Abstract
Founded in 1983 in Paris by Professor R. Modigliani, the Groupe d’ETude des Affections Inflammatoires Digestives (GETAID) was established to develop collaborative clinical research on inflammatory bowel disease (IBD). This was innovative 40 years ago and remains challenging today. From the beginning, this multicenter group has aimed to address clinical questions arising from patients, physicians, and the IBD community by conducting clinical research on treatments through randomized controlled trials, prospective cohorts, index creation, and observational studies. GETAID has advanced IBD knowledge by publishing over 140 original articles in peer-reviewed journals. This review explores the history of the GETAID, how it functions, and its contribution to IBD knowledge over the past four decades. It illustrates the disruptive and innovative academic research conducted by an independent group of researchers and its potential future impact through examples.
Introduction
Approximately one century ago, ulcerative colitis (UC) followed by Crohn’s disease (CD) were recognized as forms of inflammatory bowel diseases (IBD). They were among the first diseases to be studied in a controlled trial, and the seminal study by the Oxford group, led by Professor Sydney Truelove in 1955, demonstrated the efficacy of intravenous steroids for treating severe UC attacks. 1 This study paved the way for clinical research in IBD. In this pioneering context, Professor Robert Modigliani created the Groupe d’Etude Thérapeutique des Affections Inflammatoires du Tube Digestif (GETAID) in Paris in 1983 with the aim of developing collaborative clinical research on IBD. This was an innovative approach 40 years ago, and it remains challenging today. Since the beginning, the objective of this multicenter group has been to address clinical questions arising from patients and physicians in their daily practice, as well as from the IBD community, by focusing on clinical research on treatments through randomized controlled trials, prospective cohorts, the creation of indices, and observational studies. This review will explore the history of GETAID, how it functions, and its contributions to IBD knowledge over the past four decades, as well as its potential future impact.
A brief story of the GETAID since the beginning
The pioneers’ era: 1983–2000
In the 1980s, the medical armamentarium was limited to salicylates, steroids, and immunomodulators, and strategies with induction and maintenance phases were emerging. In this context, the first GETAID trial aimed to assess the effectiveness of levamisole, an anti-parasitic agent with immunomodulatory properties, to avoid relapses in patients with CD.2,3 This multicenter, double-blind, placebo-controlled randomized trial enrolled 167 patients. Despite its negative results, this seminal trial, now completely forgotten by the IBD community, illustrates the GETAID standards that are still followed today: answering a relevant clinical question through independent, collaborative, academic research with robust methodology and strong statistical analysis, sometimes yielding negative results. The basis of GETAID was already there.
In the following years, the GETAID conducted other trials in CD evaluating the use of steroids or lymphocyte apheresis as induction therapy,4,5 mesalamine as maintenance therapy and for preventing postoperative recurrence,6,7 and mesalamine versus ciprofloxacin. 8 At the same time, the GETAID sought to standardize endoscopic findings in CD to build the first endoscopic index, the Crohn’s Disease Endoscopic Index of Severity (CDEIS), with the help of Jean-Yves Mary’s statistical expertise. 9 Until the recent development of the EASE-CD score, 10 the CDEIS was one of the only validated endoscopic scores in CD, alongside the SES-CD. Moreover, when compared with the Crohn’s Disease Activity Index to measure disease clinical activity, the group was the first to identify the poor correlation between clinical and endoscopic findings in CD. 11
Growth: 2000–2012
The advent of biologics dramatically changed the landscape of clinical research in IBD. New questions emerged regarding the combination of old and new drugs, treatment duration, and safety. Under the leadership of Marc Lémann and Jean-Frédéric Colombel, the GETAID grew rapidly during this period, expanding the number of centers, projects, and funding.
Among the GETAID studies from this period, the most remarkable randomized controlled trials were those evaluating a bridge strategy with infliximab or a placebo with azathioprine, 12 infliximab versus cyclosporine in patients admitted for an acute severe UC episode refractory to intravenous steroids, 13 methotrexate versus placebo in steroid-dependent UC, 14 and probiotics versus placebo for preventing endoscopic CD postoperative recurrence. 15
At the same time, the GETAID was one of the first to explore the concept of drug withdrawal in IBD through prospective cohorts of patients stopping thiopurines,16,17 a randomized placebo-controlled withdrawal of thiopurines trial in CD, 18 and the pioneer infliximab withdrawal study (STORI). 19 Importantly, some of these studies involved also academic centers from other European countries, increasing the sample size and the significance of the results through this international collaborative approach.
