Abstract
Background:
Mirikizumab is a first-in-class IL-23p19 inhibitor, which is approved for the treatment of adults with moderate to severe ulcerative colitis (UC).
Objectives:
We aimed to assess the effectiveness and safety of mirikizumab in moderate to severe UC in a real-world cohort from two inflammatory bowel disease referral centers.
Design:
This was a two-center international retrospective observational cohort study.
Methods:
This study aimed to assess the effectiveness and safety of mirikizumab for 12 weeks of induction. Clinical response and remission were defined as a reduction in the Simple Clinical Colitis Activity Index (SCCAI) of ⩾3 points, and SCCAI ⩽ 2, respectively. The primary outcome was the clinical remission rates after 12 weeks of induction.
Results:
We included 74 adult patients (58.1% female, 56.8% pan-colitis extent). Most patients (69/74, 93%) were previously exposed to a biological or a small-molecule therapy, and 39 (52.7%) started mirikizumab while on corticosteroids. By the end of induction, 8.1% discontinued therapy due to either lack of efficacy or an adverse event. Overall, clinical response, clinical remission, and corticosteroid-free-remission were 70.3%, 17.6%, and 16%, respectively. Week-12 clinical response and clinical remission rates were comparable between exposed and naïve patients for anti-tumor necrosis factor agents, ustekinumab, vedolizumab, and Janus-kinase inhibitors.
Conclusion:
Mirikizumab was effective for inducing clinical response and well-tolerated in a substantial cohort of treatment-experienced patients with UC. This positions mirikizumab as a valuable option to the expanding therapeutic armamentarium for UC.
Plain language summary
1. Why was the study done?
Many people with UC do not get full relief from existing treatments. We wanted to see if mirikizumab helps patients in routine clinical care, especially those who already tried other advanced therapies.
2. What did the researchers do?
In this study they reviewed medical records from two specialist IBD centers from Israel and the Czech Republic. Seventy-four patients who started a 12-week mirikizumab induction were included. They measured symptom improvement and whether patients reached remission of their symptoms. They recorded steroid use and reasons for stopping therapy.
3. What did the researchers find?
Most patients (93%) were previously treated with biologic or small-molecule drugs. After 12 weeks of mirikizumab induction, 70% had a clear improvement in symptoms. Of those, 18% reached symptomatic remission, 88% of those were in remission without corticosteroids. In addition, only 8% of all patients stopped treatment because of side effects or lack of benefit. Results were similar whether patients had previous experience treated with anti-TNF agents or JAK inhibitors.
4. What do the findings mean?
Mirikizumab may help people with difficult-to-treat UC, including those who failed earlier therapies. The induction treatment was generally well tolerated and safe in this real-world group. These results support mirikizumab as an additional option for patients with UC, but further studies will help confirm long-term benefits and safety.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory bowel disease (IBD), which is characterized by mucosal inflammation of the colon with a relapsing-remitting pattern that may lead to complications and significantly impact patients’ quality of life.1–3 Despite therapeutic advances, up to one-third of patients are primary non-responders and 50% are secondary non-responders to any biological agent or small molecule.4–7 Recent advancements in understanding the inflammatory bowel immunopathogenesis have led to the development of novel targeted therapies. 8
Interleukin-23 (IL-23) plays a critical role in sustaining inflammation in the intestinal mucosa by promoting pathogenic T-helper 17 (Th17) responses. 9 This cytokine has emerged as a promising therapeutic target, with research suggesting that selective inhibition of the p19 subunit of IL-23 might offer improved efficacy over dual IL-12/23 inhibition, 10 such as mediated by ustekinumab. In addition, focusing solely on IL-23 can potentially reduce safety concerns associated with IL-12 pathway blockade.11,12
Mirikizumab is an IgG4-variant monoclonal antibody that selectively binds to the p19 subunit of IL-23 without inhibiting IL-12.13,14 In October 2023, the FDA approved mirikizumab as the first-in-class IL-23p19 inhibitor for the treatment of adults with moderate to severe UC. 15 This approval was based on the LUCENT phase III randomised controlled trials, which evaluated 12-week induction therapy followed by 40-week maintenance therapy. 16 These pivotal trials demonstrated significant efficacy of mirikizumab compared to placebo, with 63% of patients achieving clinical response and 24% achieving clinical remission after 12 weeks of induction therapy. 17
While clinical trials provide essential efficacy and safety data, their patient populations may not reflect real-world heterogeneous patient populations, especially patients with a history of non-response to multiple lines of advanced treatment. Thus, real-world studies are more reflective of how therapies perform in typical clinical practice. However, the literature exploring real-world experience with mirikizumab is scarce. In this study, we aimed to evaluate the real-world effectiveness and safety of mirikizumab among patients with UC in a cohort from two tertiary IBD centers.
