Abstract
Background:
Inflammatory bowel disease (IBD) is associated with a considerable burden to the patient and society. However, current data on IBD incidence and burden are limited because of the paucity of nationwide epidemiological studies, heterogeneous designs, and a low number of participating centers and sample size. The EpidemIBD study is a large-scale investigation to provide an accurate assessment of the incidence of IBD in Spain, as well as treatment patterns and outcomes.
Methods:
This multicenter, population-based incidence cohort study included patients aged >18 years with IBD (Crohn’s disease, ulcerative colitis, or unclassified IBD) diagnosed during 2017 in 108 hospitals in Spain, covering 50% of the Spanish population. Each participating patient will attend 10 clinic visits during 5 years of follow up. Demographic data, IBD characteristics and family history, complications, treatments, surgeries, and hospital admissions will be recorded.
Results:
The EpidemIBD study is the first large-scale nationwide study to investigate the incidence of IBD in Spain. Enrollment is now completed and 3627 patients are currently being followed up.
Conclusions:
The study has been designed to overcome many of the limitations of previous European studies into IBD incidence by prospectively recruiting a large number of patients from all regions of Spain. In addition to epidemiological information about the burden of IBD, the 5-year follow-up period will also provide information on treatment patterns, and the natural history and financial burden of IBD.
Introduction
The relapsing–remitting nature of inflammatory bowel disease (IBD) and its lifelong course make this condition one of the most burdensome gastrointestinal disorders. 1 In fact, in addition to the financial burden on healthcare systems, IBD has a significant impact on quality of life. 1 Most patients with IBD are diagnosed during their 20s or 30s, 2 so IBD often has a significant effect on patients’ lives at a time when they are completing their education, establishing their careers, embarking on intimate relationships, or starting a family.3,4
The incidence of IBD seems to vary widely between countries.
5
Traditionally, a north-south gradient had
been reported, so that the diagnosis of IBD was more frequent in northern than in southern
countries.
6
More
recently, the existence of an east-west gradient has been suggested, with a higher incidence
of IBD in western than eastern countries.7,8 Also there is a higher prevalence of IBD in high-income
Most authors agree, however, that the overall incidence of IBD has been increasing during recent decades. For example, in a systematic review of 238 articles on the incidence and 122 on the prevalence of IBD, 75% of the studies of patients with Crohn’s disease (CD) and 60% of the studies of patients with ulcerative colitis (UC) showed a significant increase of the incidence of these diseases over time. 5 The annual increase in the incidence ranged from 1% to 23% for CD and from 2.5% to 18% for UC. 5 Another systematic review that included 147 studies published between 1990 and 2016 concluded that the incidence of CD and UC in the developed countries of Europe, North America and Oceania has stabilized and may even be decreasing. 10 However, with improved treatments and growing populations, the prevalence of CD and UC in these countries continues to rise. At the same time, the incidence of IBD in the developing countries of Africa, Asia, and South America is increasing. 10
In Europe, the European Crohn’s and Colitis Organisation-Epidemiological Committee (ECCO-EpiCom) initiated a study in 2010 to investigate differences in the incidence, disease characteristics, and therapeutic management of IBD between eastern and western Europe. The EpiCom study included 1515 cases of IBD diagnosed over 1 year, with an overall incidence rate of 15 cases/100,000 person-years: 5.4 for CD and 8.2 for UC. 7 These estimates fall within the range of IBD incidences in systematic reviews of the European literature, which report a UC incidence of between 0.6 and 57.9/100,000 person-years, and CD incidence of between 0.0 and 15.4/100,000 person-years.5,10
These previous studies highlight the broad range of IBD incidences reported, possibly due
to heterogeneous study designs, which may involve study sites, patients selection,
denominators to estimate population incidence, and protocols or criteria for IBD diagnosis,
as well as other methodological differences.
11
In Europe, there is a paucity of
nationwide epidemiological studies, with most of them being conducted in countries with
relatively small populations in northern or central Europe (Denmark, Hungary, and
Iceland).12–14 Nationwide studies are important to
enhance representativeness and limit the impact of incidence variability within a
country.
15
However, even nationwide studies can be flawed; for example, those using administrative
databases may be subject to certain classification bias.
