Abstract
Crohn’s disease is a chronic inflammatory, relapsing-remitting, and progressive gastrointestinal disorder with an often-negative impact on the physical, emotional, and psychological well-being. Over the past two decades, the medical compendium for the treatment of Crohn’s disease has increased significantly, enabling treatment beyond symptoms. Indeed, early and timely use of effective medical therapy has been reflected by improved outcomes with reduction in surgery and ability to achieve clinical and endoscopic remission, reduce corticosteroid dependance, and prevent long-term complications in more patients. In this review, we discuss the key milestones in the medical management of Crohn’s disease.
Introduction
Crohn’s disease (CD) is a chronic inflammatory, relapsing-remitting, and progressive gastrointestinal disorder with variable disease location and behaviour. 1 It was first recognised as a separate entity from ulcerative colitis (UC) in the landmark publication by Crohn et al. in 1952, 2 although there have been previous reports describing ‘regional ileitis’ or ‘regional enteritis’. 3 Currently, it is believed that CD is caused by the interaction between the environment we live in, our immune system, genetics, and the microbiome 4 .
Over a course of 20 years, the actuarial rate of developing inflammatory, structuring, and penetrating disease is 12%, 18%, and 70%, respectively. 5 These complications will likely result in multiple surgeries and eventual disability, affecting patients’ physical and psychosocial functioning. 6 Encouragingly, recent evidence indicates that surgery rates are declining in CD, which may partly be associated with the early and timely use of medical therapy. 7
Over the last five decades, rapid strides in our understanding of the immuno-pathogenesis of CD and consequent improvements in pharmacological therapies has enabled clinicians to realise what can be achieved through abrogation of the immuno-inflammatory pathway. Our paradigms have evolved from symptom management to clinical and endoscopic remission, with the aim of reducing the long-term use of corticosteroids and preventing long-term complications and disability. In this review article, we discuss the key milestones in the medical management of CD, including current therapies (See Figure 1), a look at the immediate future with promising therapies on the horizon.

(a) The timeline of the introduction of immunosuppressive medication prior to the use of biologics. (b) The timelines of the milestone studies that introduced the biological medication used today for CD.
Corticosteroids
Truelove and Witts first demonstrated the efficacy of corticosteroid treatment in acute severe UC in 1955. 8 In 1979 and 1984, two landmark studies, the National Cooperative Crohn’s Disease Study (NCCDS) and the European Cooperative Crohn’s Disease Study (ECCDS) established the efficacy of prednisolone and 6-methylprednisolone, respectively, in inducing remission in patients with active moderate to severe CD.9,10 When evaluating 192 patients with CD over a 2-year period, 6-methylprednisolone was shown to be the most effective drug overall compared with 6-methylprednisolone and sulfasalazine combination, sulfasalazine alone, or placebo. The superiority of this drug was shown in overall comparison of all patients (p < 0.001), isolated small bowel disease (p < 0.05), and in those with small and large bowel disease (p < 0.05). 10
Corticosteroids, however, have numerous unwanted side effects, such as metabolic (steroid-induced diabetes, cushingoid appearance, and hepatic steatosis), central nervous system (psychosis, insomnia, and emotional disturbances), gastrointestinal (dyspepsia and peptic ulcer), musculoskeletal (osteonecrosis of the jaw and hip, osteoporosis, and growth failure), skin (easy bruising, skin thinning, weight gain, acne, hirsutism, striae, and purpura), and ocular effects (glaucoma and cataracts).11,12 Long-term use can also increase the risk of infection, lead to impaired wound healing, and can result in steroid dependence. Furthermore, patients may suffer with glucocorticoid withdrawal syndrome when attempting to stop or wean this medication. More recently, prolonged corticosteroid therapy has shown to be associated with an increase in mortality in patients with CD. 13 In a prospective study, the TREAT registry 14 found that prednisolone was linked to increased mortality risk (hazard ratio (HR): 2.14, 95% confidence interval (CI): 1.55–2.95; p < 0.001) with similar findings from the European ENCORE registry 15 (HR: 3.58, 95% CI: 1.49–8.61).
In 1994, a newer glucocorticoid formulation, budesonide, was shown to have equal efficacy to prednisolone, 16 with a 15 times greater affinity for glucocorticoid receptors, such that 5 mg of budesonide is equivalent to 12 mg of prednisolone. 17 Budesonide has an added advantage of a high first pass liver metabolism and rapid elimination, resulting in minimal systemic absorption and thereby reducing the risk of steroid-induced side effects. 18 While budesonide 9 mg once daily has been shown to be superior than placebo in inducing remission for patients with mild to moderate ileocaecal CD (OR: 2.92, 95% CI: 1.52–5.39),6,19 budesonide was inferior to prednisolone for the induction of clinical remission in severe ileocaecal CD or in colonic CD (relative risk (RR): 0.52, 95% CI: 0.28–0.95). 19
Corticosteroids remained the mainstay for induction therapy until the late 1990s 12 when evidence began showing that they only induce complete clinical remission in 48% and partial clinical remission in 32% of patients with active CD. 20 However, 20% of patients were found to be resistant from the onset, and at 1-year follow-up, 45% of the patients who responded initially became steroid-dependent with only 32% of patients maintaining a prolonged clinical response. 21 It became rapidly apparent that corticosteroids were ineffective at maintaining remission, reducing flares, or disease recurrence.9,22 –24 The French GETAID study provided further proof that corticosteroids were not disease-modifying agents with limited evidence in their ability to achieve endoscopic mucosal healing or preventing endoscopic relapse. 25 In this study, patients were given prednisolone 1 mg/kg for 7 weeks, and only 29% achieved endoscopic and clinical remission, with 71% showing active endoscopic lesions. In fact, 9% of patients had worsening endoscopic lesions despite symptomatic improvement. 25 All of the above limitations associated with prolonged corticosteroid use make a compelling case for using newer safer therapies that maintain remission without exposing patients to unwanted side effects.
