Abstract
Background:
The increasing availability of biological drugs (originators and biosimilars) in the last decade for inflammatory bowel diseases (IBD), such as Crohn’s disease (CD) and ulcerative colitis (UC), has led to complex switching patterns in real-world settings.
Objectives::
To describe the switching/swapping patterns of biological drugs in IBD patients in Italy over the last decade.
Design:
A retrospective cohort study was conducted using administrative data from 14 Italian regions (2010–2023) in the VALORE distributed database network.
Methods:
Patients with at least 1 year of look-back and follow-up, who initiated biological therapy with ⩾2 dispensations for IBD, were included. Switches, swaps (between biologic classes), multiple switches (⩾2), switch-backs, and re-transitioning (biosimilar to originator) were described. Predictors of multiple switches at 3 years were identified through COX regression analysis.
Results:
Among 28,073 first-ever users (55.8% Crohn’s disease and 44.2% ulcerative colitis), most started with adalimumab (45.3%) or infliximab (39.6%). The F/M ratio was 0.79, with a median age of 41.0 years (IQR: 27.0–54.0). At 1, 3, and 5 years, switch/swap rates were 12.0%, 35.6%, and 52.6%, respectively, while multiple switches occurred in 18.7% at 5 years. Re-transitioning from biosimilar to originator occurred in 10% of patients who initially switched from originator to biosimilar of the same molecule. Tumor necrosis factor alpha (TNF-α) inhibitors switched more frequently and more rapidly than ustekinumab or vedolizumab. Depression and corticosteroid use were identified as predictors of multiple switches at 3 years of follow-up.
Conclusion:
About half of first-ever users of biological drugs who were treated because of IBDs switched or swapped within 5 years from treatment start. TNF-α drugs were more likely to switch or swap. They also swapped or switched more rapidly than vedolizumab and ustekinumab. Notably, 1 out of 5 had changed biologic therapy more than once at 5 years and, among those who switched to a biosimilar, 1 out of 10 re-transitioned to the originator.
Plain language summary
Over the past decade, the availability of different biologic treatments for inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, has increased. As more new biologic drugs and biosimilars become available, the way IBD patients switch between these treatments is becoming more complex. This study looked at real-world data from over 28,000 IBD patients across 14 Italian regions between 2010 and 2023 to understand how often patients switch treatments and what factors might influence these switches. We found that treatment switches are common: 12% of patients switched drugs within a year, 36% within three years, and 53% within five years. Many patients switched from originator drugs to biosimilars, although about 10% later on returned to the originator drug. Nearly 1 in 5 patients had two or more switches over five years. Those with a history of corticosteroid use or depression were more likely to switch multiple times, highlighting the need for more tailored care and closer follow-up of specific categories of patients.
Introduction
To date, in Europe, seven biological drugs belonging to tumor necrosis factor alpha (TNF-α) inhibitors (adalimumab, golimumab, and infliximab), anti-interleukin agents (anti-IL-12/23: ustekinumab; anti-IL-23: risankizumab, mirikizumab), or anti-integrins (vedolizumab) are licensed for treating inflammatory bowel diseases (IBDs), such as Crohn’s disease (CD) and ulcerative colitis (UC); of those, all have been authorized for both indications, except for golimumab and mirikizumab approved only for ulcerative colitis.1–3 The choice of first-line or subsequent line of biological drugs can rely on several factors, including disease characteristics, comorbidities, treatment response, tolerability, and costs. Patients with IBDs may also switch multiple times between different biological drugs to induce and maintain disease remission 4 due to loss of efficacy or onset of poorly tolerated adverse drug reactions. 5
While switching biological drugs in IBD patients is common in clinical practice (from 5% to 30% in the first year, depending on the study setting and patient characteristics),6,7 the treatment landscape has become more complex with the patent expiry of infliximab (in 2015) and adalimumab (in 2018), followed by the introduction of their biosimilars into the European market. Currently, 10 adalimumab biosimilars and 4 infliximab biosimilars have been approved for CD and UC treatment in Europe. In September 2022, the European Medicines Agency (EMA) stated that biosimilars approved in Europe are to be considered interchangeable with the respective originators. 8 Thus, in the attempt to reduce costs and to widen the number of patients eligible to be treated with the best available biological drugs, after the approval of biosimilars, drug policy interventions at the loco-regional level have been taken to promote a switch toward the less expensive molecule among interchangeable biologics originator/biosimilar. Thereafter, a large body of published evidence confirmed biosimilars’ effectiveness, safety, and interchangeability in IBD patients in a real-world setting. However, non-medical switching, that is, switching from a biologic originator to a biosimilar or vice versa, was also shown to occasionally induce a nocebo effect in patients who have not been fully engaged and informed about the comparability of originator–biosimilar benefit–risk profiles. 9 In line with that, a systematic review of real-world studies reported that re-transitioning (i.e., switch-back to the originator after a previous switch from originator to biosimilar) occurred in about 7% of IBD patients who initially switched between adalimumab or infliximab originator and respective biosimilar. 10
Considering the rapidly increasing number of newly marketed biological drugs, including biosimilars, the switching pattern of biological drugs in IBD patients might be very complex, and it may vary across geographic areas, calendar time, and active ingredients in a real-world setting. To date, scarce real-world data about switches between different biological drugs in IBD patients are available.