Methods
Study design
A retrospective, observational two-center study conducted in two tertiary IBD referral centers in Israel and in the Czech Republic.
Population
Adult patients (>18 years old) starting mirikizumab treatment for UC before 1st of May, 2025, were included. Patients with Crohn’s disease, IBD—unclassified, or those who had undergone total colectomy were excluded from the study.
Participants were initiated on a standard dose of mirikizumab (300 mg intravenous infusions at weeks 0, 4, and 8), followed by standard maintenance dosing of 200 mg subcutaneously every 4 weeks.
Data collection
Data on patients’ demographics, including age of diagnosis, age of mirikizumab initiation, gender, BMI, smoking status, extraintestinal manifestations, disease extent according to Montreal classification, previous and concomitant therapies, prior use of biologics or small molecules, and corticosteroid use at each time point, were captured. Clinical disease activity was assessed using the Simple Clinical Colitis Activity Index (SCCAI), which was captured routinely in every clinic visit in the two centers. Relevant clinical data, including C-reactive protein (CRP) and fecal calprotectin (FC), were collected. Endoscopic data, including the Mayo endoscopic subscore (MES), were collected from baseline and maintenance visits. Details on mirikizumab use, including start and stop dates, were recorded. Patients’ data were retrieved up until the last standard mirikizumab induction administration or the last follow-up visit, the latest of these two.
The reporting of this study conforms to the Harmonized protocol for real-world evidence reporting (HARPER) guideline (Supplemental Figure 1). 18
Definitions of response and remission
Clinical remission and response were determined by SCCAI at baseline and after 12 weeks of induction. Clinical response was defined as a reduction of SCCAI ⩾ 3, and clinical symptomatic remission was defined as a SCCAI ⩽ 2. Corticosteroid (CS)-free remission was defined as remission without CS use at that time point, irrespective of initial CS status. Endoscopic response was defined as a MES of 0–1. CRP remission and FC remission were defined as a CRP level of <5.0 mg/L and a FC level of <150 μg/mL, respectively.
Outcomes
Primary outcome: Clinical symptomatic remission rates after 12-week induction.
Secondary outcomes: Clinical response and CS-free remission rates after 12-week induction, CRP and FC remission, endoscopic response, and AEs during induction therapy.
Adverse events
AEs were extracted from patients’ clinical charts as reported by the treating gastroenterologists. All hospitalizations and surgeries were recorded.
Subgroup analysis
We performed several subgroup analyses to investigate clinical outcomes among patients with prior exposure to specific drug classes, specifically anti-TNFs, ustekinumab, JAK inhibitors, and vedolizumab. In addition, we compared outcomes between patients receiving mirikizumab as an early-line therapy (first or second line) and late-line therapy (third line and above).