11
Many studies (including EpiCom) were
conducted at a limited number of centers within each country,7,16 which may limit generalizability, or used
small sample sizes, which reduces the precision of the incidence estimates.11,15 For example, only one Spanish center was
included in the EpiCom study,
17
and this center was in Galicia, a region with a known association
between CD susceptibility and the presence of
The studies conducted on the incidence of IBD in Spain are summarized in Supplementary Table 1.17,19–45 Most of these studies were carried out more than a decade ago in selected areas and included a small number of patients. Differences in IBD incidence between regions have been reported, with variability between urban and rural centers and between inland and coastal areas even within a single region. 46 Therefore, the results of these studies, or the EpiCom data for Spain, 17 may not accurately reflect the current incidence of IBD in the whole country.
Since IBD is a chronic disease that is diagnosed at a young age and the incidence appears to be increasing, the current IBD prevalence in Spain might be considerably higher than that reported previously. However, no up-to-date information about this is available. Thus, the present (EpidemIBD) study was designed to address the information gap on the nationwide burden of IBD in Spain. Nationwide studies provide a representative estimate of an outcome, while limiting the impact of incidence variability within a country. Data obtained from such studies can inform health policy and approaches to disease management. The primary objective of the EpidemIBD study is to measure the incidence of IBD in Spain, a country with almost 50 million people. Secondary aims are: (1) to determine the geographical distribution of IBD in Spain; (2) to describe the characteristics of patients at the time of IBD diagnosis; (3) to register the use of immunosuppressive treatments and biological drugs, surgeries and hospital admissions during the first 5 years after diagnosis of IBD; and (4) to measure the delay from the onset of symptoms to diagnosis. At the time of writing, patient enrollment has been completed and the study is at the follow-up stage.
Methods
Study design
EpidemIBD is a multicenter, nationwide, prospective, population-based incidence cohort study of adult patients diagnosed with IBD (CD, UC, or unclassified IBD) during 1 year in Spain. In addition, each incident case is being followed to determine changes in disease phenotype or location, the need for immunosuppressive and biologic treatments, and the need for hospital admission or surgery during the first 5 years after diagnosis.
Study population
The study included all incident patients diagnosed with IBD in the 108 study centers
between 1 January 2017 and 31 December 2017 (
Patients were eligible to be included in the IBD incident cohort if they belonged to the reference area of the participating centers. A patient diagnosed at one center (e.g. a particular hospital in Madrid) but who was part of the reference population of another center (e.g. a different hospital in Madrid) was assigned to their own reference population (in this example, that of their usual hospital).
Recruitment
This study is being conducted at 108 centers providing free access to healthcare within the National Health System. In Spain, healthcare is provided mostly by public services; about 15% of the Spanish population have private health insurance, but only about 15% of insured individuals do not use the public services concurrently. 48 Thus, given the severity and relapsing–remitting nature of IBD, it is unlikely that the incidence of IBD is substantially underestimated by including only patients seen at public health centers.