Using the Italian, large-scale, VALORE distributed database network, this study aimed (1) to describe the frequency of switches, multiple switches, switch-back, as well as re-transitioning over a long-term period, and (2) to explore the predictors of multiple switches in IBD patients who were newly treated with biological drugs.
Methods
Study design
This is a population-based, retrospective cohort study. The protocol was submitted to the Heads of Medicine Agencies (HMA)-EMA Catalogue (EUPAS1000000412) and, as part of the VALORE project, it was approved by the Ethical Committees of Verona and Messina Academic Hospital.
Data source
This study analyzed the claims databases from 14 Italian regions (Veneto, Lazio, Lombardy, Apulia, Friuli-Venezia-Giulia, Sardinia, Emilia-Romagna, Campania, Calabria, Tuscany, Marche, Sicily, Abruzzo, and Umbria) covering an underlying population of 51 million inhabitants (87% of the Italian population) and which participated in the VALORE project. Further details regarding the data source are described elsewhere.11,12 The following regional claims databases were considered: (1) inhabitant registry, including demographic information about the date of birth, sex, and date of registration in the regional healthcare system; (2) drug dispensing from pharmacy claims database; (3) hospital discharge records; (4) exemptions from healthcare service co-payment database; and (5) outpatient diagnostic tests and specialist’s visits database. Coverage of all these healthcare services is very high since Italy has a National Health Service offering universal care for all residents in each region. Drugs were coded using the Anatomical Therapeutic Chemical (ATC) classification system and the national drug code, while indication of use and causes of hospitalization were coded using the International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM). An R-based tool for distributed analyses developed by the Italian National Institute of Health (TheShinISS) was employed by each center to locally elaborate claims data using a common data model, sharing only a fully anonymized dataset for central analysis, in compliance with the EU General Data Protection Regulation regulations. 13
Cohort selection
From the VALORE distributed database network, subjects were included in the study based on the presence of all the following criteria: (1) at least two records of biological drug (approved for IBDs) dispensing during the study period (January 2010–June 2023); (2) at least 1 year of look-back period in the database and at least 1 year of follow-up after the index date; (3) patients treated because of CD or UC, identified through a validated META-algorithm; 14 (4) only first-ever users of biological drugs were included, that is, biological drug users with no prior dispensing of a biological drug. The index date was defined as the first dispensing of a biological drug. Each patient included in the study was followed until the last data available (e.g., last dispensing in the study period), death, or emigration from the region, whichever occurred first.
Drug exposure
The following biological drugs approved for the treatment of CD or UC were considered: TNF-α inhibitors: adalimumab (L04AB04), golimumab (L04AB06), and infliximab (L04AB02); anti-IL: ustekinumab (L04AC05); anti-integrins: vedolizumab (L04AA33). For the above-mentioned drugs, both the originator and the currently marketed biosimilars of adalimumab and infliximab were included in the analysis. Risankizumab and mirikizumab were not included in the study as these drugs were granted approval for UC treatment in Italy only after the end of the study period.
Definition of single switch, single swap, multiple switches, and switch back
According to the treatment exposure, patients were categorized as follows:
- Single switchers: patients switching from the index biological drug to only one different biological drug, which belonged to the same pharmacological class, during the follow-up.