Statistical methods
Descriptive statistics for demographic and clinical characteristics include median and interquartile range (IQR) for continuous variables and frequency distributions for categorical data. Non-parametric testing was performed for before-and-after variables using the Wilcoxon matched-pairs signed rank test. The Fisher’s exact test and the Mann–Whitney U test were used for comparisons between groups of categorical and continuous variables, respectively. To evaluate whether baseline disease severity influenced outcomes, we analyzed SCCAI, CRP, and FC with 12-week clinical outcomes. In addition, we included a multivariable logistic regression analysis with pre-specified covariates to account for potential confounding. Pre-specified covariates were selected based on clinical relevance, including prior advanced therapy exposure, corticosteroid use at mirikizumab initiation, baseline disease severity variables (SCCAI, CRP), age, sex, and disease extent per Montreal classification. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated. Model fit was assessed using the Hosmer–Lemeshow test. A two-sided p-value of <0.05 was considered statistically significant. Statistical analyses were performed using SPSS software (IBM SPSS Statistics for Windows, Version 26; IBM Corp., Armonk, NY, USA, 2019).
Results
Patient cohort
We collected 84 patients who initiated mirikizumab treatment. Ten patients were excluded from this study—five after total colectomy, three patients with IBD—unclassified, and two with insufficient data (Figure 1). Thus, 74 patients, who received mirikizumab for moderate to severe UC, were included in the study (38 in Israel, 36 in the Czech Republic). Patients’ characteristics are depicted in Table 1. Overall, 58.1% (43/74) of patients were female, and the median age of UC diagnosis was 26 