Abstract
Background:
Prospectively and systematically collected real-world data on vedolizumab are scarce. We aimed to assess the long-term clinical effectiveness of vedolizumab in inflammatory bowel disease (IBD).
Methods:
This study was a prospective, observational, multicentre study. Overall, 286 patients with active IBD were included (Crohn’s disease, n = 169; ulcerative colitis, n = 117). The primary outcomes were clinical response at week 12 and clinical remission at week 52, based on the Harvey Bradshaw Index and the partial Mayo Clinic score. Secondary outcomes included clinical remission at week 12, clinical response at week 52, corticosteroid-free clinical remission at week 52, changes in biochemical measures, and health-related quality of life (HRQoL).
Results:
At baseline, 88% of the patients were exposed to anti-TNF and 41% of the patients with Crohn’s disease had undergone ⩾1 surgical resection. At week 12, clinical response was 27% and remission 47% in Crohn’s disease; corresponding figures in ulcerative colitis were 52% and 34%. Clinical response, remission and corticosteroid-free remission at week 52 were 22%, 41% and 40% in Crohn’s disease and 49%, 47% and 46% in ulcerative colitis, respectively. A statistically significant decrease in median faecal-calprotectin and C-reactive protein was observed at 12 and 52 weeks in patients with Crohn’s disease and ulcerative colitis. The HRQoL measures Short Health Scale and EuroQol 5-Dimensions improved in both Crohn’s disease and ulcerative colitis patients (p < 0.001). Clinical disease activity at baseline was inversely associated with clinical remission at week 52.
Conclusion:
Vedolizumab proved effective for the treatment of refractory IBD in clinical practice.
Introduction
Biological agents have become the mainstay of therapy for patients with moderately to severely active inflammatory bowel disease (IBD) who fail or are intolerant to conventional treatment. For many years, the only class of biological drugs available in IBD was anti-tumour necrosis factor (anti-TNF) agents. Many patients, however, do not respond to anti-TNF therapy, lose response over time, or discontinue the treatment because of excessive or intolerable adverse effects. 1
Vedolizumab is a humanised monoclonal antibody targeting the α4β7 integrin expressed on gut-homing lymphocytes, thus inhibiting leukocyte migration from the blood to the intestinal mucosa. In 2014, the European Medical Agency and the US Food and Drug Administration approved vedolizumab for patients with moderately to severely active Crohn’s disease and ulcerative colitis who have had an inadequate response with, lost response to, or were intolerant to either conventional therapy or a TNFα antagonist based on evidence from three randomised controlled trial (RCTs). The RCTs demonstrated the efficacy of vedolizumab in inducing and maintaining remission in patients with moderately to severely active IBD and previous unsuccessful treatment with one or more conventional treatment or anti-TNF.2–4
However, many patients evaluated in routine clinical practice would not qualify for enrolment in a clinical trial because of strict inclusion and exclusion criteria. 5 In addition, both patients and doctors may be reluctant to accept placebo control. Finally, many important outcomes, such as the effect on extra-intestinal manifestations, often remain beyond the scope of RCTs.2–4
Therefore, real-world data are vital to complement the knowledge gained from traditional RCTs. Real-world studies increase our understanding of how biologics work in a real-life clinical setting. Moreover, they may shed light on previously undetected safety signals.
Observational studies of vedolizumab performed at tertiary referral centres demonstrated response and remission rates comparable to those reported in the pivotal trials.6,7 However, data from unselected population-based cohorts, long-term prospective follow-up and data on the effect on quality of life are sparse.8–11
This prospective multicentre national study aimed to (i) assess the long-term clinical effectiveness of vedolizumab in clinical practice, (ii) identify predictors of treatment outcomes and (iii) describe the vedolizumab-treated patient population in Sweden using data collected prospectively by an electronic case report form (eCRF) linked to the Swedish National Quality Register for IBD (SWIBREG).
Materials and methods
Patients
SVEAH was a prospective, observational, single-arm, multicentre study conducted at 21 Swedish hospitals (13 regional and eight university hospitals) between 1 June 2015 and 31 November 2018. Eligible patients were ⩾18 years old and had active IBD at the onset of vedolizumab. Active Crohn’s disease was defined as presence of symptoms based on patient reported outcome measures in combination with ulcers at colonoscopy, signs of active disease at magnetic resonance imaging (i.e. contrast enhancement, bowel thickening or combs sign), C-reactive protein (CRP) higher than the lower limit of detection, high-sensitivity CRP >2.87 mg/l or faecal calprotectin >200 µg/g not more than 4 weeks before onset of treatment. The lower limit of detection defines normal CRP concentration in clinical practice. According to the manufacturers, the threshold was 4.0 mg/l for most CRP assays. Correspondingly, active ulcerative colitis was defined as the presence of symptoms accompanied by a Mayo endoscopic subscore of ⩾2 not more than 4 weeks before initiation of vedolizumab. 12 Exclusion criteria were concurrent participation in a clinical trial in which IBD treatment was dictated by an (interventional) study protocol, contraindications to vedolizumab (i.e. patients with known hypersensitivity to vedolizumab or any of its excipients), prior exposure to vedolizumab or planned cessation of treatment within 12 months from initiation (e.g. planned pregnancy). Written informed consent was obtained from all individual participants included in the study. Vedolizumab therapy was initiated according to its summary of product characteristics, that is, at a dose of 300 mg at weeks 0, 2 and 6, followed by 300 mg every 8 weeks. Patients with Crohn’s disease who had not responded by week 10 were allowed to receive an additional dose at week 10. However, as this was an observational study, the treating physician was permitted to adjust the dose or dosing interval at any time.
