Abstract
Background:
Intraepithelial eosinophils have been described in patients with esophageal motor disorders. Conversely, motor disorders and even achalasia have been reported in patients with eosinophilic esophagitis (EoE).
Objectives:
This prospective study aimed to further investigate the association between eosinophilia and achalasia.
Design:
This is a prospective study.
Methods:
A series of 30 consecutive treatment-naïve achalasia patients (mean age: 45.5 years, 53% female) undergoing laparoscopic myotomy were prospectively enrolled. Preoperative demographic and clinical data, radiological findings, and high-resolution manometry (HRM) features were collected. Patients underwent upper endoscopy with biopsy sampling before surgery, and two samples of distal esophageal muscle were obtained during myotomy. Histopathologic findings were analyzed, and clinical characteristics were compared based on the presence and absence of eosinophils on biopsy samples.
Results:
Intraepithelial eosinophils were found in seven patients (23%; mean 8 eos/high-power field, 95% CI 0–15), but only one patient demonstrated >15 eos/high-power field, and one had eosinophilic infiltration of the esophageal muscle. Patients with intraepithelial eosinophilia had a narrower esophageal diameter on barium radiography (2.4 vs 3.6 cm without eosinophilia, p < 0.05). There were no additional differences in histopathology or preoperative and postoperative data between patients with and without intraepithelial eosinophilia.
Conclusion:
Our study confirmed that intraepithelial and intramuscular eosinophils are present in some patients with achalasia, but seldom meet the criteria for EoE. Only a narrower esophagus was found in patients with intraepithelial eosinophils as compared to those without, possibly reflecting an earlier stage of the disease.
Introduction
Background/rationale
The role of eosinophilic infiltrates in the pathogenesis of esophageal disorders is an intriguing area of esophageal research. The esophagus is normally devoid of eosinophils. The prototypical eosinophilic disorder of the esophagus is eosinophilic esophagitis (EoE), a chronic, immune/antigen-mediated esophageal disease with histologic evidence of eosinophil-predominant inflammation and symptoms related to esophageal dysfunction.1,2 Although pathogenesis remains unclear, genetic, environmental, and immune-mediated factors have been implicated.3–5
Although EoE is predominantly a mucosal disorder, several studies have described esophageal motility disorders, including achalasia, in patients fulfilling EoE criteria.6–13 Since eosinophils have been found in the muscularis propria in achalasia and can release neurotoxic products, these cells have been hypothesized to participate in achalasia pathogenesis.6,14–17 A recent study reported a relative risk as high as 32.9 (95% confidence interval: 24.8–42.8) for the association of achalasia with EoE and other allergic disorders. 18 Interestingly, both symptoms and motor abnormalities may revert to normal with anti-inflammatory treatments in some EoE patients, unlike traditional achalasia, which is progressive and irreversible by definition.6,7,19 However, scarcity and heterogeneity of available data have prevented further speculations to date.
Objectives
We hypothesized that a true association between eosinophilic infiltration and achalasia is plausible in some instances through shared eosinophilic inflammation. We therefore aimed to clarify the prevalence and the degree of esophageal eosinophilia in patients with achalasia, considering that EoE in achalasia cohorts seems far less frequent than vice versa.4,20 We prospectively evaluated histological samples from the esophageal mucosa and muscularis propria of consecutive patients with achalasia undergoing laparoscopic Heller myotomy (LHM). Secondary aims were to evaluate demographic, clinical, and endoscopic features as well as response to treatment in these patients.
Methods
Study design and setting
This was a prospective study conducted at the University of Padua, between the Chirurgia Generale 1 and the Gastroenterology Departments. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 21 All the diagnostic tests are standard in any department dealing with esophageal achalasia, and also the surgical procedure, introduced more than 30 years ago, has become a standard procedure performed in the majority of surgical centers. 22
Participants
Consecutive patients presenting with dysphagia, diagnosed with treatment-naïve achalasia, and undergoing LHM for definitive management over a 6-month period (February–July 2022) were evaluated for inclusion in the study. Exclusion criteria consisted of age <18 or >80 years, previous endoscopic or surgical management, including botulinum toxin injections, pneumatic dilation, or per oral endoscopic myotomy (POEM), radiological stage IV of the disease, and refusal to participate in the protocol. The study protocol was approved by the Ethical Committee of the Azienda Ospedale Università di Padova (5191/AO/21). This study has been approved by the appropriate ethics committee and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. Written informed consent was obtained from each patient prior to participation in this prospective study, as well as consent to eventual publication of the results.