years (IQR 21–38). Median disease duration until mirikizumab initiation was 6.5 years (IQR 3–15.25). Disease extent, according to the Montreal classification, was limited to the rectum (E1) in 2.7% (2/74), left-sided (E2) in 40.5% (30/74), and extensive or pancolitis (E3) in 56.8% (42/74). The median age of mirikizumab initiation was 38 years (IQR 27.6–49.9). Extra-intestinal manifestations were present among 33.8% (25/74) of patients. A total of 28 (37.8%) patients had previously received an immunomodulator, and 80% (59/74) were previously treated with a 5-ASA drug. Ninety-three percent (69/74) of patients were previously exposed to a biological or a small-molecule therapy. Baseline endoscopic data were available for 85% (63/74) of patients. Mayo endoscopic score (MES) 0 was demonstrated in 1/63 (1.6%), MES 1 in 5/63 (7.9%), MES 2 in 22/63 (35%), and MES 3 in 35/63 (55.6%).
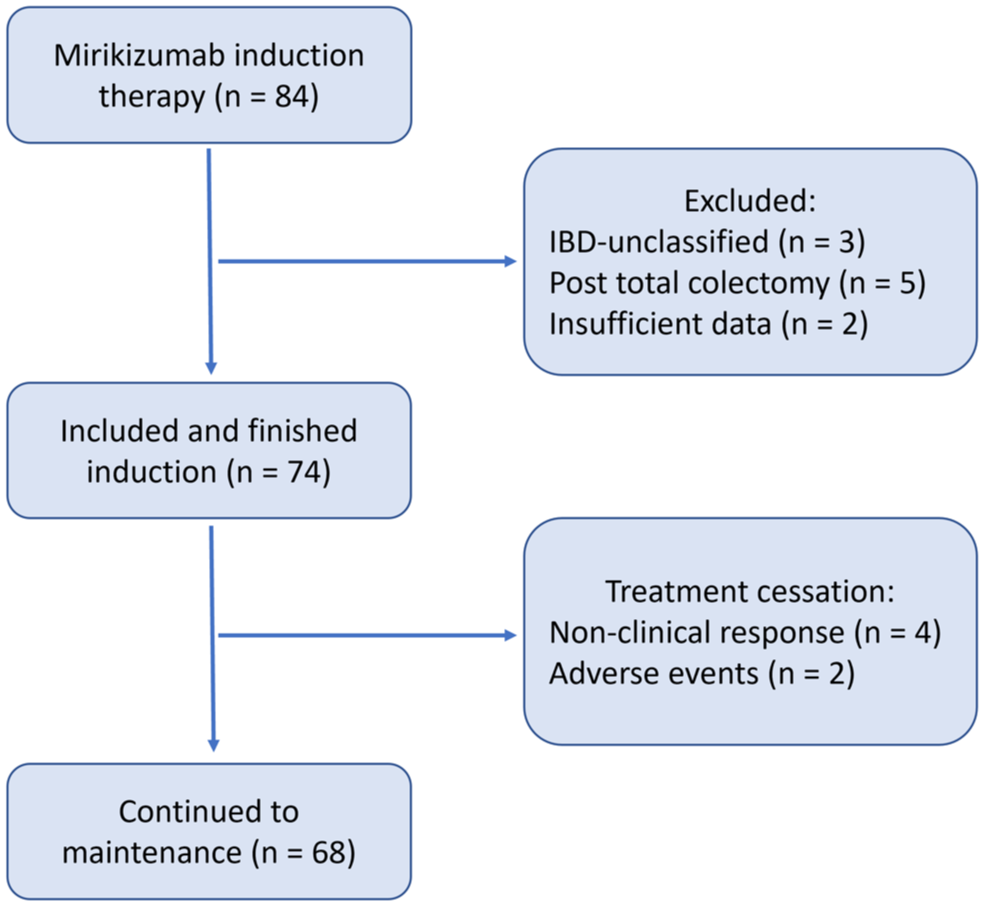
Study layout.
Baseline characteristics of the study population.
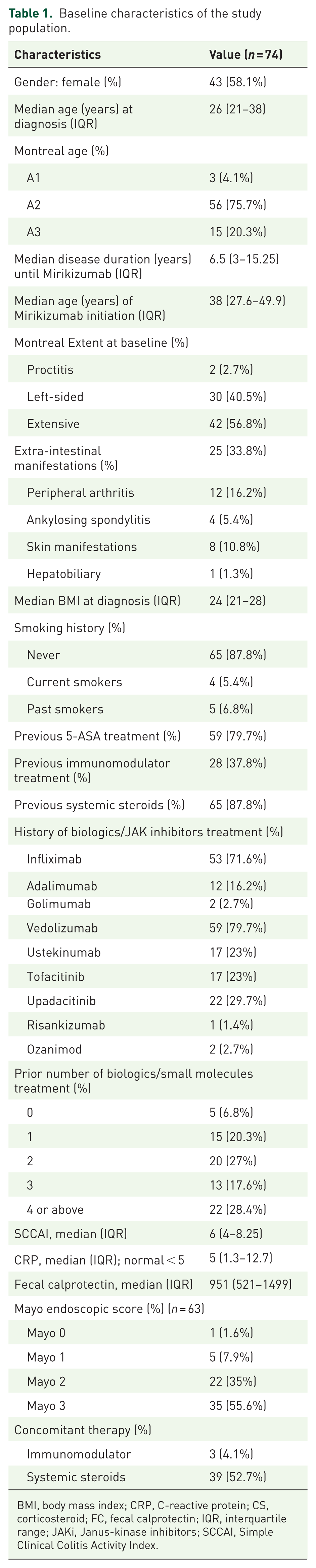
BMI, body mass index; CRP, C-reactive protein; CS, corticosteroid; FC, fecal calprotectin; IQR, interquartile range; JAKi, Janus-kinase inhibitors; SCCAI, Simple Clinical Colitis Activity Index.
Clinical outcomes
All of the 74 patients who started mirikizumab therapy received the three IV doses of the drug. After completing induction, 6/74 (8.1%) of patients discontinued therapy (did not receive the first subcutaneous dose) due to either lack of efficacy (n = 4) or an AE (n = 2, Figure 1). Of the 74 patients who were included in the study, 52 (70.3%) responded, 13 (17.6%) achieved clinical symptomatic remission, and 12 (16%) patients achieved CS-free remission (Figure 2). The rate of CS-free remission after 12-week induction among the 39 patients who received CS when commencing mirikizumab was 5% (2/39).