Because most of the investigators were also involved in translational research in their university hospital, the GETAID network also made significant contributions to basic research on IBD, leading to the discovery of NOD2/CARD15 polymorphisms associated with CD. 20 This led to the creation of a collaborative French basic research group on IBD called REMIND in 2006. At the beginning of the century, GETAID also supported the French prospective, population-based CESAME cohort, which aimed to describe the safety of drugs, especially thiopurines, which were the standard treatment for refractory IBD at that time. 21
During this productive period, the GETAID published more than 20 papers, most of which appeared in highly ranked peer-reviewed journals. Sadly, Marc Lémann passed away suddenly at the end of summer 2010, leaving the GETAID orphan. Marc not only managed the group, but he also mentored many emerging French IBD leaders. He developed new concepts in IBD such as drug de-escalation, disability, and bowel damage—he gave his name to the index of cumulative bowel destruction in CD. 22
Adulthood: 2012–2025
During this period, the GETAID board, first led by Edouard Louis and then by Laurent Peyrin-Biroulet, succeeded in continuing high-level clinical research in IBD with new projects. Notable studies from this period include cohort studies demonstrating the efficacy of anti-TNF in complicated CD,23,24 randomized controlled trials evaluating fibrin glue and fistula plug versus sham procedures in ano-perineal fistulating CD,25,26 proactive therapeutic drug monitoring with infliximab, 27 and infliximab versus azathioprine in patients with acute severe UC responding to intravenous steroids. 28
In line with previous GETAID studies, the SPARE trial, an international randomized controlled trial, explored anti-TNF de-escalation in CD. 29 Building on the CESAME cohort experience, the group decided to conduct, with ECCO’s support, a European prospective longitudinal observational cohort study to describe the safety of anti-IBD agents, including biologics, in a large real-life sample of patients. 30
The involvement of the GETAID in endoscopy continued with the description of elementary CD lesions, 31 reproducibility of the Rutgeerts score for postoperative recurrence, 32 comparison of endoscopic response to anti-TNF according to segmental CD location, 33 and prospective cohorts of patients screened for colorectal or small bowel dysplasia.34,35 Input from GETAID members and statisticians also helped develop and validate the Ulcerative Colitis Endoscopic Index of Severity (UCEIS). 36
Beyond clinical research, the GETAID developed educational activities and resources spread through annual meetings and masterclasses, and its website. The group issued French recommendations for UC in 2022 and for CD in 202437,38 in line with current knowledge and French specificities.
During this decade, associate GETAID groups of surgeons (GETAID-chirurgie) and pediatricians (GETAID-pédiatrique) were created. In collaboration with the GETAID, these groups developed specific projects through prospective cohort studies and trials.39,40
How does it work?
Initially, GETAID began with 20 academic tertiary IBD centers in France and Belgium. Over time, it expanded to include 64 active centers, including one out of four secondary French centers and even two from Switzerland (https://www.getaid.org).
The GETAID board is elected every 3 years by the centers and is composed of a president, vice president, general secretary, and treasurer. The board interacts with the scientific (SciCom) and educational (EduCom) committees, as well as the clinical operations team that now comprises 14 people coordinated by Mrs Charlotte Mailhat, who works in the GETAID office in Paris (Figure 1). The growth of the team over the past 10 years reflects the efforts made to meet the quality requirements expected by regulatory authorities when conducting human clinical research, as well as the increase in GETAID studies in response to the needs of the network.

GETAID organization in 2025.
The general principles of how GETAID functions have not changed since the beginning: four to six in-person meetings in Paris annually to discuss current and future IBD clinical projects, from conception to publication. These meetings include volunteer and unpaid investigators from all the centers. A project always starts with a “good clinical question.” This process is open to all GETAID members. Particular attention is paid to the training and inclusion of young gastroenterologists in all GETAID projects. Importantly, the GETAID statistician interacts with the board and each principal investigator at every stage of project development. Once the board has approved it, the principal investigator writes the study protocol, which is developed with the statistician and discussed step by step with the GETAID board and in the general assembly. At the same time, funding sources are sought, either from national or international academic grants, when pharmaceutical companies are uninterested or hostile. A lack of financial support meant that several projects could not be carried out. Alternatively, funding is sought from industry sponsors, but the studies are still promoted by GETAID (investigator-initiated studies).