Variables and data sources
a. Preoperative assessment. Each patient was initially evaluated clinically and underwent endoscopy, barium esophagography, and high-resolution manometry (HRM) prior to consideration of LHM. Preoperative demographic data, body mass index (BMI, kg/m2), Eckardt symptom score, and clinical presentation were collected prospectively in a dedicated database. Details of HRM findings, endoscopic and radiological features were also collected and recorded.
b. Radiologic and endoscopic examination. As part of the preoperative assessment, all study patients underwent a barium swallow in the upright position. The maximum esophageal diameter was measured at the barium–air interface in the standard anteroposterior image. Patients were classified according to their maximum esophageal diameter and the shape of the esophagogastric passage as follows: stage I (4 cm or less), stage II (4–6 cm), stage III (6 cm or more), and stage IV (6 cm or more and/or a sigmoid-shaped esophagus). 23 Endoscopy was performed in each patient to assess for macroscopic lesions related to EoE using the EoE Endoscopic Reference Score (Edema, Rings, Exudate, Furrows, Stricture, EREFS score), 24 to assess for food stasis, esophagitis, and candidiasis, and to rule out structural processes, including malignant disease.
c. High-resolution manometry. All patients underwent HRM as part of dysphagia evaluation, performed according to the Italian National Guidelines, 25 and interpreted using Chicago Classification v. 4.0. 26 Acquisition and interpretation of HRM studies were performed using ManoScan™ Acquisition Software and ManoView™ Analysis Software (Medtronic, MN, USA). The diagnosis of achalasia required abnormal supine median integrated relaxation pressure (IRP) and absence of normal peristalsis. 27 Achalasia was subtyped into three types based on absence (type 1) or presence of ⩾20% pan-esophageal pressurization (type 2) or ⩾20% premature contractions (type 3).
d. Endoscopic and surgical sampling. After the induction of general anesthesia, a preoperative endoscopy was performed routinely as part of the surgical procedure. In all patients, four biopsies were taken, from the proximal, mid, and distal esophagus, and from the Z line, respectively. In order not to increase the risk of perforation during the following myotomy, these biopsies were performed on the posterior esophageal wall. Intra-operatively, after completing the myotomy, two samples of esophageal muscle were also obtained, one from the distal esophagus and one from the gastro-esophageal junction, as shown in Figure 1(a) and (b). This procedure was performed using laparoscopic scissors after placing a marking stitch (Vicryl 3/0, needle 17) at the margin of the myotomy to better orient the specimen. Thorough hemostasis was confirmed after the esophageal muscle samples were obtained.
e. Histological examination. The hematoxylin and eosin-stained slides of the biopsy samples taken from the study patients were jointly reviewed by two gastrointestinal pathologists (M.F. and V.A.). The pathologists evaluated each sample separately and, in case of discrepancy, together. Of note, the pathologists knew that the patients had achalasia, since the aim of the study was to find intraepithelial or muscular eosinophilic infiltrate, if any. The biopsies were specifically assessed for intraepithelial lymphocytes, neutrophils, and eosinophils, inflammatory infiltrate within the lamina propria, muscle fibrosis, muscle fiber disarray, and presence of perivascular, perineural, and intraneural inflammatory infiltrate, as well as non-specific alterations of the epithelium associated with esophagitis (i.e., basal cell hyperplasia, spongiosis, erosion). Inflammatory infiltration of the epithelium and lamina propria, muscle fibrosis, and muscle fiber disarray were graded as mild, moderate, or intense (Figure 2(a)–(e)). The hotspot count of intraepithelial eosinophils per high-power field (40× magnification) was reported. According to the international guidelines, the diagnosis of EoE requires an eosinophil count ⩾15/high-power field (eos/high-power field). 28 Mast cells were then determined by analyzing the c-kit gene product (CD117) on both the esophageal biopsies (obtained during endoscopy) and the samples from the esophageal myotomy.
f. Main outcome. The main outcome was the finding of IE in the esophageal biopsies and correlating it with clinical parameters and outcomes.