5-Aminosalicylic acid compounds
5-Aminosalicylic acids (5-ASAs) play a fundamental role in inducing and maintaining remission for patients with UC. However, despite their extensive use, multiple studies over many years have demonstrated that there is limited evidence of benefit, if any, in CD (Figure 2). The initial study conducted by Gendre in 1993 demonstrated effectiveness with 5-ASAs in achieving and maintaining remission in CD (45% 5-ASA vs 29% placebo for 2-year remission rate). 26 However, this efficacy shown by Gendre could not be replicated in subsequent studies.27 –29 Systematic literature reviews also have not demonstrated efficacy of oral 5-ASAs compared to placebo in inducing or maintaining clinical remission in CD, with relapse rates at 12 and 24 months to be 53% versus 54% (RR: 0.98, 95% CI: 0.91–1.07) and 54% versus 58% (RR: 0.94, 95% CI: 0.68–1.29), respectively.30,31 A modest benefit in using 5-ASAs to prevent post-operative CD relapse, however, was demonstrated in a recent meta-analysis showing RR of relapse with 5-ASA versus placebo to be 0.86 (95% CI: 0.74–0.99). 32 Despite international guidelines from the British Society of Gastroenterology (BSG), 1 European Crohn’s and Colitis Organisation (ECCO), 33 American Gastroenterological Association (AGA), 34 and the United Arab Emirates (UAE IBD) association 35 advising against their use in CD, 5-ASAs remain widely prescribed. Notably, 5-ASA’s may also be associated with adverse effects including, headaches, nausea, malaise, rash, and the rare side effects such as interstitial nephritis, haemolytic anaemia, hepatitis, pancreatitis, and paradoxical worsening of colitis. 36

The initial study conducted by Gendre in 1993 showed efficacy with 5ASAs with further support by Ford et al. in their use for post-operative CD. However, there are multiple studies over the years that counteracted these studies and as such, 5ASAs are not currently recommended for use in induction or maintenance of CD.
Antibiotics
It is widely accepted that bacteria play a role in the pathogenesis of certain manifestations of CD, such as abscesses and fistulae. 12 There is also established evidence that bacterial overgrowth secondary to strictures and blind loops may respond well to antibiotic therapy. 12 Although the precise mechanisms of how broad-spectrum antibiotics work are uncertain, it is thought to consist of immunosuppressive activity (i.e. metronidazole), treatment of bacterial overgrowth and suppression of a bacteria-induced antigenic stimulus. 37
Metronidazole
Metronidazole, a nitroimidazole compound, was first reported to be effective in CD in 1975 who reported a response in four out of five patients with large bowel disease after 2 to 4 weeks treatment. 38 Metronidazole has high antimicrobial activity against anaerobes, which are present in high quantities in the neo-terminal ileum. As such, this drug has the greatest effect in patients with ileocolonic disease, which was first illustrated in a placebo-controlled trial of 56 patients. 39 Initial studies also suggested a benefit with metronidazole in treating perineal disease, although 100% of patients had recurrence of disease when discontinuation or dosage reduction was attempted.40,41 A small double-blind placebo-controlled study of 60 CD patients showed that metronidazole reduced the incidence of severe endoscopic recurrence 3 months post-operatively (13% in metronidazole vs 43% in placebo group; p = 0.02). 42 The results were borderline significant at 1 year (4% vs 25%) and was not significant at 2 (26% vs 43%) and 3 years (30% vs 50%) after resection; however, this may be due to the small sample size rather than clear lack of benefit from metronidazole.
The main drawback of metronidazole is the frequency of adverse effects, including gastrointestinal intolerance, metallic taste, and neurotoxicity. 12 It is often poorly tolerated, particularly at the high doses required to treat these patients. Thus, long-term therapy is not recommended.