- Single switch was further classified as a medical switch, that is, switching to an active ingredient different from the previous one, or a non-medical switch, that is, switching from the originator to a biosimilar (and vice versa) or from one biosimilar to another biosimilar of the same active ingredient.15,16
- Single swappers: patients who switched from the index biological drug only to one different biological drug that did not belong to the same pharmacological class.
- Multiple switchers: patients who switched/swapped at least two times during the follow-up to more than one biologic drug. Among multiple switchers, switch back was defined as receiving again the initial biological drug after switching to another biological drug (i.e., from drug A to drug B and then from drug B back to drug A).
Finally, re-transitioning was defined as switching back to the originator after the initial switch from the originator to a biosimilar of the same active ingredient. More details about the definition of single and multiple switches, swap, re-transitioning, and switch back are reported in Figure 1.

Definitions of switch, swap, and multiple switches.
Statistical analysis
Descriptive analyses were conducted to assess the demographic and clinical characteristics of the biological drug users included in the study. Continuous variables were reported as mean and standard deviation (SD) or median with interquartile range (IQR) as appropriate, while categorical variables were expressed as frequencies and percentages. Frequency of switch, swap, multiple switches, and switch back in CD and UC patients was assessed at 1, 3, and 5 years of follow-up. Sunburst diagrams were plotted to analyze the pattern of switch and swap by pharmacological class and active ingredient for each indication for use. A stratification by index date (2010–2016 vs 2017–2023) was also performed according to each indication for use. Time to switch and swap was explored using inverse Kaplan–Meier curves, stratified by index biological drug class and indication for use. Differences in the timing of switch and swap across groups were assessed using the log-rank test. Multivariable COX proportional hazards regression models were used to analyze predictors of multiple switches at 3 years of follow-up for each indication for use. Cox models’ results were reported as hazard ratios (HR) with 95% confidence interval (CI) and represented using forest plots. Variables included in the models were age class, sex, region, index drug, comorbidities (diabetes mellitus, hypertension, major cardiovascular events, depression, previous intestinal infections, other infections), and previous use of other immunosuppressant drugs (conventional immunosuppressants and glucocorticoids) or non-steroidal anti-inflammatory drugs. To exclude not statistically significant covariates, a stepwise selection method, based on the Akaike information criterion, was used. Frequency analyses of switch/swap/re-transitioning were conducted at 1, 3, and 5 years of follow-up (whenever applicable). All statistical analyses were performed using R software version 4.3.0 (R Foundation for Statistical Computing, Vienna, Austria).
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (see Table S1). 17
Results
From a source population of around 51.2 million Italian inhabitants, 123,621 (0.2%) users of biological drugs approved for IBD treatment were identified during the study period (January 2010–June 2023). The prevalence of use of biological drugs by active ingredient and type (originator /biosimilar) was reported in Figure S1. Among them, 28,073 (22.7%) first-ever biological users who were treated because of an IBD were finally selected (Figure 2). Demographic and clinical characteristics of the first-ever users of biological drugs included in the study cohort are reported in Table 1.

Flow chart of first-ever users of biological drugs included in the study.
Characteristics of first-ever users of biological drugs treated for IBDs by active ingredient at index date.

All look-back period.
Concomitant use evaluated in 3 months before and after the index date.
IBD, inflammatory bowel disease; IQR, interquartile range; MACE, Major adverse cardiovascular events; NSAIDs, non-steroidal anti-inflammatory drugs; SD, standard deviation.
Overall, 12,725 (45.3%), 11,119 (39.6%), 2692 (9.6%), 1212 (4.3%), and 325 (1.2%) users of adalimumab, infliximab, vedolizumab, golimumab, and ustekinumab, respectively, were included. The female/male ratio was 0.79, with a median age of 41 years old (IQR: 27.0–54.0). Users of vedolizumab had the highest median age (60 (IQR: 44, 70)). Most of the biological drug users were treated for CD (55.8%). Among new users of adalimumab and infliximab, 29.8% and 60.8%, respectively, initiated treatment with a biosimilar. Around 60% of users of biologics had previous use of conventional immunosuppressants, and 49% of corticosteroids. The median time of follow-up was 4.5 years. The longest median follow-up was reported for infliximab and adalimumab users: 5.0 years and 4.7 years, respectively, while the shortest was reported for those starting with ustekinumab (2.0 years).