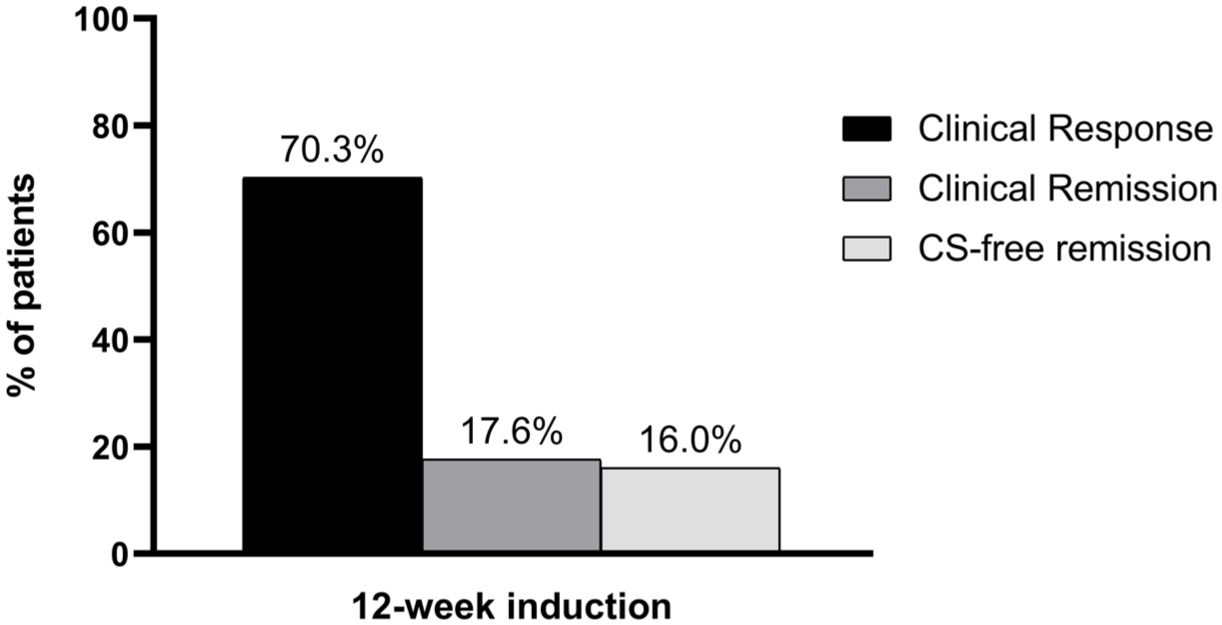
Clinical response, remission, and CS-free remission after 12 weeks in all patients.
Median serum CRP was low and unchanged from a baseline of 5 (IQR 1.3–15.7, n = 50) to 3.55 (IQR 1.2–7, n = 32) mg/dL after induction, p = 0.7. By contrast, median FC at baseline was 951 µg/g (IQR 521.7–1499.2, n = 36), and decreased to 202 µg/g (IQR 20–589, n = 34) after induction, p = 0.007. Endoscopic evaluation after induction was available in 20 patients. MES 0 was demonstrated in 1/20 (5%), MES 1 in 5/20 (25%), MES 2 in 10/20 (50%), and MES 3 in 4/20 (20%). A lower SCCAI score (<6) at mirikizumab initiation was significantly associated with higher clinical remission rates at 12 weeks compared to a higher SCCAI score (30.3% vs 9.8%, p = 0.036). In addition, a lower FC (<250) at mirikizumab initiation was only numerically associated with higher clinical remission rates at 12 weeks compared to a higher FC (60% vs 16%, p = 0.06). Baseline severity parameters (SCCAI, CRP, FC) were not associated with 12-week clinical response rates.
In the multivariable analysis, initiation age was independently associated with 12-week clinical response. Younger patients had significantly higher odds of achieving week-12 clinical remission (adjusted OR (aOR) 0.94, 95% CI 0.89–0.99, p = 0.023). None of the pre-specified covariates were independently associated with 12-week clinical remission or CS-free remission.