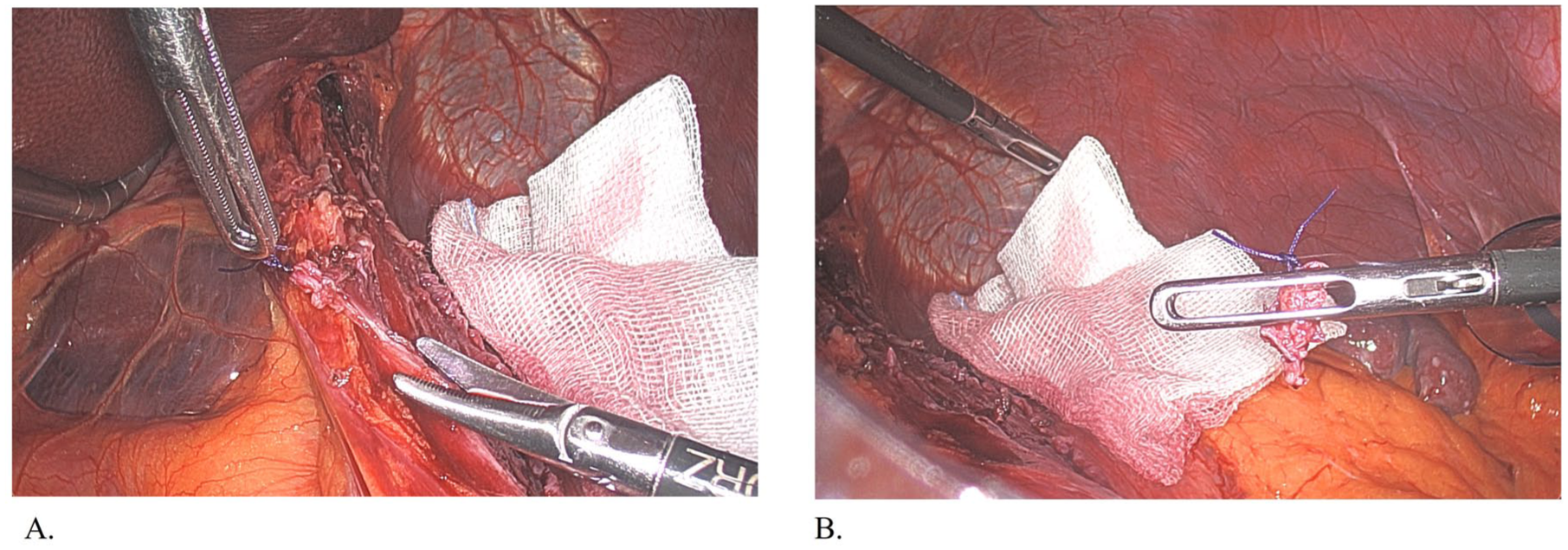
(a) After the induction of general anesthesia, an endoscopy was performed, and four biopsies of the esophageal mucosa were taken. After completing the myotomy, two full-thickness samplings of the esophageal muscle were taken from the margins of the myotomy. (b) The muscular specimen (arrow) is retrieved: a temporary stitch on the proximal margin allows a correct orientation of the specimen.

Representative images of (a) moderate intraepithelial lymphocytic infiltration, (b) perineural/intraneural eosinophilic infiltration, (c) intraepithelial esophageal infiltrate, (d) mild muscle fiber disarray, and (e) perivascular lymphocytic infiltrate.
Bias
The only possible bias was that our unit is a surgical one; thus, most of the patients were sent to our unit by their physician or gastroenterologist with the possible indication for LHD: thus, patients undergoing pneumatic dilations (PD) or other forms of treatment (POEM) were not considered.