Once the project is accepted by the group and receives financial support, interested centers are selected. Administrative procedures, including submissions to the ethics committee, insurance, health authorities, and hospital administrations, are carried out by the GETAID and are subject to an agreement between hospital research centers and GETAID to conduct the clinical trial. With the help of the operational team, the advancement of the study is followed and presented at each GETAID meeting until the end of recruitment. After the statistical analysis, a draft of the results is presented and validated before the congress submission and publication. The entire process, from conception to completion, takes 2–10 years, sometimes more. Importantly, the voluntary participation of investigators is key to developing the project and promoting young gastroenterologists.
How did GETAID impact the IBD landscape?
Over the past four decades, the group has aimed to address clinical questions raised by patients and/or their gastroenterologists, by conducting clinical research into treatments through randomized controlled trials, prospective cohorts, and cross-sectional studies, quantifying the severity of the disease when necessary, in these studies (Figure 2). This very innovative approach has advanced knowledge of IBD, with more than 140 original articles published in peer-reviewed journals—primarily in the best gastroenterology journals (Figure 3). We will provide a few examples to illustrate the disruptive and innovative academic research conducted by GETAID, an independent group of researchers.
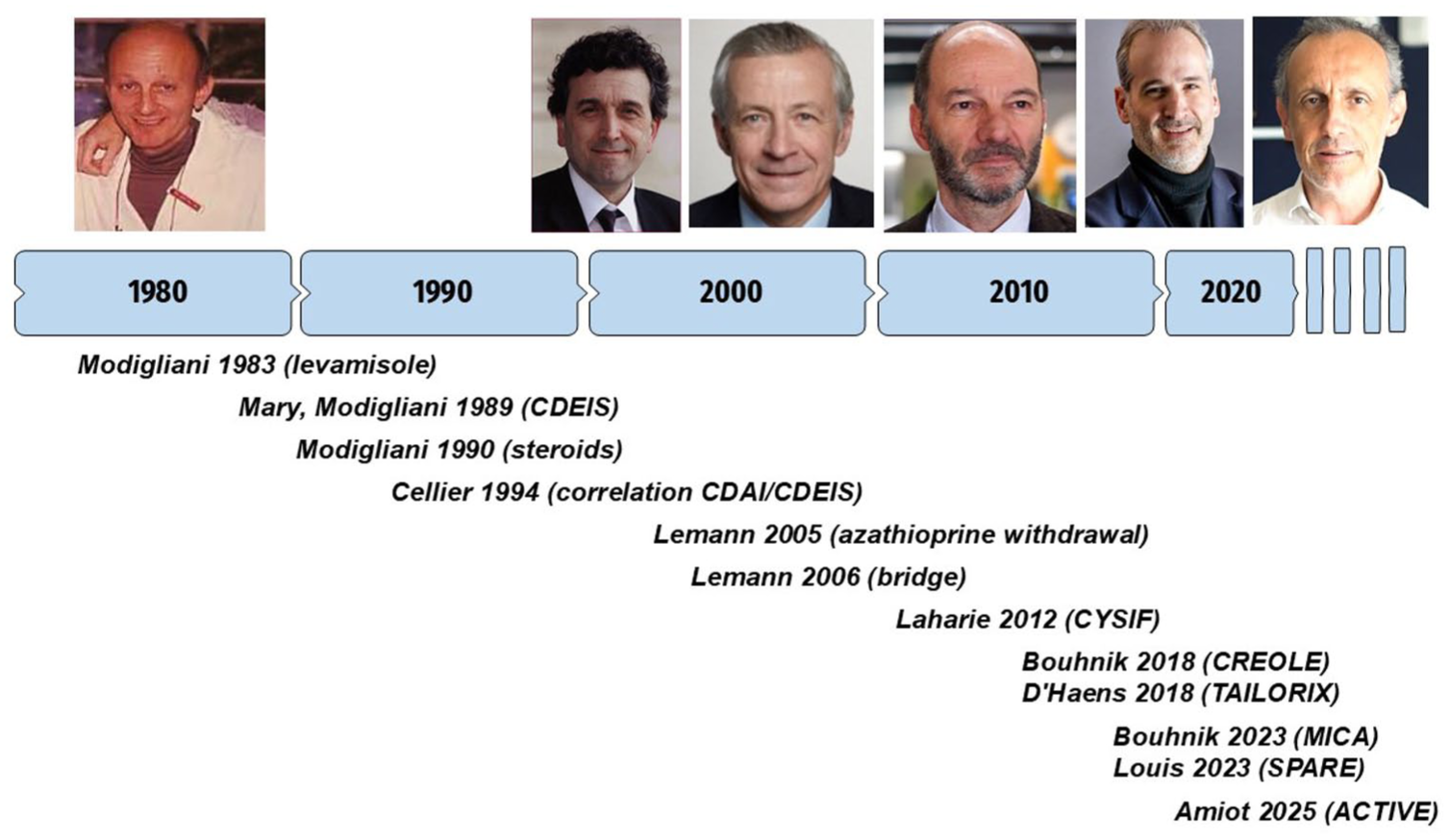
Timeline showing the most impactful studies conducted by the GETAID from 1983 to 2025, with pictures of successive presidents (Robert Modigliani, Marc Lémann, Jean-Frédéric Colombel, Edouard Louis, Laurent Peyrin-Biroulet, David Laharie).

(a) Number of publications from the GETAID per year. (b) Cumulative number of original articles published by the GETAID since 1983.
Endoscopic evaluation of CD
As endoscopy technology advanced in the 1980s, it was suspected that the extent and severity of the lesions observed during ileo-colonoscopy might impact the clinical course of CD. Before demonstrating the impact of mucosal lesions on the course of the disease, it was first necessary to define and assess the severity of endoscopic lesions using a validated index of severity. Strikingly, the future CDEIS was conceived not by endoscopists, but by a couple of eminent clinicians, Robert Modigliani, and a statistician, Jean-Yves Mary. They realized earlier than anyone else that we should quantify the overall severity of the disease and also consider the location of the lesions in the different segments, from the terminal ileum to the rectum, as the same lesions were unlikely to have the same impact, depending on their location. To construct this index, all elementary lesions were considered without any a priori, and each of them was precisely described in terms of number and extent by endoscopists in each segment. The CDEIS was developed using an extremely robust methodology, and it remains the only perfectly validated endoscopic severity index to date. 9 After the creation of this severity index, the group demonstrated that the available treatment at this time, prednisolone, was unable to heal the intestinal mucosa despite its benefit on clinical symptoms. 4 The weak correlation, or even absence, between symptoms and endoscopic lesions laid the foundations for modern patient care and future STRIDE criteria. 11