Prior advanced-therapy exposure on 12-week mirikizumab induction outcomes
Baseline characteristics of the advanced-therapy naïve and advanced-therapy exposed groups are depicted in the Supplemental Material (Table S1). Patients in the anti-TNF-exposed group had significantly higher rates of extensive colitis at baseline (38/56, 67.9%) than those in the anti-TNF-naïve group (4/18, 22.2%, p = 0.03). The great majority of the JAK inhibitor-exposed patients (97%) and the Ustekinumab-exposed patients (94.1%) had been likewise exposed to at least one anti-TNF drug. The patients in the JAK inhibitors-naïve group were older at the age of initiating mirikizumab compared to the exposed group (43 years, IQR 29.8–61.6 vs 31.7 years, IQR 26–45.1, respectively, p = 0.02). Prior exposure to other biologics or small molecules did not materially influence mirikizumab effectiveness. Week-12 clinical response and remission rates were comparable between exposed and naïve patients for anti-TNF agents, ustekinumab, vedolizumab, and JAK inhibitors (Tables 2 and 3 and Figure 3). In the multivariable analysis adjusted for specific prior exposure to advanced therapy, initiation age was the only covariate independently associated with 12-week clinical response (aOR 0.94, 95% CI 0.88–0.99, p = 0.03). None of the pre-specified covariates were independently associated with 12-week clinical remission or CS-free remission.
Clinical comparison at 12 weeks among anti-TNF or ustekinumab-naïve and anti-TNF or ustekinumab-exposed groups.

CI, confidence interval; CRP, C-reactive protein; CS, corticosteroid; FC, fecal calprotectin; IQR, interquartile range; JAKi, Janus-kinase inhibitors; OR, odds ratio; SCCAI, Simple Clinical Colitis Activity Index.
Clinical comparison at 12 weeks among JAK inhibitors or vedolizumab-naïve and JAK inhibitor- or vedolizumab-exposed groups.

CI, confidence interval; CRP, C-reactive protein; CS, corticosteroid; FC, fecal calprotectin; IQR, interquartile range; JAKi, Janus-kinase inhibitors; NA, not applicable; OR, odds ratio; SCCAI, Simple Clinical Colitis Activity Index.

Clinical response, remission, and CS-free remission after 12 weeks by previous treatment exposure (anti-TNF, ustekinumab, JAK inhibitors, vedolizumab).
Early-line mirikizumab versus late-line mirikizumab patients
A total of 20/74 (27%) of patients were defined as early-line mirikizumab treatment; five patients were naïve to biologic/small-molecule therapy, and 15 patients received mirikizumab as second-line therapy. The other 73% of the cohort received mirikizumab as third-line therapy (27%), fourth-line (17.6%), and above (28.4%). The comparison of clinical outcomes at 12 weeks of treatment between the groups is depicted in Table 4. Rates of clinical remission, clinical response, and biochemical markers remission (CRP, FC) did not differ between the groups. In a multivariable analysis adjusted for early versus late-line mirikizumab treatment, initiation age was the only independent covariate associated with 12-week clinical response.
Clinical comparison at 12 weeks between early-line mirikizumab and late-line mirikizumab treatment groups.

CRP, C-reactive protein; CS, corticosteroid; FC, fecal calprotectin; IQR, interquartile range; SCCAI, Simple Clinical Colitis Activity Index.
Safety
Between induction and maintenance evaluation visits, 6/74 (8.1%) of patients discontinued therapy. Four patients (5.4%) discontinued treatment due to lack of clinical response. UC exacerbated in one patient (1.4%), who needed hospitalization due to Clostridium difficile infection. No patient was referred for colectomy. Two patients (2.7%) discontinued therapy due to AEs; one patient due to elevated liver enzymes, which resolved after cessation, and another patient who suffered from myalgias. In both cases, rechallenged was not attempted. There were no cases of infection or rash in the 12-week follow-up of this study.
