Abstract
Background:
Iron deficiency anemia (IDA) is a common extraintestinal manifestation of inflammatory bowel disease (IBD), affecting around one-third of patients.
Objective:
To compare IBD progression and healthcare resource utilization in patients with and without a co-diagnosis of IDA in a real-world setting.
Design:
A retrospective comparative study was conducted using Italian entities’ administrative databases, covering 9.3 million health-assisted individuals.
Methods:
Adult IBD patients diagnosed with ulcerative colitis and/or Crohn’s disease were enrolled between January 2010 and September 2017. Within 12 months from IBD diagnosis, IDA was identified by at least one prescription for iron and/or IDA hospitalization and/or blood transfusion (proxy of diagnosis). IBD population was divided according to the presence/absence of IDA. Given the nonrandom patients’ allocation, propensity score matching (PSM) was applied to abate potential unbalances between the groups. Before and after PSM, IBD progression (in terms of IBD-related hospitalizations and surgeries), and healthcare resource costs were assessed.
Results:
Overall, 13,475 IBD patients were included, with an average age at diagnosis of 49.9 years, and a 53.9% percentage of male gender. Before PSM, 1753 (13%) patients were IBD–IDA, and 11,722 (87%) were IBD–non-IDA. Post-PSM, 1753 IBD–IDA patients were matched with 3506 IBD–non-IDA. Before PSM, IBD progression was significantly higher in IBD–IDA (12.8%) than in IBD–non-IDA (6.5%) (p < 0.001). After PSM, IBD progression and IBD-related hospitalizations were significantly (p < 0.001) more frequent in IBD–IDA patients (12.8% and 12.0%, respectively) compared to IBD–non-IDA (8.7% and 7.7%). Consistently, healthcare expenditures resulted significantly higher among IDA patients (p < 0.001), with an overall mean annual cost of €5317 compared to €2798 for patients without IDA. These results were confirmed after PSM matching, as the mean annual total cost/patient in IBD–IDA versus IBD–non-IDA were €3693 and €3046, respectively (p < 0.001).
Conclusion:
In a real-life setting, IDA co-diagnosis in IBD patients was associated with disease progression and higher related economic burden.
Keywords
Introduction
Inflammatory bowel diseases (IBDs) are a group of chronic inflammatory disorders characterized by repetitive episodes of inflammation of the gastrointestinal tract throughout their clinical course. 1
IBDs comprise Crohn’s disease (CD) and ulcerative colitis (UC): CD is characterized by chronic inflammation that primarily affects ileum and colon; however, it can involve any part of the gastrointestinal tract, while the inflammation caused by UC is limited to the colonic mucosa.2,3 CD and UC severely impair the quality of life of patients affected, with negative effects on psychosocial, social, and economic well-being. 4 Moreover, due to their chronic nature, CD and UC pose a heavy economic burden that could likely increase with the progression of the diseases and clinical worsening. 5
Extraintestinal manifestations are quite common in both CD and UC, affecting 25–40% of patients. 6 One of the most common manifestation is the presence of anemia: around one-third of IBD patients displays hemoglobin (Hb) levels <12 g/dL for females and <13 g/dL for males.7–9 In a large US IBD cohort, the 5-year anemia prevalence was found to be 50%. 10 Most cases of anemia in patients with IBD result from functional and/or absolute iron deficiency [i.e., iron-deficiency anemia (IDA)].8,11
However, in some rare cases, anemia can be due to B12 and folate deficiency, medication toxicity, and other causes. 11
The prevalence of anemia in IBD patients has been recently estimated of 24% [95% confidence interval (CI): 8–31] in European countries. 12 In patients with chronic conditions, the anemic state is also associated with a decreased quality of life 4 and represents an important factor to be considered in therapeutic management. 8 However, recent studies suggest that iron deficiency is underdiagnosed and undertreated in IBD in clinical practice.13,14
The consensus statement of the SPIRIT–IOIBD (Selecting End PoInts for Disease-ModIfication Trials–International Organization for the Study of Inflammatory Bowel Diseases) indicated some endpoints to define disease progression in IBD. The expert panels agreed that the optimization in the management of both CD and UC should encompass a careful monitoring of short-midterm complications, as well as long-term complications. Thus, several culprits of IBD progression have been implicated, including the detrimental rebounds on health-related quality of life (i.e., disability, fecal incontinence), IBD-related surgery and hospitalizations, extraintestinal manifestations, including anemia, permanent stoma, short bowel syndrome, gastrointestinal and extraintestinal dysplasia or cancer, and mortality. 15 Anemia in IBD has many underlying causes, above all the combination of chronic iron deficiency, anemia of chronic disease, followed by vitamin B12 and folate deficiency, and medication toxicity. 11
Moreover, the active disease appears to be a risk condition for anemia: the 2019 consensus guidelines of the British Society of Gastroenterology, based on an extensive systematic literature review of 88,247 publications, emphasized how about one-third of patients with active IBD develop IDA. 16 Hence, further studies on the association between anemia/iron deficiency and disease progression in IBD are needed to improve the management of these complex patients. It has been reported that the persistent and recurrent manifestation of anemia in IBD patients correlates with more aggressive or disabling disease, 17 and the evaluation of plasma biomarkers, such as Hb or hematocrit, has been suggested as a predictor of disease complication in CD patients. 18 In addition, comorbid anemia in IBD is largely correlated with inflammatory activity 19 representing a significant predictor of increased risk of hospitalization and even increased patient mortality.20–24
A retrospective analysis carried out among the US population affected by several chronic conditions has shown that IBD patients with IDA represent about 10% of the overall anemic population and carry an economically relevant healthcare burden. 25
The goal of this analysis, conducted in a real-world clinical setting, was to assess the impact of IDA on the progression of IBD, measured in terms of IBD-related hospitalizations and IBD-related surgery as outcome indices. The healthcare resource consumption and the related costs sustained by the Italian National Health Service (INHS) were also examined through a comparative evaluation of IBD patients with and without IDA.