An IBD specialist who is a member of GETECCU (Spanish Working Group on Crohn’s Disease and Ulcerative Colitis) was selected from each of the 17 Autonomous Communities (administrative regions) in Spain to serve as the study coordinator in their region. An initial selection of centers was made from the database of health centers of the Ministry of Health, which included 893 centers in 2016. The regional coordinators completed a questionnaire to determine the feasibility for each center to participate in the study. Initially, centers that did not have a gastroenterology service at their hospital were excluded. Of the centers that did have such a service, some were excluded because the physicians did not respond to the invitation emails, refused to participate, or the study coordinator together with the principal investigator considered that it was not possible to identify all IBD cases in the hospital reference area. After this initial phase, 142 centers were selected to start the study. Centers that did not adequately follow the study procedures or ensure the inclusion of all patients diagnosed with IBD in their area were excluded. During the first 12 months, 34 centers had withdrawn from the study; the main reasons were inability to follow the study protocol and difficulties in including all patients with incident IBD. At the time of writing, 108 centers continue to be involved in the study (Figure 1), covering a referral area with a population of 21,943,160 inhabitants (approximately 50% of Spanish population, which was 46,659,302 as of 28 June 2018). 49
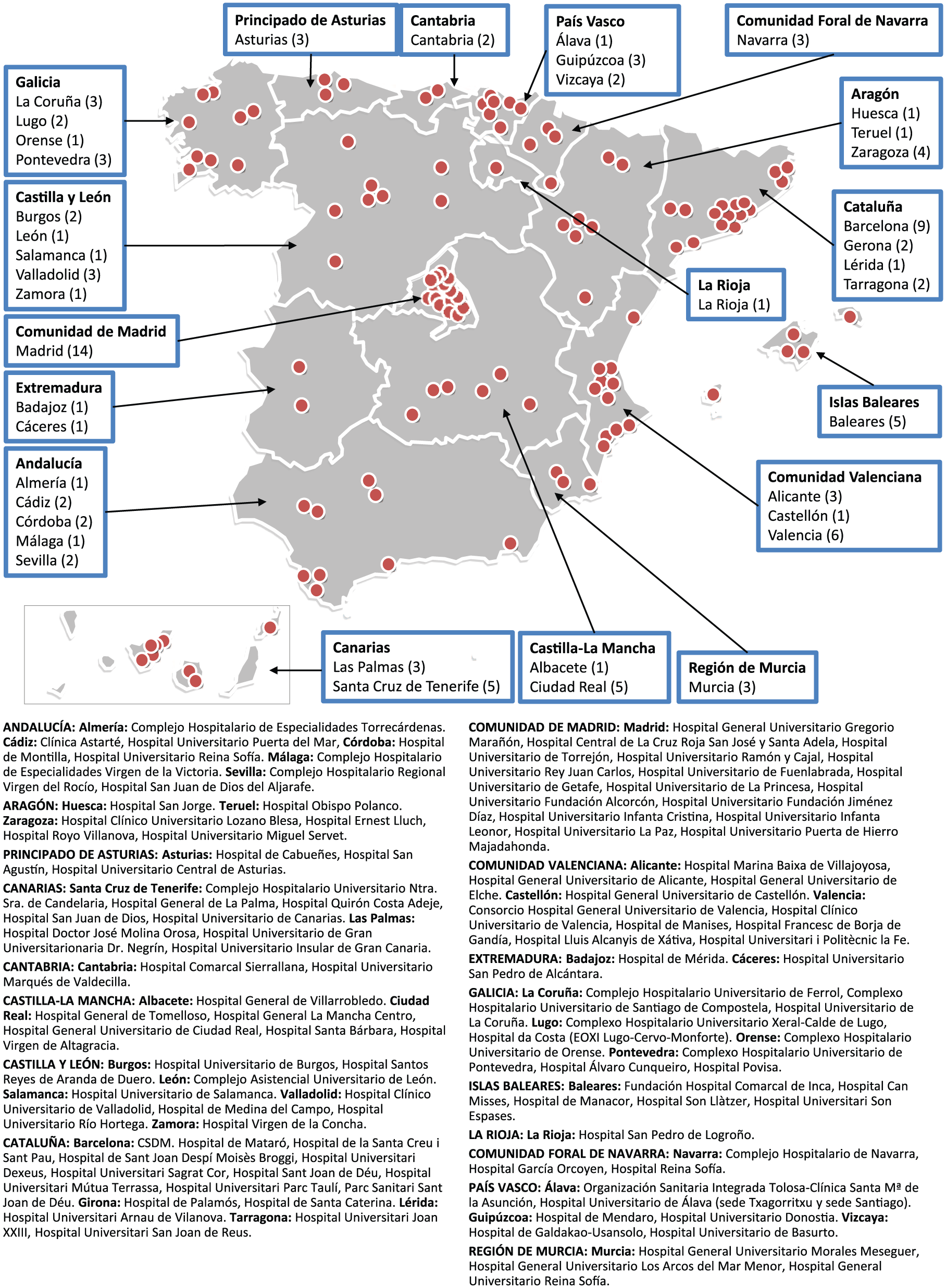
Study sites.
The EpidemIBD protocol allowed each center to use the most appropriate procedure to ensure complete identification of incident IBD cases. The databases of the endoscopy units and pathology departments were also reviewed at all participating sites during the year of enrollment (2017) to detect cases that might not have been identified initially. In these instances, the inclusion period was extended for 6 months, up to 30 June 2018.