Others
In a small pilot study of 33 patients, ciprofloxacin did not show much benefit over placebo 43 and trials with the gut-specific antibiotic rifaximin are currently awaited in the post-operative setting. 44 In a randomised placebo-controlled trial lasting 2 years with 213 patients receiving treatment with either clarithromycin, rifabutin, or clofazimine, early benefit of antibiotics was noted. However, there were no significant differences in relapse rates during follow-up. 45 In another phase-III trial, 331 patients with moderate to severe CD were randomised to receive either five capsules of RHB-104 (clarithromycin 95 mg, rifabutin 45 mg, and clofazimine 10 mg) twice daily or placebo. Remission was achieved at 26 weeks in 37% versus 23% on placebo; p = 0.019. A more recent study compared oral budesonide with a combination treatment of ciprofloxacin, doxycycline, and hydroxychloroquine. 46 Results showed that although budesonide had higher induction rates than antibiotics at 10 weeks (25% vs 7.4%), their efficacy reduced at 24 (3.1% vs 7.4%) and 52 weeks (3.1% vs 3.7%), respectively. Thus, this study illustrated that there may be some benefit for antibiotics in long-term remission use. However, further studies are needed before it can be said that antibiotics have a definitive role to play in CD management.
Thiopurines
Thiopurines (6-mercaptopurine (6MP) and azathioprine (AZA)), which act as antimetabolites and immunomodulators, were first discovered in the 1950s and originally used for the treatment of childhood leukaemia. 47 Although the first IBD patient to be treated with thiopurines was in 1962 and for UC, 48 studies in the early 1990s demonstrated the advantages of using thiopurines in CD. 49 Present et al. 49 showed an improvement with 6MP compared to placebo in the treatment of fistulas (31% vs 6%) and in allowing discontinuation of corticosteroids (75% vs 36%) over a 2-year double-blind crossover study of 83 patients (p < 0.001). The advantages of using thiopurines are its steroid-sparing effects; however, its slow onset of action (8–12 weeks) makes them ineffective for short-term induction in active, symptomatic disease.50,51 Regardless, thiopurines have been shown to be more effective than placebo for maintenance of remission in CD, although the quality of evidence for this has been reported as low (number needed to treat (NNT) = 9).
Candy et al. added further weight to the growing evidence of thiopurines in the use of CD. In their double-blind placebo-controlled trial of 63 patients, they were able to demonstrate a clinically significant advantage of using AZA at a dose of 2.5 mg/kg for the maintenance of remission while reducing the dose of prednisolone (42% vs 7% at 15 months, p = 0.001). 52 In the study by Ewe et al., 53 76% of patients on a combined regimen of azathioprine and corticosteroid went into clinical remission earlier (CDAI < 150) and maintained remission longer (12 weeks vs 4 weeks) than in the placebo-treated group (38%); p = 0.03.
As per any medication, thiopurines have their own collection of adverse effects including nausea, infections, allergic reactions, pancreatitis, myelosuppression, hepatotoxicity, and malignancy, particularly lymphoma and nonmelanoma skin cancer.54,55 In 1999, Cuffari et al. 56 first observed the link between leucopenia to thiopurine methyltransferase (TPMT) genetic polymorphism and bone marrow toxicity in adolescent CD. Sandborn 57 and Belaiche et al. 58 then validated the use of 6-thioguanine nucleotide (6-TGN) levels when prescribing thiopurines. 2018 brought about the discovery of NUDT15 gene testing, which if found to be deficient in individuals indicates susceptibility to thiopurine-related toxicity. 59 These studies were important as it allowed clinicians to use drug-level monitoring and genetic testing to personalise prescriptions and determine which patients thiopurines were safe to use in. They not only helped to confirm therapy compliance but also allowed thiopurine dose optimisation in non-responders first before escalating or changing treatment.
Methotrexate
Although thiopurines were the first medication to demonstrate they can maintain remission in CD, there was still concern among clinicians regarding their side effects and potential toxicity. Approximately 15% of patients are intolerant to thiopurines from non-specific nausea and malaise 12 and up to 50% will discontinue treatment within the first 2 years either due to adverse drug events or therapy failure. 60 Methotrexate (MTX) was a medication that attempted to bridge this gap. Although first developed for use in the 1950’s as a chemotherapeutic agent in cancers such as leukaemia, lymphoma and choriocarcinoma, 60 it was subsequently established in treating two chronic inflammatory diseases: rheumatoid arthritis and psoriasis. 61
Kozarek et al. first reported the use of MTX in 1989 in a pilot study of 21 patients with IBD (14 with CD). This study reported a clinical improvement in 79% of patients with CD with a reduction in steroid requirement (p = 0.006). 62 In a larger study of 141 patients, Feagan et al.63,64 further demonstrated that when compared to placebo, MTX was more effective at inducing (19.1% vs 39.4%, p = 0.025) and maintaining remission (39% vs 65%, p = 0.04) in patients with active CD. In addition, despite concerns regarding hepatic toxicity with MTX, no severe adverse events were recorded in these studies, concluding it to be a safe and effective drug.
Thus far, conventional management typically involved the use of broad-spectrum anti-inflammatory agents and immunosuppressants, often sequentially with the aim of relieving symptoms and preventing long-term complications in CD. Although we achieved modest and real benefits with steroid-sparing agents in inducing and maintaining clinical remission in CD, the advent of the biological era would bolster ambitions with treatment goals (Table 1).