Methods
Data source
Data were retrospectively collected from the administrative databases of a sample of Italian Healthcare Departments, geographically distributed across the Country and covering around 9.3 million health-assisted individuals. These databases are large repositories of information to be used for reimbursement purposes regarding all healthcare services provided by the INHS, which is funded on the principle of universal coverage of healthcare expenses for all national and legal foreign residents. For the current study, Italian Healthcare Departments database were selected by their geographical distribution, by data completeness, and by the high-quality linked datasets. Specifically, data linkage was performed among the following databases: (1) demographic database, which collects patients’ demographic as gender, age and date of death; (2) pharmaceuticals database, which collects information regarding medicinal products reimbursed by the INHS, namely the Anatomical Therapeutic Chemical (ATC) code, number of packages, number of units per package, unit cost per package, and prescription date; (3) hospitalization database, which contains all data related hospitalizations, like discharge diagnosis codes classified according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM), Diagnosis Related Group (DRG) and DRG-related charge (provided by the INHS); (4) outpatient specialist services database, which contains all information about diagnostic tests and specialist visits (date and type of service delivery, activity description and tariff for laboratory test or specialist visit); (5) exemption database, gathering all payment waiver codes that exempt patients from contribution charge for services/treatments in case of specific disease diagnoses. The reporting of this study conforms to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement. 26
To guarantee patients’ privacy, an anonymous univocal numeric code was assigned to each subject included in the study, in full compliance with the European General Data Protection Regulation (2016/679). The patient code in each database allowed electronic linkage between all different databases. The results are exclusively in aggregated form and are not attributable to a single institution, department, doctor, individual, or individual prescribing behavior. Based on the Data Privacy Guarantor Authority (General Authorization for personal data treatment for scientific research purposes—n.9/2014), informed consent was not required, as the data collected were anonymized and could not be linked to individual patient identification. According to Italian law on the conduction of observational analyses, the ethics committee of each participating Health Department was notified and approved the analysis.
Study design and study population
In the present retrospective observational study, all adult (⩾18 years old) patients with a diagnosis of UC (identified by at least one hospitalization with primary or secondary diagnosis with ICD-9-CM code 556 and/or active exemption code 009.556) and/or CD (identified by at least one hospitalization with primary or secondary diagnosis with ICD-9-CM code 555 and/or active exemption code 009.555) and/or that have received at least one prescription for vedolizumab (ATC code L04AA33) between January 1, 2010, and September 30, 2017 (inclusion period), were consecutively included. This last criterion was used to include patients with potential moderate-to-severe IBD, as vedolizumab is commonly used specifically in this setting of patients, as first or second-line therapy after conventional therapy, and in specific special situations where anti-tumor necrosis factor (anti-TNF) are hardly prescribed (i.e., elderly patients, patients with latent tuberculosis or hepatitis B virus infections, patients with previous cancer). The date of the first match with one of the inclusion criteria was defined as the IBD-diagnosis date. A 12-month period before the IBD-diagnosis date was set to characterize patients (characterization period). IDA diagnosis was assessed 12 months after IBD-diagnosis date (IDA evaluation period) by the presence of at least one prescription for iron preparations (ATC code B03A) and/or at least one hospitalization for IDA (primary or secondary diagnosis with ICD-9-CM code 280) and/or at least one blood transfusion (ICD-9-CM code 99.0, primary or secondary procedure, ICD-9-CM code V58.2, primary or secondary diagnosis, specialistic code 99.07.1). For all patients with or without IDA, the index date corresponded to 12 months after IBD-diagnosis date, and marked the beginning of follow-up, which lasted 1 year. Patients without continuous data availability during the study period (i.e., moving to another healthcare entity) and those who died before index date or during follow-up period were excluded. A schematic representation of the timetable of the analysis is provided in Figure 1.