Study size
Since achalasia is a rare disease, and allowing for an extensive period of recruitment of the patients could lead to intrinsic further biases (changes in protocols, surgical techniques, number of operators), we chose to limit the number of patients to around the patients normally operated during a 6-month period in our department for the disease.
Statistical methods
Continuous data are expressed as average and 95% confidence interval (95% CI), and categorical data as numbers and percentages. Comparisons were performed using the Mann–Whitney test, the Chi-square test, and Fisher’s exact test. A p < 0.05 was considered statistically significant. All statistical analyses were performed using the “R” statistical software. 29
Results
Participants
During the study period, 68 patients with a diagnosis of esophageal achalasia were observed. Of these, 15 patients had a previous esophageal treatment (i.e., pneumatic dilations, botulin toxin injection, POEM), 6 were younger than 18 or older than 80 years old, 6 had a radiological stage IV achalasia, and 11 refused to take part in the protocol. A total of 30 patients (14 F, 16 M) with an average age of 44 years (95% CI 38–51 years) were enrolled during the study period (Table 1). The study flow diagram of the patient enrolment is described in Figure 3.
Demographics, clinical, and instrumental characteristics of the study population prior to and after surgical intervention.
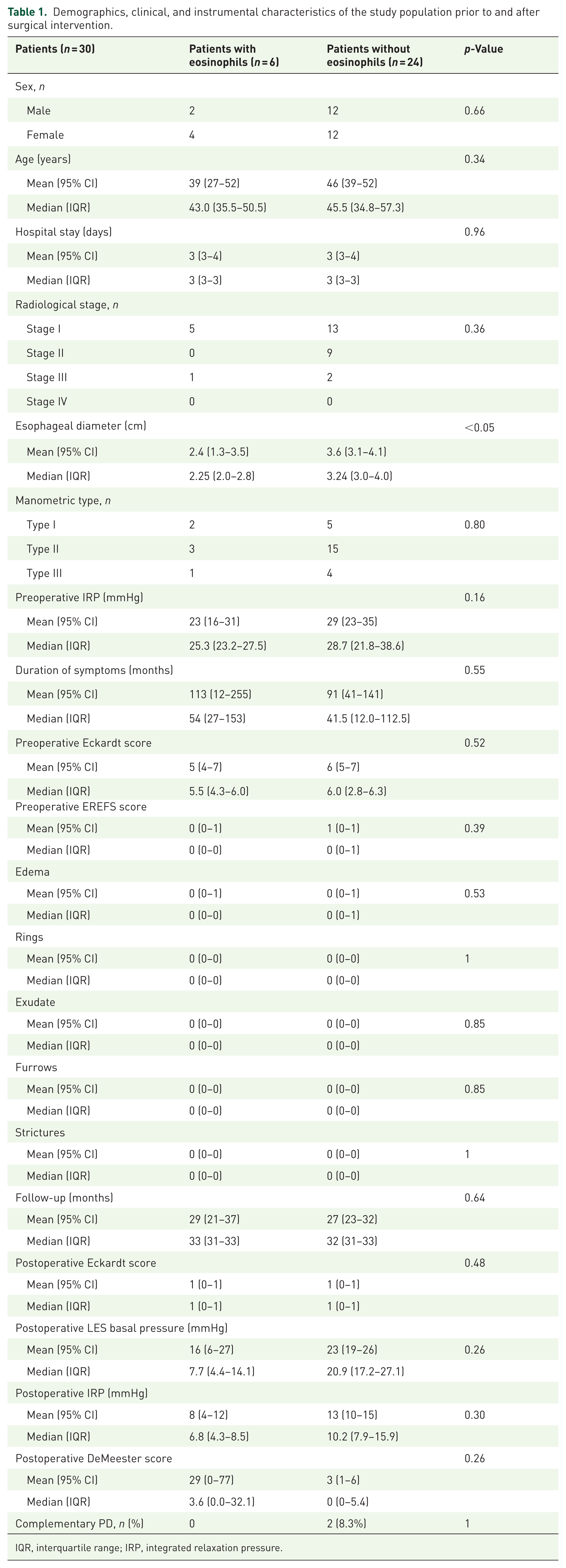
IQR, interquartile range; IRP, integrated relaxation pressure.