Each participating investigator (see Supplementary Data) was required to confirm the diagnosis at the time of patient inclusion and 3 months later to ensure the correctness of the diagnosis and the phenotypic characteristics of the disease. External monitoring of incident patients included in the registry is also being performed by the research staff.
Definitions
IBD location and phenotype were defined according to the Montreal classification. 50 European Crohn’s and Colitis Organisation (ECCO) and European Society of Gastrointestinal and Abdominal Radiology (ESGAR) Guideline for Diagnostic Assessment in IBD was used to assess the extent of disease.51,52 Briefly, in patients with UC, a colonoscopy was mandatory. Patients with CD had to undergo colonoscopy and a cross-sectional image examination to assess the presence of small bowel involvement. In patients with CD and suspicion of perianal disease, magnetic resonance imaging or transanal ultrasound was performed. Time to diagnosis was defined as the time from the first medical consultation attended by a symptomatic patient to the diagnosis of IBD. Change in phenotype was defined as the appearance of new lesions not present at diagnosis, after the initial tests performed to determine disease extent and severity. An emergency surgical procedure was defined as any surgery performed within 24 h of admission, while an elective procedure was that performed after the first day of admission and by the scheduled (nonemergency) surgical team.
For each patient, the type of population center (urban or rural) at the patient’s birth and at diagnosis of IBD were recorded. Whether the population center of origin was considered rural or urban was based on the classification of the National Statistics Institute (INE) of each municipality. Socioeconomic level was assessed from the patient’s educational level (primary or lower, secondary, higher education), occupational status (self-employed, employed, unemployed, retired), professional status (nonsalaried or salaried), and work hours (full-time or part-time). The number of cohabitants at the patient’s home during their childhood (up to 16 years) and at diagnosis of IBD was also recorded.
Smoking status was categorized at the time of diagnosis of IBD as ‘nonsmoker’, ‘smoker’, or ‘ex-smoker’. Patients were considered ‘smokers’ if they smoked more than seven cigarettes per week for at least 6 months and smoked at least one cigarette in the 6 months prior to diagnosis. Patients were considered ‘ex-smokers’ if they quit smoking at least 6 months before diagnosis. Patients were considered ‘nonsmokers’ if they never smoked or did so in very small amounts or occasionally. 53
Data collection and follow up
Study data were collected and managed using an electronic data capture tool (Research Electronic Data Capture; REDCap), 54 which is hosted at Asociación Española de Gastroenterología (AEG; www.aegastro.es), a nonprofit medical society. AEG provided this service free of charge, with the aim of promoting investigator-driven research. REDCap is a secure, web-based application designed to support data capture for research studies that provides the following: (1) an intuitive interface for validated data entry; (2) audit trails for tracking data manipulation and export procedures; (3) automated export procedures for seamless data downloads to common statistical packages; and (4) procedures for importing data from external sources. 54
Demographic data, family history of IBD, socioeconomic characteristics, IBD type, pattern, and location, and presence of extraintestinal manifestations at diagnosis were collected from each patient at the time of inclusion in the study. The occurrence of complications (fistulas, stenoses, abscesses), changes in disease location, treatments for IBD, surgeries for IBD, and hospital admissions during 5 years after diagnosis will be also recorded.
After the patient is included, two other visits will be conducted during the first 12 months, followed by a visit every 6 months, until 5 years of follow up are completed. Thus, over the course of the study, the following 10 visits will be conducted (Figure 2):
Visit 0 (baseline): inclusion of a patient in the study and collection of socioeconomic data and diagnosis of IBD.
Visit 1 (month 3): confirmation of IBD diagnosis and updating of data related to treatment, changes in phenotype, hospital admissions, and surgery.
Visit 2 (month 12) to visit 10 (5 years): confirmation of IBD diagnosis and updating of data related to treatment, changes in phenotype, hospital admissions, and surgery.

Program of clinic visits over the course of the study.
Only the first prescription of each therapeutic group will be recorded. When a change in phenotype is observed, the phenotype, the complication leading to reclassification of the patient’s phenotype, and date of occurrence of the complication will be recorded.