Data collection
Data were recorded at baseline and prospectively using an eCRF integrated with the SWIBREG. 13 Study visits were scheduled at week 0 (±2 weeks), week 12 (±2 weeks) and week 52 (±4 weeks).
The following information was collected: age, sex, smoking status, duration of disease, data to classify the patient according to the Montreal classification, 14 medical and surgical treatment, use of antibiotics, clinical measures [Harvey–Bradshaw Index (HBI), partial Mayo Clinic score],12,15 biochemical measures (haemoglobin, CRP, faecal calprotectin), endoscopic activity (Mayo endoscopic subscore, presence of ulcers) and health-related quality of life (HRQoL) measures [Short Health Scale and EuroQol 5-Dimensions, 5-Levels (EQ5D-5L)].16,17 Reasons for termination of vedolizumab therapy were determined according to the following criteria: lack of or loss of response, adverse drug reaction, other reasons, that is, pregnancy and patient’s request.
The HBI consists of five clinical components reflecting the activity of Crohn’s disease (general wellbeing, abdominal pain, number of liquid stools per day, abdominal mass, complications), with higher scores indicating more severe disease activity. 15
The Mayo Clinic score for ulcerative colitis comprises four items (stool frequency, rectal bleeding, physician’s global assessment, the endoscopic subscore). The score ranges from 0 to 12, with higher scores indicative of more active disease. 12 A version that includes only the non-invasive (excluding the endoscopic subscore) component is called the partial Mayo Clinic score. 18
The Short Health Scale is a self-reported questionnaire that evaluates four dimensions of HRQoL in patients with IBD (symptom burden, disease-related worry, general wellbeing, social function). Each item is scored from 0, representing no problem, to 5, reflecting the worst imaginable state. 17
The EQ5D-5L assesses five generic dimensions of HRQoL, including mobility, self-care, usual activities, pain/discomfort and anxiety/depression, separately rated from 1 (no problems) to 5 (severe problems). The responses could be converted into a single number called the index value where 1.0 represents best possible wellbeing. The EQ5D-5L also comprises a visual analogue scale (VAS) that ranges from 0 to 100 to assess the current health state. 16
Outcomes
The two primary outcomes were clinical response at week 12 and clinical remission at week 52. Clinical response was defined as a decrease in the HBI of ⩾3 from baseline in patients with Crohn’s disease and as a decrease in the partial Mayo Clinic score of ⩾2 and a reduction of ⩾25% from baseline, with a reduction ⩾1 on the rectal bleeding score or an absolute rectal bleeding score of 0 or 1, in patients with ulcerative colitis. Clinical remission was defined as a HBI of <5 in patients with Crohn’s disease and as a partial Mayo Clinic score <2 in patients with ulcerative colitis.
Secondary outcomes included clinical remission at week 12, clinical response at week 52, corticosteroid-free clinical remission at week 52, the effect of vedolizumab on endoscopic activity (ulcerative colitis patients only), biochemical measures (haemoglobin, CRP, faecal calprotectin), extra-intestinal manifestations and HRQoL (Short Health Scale and EQ-5D-5L). Finally, we assessed the proportion of patients on concomitant corticosteroid treatment during the study period.
Predictors of response
We stratified the main results by the reason for initiating treatment with vedolizumab according to the following criteria: (1) patients with a primary non-response to the most recent anti-TNF therapy; (2) patients who discontinued the most recent anti-TNF due to adverse drug reactions, loss of response or other reasons; and (3) patients naïve to anti-TNF. In addition, we assessed predictors for clinical remission at week 52 in patients with Crohn’s disease and ulcerative colitis.