Flow diagram of the patient enrolment in the study.
Descriptive data
All patients complained of dysphagia, but none had episodes of food impaction requiring endoscopic bolus removal. The mean preoperative Eckardt score was 6 (95% CI 5–7) and the mean duration of symptoms was 95 months (95% CI 45–146). In addition, none of the patients were on proton pump inhibitor (PPI) or corticosteroid therapy. Only two patients had Th2-mediated comorbidities of rhinitis or asthma. Most of the patients (18, 60%) were radiologic stage one on barium radiography, 9 (30%) were stage II, and 3 (10%) were stage III. The dominant achalasia subtype was type 2 (18, 60%), while 7 (23%) had type 1 and 5 (17%) had type 3 achalasia (Table 1). No patients had missing relevant data.
Outcome data
All 30 patients underwent LHM performed by the same surgeon, and the surgery included a Dor fundoplication in all instances. There were no intraoperative or postoperative complications. The patients underwent the scheduled follow-up at our institution with outpatient office visits at 1 month and 6 months, barium swallow at 1 month, HRM and 24-h pH monitoring at 6 months, and an upper endoscopy 1 year after LHM. Since the caliber of the esophagogastric junction was noted to be narrow on barium radiography, one patient underwent a postoperative pneumatic dilation 9 months after LHM. The mean follow-up was 28 months (95% CI 23–32). All study patients had a good outcome with a mean postoperative Eckardt score of 1 (95% CI 0–1).
Main results
Of the 30 patients, seven (23%) had eosinophilia on histopathology, six within the mucosa, and one additional in the muscular layer. There was no difference in demographics, clinical presentation, manometric achalasia subtype, and mean supine IRP between the patients with and without eosinophilia (Table 1). Duration of symptoms and degree of weight loss were also similar. However, the maximum diameter of the esophagus prior to LHM was narrower in patients with eosinophilia compared to those without eosinophilia (2.4 vs 3.6 cm, respectively, p = 0.04). There were numerically more patients with no esophageal dilation among patients with eosinophilia (86% vs 26%, p = 0.1), and only one patient with eosinophilia had a dilated esophagus. There were no differences in postoperative lower esophageal sphincter (LES) metrics, reflux burden, and symptomatic outcome between patients with and without eosinophilia (Table 1). None of the patients had candidiasis at the preoperative endoscopy. The EREFS score was low in all patients, and no differences were found between patients with IE and patients without.
The histopathological findings in biopsy samples of Z-line, distal, mid, and proximal esophageal mucosa are summarized in Table 2. The k agreement was 0.9 between the two pathologists. Biopsies from at least two sites were available in all 30 patients, and from all four esophageal sites in 27 patients. Intraepithelial eosinophils were found in six patients (20%), ranging from 1 to 10 eos/high-power field in at least one of the four esophageal biopsy samples. In one case, intraepithelial eosinophils were found within three esophageal segments, in two cases, they were found within two esophageal segments, and in three cases, they were found within one esophageal segment. Only one of these patients had 20 eos/high-power field. Another patient had intramuscular eosinophilia. There were no differences in inflammatory changes, intraepithelial granulocytes and lymphocytes, interstitial muscle fibrosis, muscle fiber disarray, and perineural inflammation between patients with and without eosinophilia (Table 3). Overall, 4/22 (18%) patients had perineural inflammation in the proximal and/or distal muscle, but only one of these had a perineural and intraneural eosinophilic infiltrate. This patient had no intraepithelial eosinophilia on the endoscopic biopsies. Mast cells were found in esophageal biopsies of 21/24 patients without eosinophilia, and in 5/6 patients with eosinophilia (p = ns), although the thickness of individual biopsy samples was not homogeneous, and this could have impacted the results. There was a higher number of mast cells in the proximal esophagus in patients without eosinophilia (Table 4). Finally, no features of candidiasis were detected on any of the endoscopic examinations or biopsies.
Summary of histopathologic findings in biopsy samples of Z-line, distal, mid, and cervical esophagus prior to surgical intervention.