Hospital admissions that occur during 5 years after IBD diagnosis will be recorded, including the dates of admission and discharge, and the cause of admission (related to IBD or not). Data will also be collected on surgical procedures performed on the patient since the diagnosis of IBD (including those which led to its diagnosis), the indication for surgery, and its date.
In order to ensure adherence to the hospital visits schedule, investigators at each center will receive monthly reminders about the patients who are expected to attend hospital visits during the upcoming month and the information that will need to be collected. In addition, research staff will regularly review compliance.
Statistical analysis
The reference population (based on estimates form the National Statistics Office) for the analyses is the population of the catchment areas of the participating centers. The incidence rate (number of incident cases per 100,000 inhabitants) during 1 year will be calculated, using the reference population as denominator. Also, age-standardized incidence rates adjusted to the European population will be calculated. 55
The Kaplan–Meier method will be used to estimate the time course of the use of treatments, hospital admissions, surgery, and of complications; the differences between the curves will be assessed with the log-rank test. A Cox regression model will be used to identify variables associated with the rate of complications, progression to more severe phenotypes, use of immunosuppressive or biological drugs, hospital admission, and performance of surgery.
Ethics
The study is being conducted in accordance with the 1964 Declaration of Helsinki and its subsequent revisions. The study protocol has been approved by the Ethics Research Committee of the coordinating hospital (Hospital Universitario de La Princesa, Madrid, Spain). In accordance with the Spanish regulations for observational studies, approval from the ethics committees at individual cites is not required. All patients provided informed written consent to participate.
Discussion
In order to plan healthcare for patients with IBD, it is important to know the current incidence of IBD, to evaluate the characteristics of patients at diagnosis, and to analyze their disease course and need for appropriate treatments, mainly in the first years after diagnosis. This information will be provided by the present (EpidemIBD) study.
A key strength of the EpidemIBD study is the inclusion of a high proportion of the Spanish population (approximately 50%) from all areas of the country. Another strength is that patients have been prospectively identified from a range of hospital settings. While there are a number of IBD units in Spain, most Spanish hospitals do not have IBD-specific units and patients are usually treated, depending on the circumstances of each case, by departments with limited training in this subspecialty. By including a range of hospitals with and without IBD units, the EpidemIBD study will assess diagnostic and treatment practices across a range of settings and will allow comparison of patient outcomes between settings and regions.
Another strength of this study is the 5-year follow up, that will identify patients diagnosed with CD or UC whose diagnosis, location, or phenotype change over time.56,57 The 5-year follow up also allows for assessment of treatment patterns and outcomes among IBD patients in Spain, and for a comparison of findings against those of the previously mentioned EpiCom study, which provides Europe-wide data on the treatment and outcomes in patients with IBD.58–61
Various studies suggest that the economic burden of IBD is increasing over time, either because of drug-associated costs or the greater sophistication of the diagnostic tests and therapies required by patients with IBD. 62 In Spain, as in several other developed countries, universal healthcare coverage is provided by public healthcare services. Therefore, the introduction of ever more costly techniques and drugs is causing a dramatic increase in the public resources devoted to health. This rate of increase is not sustainable for most developed countries; therefore, evaluation of diagnostic and therapeutic interventions in terms of their efficiency and cost–benefit ratio is being increasingly required by healthcare authorities. In the case of IBD, which is costly to manage and primarily affects young people, the lifelong resource utilization per patient can be considerable. Therefore, for countries like Spain, it is important to be able to accurately identify the incidence and prevalence of the condition (for healthcare planning), and to identify ways to optimize management and reduce burden (for maximizing patients’ contribution to society). This is particularly important for expensive biologic therapies, which must be used wisely to optimize their cost-effectiveness. In this regard, better care may be provided at a lower cost if patients are treated by physicians specialized in their particular disease, which points to further subspecialization as a way forward in the search for efficiency in medical practice. This may only be possible through the formation of teams of healthcare professionals who can provide uniform and specific care for these conditions. Data from our study, which includes both specialist IBD units and general gastroenterology departments, will provide additional insight into this question.