Landmark studies that helped define the use of biologics in CD management

The millenium and the dawn of a new (biological) era in IBD therapeutics
In 1985, Beutler et al. 65 showed that the pro-inflammatory cytokine, tumour necrosis factor (TNF), played a significant role in endotoxin-mediated shock. Keffer et al. 66 subsequently demonstrated the role of TNF in the pathogenesis of inflammatory arthritis in mice; but it was not until 1990 when MacDonald et al. 67 found an increase in TNF concentration in tissue inflammation in rheumatoid synovial membranes and interestingly, in the mucosa and lamina propria of patients with CD that the prospect of anti-TNF therapy received attention. Elliot et al. 68 conducted the first human trial for rheumatoid arthritis using a TNF-alpha-directed chimeric mouse/human monoclonal antibody, which was followed shortly after by the first human trial successfully using this drug in a small sample size of 10 CD patients. 69 The advent of anti-TNF therapy and its ability to effectively induce and maintain remission while boasting of its corticosteroid-sparing effects, mucosal healing and reduced hospitalisation and surgery rates represent a defining moment in IBD therapeutics.
Anti-TNF biologics
Infliximab
Infliximab (IFX), a chimeric anti-TNF-alpha monoclonal antibody was the first anti-TNF agent to gain a licence for CD therapy. This was demonstrated in the landmark ACCENT I study, whose primary aim was to demonstrate that maintenance IFX therapy for CD patients can provide better long-term efficacy than no further treatment after a single-dose infusion. 70 Results showed that patients who received maintenance IFX therapy were two times more likely to maintain clinical remission compared with placebo treatment (OR: 2.7, 95% CI: 1.6–4.6). The median time to loss of response was 46 weeks (interquartile range (IQR): 17 to >54) in the treatment group versus 19 weeks (10–45) in the placebo group. This study was important for many reasons. Steroid dependent and immunomodulatory refractory patients now had a realistic medical option, with evidence that IFX use was safe and well-tolerated. Another important treatment advance with this study was patients on maintenance IFX were able to reduce steroid use, with a third of patients being able to stop steroids completely (29% vs 9% in placebo; OR: 4.2, 95% CI: 1.5–11.5; p = 0.004). Furthermore, the study also supported the concept that combination therapy with AZA or MTX might have an additional or synergistic efficacy with IFX, with 50% of patients who received a concomitant immunosuppressive maintaining a clinical response at week 54 compared with 41% not receiving these drugs. 70
Fistula development in CD is common and, prior to the use of biological therapy, was notoriously difficult to medically manage with surgery usually as the main treatment option. The ACCENT-II study demonstrated that IFX treatment was effective in 72.2% of patients in inducing closure of rectovaginal fistulas when given at 0, 2, and 6 weeks. 71 Furthermore, IFX provided a 3 months longer duration of closure than placebo when IFX induction was followed by a maintenance regimen. However, a limitation to this study was that fistula closure was assessed with only physical examination and symptom report. Optimism around fistula response from initial studies should be tempered by evolving paradigms and objective assessments with examination under anaesthesia (EUA), pelvic magnetic resonance imaging (MRI), or anorectal endoscopic ultrasonography (EUS), which often show persistent fistula tracts even when fistula drainage has ceased. 71
Building on early evidence supporting combination therapy using thiopurines with IFX from ACCENT-I, 70 the SONIC trial investigated the efficacy of IFX, AZA, and a combination of the two drugs for inducing and maintaining corticosteroid-free clinical remission in patients with moderate to severely active CD. 72 The primary aim of this study was to evaluate the rate of corticosteroid-free clinical remission at week 26. Disease severity was evaluated using the CDAI and IBD-Questionnaire (IBDQ) scores, as well as with direct visualisation of mucosal healing with ileocolonoscopy at week 26. Corticosteroid-free remission was achieved greatest with combination therapy (43.9%) although results were significantly better with IFX-based strategies (30.1%) as compared with AZA alone (16.5%). Furthermore, antibodies to IFX were detected at week 30 in only 1 of 116 patients receiving combination therapy versus 15 of 103 patients receiving IFX alone, suggesting that AZA may have a protective effect against IFX antibodies. 72 In a post hoc analysis of the SONIC trial, it was highlighted that those who were on combination therapy (IFX and AZA) had significantly greater composite remission rates (mucosal healing and clinical) than those on monotherapy (IFX) (range: 52.3%–63.6% vs 12.9%–29.0%; p ⩽ 0.013 for all comparisons). Furthermore, this study highlighted that those who achieved this composite endpoint were more likely to have higher IFX trough levels hence the suggestion that AZA may help boost the effectiveness of IFX through increasing IFX trough levels but as yet this remains association rather than causation. 72
Adalimumab
An important limitation with IFX is the development of anti-chimeric antibodies which can occur in 7–10% of patients regularly receiving 4- or 8-weekly maintenance infusions. 73 Antibody formation can lead to infusion reactions, loss of efficacy and delayed hypersensitivity reactions. While IFX was developed as a humanised murine antibody, there was resurgent interest in a molecule of entirely human origin, and therefore less likely to cause adverse allergic reactions. 74 Adalimumab was developed as a subcutaneous injection which can be self-injected by the patient. The CLASSIC-I trial was a short 4-week dose-ranging study evaluating the efficacy of another anti-TNF drug, adalimumab in the Crohn’s cohort. A small number of patients (299) were included who were randomly assigned to receive adalimumab induction treatment doses at weeks 0 and 2 of either 40 mg/20 mg, 80 mg/40 mg, 160 mg/80 mg, or placebo. Compared to placebo, the only induction-loading dose regimen that achieved statistical significance for remission rates was 160 mg/80 mg. Furthermore, only 1 out of 225 patients developed antibodies against adalimumab, although this may have been an underestimation considering the short course of the study duration. 75