Data not available in atwo and bone cases.
Summary of histopathologic findings in biopsy samples of distal and proximal muscle prior to surgical intervention.
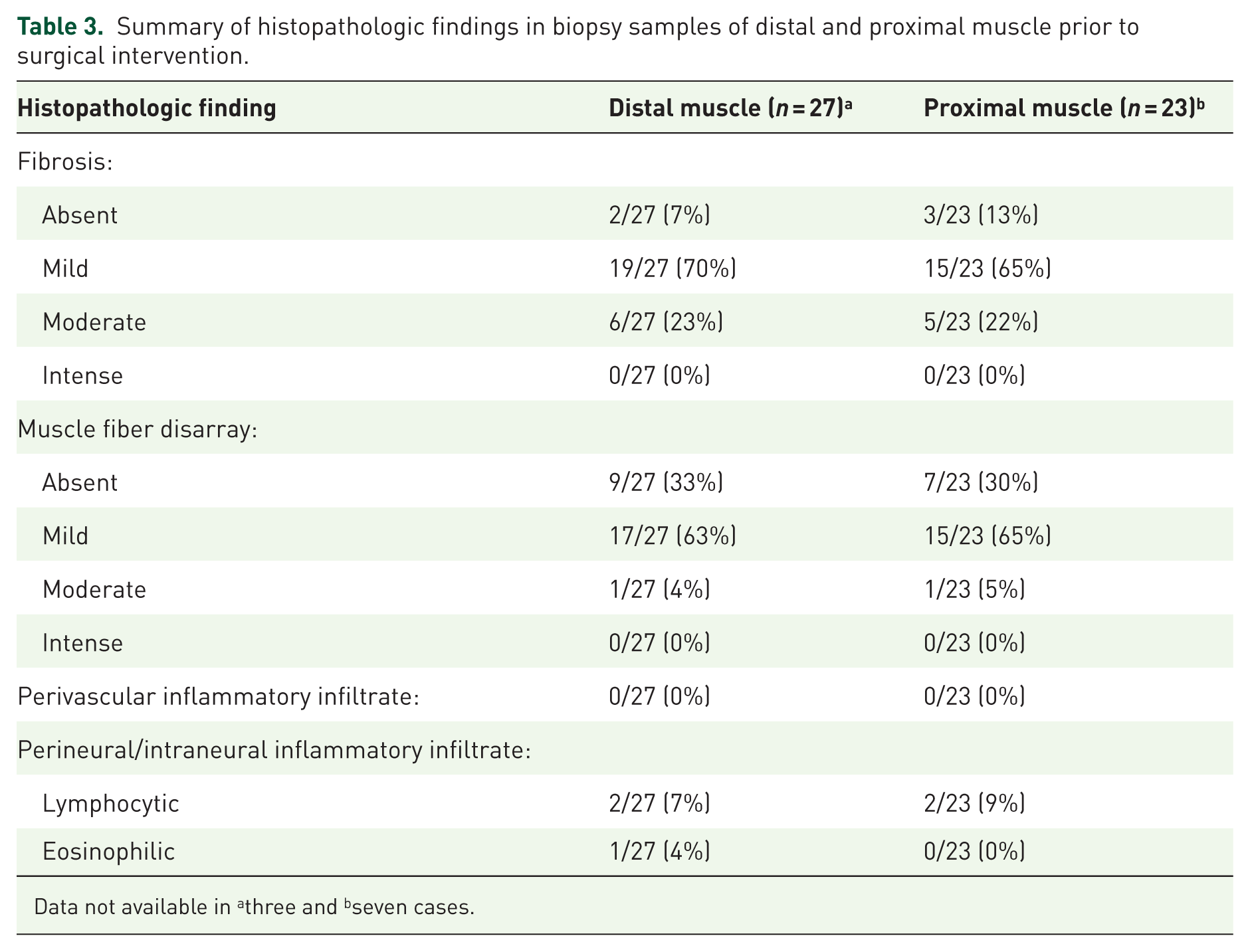
Data not available in athree and bseven cases.
Summary of mast cell findings in biopsy samples prior to surgical intervention.