Pattern of switch/swap
The frequency of single switch, single swap, multiple switches, and switch back at 1 year, 3 years, and 5 years of follow-up by active ingredient and indication is reported in Figure 3.

Frequency of switch, swap, multiple switches, and switch back in Crohn’s disease and ulcerative colitis patients.
Overall, IBD patients with ⩾1 switch/swap were 15.7%, 38.0%, and 53.1% at 1 year, 3 years, and 5 years of follow-up, respectively. UC patients reported higher frequencies of ⩾1 switch/swap (20.3% at 1 year, 41.3% at 3 years, and 54.0% at 5 years) than CD patients (12.0%, 35.6%, and 52.6%, respectively).
UC patients initiating TNF-α inhibitors had a higher frequency of experiencing ⩾1 switch/swap at 1 year than anti-IL-12/23 and anti-integrin new users (adalimumab: 21.3%; golimumab: 27.4%; infliximab: 20.3% vs ustekinumab: 8.5%; and vedolizumab: 13.6%). For patients with CD, the lowest frequency of switch/swap at 1 year was observed for ustekinumab (5.8%) compared to adalimumab (9.7%), infliximab (16.7%), and vedolizumab (10.1%). At 3 and 5 years of follow-up, the switching frequency was also lower for ustekinumab and vedolizumab users than for TNF-α inhibitors for both indications.
Overall, at 5 years, 18.7% switched at least twice (i.e., multiple switchers); among them, those who switched back to the initial biological drug were 18.8% (438/2329). In Figure 4, a sunburst chart describes the pattern of switch/swap during the entire follow-up stratified by index date (2010–2016 vs 2017–2023) and indication for use. In both UC and CD patients, the first-line use of the infliximab originator significantly declined from 2010–2016 to 2017–2023, being largely replaced by its biosimilar. Between 2017 and 2023, unlike infliximab, which was largely replaced by its biosimilar, a greater proportion of patients started adalimumab with the originator drug. However, many switched to its biosimilar during follow-up (18% and 21.2% for UC and CD, respectively).

Sunburst chart illustrating the use of biological drugs across different lines of treatment, stratified by active ingredient and type (originator vs biosimilar).
The same pattern of switch/swap is also displayed in Sankey plots (see Figure S2).
Among the first-ever users of TNF-α inhibitors with CD who switched from originator to biosimilar (same active ingredient) during the follow-up, 11.6% of adalimumab and 12.6% of infliximab users re-transitioned to the originator (Table 2). Similar results were found for adalimumab and infliximab users with UC (11.6% and 11.0%, respectively). Differences in terms of the percentage of re-transitioning were also found according to the type of biosimilar: as for adalimumab, it ranged from 7.2% to 16.9% and from 8.3% to 15.5% in patients with CD and UC, respectively. As for infliximab, it ranged from 6.9% to 19.6% and from 3.7% to 18% in patients with CD and UC, respectively. In patients with CD, the median time from switching to biosimilar to re-transitioning was 112 and 60 days for adalimumab and infliximab users, respectively. Similar results were observed for patients with UC.
Re-transitioning frequency according to the indication for use and the type of biosimilar.

Time from biosimilar switching to re-transitioning to the originator.
Number of patients starting with the originator.
Number of patients with a switch to the biosimilar of the same active ingredient during the follow-up (adalimumab to adalimumab/infliximab to infliximab) on top of those starting with originator.
IQR, interquartile range.
Finally, the median time to first switch/swap was shorter for golimumab (UC: 4.2 years) followed by infliximab (for both CD and UC: 5.0 and 5.2 years, respectively) and then adalimumab (for both CD and UC: 7.6 and 5.7 years, respectively) vs ustekinumab and vedolizumab for which median time was not reached in the Kaplan–Meier curves (see Figure S3).
Predictors of multiple switches
Predictors of multiple switches at 3 years of follow-up for each indication for use were reported in Figure 5.