While the CLASSIC-I study demonstrated that adalimumab was effective in inducing remission by week 4, the CLASSIC II study went on to show that adalimumab was equally effective in maintaining remission in patients with moderate to severe CD, either at a dose of 40 mg weekly or every other week. At week 56, those on adalimumab were 1.5–2 times more likely to have maintained remission compared to placebo. The CLASSIC-II study was praised for selecting remission as the primary outcome measure as opposed to maintenance of response. However, a significant limitation to this study is the relatively small sample size (<20 patients in each group) such that it was not powered to detect statistical significance. It is also important to mention that patients who were randomised into the study were from a highly selected cohort that had rapidly responded to the drug within 4 weeks of treatment. 76
The CHARM trial supported and extended the findings of the CLASSIC-I and -II studies by confirming with a greater sample size that adalimumab was effective in inducing and maintaining long-term clinical remission in CD patients who initially responded to induction therapy with adalimumab. Specifically, patients given adalimumab 40 mg every other week or weekly had greater remission rates compared to placebo at week 26 (40%, 47% and 17% respectively, p < 0.001) and week 56 (36%, 41%, and 12% respectively, p < 0.001). In contrast to CLASSIC-II, this study demonstrated a statistical difference in lowering disease activity and improving quality of life (QoL) in the adalimumab treatment group compared to the placebo group. The study also demonstrated that adalimumab dosing of either weekly or alternate weeks were equally effective in maintaining remission in patients with CD. Importantly, the efficacy results from this study showed similar response and remission rates to IFX, not only in maintaining corticosteroid-free remission but also in the complete closures of fistulas. Furthermore, this study was crucial in demonstrating that adalimumab was effective in patients who previously lost response or were intolerant to infliximab. However, the patients who were naïve to anti-TNF therapy had numerically greater remission rates at week 26 and week 54 as compared to those with a previous history of anti-TNF use. 77
Following on from the CHARM study, the GAIN study was the first randomised, double-blind placebo-controlled trial in any immune-mediated disease to demonstrate the efficacy of a second TNF antagonist where the first TNF antagonist had failed. Compared to placebo (7%), patients with moderate to severe CD who were given adalimumab (21%) had a superior response in inducing remission in patients who were previously intolerant or lost response to infliximab; p < 0.001. The main limitation to this study was the short 4-week duration, although the efficacy for maintenance treatment was previously demonstrated in the 52-week long CHARM study. 78
Up to now, studies on adalimumab used clinical response with CDAI and IBDQ mean scores to assess remission rates as primary treatment goals for patients with CD. EXTEND was the first study designed to evaluate mucosal healing as the primary end point. Results demonstrated that adalimumab can provide early and sustained mucosal healing in patients with moderate to severe ileocolonic CD. Higher rates of mucosal healing with adalimumab compared with placebo were observed by week 12 (27% vs 13%, p = 0.056) and week 52 (24% vs 0%, p < 0.001). Patients were eligible if they had longer disease duration, in whom conventional therapy had failed, with 52% of patients receiving adalimumab as their second TNF antagonist, and they were allowed to remain on corticosteroids if they were receiving them at baseline. Recruitment of these patients were reflective of the ‘real-world’ and was a main strength of this study. 79
The SERENE-CD trial is currently investigating high (160 mg at week 0, 1, 2, and 3) versus standard (160 mg at week 0 followed by 80 mg at week 2 and 40 mg at week 4) adalimumab induction dosing regimens. Although the study is ongoing, early results have not shown a significant difference in the rate of clinical remission at week 4 or endoscopic remission at week 12 between the high or standard doses. 80
Switching from infliximab to adalimumab is extremely common in clinical practice and is often a result of convenience or financial burden rather than clinical necessity. The SWITCH trial investigated the impact of electively switching treatments from intravenous infliximab to subcutaneous adalimumab in patients with well-controlled CD. Results demonstrated that this elective switch was associated with loss of tolerance and loss of efficacy within 1 year and as such, led to worse outcomes in the CD patients. Due to the limited number of approved biological agents, this study highlighted the importance to adhere with the first anti-TNF agent unless there is loss of response or tolerance. 81
Immunogenicity
Anti-TNF drugs are highly effective in the management of CD but treatment failure is a common downfall to these medications. The personalised anti-TNF therapy in Crohn’s disease study (PANTS) aimed to identify specific clinical and pharmacokinetic factors that predicted primary non-response. Their multivariate analysis demonstrated that the only factor independently associated with primary non-response was low drug concentration at week 14 for both infliximab and adalimumab. For both drugs, suboptimal drug concentrations at week 14 predicted immunogenicity, with the formation of anti-drug antibodies. 82
