current, previous, and never smokers) in the entire cohort, No SLD, MASLD, and MetALD cohorts. The table includes HRs both before and after IPTW adjustment.
CI, confidence interval; HR, hazard ratio; MASLD, metabolic dysfunction-associated steatotic liver disease; MetALD, metabolic dysfunction and alcohol-associated liver disease; SLD, steatotic liver disease.
Incidence of HCC and smoking in various SLDs
Furthermore, we analyzed the effect of smoking status on the incidence of HCC between different SLD cohorts. In the No SLD cohort, there was no significant difference in the incidence of HCC between smoking statuses (p = 0.490; Supplemental Figure 9(A)). In the MASLD cohort, there was a significant difference in the incidence of HCC between smoking statuses, with the lowest incidence in the never smoking group (p < 0.001; Supplemental Figure 9(B)). In the MetALD cohort, there was no significant difference in the incidence of HCC between smoking statuses (p = 0.077; Supplemental Figure 9(C)). When analyzing HCC risk relative to current smokers, never smokers in the entire cohort presented a significantly lower risk. However, in the No SLD cohort, neither never nor previous smokers showed a significant difference in HCC risk. By contrast, within the MASLD cohort, never smokers had a significantly reduced risk of HCC, whereas in the MetALD cohort, no significant differences were observed between smoking groups (Supplemental Table 9). These data showed that in the No SLD and MetALD cohorts, there was no significant difference in the incidence of HCC between smoking statuses, while in the MASLD cohort, never smokers had the lowest incidence of HCC with a significant difference between smoking statuses.
Discussion
This study provides a comprehensive analysis of the detrimental effects of smoking on clinical outcomes in MASLD and MetALD. Utilizing data from the UK Biobank, we evaluated the effects of smoking status on overall and liver-related survival, incidences of liver cirrhosis, hepatic decompensation, and cardiovascular and cerebrovascular events across different liver disease cohorts. Our findings indicate that current smokers generally had significantly higher risks of adverse health outcomes compared to previous and never smokers. However, some cohorts, particularly MetALD and No SLD, displayed insignificant results that require further discussion. Previous studies have shown that smoking exacerbates liver fibrosis and increases the risk of mortality in patients with NAFLD, which suggests a synergistic effect between smoking and liver disease progression. 21
Smoking exacerbates liver disease through several cellular and molecular pathways. Nicotine and the metabolites in cigarettes induce oxidative stress, resulting in the formation of reactive oxygen species (ROS) that could damage hepatocytes and promote liver fibrosis.22 –25 ROS lead to lipid peroxidation, protein oxidation, and DNA damage, inducing the development of a pro-inflammatory and fibrogenic environment. 26 Smoking also stimulates inflammatory pathways by activating pro-inflammatory cytokines such as tumor necrosis factor-alpha, interleukin-6, and interleukin-1 beta, which contribute to liver injury and fibrosis progression. 27 In addition, smoking impairs immune function, further exacerbating liver inflammation and damage. 28 One crucial aspect of smoking-induced liver damage is its effect on hepatic stellate cells (HSCs), which are the main collagen-producing cells in the liver. Activation of HSCs is a key event in the development of liver fibrosis. 29 Smoking has been shown to promote HSC activation by oxidative stress and inflammatory mediators. A study demonstrated that smoking-induced oxidative stress-activated HSCs, leading to increased collagen production and fibrosis. 30 This activation of HSCs by smoking contributes significantly to the fibrogenic response in the liver, exacerbating the progression of liver disease.
The findings of this research are consistent with previous studies that demonstrated smoking as a major risk factor for increased mortality in patients with liver disease.11,14,31 In the entire cohort, No SLD, and MASLD cohorts, the incidence of liver cirrhosis was significantly lower in previous and never smokers compared to current smokers. However, in the MetALD cohort, the incidence of liver cirrhosis did not differ significantly between smoking statuses. This lack of significance could be attributed to the confounding effects of alcohol consumption, which is a major risk factor for liver cirrhosis and may mask the adverse effect of smoking. This is particularly evident in the MetALD cohort, where the dominant pathogenic influence of alcohol may dilute the relative contribution of smoking to liver disease progression. Previous studies have suggested the overwhelming effect of alcohol on liver disease, indicating that even moderate alcohol consumption significantly exacerbates liver injury, independent of other factors.32,33 Alcohol consumption induces similar oxidative stress and inflammatory pathways as smoking, potentially masking the differences between smoking statuses. 34
The protective effect of never smoking was particularly evident in the MASLD cohort, where never smokers had the lowest incidence of liver cirrhosis. This revealed the compounded risks of smoking and metabolic dysfunction in accelerating liver disease progression. Previous studies have also reported that smoking exacerbates liver fibrosis and cirrhosis, reinforcing the requirement for smoking cessation in managing chronic liver diseases.12,35 On the other hand, current smokers had significantly higher incidences of CVD across all liver disease cohorts compared to previous and never smokers. The data are consistent with the established role of smoking as a risk factor for cardiovascular disease.36,37 The adverse effects of smoking on endothelial function, platelet aggregation, and lipid metabolism contribute to the increased risk of cardiovascular events. 36 Notably, in the MASLD and MetALD cohorts, the incidence of cardiovascular disease was similar between current and previous smokers, despite a significantly lower incidence in never smokers. This suggests that while smoking cessation reduces cardiovascular risk, never smoking provides the greatest benefit. The persistence of elevated risk in previous smokers may be due to lasting vascular damage from prior smoking.