IQR, interquartile range.
Other analyses
No further analyses were obtained from the study.
Discussion
Achalasia and eosinophilic esophagitis are two different pathological entities, both requiring disease-focused treatment. Even so, both disorders share almost identical clinical features, especially dysphagia and abnormal bolus transit, making it necessary to thoroughly evaluate any patient presenting with these symptoms. Consequently, understanding pathophysiology and temporal progression of these disorders is clinically relevant, and could potentially lead to further research and impact on the clinical approach to each of these disorders.
Recently, interest in the association between esophageal motility disorders and EoE has escalated, but available literature remains limited to retrospective studies and a few prospective data. 13 In most of these reports, some abnormality in esophageal motor function has been reported in 25%–75% of EoE patients. 6 More recently, a prospective study of 109 consecutive EoE patients who underwent routine HRM as part of dysphagia evaluation reported achalasia in 7.3% and obstructive motility disorders in 14.7%, despite almost identical clinical, endoscopic, and histological features as EoE-only patients. 7 In addition, eosinophilic infiltrate has been retrospectively identified in both esophageal muscle and mucosal layers in surgically treated achalasia patients, sometimes meeting and exceeding EoE diagnostic thresholds.15,16,19,28–30 In the large series from Cools-Lartigue et al., 28 of 50 patients with a confirmed diagnosis of achalasia demonstrated esophageal eosinophilia, four of which (8%) met the pathologic diagnostic criteria of EoE. 30 Our data, although limited by the sample size, suggest that eosinophilia meeting EoE thresholds is not as common as previously suggested in achalasia. Only one patient had the eosinophil count higher than 15/high-power field, and in a single biopsy specimen taken from the Z-line. Although this finding could be compatible with EoE, it is impossible to rule out other causes completely, especially esophageal stasis.
Despite the low prevalence of eosinophilia in achalasia in our study, a review of literature from Schizas et al. concludes a possible association between EoE and achalasia, but it is unclear as to which disorder is primary, and which is a consequence. 31 There are data supporting an atopic or allergic phenotype of achalasia, including a case–control study by King et al. that demonstrated a 40% increase in risk of atopy in patients with achalasia. 32 More recently, Reddy et al. 18 evaluated a cohort of 844 patients with achalasia and reported 402 (47.6%) with ⩾ 1 allergic disorder. In particular, 55 patients with achalasia (6.5%) had EoE (1.67 EoE cases expected), for a RR of 32.9 (95% confidence interval, 24.8–42.8; p < 0.001). Furthermore, in 208 patients with achalasia aged ⩽40 years, the RR for EoE was 69.6 (95% confidence interval, 46.6–100.0; p < 0.001). Thus, the authors concluded that achalasia was strongly associated with EoE and other allergic disorders, supporting the hypothesis that achalasia sometimes might have an allergic etiology. 18 In our study, however, only two patients (6.7%) had a previous history of Th2-mediated associated comorbidities (one had asthma and one had rhinitis), but neither had eosinophilic infiltration in the muscular or mucosal biopsies. Consequently, our findings are discordant from these reports, potentially related to the small sample size. Other possible factors may explain these results. One of these could be the different epidemiology: in fact, according to a work from Dellon et al., 33 incidence rates for EoE range from a low of 2.1/100,000/year in the Netherlands to a high of 12.8/100,000/year in Ohio in the United States. Another possible confounder could be related to ethnicity and geographical location. Moreover, the different dietary patterns, very different between American and European subjects, may represent another explanation.
Three potential pathophysiological relationships have been suggested between EoE and achalasia 12 : the stasis of retained food material triggering mucosal inflammation, eosinophil-mediated destruction of esophageal intramural neurons in Auerbach’s plexus, and eosinophilic products causing achalasia-like motility abnormalities. This last hypothesis leads to a plausible mechanism for the resolution of symptoms and the motility disturbance after treatment targeting eosinophilic inflammation. However, to date, data remain inconsistent regarding a finite relationship between achalasia and EoE and the underlying pathophysiological mechanism if a true relationship does exist.