Strategies for drug de-escalation
Demonstrating the efficacy of a new molecule is a lengthy process, from the laboratory to clinical practice, which culminates in positive results from large phase III trials. While this makes the new drug available to physicians and their patients in specific situations, it does not provide information on how to optimally use the drug. As an independent group of investigators, GETAID has made significant contributions to the better use of anti-TNFs and to answering several clinical questions in post-marketing trials.
In the early 2000s, phase III trials demonstrated the efficacy of infliximab in inducing remission in patients with active CD. At that time, thiopurine was the mainstay of treatment for steroid-dependent patients. Marc Lemann and the GETAID investigated the benefits of adding infliximab to induce clinical remission without steroids in these patients. 12 Although the results were published in 2006, it is important to note that patients were enrolled between 2000 and 2002, at the very beginning of the anti-TNF era. The study demonstrated that infliximab plus azathioprine was more effective than azathioprine alone in steroid-dependent CD patients, and the authors concluded that infliximab could be used as a bridge therapy while awaiting the delayed effect of thiopurines. Results from the “Bridge trial” raised new questions for the medical community. Indeed, at that time, it became clear that patients should be maintained on long-term anti-TNF agents. However, concerns about safety and tolerability, particularly in patients treated with thiopurine and infliximab in combination, raised questions about treatment de-escalation. The first GETAID trial exploring drug withdrawal was conducted with azathioprine in patients with CD in clinical remission. 18 Later, Edouard Louis and the GETAID investigated the possibility of interrupting infliximab in patients with CD in long-term remission treated with in combination with a conventional immunosuppressant in the STORI cohort study. 19 Patients included were in steroid-free remission and were followed for up to 30 months after stopping infliximab or until relapse. The main goal of the STORI study was to identify predictive factors of relapse, including biological markers of inflammation, endoscopy, and trough levels. The STORI study was the first to observe that up to 50% of patients experienced relapse within 2 years of stopping infliximab, primarily in the first year. By investigating predictors of relapse, the STORI study was one of the first to develop the concept of deep remission. It also helped lay the foundation for new patient monitoring approaches as biomarkers and infliximab trough levels were identified as predictors of relapse. Several years later, it is widely accepted that these biomarkers are reliable indicators prior to treatment discontinuation in patients with long-term clinical remission. Conversely, a progressive increase in fecal calprotectin measurements predicts clinical relapse after treatment discontinuation.