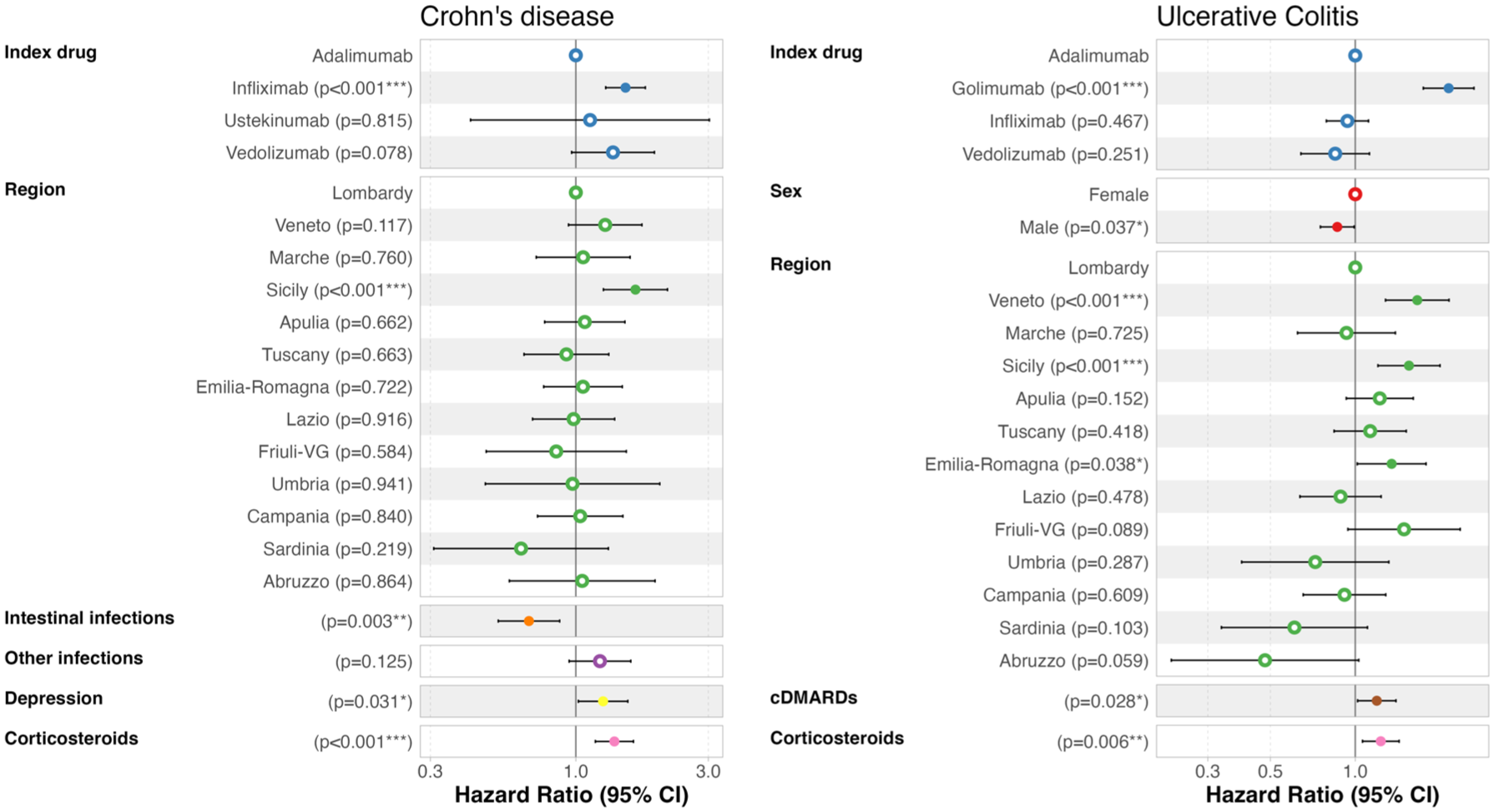
Adjusted predictors of multiple switches (at least two changes of active ingredient) at 3 years stratified by indication for use.
Among UC patients, those initiating treatment with golimumab had a higher risk of multiple switches (HR: 2.32, 95% CI: 1.88–2.86) than those initiating adalimumab. Similarly, among CD patients, those starting with infliximab had an increased risk (HR: 1.51, 95% CI: 1.28–1.78). A previous use of corticosteroids was associated with a higher risk of multiple switches for both patients with CD (HR: 1.38, 95% CI: 1.18–1.62) and UC (HR: 1.23, 95% CI: 1.06–1.43), while a previous use of conventional immunosuppressants was associated with a higher risk (HR: 1.20, 95% CI: 1.03–1.40) only for UC patients. Depression in CD patients was identified as a predictor of multiple switches (HR: 1.25, 95% CI: 1.02–1.54).