Certolizumab pegol
The anti-TNF therapies, IFX and adalimumab, were shown to be effective for induction and maintenance of moderate to severe CD. However, 40–50% of anti-TNF ‘primary responders’ develop either a loss of response and/or develop acute or delayed hypersensitivity reactions within 6–12 months. 83 Certolizumab pegol is a pegylated humanised Fab’ fragment of the anti-TNF monoclonal antibody which has a high affinity for TNF-alpha. Similar to adalimumab, it has the added advantage that it can be administered subcutaneously. The PRECiSE 1 study was a double-blind randomized controlled trial (RCT) that compared the efficacy and safety of certolizumab pegol against placebo in patients with moderate to severe CD. 83 Results showed a modest benefit with statistically more patients displaying >100-point CDAI reduction (CR100) at week 6 (37% certolizumab vs 26% placebo) but not at week 26 (22% certolizumab vs 12% placebo). The PRECiSE-284 study was undertaken in parallel with PRECiSE-1, with similar inclusion criteria but conducted at separate sites. Interestingly, 64% had a response at week 6 and thus continued into the trial with 43% in remission (defined as CDAI score of less than or equal to 150) following induction therapy. By week 26, there was a statistically significant response to certolizumab (48%) as compared to placebo (29%). This study highlighted the superior response of subcutaneous certolizumab; however, there is a clear discrepancy in the response and remission rates at week 6 from the PRECiSE-1 and -2 studies, for reasons that remain unexplained. Thus, although the PRECiSE-1 study indicates a lack of efficacy with certolizumab, the PRECiSE-2 data suggest the opposite in the induction of CD remission. As a result, the European Medicines Agency (EMA) cited concerns about the insufficient evidence of the efficacy of certolizumab and the short study duration of the PRECiSE trials. Thus, it is currently only approved for use in CD in the United States, Switzerland, and Russia. 84
Biosimilars
Due to the recent expiry of infliximab and adalimumab patents, several biosimilars have been approved for use in IBD by the EMA and US Food and Drug Administration (FDA).85,86 These agents are lower priced than the original compounds by up to 70% and by definition, are highly similar to the reference drug such that any molecular and/or structural differences should not affect its quality, safety, or efficacy. 85 The use of biosimilars provides several advantages including easing the economic burden of anti-TNF treatment, increasing access to anti-TNF therapy, thereby allowing earlier access to treatment, and availability of assays for measuring drug concentrations to optimise patient care. 87 This paves the way for reducing complications and functional disability associated with IBD. 86
In 2021, Schreiber et al. conducted an open-label study investigating the use of subcutaneous IFX biosimilar compared to its intravenous route. A total of 131 patients were recruited and results showed comparable clinical remission rates between the two treatment modes. Efficacy, safety, and immunogenicity assessments were also comparable. A subcutaneous option for IFX may have multiple advantages including ease of administration and reduced requirement for medical visits and associated travel, in addition to optimising medical resources and improving treatment options for our IBD patients. 88
In summary, anti-TNF therapies have transformed the care of patients with IBD, re-defining our perceptions around meaningful disease control, moving beyond symptom control to bolder definitions such as mucosal healing, histological and deep remission, and an improvement in QoL.89,90 Even as anti-TNF agents fuelled our ambitions with hitherto unachievable outcomes, it became obvious that they are not universally effective, with 30–50% of patients being primary non-responders and with further attrition from subsequent loss of response (mechanistic escape, immunogenicity, or intolerance). 91 There is also the real risk of infectious complications attributable to non-specific inhibition of TNF-mediated immunologic cascades.91,92
Recently developing biologics
Vedolizumab
Evolution in our understanding of the involvement of T-lymphocyte biology orchestrating gut inflammation has paved the way for the development of several agents directed against trafficking of effector T-lymphocytes towards the gut mucosa. 93 In 2014, vedolizumab was introduced into the biologic armamentarium. Vedolizumab is a highly selective monoclonal antibody that blocks lymphocytic gut migration via antagonism of α4β7 integrin on lymphocytes. 94 This inhibits binding to mucosal addressin cell adhesion molecule-1, which is overexpressed in the intestinal vasculature in IBD, and thus reduces trafficking to the gut. 94 The GEMINI-II study 95 investigated the efficacy of vedolizumab in inducing and maintaining remission in CD patients. The primary endpoint was to assess clinical remission (CDAI score of less than or equal to 150) at week 6. The study showed that of the 368 randomised patients, vedolizumab induction therapy was more likely than placebo to result in remission at week 6 (14.5% vs 6.8%; p = 0.02). However, by week 52, patients who had an initial response to induction therapy had higher rates of clinical remission, CR100 response and glucocorticoid-free remission than placebo (21.6%) when vedolizumab was given 4- or 8-weekly (36.4% and 39% respectively); p = 0.004 and p < 0.001, respectively. The modest effect of vedolizumab induction at week 6 could be attributed to the severity of disease in the study population, where a large proportion of patients had fistulising disease (37%), undergone previous surgery (42%), previous treatment failure with one or more TNF antagonists (50%), or treatment failure with two or more TNF antagonists (30%).