Discussion
In this real-world international two-center study, we provide insight into the effectiveness and safety of mirikizumab in patients with moderately to severely active UC in routine clinical practice. In this cohort of 74 refractory UC cases, mirikizumab induced a 70.3% clinical response rate; clinical remission and steroid-free remission remained modest at 17.6% and 16.0%, respectively. All of the 74 patients who started mirikizumab therapy completed induction therapy. Furthermore, AEs leading to drug cessation at the end of induction were noted in only 2.7% of patients.
Our results are comparable with a few other real-world mirikizumab studies. A recent multicenter study by Takagi et al. reported a clinical remission rate of 44.2% at week 12, which is substantially higher than our observed clinical remission rates of 16%. 19 However, this comparison must be interpreted cautiously due to different outcome definitions—Takagi et al. used partial Mayo score < 2 with rectal bleeding subscore of 0, while our study employed different clinical remission criteria. In addition, differences in outcomes may be attributed to the varying baseline disease characteristics in the Japanese study—relatively low baseline disease activity and a rather high 31% concomitant treatment rate with tacrolimus.
A study by St-Pierre et al. 20 demonstrated even higher clinical remission rates, with 83% of patients achieving clinical remission (SCCAI < 3) at week 12 and 78% achieving CS-free remission. 20 However, these superior clinical outcomes may reflect several factors, including different disease activity assessment tools, a smaller sample size (n = 20), and potential selection bias in a single-center study. Although their study population had similar rates of prior advanced therapy exposure (70% exposed to ⩾3 therapies) to our cohort, baseline clinical remission (30%) and CS-free remission (15%) rates were relatively higher compared to our cohort.
In this study, we demonstrated a modest CS-free remission rate at week 12 (16%), but closely paralleled the overall clinical remission rate, suggesting that most patients who achieved remission were successfully tapered from CS therapy. CS tapering, however, was not standardized and was undertaken at the discretion of the treating physicians. Among the 39 patients receiving corticosteroids at the start of mirikizumab therapy, 69% remained on corticosteroids after 12 weeks, while only 10% of the 39 patients were in clinical remission. These findings indicate that although CS-free remission was achieved in a proportion of patients, a substantial subset still continued corticosteroid therapy without achieving full clinical remission, highlighting the challenges of CS tapering in real-world practice.
A notable finding from our study was the effectiveness of mirikizumab in patients with prior exposure to biologics or small molecules, which could be deemed useful for positioning management decisions. In patients previously exposed to anti-TNF agents, clinical response (75.8%) and remission (15.2%) outcomes at week 12 were comparable to the anti-TNF-naïve group (65.9% and 22%, respectively). Similarly, clinical response and remission outcomes of patients previously exposed to ustekinumab (64.7% and 23.5%, respectively) were comparable to ustekinumab-naïve patients (71.9% and 17.5%, respectively). No differences were seen in the JAK inhibitors and vedolizumab analyses as well. Likewise, Takagi et al. found comparable clinical remission rates between anti-TNF-exposed and anti-TNF-naïve groups and between ustekinumab-exposed and ustekinumab-naïve groups.19,21 The consistency across treatment experience subgroups in this study corresponds with our observation of similar outcomes between treatment-experienced and treatment-naïve patients. Importantly, both studies demonstrate that mirikizumab retains effectiveness in highly refractory populations, supporting its role as a valuable option for patients with multiple prior treatment failures. Our study even reinforces this assumption through a stratified analysis of outcomes per line of mirikizumab therapy. No differences in clinical outcomes were seen between early lines of treatment (27% of patients) and later lines of mirikizumab treatment (73% of patients).