Conclusion
The EpidemIBD study will provide important information about the incidence of IBD in Spain, as well as about the management of the disease and its natural history. These data will be a vital benchmark for planning future IBD services in Spain.
Supplemental Material
Supplementary_Material – Supplemental material for EpidemIBD: rationale and design of a large-scale epidemiological study of inflammatory bowel disease in Spain
Supplemental material, Supplementary_Material for EpidemIBD: rationale and design of a large-scale epidemiological study of inflammatory bowel disease in Spain by María Chaparro, Manuel Barreiro-de Acosta, José Manuel Benítez, José Luis Cabriada, María José Casanova, Daniel Ceballos, María Esteve, Hipólito Fernóndez, Daniel Ginard, Fernando Gomollón, Rufo Lorente, Pilar Nos, Sabino Riestra, Montserrat Rivero, Pilar Robledo, Cristina Rodríguez, Beatriz Sicilia, Emilio Torrella, Ana Garre, Esther García-Esquinas, Fernando Rodríguez-Artalejo and Javier P. Gisbert in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors wish to thank A.G. McNicholl and J. Calvo for programming the e-CRF in AEG-REDCap (from the Spanish Association of Gastroenterology). The authors wish to thank A. Durán and P. Jiménez for data monitoring.
Also, the authors would like to thank Georgii Filatov and Catherine Rees of Springer Healthcare Communications who wrote the first and subsequent drafts of this manuscript. This medical writing assistance was funded by Merck Sharp & Dohme (MSD). MSD had no access to clinical data and was not involved in study design, statistical analysis or manuscript preparation.
The Author contributions are as follows:
M. Chaparro and J.P. Gisbert designed the study and are leading its development. E.G. García-Esquinas participated in study design and advised about methodological issues. M. Barreiro-de Acosta, J.M. Benítez, J.L. Cabriada, M.J. Casanova, D. Ceballos, M. Esteve, H. Fernández, D. Ginard, F. Gomollón, R. Lorente, P. Nos, S. Riestra, M. Rivero, P. Robledo, C. Rodríguez, B. Sicilia, E. Torrella, A. Garre collaborated with the center selection and are coordinating the study in each Autonomous Community. All other authors contributed to study design and critically revised the protocol, and were involved in patient enrolment and follow up. The EpidemIBD group includes the following list of investigators: A. Abraldes, J.M. Benítez, M.R. Calderón, R. Camargo, A. Hernández-Martínez, E. Iglesias, E. Leo, S. Marín, A. Monrobel, A. Núñez, N. Ruiz, M. Tejada, Y. Torres, M.J. Alcalá, E. Alfambra, Y. Ber, F. Gomollón, A. Mir, M. Montoro, J.M. Nerín, E. Peña, R. Vicente, J.M. Duque, R. de Francisco, A. Mancebo, S. Martínez-González, I. Pérez-Martínez, S. Riestra, P. Varela, I. Alonso-Abreu, D. Ceballos, N. Cruz, E. Guerra, A. Hernández Camba, L. Kolle, J.M. Marrero, N. Pérez, M. Soler, M. Vela, J.L.F. Forcelledo, M. Rivero, R. Lorente, C. Verdejo, M. Montealegre, A.J. Lucendo, O. Roncero, A Bouhmidi, D. Hervías, L. Arias, J. Barrio, L. Fernández-Salazar, P. Fradejas, A.M. Fuentes, L. Hernández, C. López, L. Mata, C. Piñero, B. Sicilia, M. Sierra, M. Vásquez, M. Aceituno, M. Bautista, X. Calvet, M. Esteve, A. Montserrat, J.A. Gómez-Valero, J. Gordillo, C. Ledezma, L. Lluis, F.J. Martínez-Cerezo, M. Menacho, M. Navarro-Llavat, S. Rodríguez, M. Sabat, M.J. Sampedro, E. Sainz, E. Sesé, A. Silva, S. Torra, L. Torrealba, C. Vila, M. Ágreda Chinea, A. Algaba, F. Bermejo, I. Blázquez, O. Bosch, B. Botella, M.J. Casanova, C. Castaño-Milla, M. Chaparro, R. de