The next step was a randomized controlled open-label trial to evaluate better therapeutic strategies for de-escalation in CD patients on combination therapy. 29 In the SPARE trial, it was demonstrated that thiopurine withdrawal was a preferable strategy compared to infliximab withdrawal in patients with CD in clinical remission on combination therapy. Interestingly, the SPARE trial was also a strategy study that demonstrated the effectiveness of retreatment with infliximab and a comparable probability of remission over 2 years in the three arms, paving the way for treatment cycling in IBD. Lastly, the same independent risk factors of relapse previously identified in the STORI study were confirmed in the SPARE trial.
Medical management of complicated CD
Historically, anti-TNF drugs have been contraindicated in CD patients who develop complications such as strictures or abscesses. The GETAID aimed to investigate the benefits of these agents in complicated CD through multicenter prospective cohort studies to identify predictors of drug response.
CREOLE was the first prospective cohort study enrolling 97 patients with symptomatic strictures. 23 Treatment success with adalimumab at week 24 was achieved in 64% of patients. Importantly, simple predictive factors for success were identified, leading to the creation of the “CREOLE score” that considered clinical patterns and MRI characteristics.
Fistulizing CD was the second critical situation studied. In the MICA study, 117 bio-naïve patients, with an intra-abdominal abscess that resolved with antibiotics ± radiological drainage, were subsequently treated with adalimumab and followed for 24 weeks. 24 Not only was this medical management feasible, but surgery was avoided in 74% of patients, and 71% of patients remained still free of surgery at the long-term follow-up.
Evolving paradigms in acute severe UC
Until Järnerot et al. 41 ’s landmark trial comparing infliximab to placebo in hospitalized patients with severe (UC) who did not respond to intravenous steroids, 41 intravenous cyclosporine has been the reference drug in this setting. Then, infliximab emerged as an alternative to cyclosporine, though comparative data between the two drugs were lacking. The GETAID made the first head-to-head trial comparing these two second-line medical therapies for acute severe UC. Based on a superiority design aiming to demonstrate that cyclosporine was more effective than infliximab, the CYSIF trial could not identify any differences between the two molecules on short-term or long-term efficacy and on safety among the 115 patients included in 27 centers from four countries.13,42 These findings, which were subsequently confirmed by the CONSTRUCT trial and observational studies, have now positioned infliximab as the standard of treatment for patients admitted steroid-refractory acute severe UC. 43
More recently, the GETAID conducted the ACTIVE trial, aiming to determine the optimal maintenance regimen for patients with acute severe UC responding to intravenous steroids. 28 This randomized open-label controlled trial compared azathioprine alone to a combination of azathioprine and infliximab. The treatment failure rate at 12 months was significantly higher in the azathioprine arm than in the combotherapy arm (82% and 53%, respectively, p = 0.03).
According to these two GETAID trials, intravenous infliximab has become a key molecule for treating patients with acute severe UC regardless of their response to steroids.
Evaluation of the safety of IBD drugs
The tolerance and the safety of medications are subjects of major importance for physicians and their patients, given their long-term use. However, clinical trials, meta-analyses, and registries are underpowered to individualize the impact of drugs used for IBD. In order to answer these questions, it is necessary to establish large enough patient cohorts to have the statistical power to identify low risks according to patients’ characteristics from a personalized medicine perspective.