Histologic studies of the esophageal muscular layer have been limited to date by a lack of access to the deeper layers underneath the mucosa and submucosa with routine endoscopic biopsies, and the high risk of perforation while performing a muscle biopsy during surgery. Chen et al. were the first to obtain and report on simultaneous biopsies of the esophageal mucosa and muscle during POEM in 20 patients with idiopathic achalasia. 34 Within a relatively small number of subjects, histologic examination did not reveal any eosinophilic infiltration in the esophageal muscle layer, so an association of esophageal eosinophilic infiltration and achalasia was not observed. To our knowledge, our study is the first with muscular biopsies surgically obtained from the edges of myotomy, thus allowing full-thickness muscular samples for improved analysis. In our series, however, only one patient showed eosinophilic inflammation in the muscle samples, thus confirming the findings by Chen and coworkers that esophageal eosinophilia is rare in achalasia.
To sum up, in this prospective study, we report that the incidence of intraepithelial and intramucosal eosinophilia is low but not negligible in consecutive achalasia patients undergoing LHM. Our study showed that patients with eosinophilia within achalasia have less esophageal dilation despite similar presentation, similar achalasia phenotypes, and similar histopathologic features, probably suggesting that the presence of eosinophilia may identify an earlier stage in the pathogenesis of achalasia. Of course, further studies with a wider population are necessary to confirm and strengthen this finding.
Our paper has, of course, some limitations. There was no EoE or non-EoE control group with similar surgical biopsies, which would be impossible to obtain. We do not have endoscopic and histological postoperative follow-up data on all the patients. Moreover, there are data suggesting that other inflammatory cells, like mast cells, may play a major role, not only in motility disorders but also in EoE development.35,36 We demonstrate a higher proximal esophageal mast cell presence in patients without eosinophilia, but the depth of the samples was not homogeneous among the different levels in the same patient and among the different patients, which may have contributed to bias in determining the role of mast cells in achalasia pathogenesis. Surely, the sample size represents an additional, important limit to our work by limiting its statistical power and possibly resulting in a Type II error. However, this work could represent a first step for future considerations and further prospective multicenter studies, with larger populations, could replicate and strengthen our findings. Nevertheless, our single-center prospective study of consecutive patients undergoing LHM adds to the understanding of the relationship between esophageal eosinophilia and achalasia. EoE is uncommon in achalasia, accounting for only 3% of this cohort, with only one patient manifesting EoE level eosinophilia. However, mild esophageal eosinophilia may be more frequent (20% in our study, i.e., six patients), and may be associated with lesser degrees of esophageal dilation, perhaps suggesting an earlier stage in the pathophysiology of the disorder.
Conclusion
In conclusion, EoE and achalasia are considered two of the most common causes of dysphagia, and an association between these disorders has recently been hypothesized. This is the first study to combine mucosal evaluation and full-thickness muscular layer histological evaluation to assess the presence of intraepithelial eosinophils in achalasia patients. This study confirmed that intraepithelial and intramuscular eosinophils are present in some patients with achalasia, but seldom meet the criteria for EoE. Only a narrower esophagus was found in patients with IE as compared to those without, possibly reflecting an earlier stage of the disease. Further studies with more patients are necessary to better elucidate the role of inflammation, particularly the role of eosinophils and mast cells in achalasia pathophysiology.
Supplemental Material
sj-doc-1-tag-10.1177_17562848251403014 – Supplemental material for Presence of eosinophils may represent an earlier stage in achalasia pathogenesis
Supplemental material, sj-doc-1-tag-10.1177_17562848251403014 for Presence of eosinophils may represent an earlier stage in achalasia pathogenesis by Andrea Costantini, Chandra Prakash Gyawali, Francesca Forattini, Valentina Angerilli, Matteo Ghisa, Luca Provenzano, Giovanni Capovilla, Loredana Nicoletti, Michele Valmasoni, Matteo Fassan, Edoardo Vincenzo Savarino and Renato Salvador in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
Artificial intelligence (AI)
AI was not used in generating text, imaging, and translation for this manuscript.
Guarantor of the article
Edoardo Vincenzo Savarino, Renato Salvador, and Matteo Fassan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