Lucas, M. Domínguez-Antonaya, M.G. Donday, A. Durán, M.L. Galve, L. García, A. Garre, J.P. Gisbert, I. Guerra, P. Jiménez, B. López-Cauce, A. López-Sanromán, P. López, I. Marín-Jiménez, M.D. Martín-Arranz, A.G. McNicholl, J.A. Olmos, V. Opio, J.L. Pérez-Calle, R. Plaza, A. Ponferrada, E. San Miguel, E. Sánchez, I. Vera-Mendoza, M. Aguas, M. García-Sepulcre, A. Gutiérrez, B. Herreros, J.M. Huguet, N. Jiménez, N. Maroto, L. Martí, M. Mínguez, M. Muñoz, P. Navarro, P. Nos, J.J. Ramírez, M. Roldán, L. Pozzati, P. Robledo, M. Barreiro-de Acosta, I. Bastón-Rey, A. Carmona, D. Carpio, E. Castro, B. Crespo, M.T. Diz-Lois, A. Echarri, J.D. Fernández-de Castro, E. Fernández, R. Ferreiro-Iglesias, V. Hernández, A. López, P. Pérez, M.J. Ruiz, P. Vega, M. Calafat, D. Ginard, E. Iyo, M.T. Novella, J. Reyes, C. Rodríguez, V. Royo, A. Sapiña, H. Fernández, M. Fraile, A. García, B. Lapeña, S. Revuelta, R. Irisarri, M. Kutz, Ó. Nantes, C. Rodríguez, S. Rubio, M. Vicuña, H. Alonso, J.L. Cabriada, A. Castiella, I. Galdona, A. Maíz, A.I. Muñagorri Santos, N. Muro, J. Ortiz, N. Otegui, I. Rodríguez-Lago, K. Spicakova, E.M. Zapata, L. Zubiaurre, J.M. Castillo-Espinosa, M.C. Martínez Bonil, C. Martínez Pascual, I. Nicolás, E. Torrella.
Funding
This research has been funded by grants from the Instituto de Salud Carlos III (FIS grant PI16/01296 and PFI17/00143), a grant from GETECCU and a grant from MSD. Funding agencies had no role in the study design, in the writing of this manuscript, or the decision to submit for publication.
Conflict of interest statement
M. Chaparro has served as a speaker, or has received research or education funding from MSD, Abbvie, Hospira, Pfizer, Takeda, Janssen, Ferring, Shire Pharmaceuticals, Dr. Falk Pharma, and Tillotts Pharma. M.J. Casanova has received education funding from Pfizer, Janssen, MSD, Abbvie, Ferring, Faes Farma, and Shire Pharmaceuticals. F. Gomollón has received honoraria for conference attendance from Janssen, Abbvie, Takeda and MSD, financial support for travel and registration to attend international conferences from Abbvie, Takeda and Janssen, and his research group has received financial grants from MSD, Abbvie and Tillots. J.L. Cabriada has served as consultant for, or received research funding from, MSD, Pfizer, Janssen, Takeda and Otsuka Pharma. M. Barreiro-de Acosta has served as a speaker, a consultant and advisory board member for, or has received research funding from, MSD, Abbvie, Janssen, Pfizer, Kern Pharma, Biogen, Takeda, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Tillotts Pharma, Chiesi, Gebro Pharma, Otsuka Pharma-ceutical and Vifor Pharma. S. Riestra has received honoraria for conference attendance from Janssen, Abbvie, Takeda and MSD, financial support for travel and registration to attend conferences from Abbvie, MSD and Janssen, and his research group has received financial grants from MSD, Abbvie and Takeda. M. Rivero has served as a speaker, a consultant and advisory board member for MSD, Abbvie and Janssen. M. Esteve acts as a consultant, assessor or scientific advisor for Abbott, MSD, Tillotts Pharma, Pfizer and Takeda and received research grants for MSD and Tillotts Pharma. J.P. Gisbert has served as a speaker, a consultant and advisory board member for, or has received research funding from, MSD, Abbvie, Hospira, Pfizer, Kern Pharma, Biogen, Takeda, Janssen, Roche, Celgene, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Tillotts Pharma, Chiesi, Casen Fleet, Gebro Pharma, Otsuka Pharmaceutical, Vifor Pharma.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