The first French collaborative effort to assess the excess risk of cancer with the use of immunosuppressors was conducted in the 2000s by Laurent Beaugerie with the national CESAME cohort, gathering 19,486 patients from 680 French investigators. Results from CESAME unquestionably identified the increased risk of lymphoma in relation to the use of thiopurines. 21 Although the GETAID did not initiate the project, its members contributed significantly to patient recruitment. Importantly, the GETAID subsequently established the European ICARE cohort during the biologic era. 30 The ICARE cohort is a longitudinal European observational cohort including 15 countries. It was designed and powered to evaluate the impact of IBD therapies such as thiopurines, methotrexate, anti-TNFs, vedolizumab, and ustekinumab on the individual risk of cancer. Between 2016 and 2019, 13,262 patients were enrolled, of whom 10,206 were included in the final analysis and followed for 3–6 years. Because the ICARE cohort has collected real-world data with a standardized longitudinal long-term follow-up and sufficient statistical power, we will soon be able to explain to patients their individual excess risk of cancer and serious infection induced by the medications prescribed for their disease in the era of biologics.
GETAID future challenges
Maintaining innovative, affordable, independent clinical research
During the last period, the IBD therapeutic armamentarium has grown rapidly. Consequently, some clinical research questions may become obsolete before a trial is completed. In this context, independent academic clinical research in IBD should be innovative, thinking one step ahead, and avoiding competition with industrial programs. Indeed, the current overwhelming pharmaceutical environment could paradoxically stifle academic research, as was recently observed in congress programs favoring post hoc analyses from phase III trials over independent research.
Several approaches may improve the speed of such academic clinical research in IBD. Collaboration between academic groups may favor the recruitment, as trials require larger sample sizes than before. This has already been done for several past GETAID studies, such as CYSIF or METEOR.13,14 This academic model has also inspired similar initiatives in Europe, in Belgium (BIRD), Spain (GETECCU), Italy (IG-IBD), or Portugal (GEDII). It is expected that collaborative projects will maintain a high level of independent research in IBD. For example, the POMEROL trial (EUDRACT Number: 2021-002369-17), for which recruitment has recently been completed, was conducted in collaboration with the GETECCU, the BIRD, and the Australian group. Innovative study designs may reduce the number of patients needed and consequently costs. 44 In studies using a Bayesian framework, the sample size is not determined a priori because the study is analyzed continuously. One of the first examples of this type of study in IBD was the REPREVIO trial, which compared vedolizumab to placebo for preventing CD postoperative recurrence. 45
Over the last 40 years, clinical research in IBD has dramatically changed with a huge increase in costs related to the regulatory environment, costs of treatments, monitoring, objective evaluations of inflammatory lesions, including now endoscopy with central reading, biomarkers, radiology, or pathology. To be as close as possible to current practice, the GETAID study protocols are designed to take into account feasibility and not only scientific relevance. Maintaining high-level academic clinical research in IBD requires greater financial support with a solid organization. While motivated French investigators and study coordinators are in place and renewing thanks to the promotion of young, the most challenging aspect remains the constant effort involved in searching for academic or industrial grants, which must be undertaken in an unfavorable economic environment.
Disseminating knowledge about IBD
Since the early 2000s, GETAID has expanded its scope to include educational activities managed by a dedicated committee. Through its website, GETAID has developed practical medication guides for both gastroenterologists and patients, as well as daily tools such as clinical and endoscopic scoring tools and guidelines for special situations. Updated recommendations for UC and for CD were published in French in 2022 and 2024, respectively,37,38 including specifics of French reimbursement and practice. In light of the increasing number of IBD cases and the rapid evolution of the therapeutic landscape, there is a greater need than ever for educational programs to help physicians provide optimal patient management in their daily practice. To this end, GETAID has organized an annual masterclass for French public and private gastroenterologists since 2023.
Conclusion
Founded over 40 years ago, GETAID is a pioneering collaborative academic group in clinical research on IBD. It has made significant contributions to advancing knowledge, aiming to answer practical questions about the new therapeutic goals beyond clinical and endoscopic remission, the combination of conventional and advanced therapies, exit strategies, and the role of surgery in complicated segmental CD. At the same time, the incidence of IBD, which was historically observed in Western countries, continues to rise and has become a global pandemic. 46 Conducting active, independent clinical research into IBD is now more important than ever, and this can be achieved through national or international collaborative projects involving independent groups, such as the GETAID.