Statistics
Because this was an observational single-arm study, the sample size was set to the minimum number of observations needed to achieve reasonable precision in the outcome estimates. A sample size of 170 patients with Crohn’s disease and 125 patients with ulcerative colitis was estimated to provide a 95% confidence interval (CI) for the 12-week response rate of 25–39% in Crohn’s disease and 38–56% in ulcerative colitis based on data from the GEMINI-trials.2,3 The corresponding figures for clinical remission at week 52 were 13–25% for patients with Crohn’s disease and 14–29% for patients with ulcerative colitis.3,4
Data on a continuous scale are represented as median with interquartile range (IQR), and differences between groups were assessed using the Mann–Whitney U-test. Paired HBI, partial Mayo Clinic score, calprotectin, CRP, haemoglobin, Short Health Scale and EQ5D-5L values were compared using Wilcoxon signed-rank test, to assess changes between baseline and follow-up. The proportion of patients with response, remission and with extra-intestinal manifestations at different time points were assumed to follow a binomial distribution and 95% CIs were calculated accordingly. An analysis was performed following the intention-to-treat principle, where missing data and discontinuation of vedolizumab were classified as treatment failure. Differences between groups were assessed using the Chi-square test for trend. Paired measures of extraintestinal manifestations were compared using McNemar’s test, to assess changes between baseline and 52 weeks. Survival plots illustrating the cumulative probability of discontinuation of vedolizumab were generated by Kaplan–Meier analysis. The EQ5D-5L index value calculator version 2.0, provided by the EuroQol group, was used to calculate EQ5D-5L index values. 19
Multivariable logistic regression was used to evaluate predictors of clinical remission at week 52, stratified by diagnosis. Covariates were selected based on their potential biological association with this outcome. All tests were two-tailed and p-values of <0.05 were considered statistically significant. Statistical analyses were performed using SPSS Statistics for Windows version 25.0 (IBM Corp., Armonk, NY, USA: 2017).
Ethical considerations
The study was performed according to the Declaration of Helsinki’s ethical guidelines and was approved by the Regional Ethics Committee in Linköping, Sweden (2015/117-32).
Results
In total, 170 patients with Crohn’s disease and 130 with ulcerative colitis were included by 31 November 2018. However, 14 patients were excluded (Crohn’s disease, n = 1; ulcerative colitis, n = 13) for various reasons (conflicting data on the date of vedolizumab initiation, n = 1; Mayo endoscopic subscore <2, n = 10; other reasons, n = 3). Each Crohn’s disease patient fulfilled at least one criterion for active inflammation, and most patients met more than one criterion. In total, 133/169 (79%) patients had signs of active mucosal inflammation at endoscopy or magnetic resonance imaging; 127/169 (75%) patients had an elevated CRP and 91/169 (54%) patients had a faecal calprotectin >200 µg/g. Correspondingly, all 117 patients with ulcerative colitis had signs of active mucosal inflammation at endoscopy (i.e. Mayo endoscopic subscore of ⩾2). Demographic and clinical characteristics of the study population are summarised in Table 1.
Baseline demographics and clinical characteristics of patients with Crohn’s disease and ulcerative colitis.

Colectomy with ileorectal anastomosis.
In total, 150 patients with Crohn’s disease and 101 patients with ulcerative colitis had previously stopped anti-TNF treatment.
anti-TNF, anti-tumour necrosis factor; IQR, interquartile range.
Effectiveness in Crohn’s disease
The 12-week drug continuation rate was 155/169 (92%) [Figure 1(a)]. At 12 weeks, 46 (27% CI: 21–35%) patients had clinical response and 80 (47% CI: 40–55%) were in remission [Figure 2(a)]. For patients who continued vedolizumab, the median HBI decreased from 6 (IQR 3–10) at baseline to 4 (IQR 1–8) at 12 weeks (p < 0.001). Correspondingly, the median faecal calprotectin decreased from 507 (200–953) µg/g to 234 (114–727) µg/g (p < 0.01) and the median CRP decreased from 5 (3–16) mg/l to 5 (2–12) mg/l (p < 0.01). The median haemoglobin concentration remained stable: 135 (124–143) g/l at baseline and 133 (124–144) g/l at week 12 (p = 0.71).

Drug survival on vedolizumab in patients with (a) Crohn’s disease and (b) ulcerative colitis.

Clinical response, remission and steroid-free remission at week 12 and 52 in patients with (a) Crohn’s disease and (b) ulcerative colitis.
The 52-week continuation rate of vedolizumab was 102/169 (60%) (Figure 1). Reasons for drug termination were evidence of its lack of or loss of response (n = 51), adverse drug reaction (n = 11) or other reasons (n = 5). The following adverse drug reactions were reported; infections (n = 2), respiratory symptoms (n = 2), skin reactions (n = 2), arthralgia (n = 4) and unspecified (n = 1). Overall, 29 (19%) patients shortened the interval between vedolizumab infusions during maintenance therapy. At 52 weeks, 38 (22% CI: 16–30%) patients had clinical response, 69 (41% CI: 33–49%) were in remission and 67 (40% CI: 32–47%) in corticosteroid-free remission [Figure 2(a)]. The proportion of patients on corticosteroid treatment over time is illustrated in Figure 3(a). When stratifying the results by the reason for initiating vedolizumab, no difference in clinical remission rate was observed between patients with primary non-response to anti-TNF (33%), bio-naive patients (47%) and patients that discontinued anti-TNF for other reasons (42%) (p = 0.96).