The GEMINI-3 study focused specifically on the efficacy of vedolizumab with previous anti-TNF failure. 96 The results did not show a significant difference between vedolizumab and placebo at week 6 (15.2% vs 12.1% respectively, p = 0.433), but there was a modest benefit at week 10 (26.6% vs 12.1% respectively, p = 0.001). The concern over vedolizumab’s inability to reduce remission at week 6 and the lack of mucosal healing data do not provide compelling evidence for its use in CD but one must consider certain caveats. It is likely that the timing of assessment was the limiting factor as evidenced by the Gemini-3 trial, wherein vedolizumab was superior to placebo for induction at 10 weeks but not at week 6, in patients with prior anti-TNF failure. For maintenance of remission at 52 weeks, vedolizumab demonstrated superiority over placebo with a magnitude of effect generally similar to that seen in UC. 97 Although the induction data appear less compelling, the clearly and clinically meaningful effect after 30 weeks suggests that vedolizumab is an appropriate option for selected patients in whom concomitant use of bridging strategies (such as co-induction with corticosteroids) is possible and where surgery may not be the appropriate option. Also, it may be an appropriate first-line biologic in patients where the focus is safety, such as in the elderly.98,99 Furthermore, several ‘real-world’ studies have gone on to showcase vedolizumab’s safety and efficacy in moderate-severe CD or those who have failed previous conventional therapy.100 –102 These studies demonstrated that vedolizumab may be more beneficial for patients who are biologically naïve and in patients with an inflammatory phenotype, as opposed to a stricturing or penetrating presentation. 103
Vedolizumab is currently licenced via the intravenous route; however, the latest VISIBLE-2 study has demonstrated that the subcutaneous form can also maintain clinical remission with a similar safety profile as per the intravenous route. 104 As stated above, subcutaneous biologics are preferred over the intravenous for both patients and healthcare services.
Ustekinumab
In 2016, ustekinumab was introduced as another out-of-class biologic option for patients with CD. Ustekinumab is a monoclonal antibody against the p40 subunit of interleukin-12 and interleukin-23, which was first approved for use in patients with psoriasis in 2009 and in 2013 for psoriatic arthritis. 105 To investigate its efficacy in CD, two 8-week placebo-controlled induction trials (UNITI-1 and UNITI-2) and one 44-week maintenance trial (IM-UNITI) were undertaken. 106 Patients who completed either UNITI-1 or UNITI-2 could then enrol in the IM-UNITI maintenance trial. Results of the three trials showed consistent superiority with ustekinumab over placebo in inducing and maintaining remission in patients with moderately to severely active CD. At week 6, patients receiving intravenous ustekinumab at a dose of either 130 mg or 6 mg/kg had significantly higher response rates than placebo (UNITI-1: 34.3% vs 33.7% vs 21.5% respectively; p < 0.003, UNITI-2: 51.7%, 55.5% and 28.7% respectively; p < 0.001). At week 44, patients receiving maintenance doses of ustekinumab every 8 or 12 weeks were more likely to be in remission than placebo (53.1% vs 48.8% vs 35.9%, respectively; p < 0.05). These results were irrespective of previous treatment or response to a TNF antagonist, and its benefit was demonstrated as early as week 3. Moreover, the rate of adverse events was not significantly different from that of placebo. 