Meanwhile, previous and never smokers exhibited significantly lower incidences of hepatic decompensation compared to current smokers in the entire, No SLD, and MASLD cohorts. However, the difference for previous smokers in the No SLD cohort was marginally significant, potentially due to the smaller sample size or the presence of other mitigating factors. In the MetALD cohort, there was no significant difference in hepatic decompensation incidence between smoking statuses, likely due to the influence of alcohol consumption, which is a predominant factor in liver decompensation.38,39 Hepatic decompensation, including esophageal varices, ascites, hepatic encephalopathy, and hepatorenal syndrome, represents a crucial turning point in liver disease progression, and the reduction in decompensation events among previous and never smokers highlights the benefits of smoking cessation.
The incidence of HCC was significantly lower in previous and never smokers compared to current smokers in the MASLD cohort, with never smokers showing the lowest incidence. This highlights the protective effects of smoking cessation and the demonstrated benefit of never smoking. However, in the No SLD and MetALD cohorts, there was no significant difference in HCC incidence between smoking statuses. In the No SLD cohort, the lack of significance may be due to the relatively lower baseline risk of HCC, while in the MetALD cohort, the confounding effect of alcohol consumption might have influenced the results. In addition, the relatively low number of HCC cases in the UK Biobank dataset may have also contributed to the lack of significance. Previous studies have documented the increased risk of HCC associated with smoking in patients with chronic liver disease, further reinforcing the importance of smoking cessation in cancer prevention.40,41 In the MASLD cohort, never smokers had the lowest incidence of HCC, emphasizing the importance of smoking prevention in reducing cancer risk in this population.
Despite the comprehensive characteristics of the research, there are several limitations in the current study. First, the reliance on self-reported smoking status may introduce recall bias, potentially affecting the accuracy of the smoking data. Second, we could not account for the duration and intensity of smoking, which could influence the severity of its effects on liver health. Further research should focus on the amount and duration of smoking, regarding the importance of liver disease. 31 Moreover, the potential confounding effects of other lifestyle factors, such as diet and physical activity, were not fully explored. The influence of genetic predispositions and the interaction between smoking and other comorbid conditions were also not examined in detail. In addition, our study was unable to account for polysubstance abuse, including cocaine, morphine, marijuana, inhalants, oral tobacco use, and passive smoking due to the lack of detailed data in the dataset. As polysubstance use is prevalent among smokers and individuals with liver disease, its potential impact on cardiovascular outcomes cannot be ruled out. 42 One limitation of our study is the inability to include hepatic encephalopathy, jaundice, and spontaneous bacterial peritonitis (SBP) in the definition of liver decompensation due to the lack of relevant data in the UK Biobank. While ascites, variceal bleeding, and hepatorenal syndrome are well-established markers of decompensation, the absence of hepatic encephalopathy and SBP could be a limitation, as they are critical clinical events in liver disease progression. In addition, jaundice alone is difficult to define as a decompensation event due to its varied etiologies. Lastly, while the IPTW adjustment aimed to balance baseline characteristics, residual confounding may still exist. Regarding IPTW adjustment, the potential influence of unmeasured time-varying confounders, such as changes in smoking behavior or disease progression, could not be accounted for in our analysis. Future longitudinal studies with detailed smoking histories and comprehensive lifestyle assessments are needed to further elucidate the relationship between smoking and liver disease progression.
Conclusion
In conclusion, this study highlights the significant adverse impact of smoking on clinical outcomes in patients with MASLD and MetALD. Current smokers may have higher risks of mortality and disease progression compared to previous and never smokers, with never smokers consistently showing the best outcomes. These findings emphasize the importance of smoking prevention and cessation programs to reduce the burden of liver disease and improve the prognosis of SLD patients.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251331315 – Supplemental material for A comprehensive analysis of the impact of smoking on adverse clinical outcomes of steatotic liver diseases
Supplemental material, sj-docx-1-tag-10.1177_17562848251331315 for A comprehensive analysis of the impact of smoking on adverse clinical outcomes of steatotic liver diseases by Keungmo Yang, Jaejun Lee, Ji Won Han, Hyun Yang, Seung Yun Chae, Beom Sun Chung and Tom Ryu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