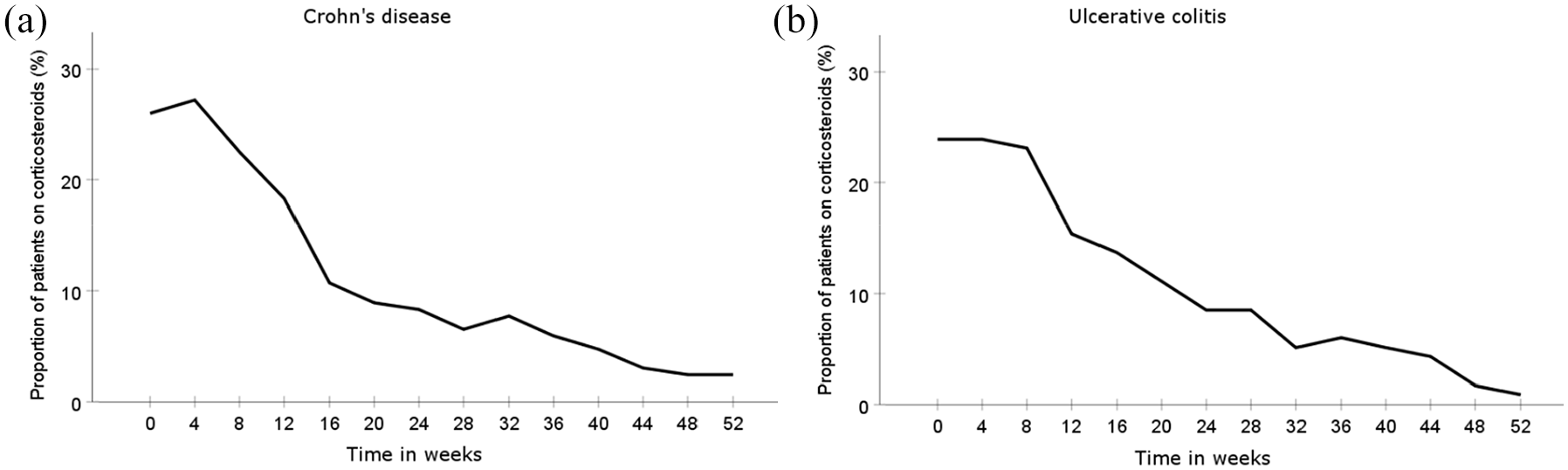
Proportion of patients on corticosteroid treatment in patients with (a) Crohn’s disease and (b) ulcerative colitis.
For patients who continued vedolizumab (n = 102), the median HBI decreased from 5 (2–9) at baseline to 3 (1–5) at 52 weeks (p < 0.001). Correspondingly, the median faecal calprotectin decreased from 475 (142–916) µg/g to 156 (65–392) µg/g (p < 0.01) and the median CRP decreased from 5 (2–11) mg/l to 4 (2–6) mg/l (p < 0.01). No statistically significant change in the median haemoglobin level was observed between baseline to week 52: 136 (IQR 125–146) g/l to 139 (IQR 129–147) g/l (p = 0.07). Among patients who continued vedolizumab, 20 (20% CI: 12–29%) had extra-intestinal manifestations at baseline (arthralgia n = 20; uveitis n = 1). The corresponding figure at 52 weeks was 25 (25% CI: 17–34%; p = 0.38; arthralgia n = 23; uveitis n = 3, erythema nodosum n = 5; and pyoderma gangrenosum n = 1).
The Short Health Scale significantly improved in all four dimensions, from baseline to week 52 in patients who continued vedolizumab therapy (Table 2). In parallel, the EQ5D-5L index value increased at week 52 versus baseline (Table 2).
Health-related quality of life in patients with Crohn’s disease.

EQ5D-5L, EuroQual 5-Dimensions, 5-Levels; IQR, interquartile range.
Predictors in Crohn’s disease
Univariate and multivariate logistic regression was used to identify predictors for clinical remission at week 52 (Table 3). In the univariate analyses female sex [odds ratio (OR): 0.49; 95% CI: 0.26–0.91], clinical disease activity, as assessed by HBI (OR: 0.87; 95% CI: 0.80–0.93), presence of extra-intestinal manifestations at baseline (OR: 0.40; 95% CI: 0.18–0.88) and a history of bowel surgery (OR: 0.51; 95% CI: 0.27–0.96) were inversely associated with clinical remission at week 52. However, after adjusting for the potential confounders listed in Table 3, high clinical disease activity at baseline [adjusted OR (aOR): 0.87; 95% CI: 0.79–0.95] was the only covariate that remained significantly associated with the outcome.
Predictors of clinical remission in patients with Crohn’s disease at week 52.

Statistically significant results are highlighted in bold.
anti-TNF, anti tumour necrosis factor; Ref., reference.