Mirikizumab was well-tolerated in our cohort. The two main AEs in our study were elevated liver enzymes and myalgias, both leading to cessation of the drug. The low discontinuation rate due to AEs (2.7%) aligns with the favorable safety profile documented in the LUCENT clinical trial program, where the incidence of serious AEs with mirikizumab was lower than with placebo.17,22,23 Nevertheless, elevations in liver enzyme levels were more frequent in patients who received mirikizumab than in placebo; these elevations resolved after the discontinuation of mirikizumab. Furthermore, our safety findings are consistent with other real-world studies, with no new safety signals identified. St‑Pierre et al. 20 reported only one serious AE (streptococcal pharyngitis) among 20 patients, with no treatment discontinuations due to AEs.. Similarly, Sawada et al. reported no AEs in their 10-patient cohort. 24 The well-accepted safety profiles across diverse patient populations and geographic regions in our study reassure mirikizumab’s favorable benefit–risk profile in real-world clinical practice.
The findings presented in our study of clinical response and remission rates are closely mirrored by those reported in the pivotal LUCENT phase III trial. 16 The results support and extend the validity of the LUCENT phase III results rather than introducing new mechanistic or prognostic findings. However, this current study provides some complementary insights. We included a highly treatment-refractory cohort consisting of 93% previously exposed to advanced therapy, a much higher rate than the 40% experienced cohort in the LUCENT program. In addition, our analysis was conducted in a real-world setting from two major referral centers, which differ fundamentally from the controlled setting of a clinical trial. Moreover, we included patients with broader baseline characteristics and analyzed specific subgroups of exposed versus naïve patients and early versus late-phase mirikizumab therapy.
This study has several limitations that should be acknowledged. First, the retrospective design and relatively small sample size limit the robustness of our findings and the ability to perform detailed subgroup analyses. Nevertheless, we were able to analyze the effectiveness of mirikizumab among treatment-experienced patients. The follow-up in the present study was limited to 12 weeks, excluding a broader evaluation of sustainable efficacy and safety. Although we report changes of disease biomarkers from baseline to week 12, our study design did not allow for analysis of temporal trends within the induction period. Further research should include larger multi-center cohorts with longer and detailed follow-up periods to better characterize the durability of response and long-term safety profile of mirikizumab. Finally, clinical response and remission were defined exclusively by SCCAI, and endoscopic outcomes were available for only a minority of patients. Thus, the risk of overestimating true endoscopic response cannot be excluded.
Conclusion
In conclusion, this real-world two-center study demonstrates that mirikizumab is an effective treatment for inducing clinical response in moderately to severely active UC in clinical practice, including in patients with prior exposure to biologic agents or small molecules and in those with multiple advanced therapy failures. Although the modest rates of clinical remission indicate the need to develop more combination strategies to enhance deeper control of inflammation, the high rates of clinical response observed, along with a favorable safety profile, position mirikizumab as a valuable addition to the evolving landscape of UC management.
Supplemental Material
sj-docx-2-tag-10.1177_17562848251392074 – Supplemental material for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study
Supplemental material, sj-docx-2-tag-10.1177_17562848251392074 for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study by Asaf Levartovsky, Martin Lukáš, Chaya Mushka Abitbol, Katerˇina Vlková, Shomron Ben-Horin, Milan Lukáš and Uri Kopylov in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848251392074 – Supplemental material for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study
Supplemental material, sj-docx-3-tag-10.1177_17562848251392074 for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study by Asaf Levartovsky, Martin Lukáš, Chaya Mushka Abitbol, Katerˇina Vlková, Shomron Ben-Horin, Milan Lukáš and Uri Kopylov in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_17562848251392074 – Supplemental material for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study
Supplemental material, sj-tif-1-tag-10.1177_17562848251392074 for Mirikizumab in ulcerative colitis: real-world evidence from an international two-center retrospective cohort study by Asaf Levartovsky, Martin Lukáš, Chaya Mushka Abitbol, Katerˇina Vlková, Shomron Ben-Horin, Milan Lukáš and Uri Kopylov in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