Discussion
This large-scale, population-based, Italian cohort study investigated the frequency and pattern of switch/swap among biological drugs, including originators and biosimilars, in a large cohort of IBD patients over a study period of more than 10 years.
The proportion of patients switching/swapping increased substantially over time, ranging from 15.7% at 1 year to 53.1% at 5 years from the treatment start. Switching rates at 1 year were consistent with previously published studies, 18 while no studies assessing the switching rate of biological drugs at 3 and 5 years were found in the literature. Overall, biological drug users with UC switched/swapped more rapidly and frequently than those with a CD indication. This may be attributed to the earlier response to immunosuppressant therapy of UC patients, 19 whereas a longer follow-up is required for CD patients to determine the effectiveness of the prescribed treatment.
Most of the patients included in the study started biological treatment with adalimumab (45.3%) and infliximab (39.6%). However, at the beginning of the study period, only infliximab and adalimumab were approved for IBDs, and now they are likely to be more frequently prescribed due to their lower costs, thanks to their biosimilar marketing. Biosimilars were used as a first-line treatment in 61% of infliximab initiators compared to 30% of adalimumab initiators. This discrepancy may be explained by the earlier market entry of infliximab biosimilars and their subsequent integration into clinical practice guidelines.
A striking finding is the higher medical switch/swap rates among users starting with TNF-α inhibitors (adalimumab, golimumab, and infliximab) compared to users of newer biologic classes, such as anti-IL agents (ustekinumab) or anti-integrin agents (vedolizumab). This result aligns with previous studies: for instance, a meta-analysis of a pooled cohort of 11,740 CD patients reported higher persistence rates at 1 year of follow-up of ustekinumab and vedolizumab than TNF-α inhibitors. 20 In our study, the differences in switching rates between TNF-α inhibitors and newer biologic classes persisted at 3 and 5 years. Differences between biological drug classes have been documented in clinical studies, generally reporting higher immunogenicity for TNF-α inhibitors (and especially for infliximab) than anti-IL, 21 which results in higher production of antibodies against TNF-α inhibitors, ultimately leading to loss of effectiveness or immune-related safety issues requiring switching or swapping. Overall, vedolizumab is also associated with a more favorable safety profile, particularly regarding infection risk and long-term tolerability in UC patients.22,23 According to EMA recommendations, vedolizumab and ustekinumab should be considered for IBD patients if conventional therapy or TNF-α inhibitors fail to provide adequate efficacy, lose effectiveness over time, or are not well tolerated;24,25 accordingly, the number of first-line ustekinumab users remained relatively low also probably due to its much more recent approval for CD treatment (as compared to TNF-α inhibitors) by the Italian Drug Agency in August 2018, followed by approval for UC treatment at even later stage.
Interestingly, among patients switching from adalimumab/infliximab originator to respective biosimilar, we found that about 10% of patients re-transitioned subsequently to the originator during the follow-up (most of the patients re-transitioned within 6 months after the switch), with marginal differences between adalimumab and infliximab as well as between UC and CD patients. This was also in line with previous evidence from patients with IBD and other immune-mediated inflammatory diseases10,26 and can be related to the well-described nocebo effect of the biosimilars 9 or their availability in hospitals. Notably, a recent survey conducted by the Italian Association of Patients with Chronic Inflammatory Bowel Diseases (A.M.I.C.I.) found that 57.6% of patients considered their knowledge of biosimilars to be insufficient, which may contribute to a higher rate of re-transitioning to the reference biologic. 27 To the best of our knowledge, this is the first study analyzing the re-transitioning frequency of adalimumab and infliximab in Italian IBD patients. Moreover, it is important to consider that switching behavior may also be influenced by factors such as regional tendering fosters competitions, which often determine the availability of specific reference biologics or biosimilars. As a result, some switches may be primarily driven by economic aspects or supply constraints rather than by clinical decisions. We found that approximately one in five biological drug users switched or swapped biological therapy at least twice over 5 years of follow-up. The need for multiple lines of biologic therapy suggests that a subset of patients does not achieve sustained disease control with currently available treatments or has low tolerability for these drugs. This highlights the ongoing challenges in IBD management, where the heterogeneity of treatment response can limit long-term therapeutic success, even after multiple treatment attempts.28,29 In addition, we found that prior use of corticosteroids was a predictor of multiple switches within 3 years after the beginning of biological drug treatment, which is probably as a proxy for more severe IBD. 30 Interestingly, CD patients starting with infliximab were found to have a higher risk of multiple switches at 3 years than adalimumab or golimumab initiators, which may suggest differences across molecules in maintaining clinical remission. Finally, in line with previously published evidence,31,32 depression was also associated with a higher risk of switching in CD patients.