Effectiveness in ulcerative colitis
The 12-week drug continuation rate was 110/117 (94%) [Figure 1(b)]. At study entry, nine patients had undergone colectomy with ileorectal anastomosis. These patients were excluded from the assessment of clinical response and remission (since the partial Mayo Clinic score is not validated in this subgroup of patients). At 12 weeks, 56 (52% CI: 43–61%) of the remaining patients had clinical response and 37 (34% CI: 26–44%) were in remission [Figure 2(b)]. For those patients who continued vedolizumab, the median partial Mayo Clinic score decreased from 4 (IQR 3–6) at baseline to 2 (IQR 1–4) at 12 weeks (p < 0.001). Correspondingly, the median faecal calprotectin decreased from 676 (325–1144) µg/g to 285 (73–885) µg/g (p = 0.01), the median CRP decreased from 5 (2–9) mg/l to 4 (2–6) mg/l (p = 0.19) and the median haemoglobin increased from 132 (122–144) g/l to 133 (125–144) g/l (p = 0.02).
The 52-week drug continuation rate was 79/117 (68%) [Figure 1(b)]. Reasons for termination of vedolizumab were lack of or loss of response (n = 32), adverse drug reactions (n = 2) or other reasons (n = 4). The following adverse drug reactions were reported; infections (n = 1), and unspecified (n = 1). In total, 15 (14%) patients shortened the interval between vedolizumab infusions during maintenance therapy.
At 52 weeks, 53 (49% CI: 40–58%) patients had clinical response, 51 (47% CI: 38–57%) were in remission and 50 (46% CI: 37–56%) in corticosteroid-free remission [Figure 2(b)]. The proportion of time on corticosteroid treatment is illustrated in Figure 3(b). Stratifying the results by the reason for initiating treatment with vedolizumab, no difference in clinical remission rate was observed (p = 0.90) between patients with primary non-response to anti-TNF (43%), bio-naive patients (40%) and patients that discontinued anti-TNF for other reasons (52%). In total, 38 (32% CI: 24–42%) patients were in endoscopic remission at week 52. However, 12 patients lacked information on the endoscopic activity and were defined as non-remitters.
Four of the patients who continued vedolizumab until week 52 had undergone colectomy with ileorectal anastomosis. In the remaining patients (n = 75), the median partial Mayo Clinic score decreased from 4 (3–5) at baseline to 1 (0–2) at 52 weeks (p < 0.001). Correspondingly, the median faecal calprotectin decreased from 646 (333–1130) µg/g to 162 (42–382) µg/g (p < 0.001), the median CRP decreased from 5 (2–8) mg/l to 4 (1–5) mg/l (p = 0.01) and the median haemoglobin concentration increased from 131 (123–147) g/L to 141 (134–150) g/l (p < 0.001). Among patients who continued vedolizumab, six (8% CI: 3–16) had extra-intestinal manifestations at baseline (arthralgia n = 6; uveitis n = 3, erythema nodosum n = 3; and pyoderma gangrenosum n = 3). The corresponding figure at week 52 was 12 (15% CI: 8–25; p = 0.07; arthralgia n = 11; uveitis n = 1, erythema nodosum n = 1; and pyoderma gangrenosum n = 1).
The Short Health Scale significantly improved in all four dimensions, from baseline to week 52 in patients who continued vedolizumab (Table 4). In addition, the EQ5D-5L index value increased at week 52 versus baseline (Table 4).
Health-related quality of life in patients with ulcerative colitis.