106 Subsequently in 2016, both the FDA and European Commission approved the use of ustekinumab for CD treatment for whom previous therapies have failed. Since then, several real-world studies have confirmed the efficacy and safety profile of ustekinumab107–109 and its efficacy in perianal disease and fistula healing. 110
The future
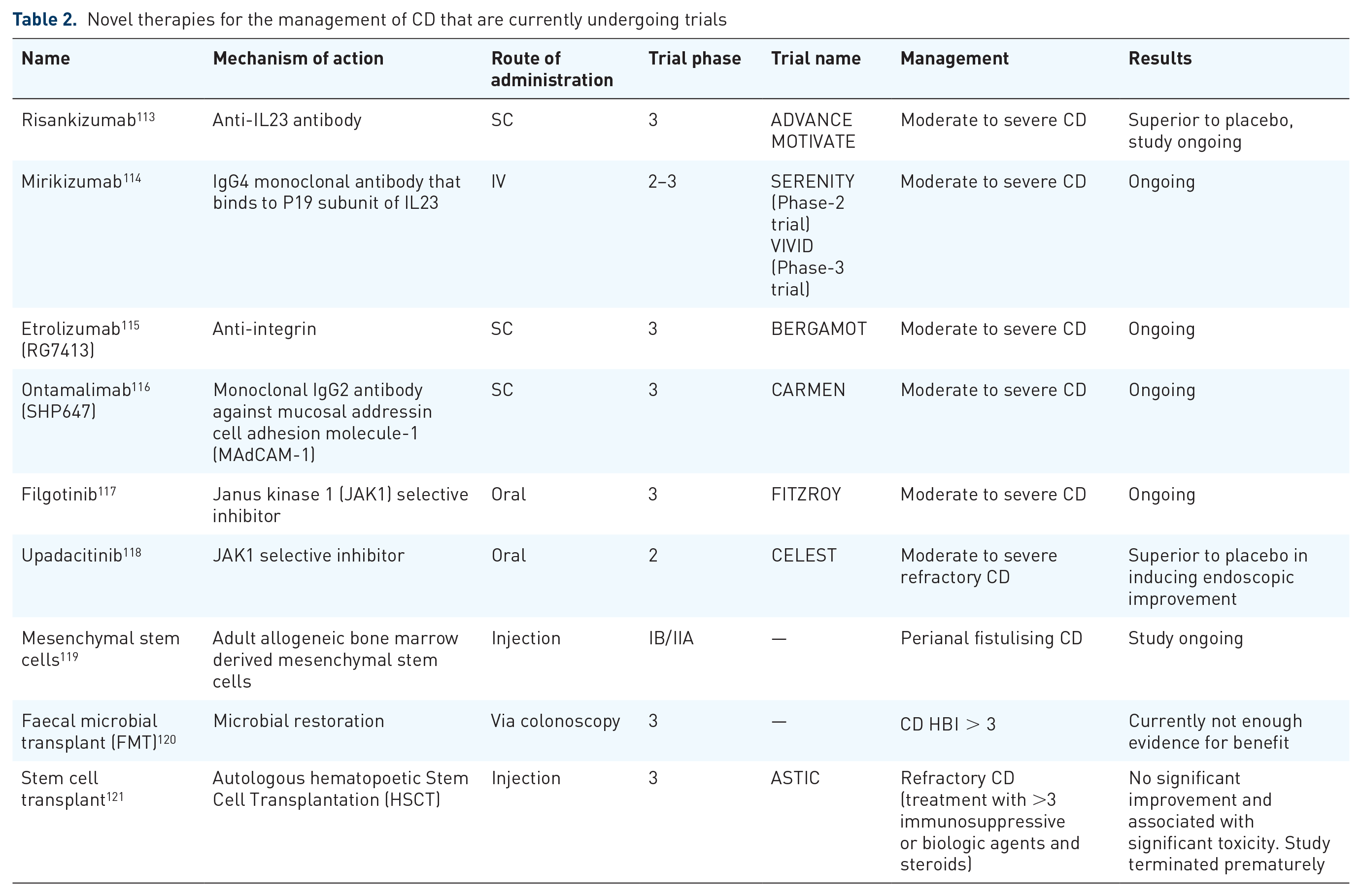
For many years, CD was managed inadequately using steroids, 5ASAs, immunomodulators, and antibiotics. The introduction of anti-TNF agents in the late 1990s created a paradigm shift in the management of this chronic incurable disease. Indeed, this was the first medication class that reduced the risk of surgery and hospitalisation, particularly if used early in the disease course. 111 Anti-TNF therapy did not come without its list of problems, including high rates of primary and secondary non-responders, and the long-term risk for complications. With the advent of vedolizumab and ustekinumab, clinicians were able to overcome these issues. However, anti-TNF agents are still first-line treatment for complex patients including fistulizing disease, pregnancy, children, post-op recurrence, and peri-operative safety. 112 Furthermore, despite the development of biologics, there is still a high rate of surgery and post-operative recurrence. Over the last decade, the probability of surgery has been reported to be between 3% and 96% within 15 years of diagnosis, with clinical relapse and reoperation rates of 50–60% and 28–45%, respectively. 7 Subsequently, there are several ongoing studies investigating new biological therapies for the treatment of CD (Table 2), which are likely to provide a greater array of medications in the armamentarium towards helping patients with CD.
Novel therapies for the management of CD that are currently undergoing trials
Conclusion
CD is a chronic relapsing-remitting disease with a high morbidity rate. Its disease complexity can result in long-lasting physical, emotional, and psychological effects on patients. Over the past two decades, the medical compendium for the treatment of CD has expanded exponentially. Although surgery continues to play a pivotal role in achieving disease control for these patients with aggressive disease, novel mechanistic approaches and deeper insights with existing therapies hold real promise. The prospect of these intellectual efforts being rewarded through meaningful outcomes for individuals living with CD is now more realistic than ever before.
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JKL has served as a speaker for Abbvie, Janssen, Takeda and Tillotts, consultant for Abbvie, Galapagos, Janssen and Pfizer and an advisory board member for Arena Pharma, Galapagos and Janssen and has received research funding from Galapagos and Takeda. PJS has served as a speaker and advisory board member for Janssen, Takeda, Tillotts, and Celltrion. JPS has received speaker fees for Takeda He has received conference funding by Tillots, Abbvie, and Janssen. He has received a research grant from Tillots.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