This study has several strengths. First, we included a large cohort of patients (about 28.000 first-ever biological drug users with IBD) with a median follow-up time of 4.5 years. Second, as we included data from 14 Italian regions, we believe that the results can be representative of the whole country. Moreover, we also explored specifically switches from originator to biosimilar (i.e., non-medical switch), which is of growing interest due to the rapidly increasing number of biosimilars being introduced in the market. Finally, we explored the predictive factors of multiple switches in a large cohort of IBD patients who were treated with biological drugs, which, to the best of our knowledge, had not previously been investigated.
Nevertheless, some limitations warrant caution. First, administrative data do not provide clinically relevant information, such as the reason for switching (e.g., lack of effectiveness, onset of adverse drug reactions) or disease severity. The lack of clinical information limits the interpretation of the patterns observed, especially when evaluating potential predictors of switching. For example, previous use of corticosteroids may be a marker of higher disease activity, which could itself drive switching behavior. Without data on treatment response or clinical activity of the disease, it is difficult to disentangle whether observed associations reflect clinical decision-making or underlying patient condition. Second, we could not investigate the pattern of use of risankizumab and mirikizumab, which were marketed after the end of the study period. Finally, the long-term frequency of the switch at 3 and 5 years of follow-up could not be carefully assessed for ustekinumab due to its more recent approval as compared to TNF-α inhibitors and vedolizumab.
Conclusion
This Italian large-scale, population-based study found that the proportion of patients switching/swapping increased substantially over time, ranging from 15.7% at 1 year to 53.1% at 5 years from the treatment start. The frequency of switch/swap was higher in UC patients than in CD patients and in those initiating TNF-α inhibitors than in vedolizumab and ustekinumab users. In addition, switching from originator to biosimilar was frequent during the follow-up, despite about 10% re-transitioned subsequently to the respective originator. Finally, approximately one in five biological drug users switched or swapped biological therapy at least twice over 5 years of follow-up; prior use of corticosteroids, which is a proxy of disease severity and depression, was identified as a predictor of multiple switches, thus pointing out categories of patients requiring even more careful monitoring from clinicians.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251378080 – Supplemental material for Single and multiple switches, swap and retransitioning among 28,073 biological drug users with inflammatory bowel diseases: results from the Italian VALORE network
Supplemental material, sj-docx-1-tag-10.1177_17562848251378080 for Single and multiple switches, swap and retransitioning among 28,073 biological drug users with inflammatory bowel diseases: results from the Italian VALORE network by Giorgia Pellegrini, Andrea Spini, Chiara Bellitto, Luca L’Abbate, Ylenia Ingrasciotta, Federica Soardo, Olivia Leoni, Arianna Mazzone, Domenica Ancona, Paolo Stella, Anna Cavazzana, Angela Scapin, Sara Lopes, Valeria Belleudi, Stefano Ledda, Paolo Carta, Paola Rossi, Lucian Ejlli, Ester Sapigni, Aurora Puccini, Rita Francesca Scarpelli, Giovambattista De Sarro, Marco Tuccori, Rosa Gini, Alessandra Allotta, Sebastiano Addario Pollina, Roberto Da Cas, Giampaolo Bucaneve, Antea Maria Pia Mangano, Francesco Balducci, Carla Sorrentino, Ilenia Senesi, Francesca Futura Bernardi, Ugo Trama, Stefania Spila Alegiani, Flavia Mayer, Marco Massari, Edoardo Vincenzo Savarino, Angela Variola and Gianluca Trifirò in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