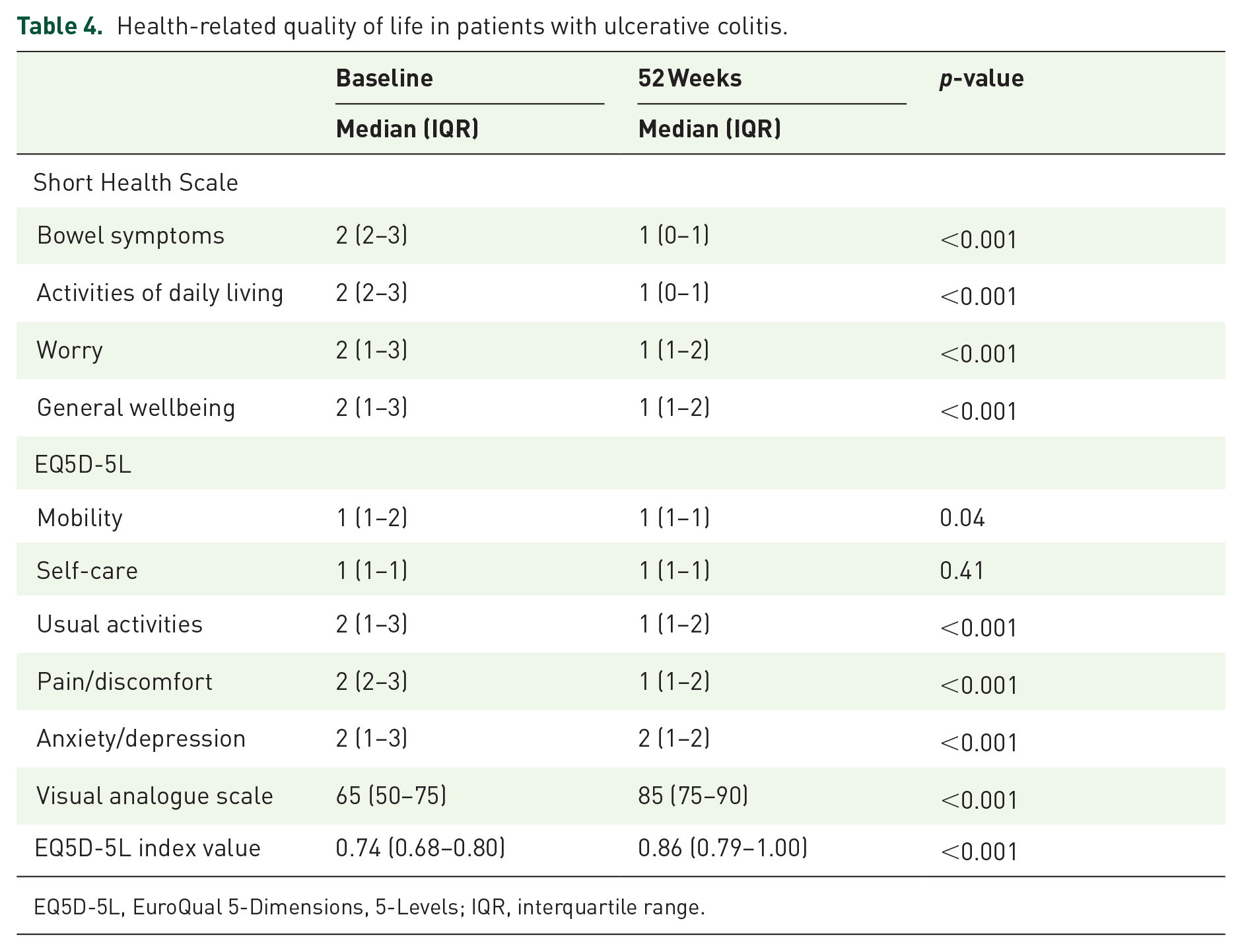
EQ5D-5L, EuroQual 5-Dimensions, 5-Levels; IQR, interquartile range.
Predictors in ulcerative colitis
Univariate and multivariate logistic regression analyses were performed to identify predictors for clinical remission at week 52 (Table 5). After adjustment for the potential confounders listed in Table 5, high clinical disease activity at initiation of vedolizumab (assessed by the partial Mayo Clinic score) was inversely associated with clinical remission at week 52 (aOR: 0.73; 95% CI: 0.58–0.92).
Predictors of clinical remission in patients with ulcerative colitis at week 52.

Statistically significant results are highlighted in bold.
Only two patients had proctitis at initiation of vedolizumab.
None of the nine patients with ileorectal anastomosis were in remission by week 52.
anti-TNF, anti-tumour necrosis factor; Ref., reference.
Discussion
In this prospective, observational single-arm, multicentre study vedolizumab showed high effectiveness and tolerability in patients with active IBD in Swedish clinical practice. Although the vast majority of patients were previously exposed to anti-TNF and almost half of the patients with Crohn’s disease had gone through at least one intestinal resection, 27% of the Crohn’s disease patients and 52% of the patients with ulcerative colitis had clinical response at week 12. The remission rate at week 52 was 41% for Crohn’s disease and 49% for ulcerative colitis. Clinical disease activity at baseline was inversely associated with the 52-week clinical remission rate in both Crohn’s disease and ulcerative colitis.
An abundance of published studies across the world have reported on the clinical effectiveness and safety of vedolizumab.6,7 However, these real-world studies are often limited by a short follow-up period,20–23 the retrospective design24–39 or a small study population. In addition, only a few prospective studies have follow-ups ⩾12 months.8–11
In the current study, data were collected prospectively using an eCRF linked to the SWIBREG. Well-established disease activity indices and objective biochemical and endoscopic measures were used to assess disease activity at multiple time points throughout the study.
Our long-term clinical remission rates are similar to those of a comprehensive meta-analysis on vedolizumab real-world data from Schreiber et al., demonstrating that approximately 30% of patients with Crohn’s disease and 46% of patients with ulcerative colitis achieve clinical remission by week 52. However, the figures are somewhat lower than in a previous study of vedolizumab in Swedish clinical practice by our group. 39 In that study the 52-week clinical remission rate was 60% in patients with Crohn’s disease and 64% in patients with ulcerative colitis. Most probably, the observed discrepancy could be explained by differences in inclusion criteria between the two studies. The previous study comprised some patients starting vedolizumab because of adverse effects of other therapies (e.g. anti-TNF) or as post-operative prophylaxis while having low clinical disease activity.
Notably, patients with Crohn’s disease had higher rates of clinical remission compared with clinical response at both week 12 and week 52. The definition of active disease at baseline could explain this finding. Active disease was defined by objective markers of inflammation at radiology, endoscopic examination or biochemical tests, that is, a patient could have active disease without a high score on the HBI, making it more challenging to achieve clinical response (which is based on a decrease in the HBI) than clinical remission.
Several real-world studies and post hoc analyses of the GEMINI data indicate better outcomes with vedolizumab in anti-TNF naïve patients compared with patients who are previously exposed to anti-TNF treatment.10,23,40,41 In the current study, most patients were anti-TNF experienced, and no difference between anti-TNF naïve and experienced patients was observed. Correspondingly, no association between the mechanism of anti-TNF failure (i.e. primary non-response, loss of response or adverse drug reaction) and outcome of vedolizumab treatment was observed. The absence of differences could potentially be explained by low statistical power. Although similar to our study, Kopylov et al. 38 observed no association between prior anti-TNF exposure and the effect on clinical outcomes of ulcerative colitis or Crohn’s disease at 52 weeks. Their study, like ours (12%), was limited by low numbers of anti-TNF naïve patients (8%). Anti-TNF naïve patients in our study were significantly older at the initiation of vedolizumab, but we found no differences in sex, disease duration or clinical activity at baseline compared with anti-TNF-exposed patients.
The association that we did find was that patients with high clinical disease activity at initiation of vedolizumab had a lower probability of achieving clinical remission at week 52. This association has previously been shown in subgroup analyses of the GEMINI trials3,4 and real-world observational cohorts8,20,21,23,29,36 have supported this finding. Whether concomitant treatment with corticosteroids affects treatment outcomes with vedolizumab is unclear. Some cohorts show an association between corticosteroid use and lower rates of response in Crohn’s disease20,21 and ulcerative colitis,8,10 which could be a confounder as corticosteroids are more often used in patients with more severe disease. Consistent with our findings, no associations between corticosteroid use and outcome of vedolizumab treatment were noted in the SWIBREG, VICTORY, Cross Pennine or Israeli cohorts.36–39
Importantly, in both patients with Crohn’s disease and patients with ulcerative colitis, we observed clinically meaningful and statistically significant improvements in all four domains of the Short Health Scale, a disease-specific HRQoL instrument. Similarly, vedolizumab treatment was associated with statistically significant decrease in the pain/discomfort and anxiety/depression dimensions of the generic instrument EQ5D-5L as well as an increase in the EQ5D-5L index value and current health status measured with the VAS. This finding is consistent with data on HRQoL from the GEMINI study, in which ulcerative colitis patients receiving vedolizumab demonstrated greater improvements in the Inflammatory Bowel Disease Questionnaire than placebo-treated patients. 3 However, the same result could not be shown in patients with Crohn’s disease in the GEMINI cohort. 4 Real-word data on the long-term effect of vedolizumab on HRQoL are limited but Stallmach et al. 10 observed a significant increase in the EQ5D-5L VAS in patients receiving vedolizumab from baseline (median 47.5) to week 14 (median 79), but no difference at 54 weeks in the 14 patients with data available.
In addition to the presence of data on HQoL, this study’s main strengths are the use of prospective data collection, the standardisation of data collection through an eCRF, the intention-to-treat approach of handling the data and using well-established outcome measures. The study comprises a wide range of patients from both regional hospitals and university centres, including patients that would have been excluded from the GEMINI trials, thus reflecting the use of vedolizumab in clinical practice.
Important limitations of this study are the absence of drug concentrations and limited endoscopic data, especially in Crohn’s disease. Despite the prospective design, only data collected in routine care were captured. We used various measures to define active inflammation at the inclusion of patients with Crohn’s disease. For some of these measures, such as CRP and f-calprotectin, the cut-off for identifying patients with endoscopically active disease is still poorly defined, and chosen thresholds may have influenced our results. In Sweden, corticosteroid tapering protocols may slightly differ between hospitals. Therefore, no valid estimate of the mean corticosteroid dose at different time-points during follow-up could be provided. Some patients lacked data on clinical disease activity during follow-up. However, attrition analyses revealed no differences in sex, age or disease duration between patients with and without data (data not shown).
In conclusion, our study demonstrates the long-term effectiveness of vedolizumab in a real-world setting. Our data show significant improvements in disease activity and HRQoL in patients with history of treatment-refractory Crohn’s disease and ulcerative colitis. Moreover, we found that high clinical disease activity at the initiation of vedolizumab was inversely associated with clinical remission at week 52, but no other predictors of response were identified. Further studies are needed to position vedolizumab in the therapeutic armamentarium of IBD and to identify molecular predictors for the development of individualised treatment algorithms.
Supplemental Material
sj-docx-1-tag-10.1177_17562848211023386 – Supplemental material for Real-world effectiveness of vedolizumab in inflammatory bowel disease: week 52 results from the Swedish prospective multicentre SVEAH study
Supplemental material, sj-docx-1-tag-10.1177_17562848211023386 for Real-world effectiveness of vedolizumab in inflammatory bowel disease: week 52 results from the Swedish prospective multicentre SVEAH study by Carl Eriksson, Sara Rundquist, Vyron Lykiardopoulos, Ruzan Udumyan, Per Karlén, Olof Grip, Charlotte Söderman, Sven Almer, Erik Hertervig, Jan Marsal, Jenny Gunnarsson, Carolina Malmgren, Jenny Delin, Hans Strid, Mats Sjöberg, David Öberg, Daniel Bergemalm, Henrik Hjortswang and Jonas Halfvarson; The SWIBREG SVEAH Study Group in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848211023386 – Supplemental material for Real-world effectiveness of vedolizumab in inflammatory bowel disease: week 52 results from the Swedish prospective multicentre SVEAH study
Supplemental material, sj-docx-2-tag-10.1177_17562848211023386 for Real-world effectiveness of vedolizumab in inflammatory bowel disease: week 52 results from the Swedish prospective multicentre SVEAH study by Carl Eriksson, Sara Rundquist, Vyron Lykiardopoulos, Ruzan Udumyan, Per Karlén, Olof Grip, Charlotte Söderman, Sven Almer, Erik Hertervig, Jan Marsal, Jenny Gunnarsson, Carolina Malmgren, Jenny Delin, Hans Strid, Mats Sjöberg, David Öberg, Daniel Bergemalm, Henrik Hjortswang and Jonas Halfvarson; The SWIBREG SVEAH Study Group in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We acknowledge Malin Olsson, Åsa Smedberg, Ida Gustavsson and the Department of Clinical Research Support, Örebro University Hospital for administrative support and all involved patients, clinicians and nurses for their efforts and contribution.
Author contributions
CE, CM and JH designed the research study. CE, SR, VL, PK, OG, CS, SA, EH, JM, JG, JD, HS, MS, DÖ, DB, HH and JH collected the data. SR, CE and RU analysed the data, SR, CE, RU and JH interpreted the findings. SR, CE and JH drafted the manuscript and all authors critically revised the manuscript. All authors approved the final version of the article, including the author contributions list. CE acts as the submission’s guarantor.
Conflict of interest statement
CE has served as a speaker, a consultant and an advisory board member for Takeda, Janssen Cilag, Pfizer, Abbvie, and has received research funding from the Regional Agreement on Medical Training and Clinical Research between Region Örebro County and Örebro University: ALF. SR has served as a speaker for Takeda and has received research funding from the research committee in Region Örebro County and the Regional Agreement on Medical Training and Clinical Research between Region Örebro County and Örebro University: ALF. OG has served as a speaker, a consultant and an advisory board member for Ferring, Janssen, Pfizer and Takeda. SA has served as a speaker, consultant or advisory board member: AbbVie, Janssen, Takeda, Tillotts Pharma, Vifor Pharma. EH has served as a speaker, consultant or advisory board member: Abbvie, Gilead, Janssen, Pfizer and Takeda. JM has served as a speaker, consultant or advisory board member: AbbVie, Bristol-Myers Squibb, Ferring, Hospira, Janssen, MSD, Otsuka, Pfizer, Sandoz, Svar, Takeda, Tillotts and UCB Pharma, and has received grant support from AbbVie, Ferring, Pfizer and Takeda. JG has served as a speaker and consultant: Abbvie, Ferring, Tillotts Pharma, Takeda. CM is an employee of Takeda. HS has served as a speaker, consultant or advisory board member: Abbvie, Ferring, Gilead, Janssen, Pfizer, Takeda and Tillotts Pharma. DB has served as a speaker, consultant or advisory board member: Ferring, Janssen, Pfizer and Takeda. HH has served as a speaker, consultant or advisory board member: AbbVie, Janssen, Pfizer, Takeda, Tillotts Pharma, Vifor Pharma, and received grant support from Ferring and Tillotts Pharma. JH has served as a speaker, consultant or advisory board member: AbbVie, Aqilion, Celgene, Celltrion, Dr Falk Pharma and the Falk Foundation, Ferring, Gilead, Index Pharma, Janssen, Lincs, MSD, Novartis, Olink Proteomics, Pfizer, Prometheus Laboratories Inc., Sandoz, Shire, Takeda, Thermo Fisher Scientific, Tillotts Pharma, Vifor Pharma, and received grant support from Janssen, MSD and Takeda. VL, RU, PK, CS, JD, MS and DÖ have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Takeda, ENCePP registration number: EUPAS22735.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