Schematic representation of the timetable of the analysis: (1) Enrollment period: January 1, 2010, and September 30, 2017, (2) Diagnosis date: date of first hospitalization, drugs or exemption for IBD during enrollment period, (3) Index date: 12 months after diagnosis date, (4) Characterization period: 12 months before diagnosis date, (5) IDA evaluation period: 12 months-period between diagnosis date and index date, and (6) Follow-up period: 12 months following index date.
Analysis of patients’ baseline characteristics
At the index date, for IBD patients without IDA and with IDA (therein named as IBD–non-IDA and IBD–IDA, respectively), the baseline demographic and clinical characteristics, namely age at IBD diagnosis, gender distribution (expressed as percentage of males and females) and Charlson et al. 27 comorbidity index were collected. Patients’ clinical profile was evaluated during the characterization period, by type of IBD, UC, or CD, and the following comorbidities: cardiovascular diseases, chronic obstructive pulmonary disease (COPD), asthma, cancer, diabetes, hypertension, kidney disease, rheumatoid arthritis, ankylosing spondylitis, psoriasis and psoriatic arthritis, menorrhagia, metrorrhagia, previous IBD-related surgery. Each disease was identified through ICD-9-CM codes for primary or secondary hospitalization discharge diagnosis and/or active exemption codes and/or ATC codes for drug prescriptions, as detailed in Supplemental Table S1.
Analysis of IBD treatment combinations
IBD treatments evaluated during the follow-up belonged to the class of biologics and included vedolizumab, infliximab, adalimumab, golimumab, ustekinumab, small molecules (tofacitinib), immunomodulating, anti-inflammatory, anti-proliferative agents, encompassing methotrexate ciclosporin, sulfasalazine, mesalazine, azathioprine, mercaptopurine, tacrolimus, corticosteroids, beclomethasone, budesonide, and antidiarrheals (namely intestinal anti-inflammatory/anti-infective agents), cholestyramine. Other treatments related to IBD examined in the study were vitamins/mineral supplements and antibacterials for systemic use, including levofloxacin, ciprofloxacin, metronidazole, and rifaximin. The ATC codes for the abovementioned drugs are provided in Supplemental Table S2.
Propensity score matching analysis
Based on the observational analysis of existing data, as IBD patients could not be randomly assigned to a specific cohort, to reduce the presence of confounding variables potentially affecting the outcomes observed, 27 A propensity score matching (PSM) method was applied. The propensity score was estimated using a logistic regression model, considering the following confounding variables: age at IBD diagnosis, gender, Charlson comorbidity index, cardiovascular disease, COPD, asthma, diabetes, hypertension, cancer, infections, kidney disease, rheumatoid arthritis, ankylosing spondylitis, psoriasis/psoriatic arthritis, previous IBD surgery, disease severity, type of IBD diagnosis, hepatic cirrhosis, and by the number of previous IBD medications prescribed (detailed in Supplemental Tables S1 and S3). Patients were matched on quintiles of propensity score calculated. To keep the maximum number of patients, a 1:2 matching algorithm was used to match patients in each quintile in the two groups under analysis to identify two balanced cohorts of patients to compare. Standardized Mean Difference (SMD) greater than 0.1 (10%) is a threshold being recommended for declaring imbalance. 28
Analysis of IBD disease progression
Before and after PSM, in both cohorts, the following variables were considered as proxies to evaluate IBD progression during follow-up: IBD-related hospitalizations (for CD or UC, by ICD-9-CM code 556 or 555); IBD-related surgical interventions (ICD-9-CM procedural codes 45.3X, 45.4X, 45.6X, 45.7X, 45.8X, 45.9X, 46.0X, 46.1X, 46.2X, 46.3X, 46.6X, 46.7X, 48.0X, 48.1X, 48.3X, 48.4X, 48.5X, 48.6X, 48.7X, 48.8X, 48.9X, 49.0X, 49.1X, 49.3X, 49.5X, 49.6X, 49.7X). IBD progression was defined as the presence of at least one of the mentioned proxies, and data were reported as the number and percentage of patients.
Analysis of healthcare resource consumption and related direct costs covered by the INHS
Before and after PSM, in alive patients, the healthcare resource utilization during the 12-month follow-up was evaluated in terms of the mean number of drug prescriptions, the mean number of hospitalizations, and the mean number of outpatient specialist service deliveries. The direct healthcare costs were evaluated over the follow-up period and were related to the following resource consumption: hospitalizations (determined by using the DRGs tariffs), drug costs (evaluated for drugs reimbursed by the INHS, and using the INHS purchase price), and the outpatient specialist service costs accordingly to regional tariffs. Data were reported in euro (€) as the mean total healthcare cost per patient.
Statistical analysis
Continuous variables were reported as mean ± SD, categorical variables were expressed as numbers and percentages. Patients were categorized into two cohorts, the IBD–IDA and the IBD–non-IDA ones. The results were compared between the two cohorts by using the Student’s t-test (to compare continuous variables) and the chi-square test (to compare categorical variables), as appropriate. After applying PSM, in the matched cohorts, a logistic regression model adjusted for the baseline confounding variables (listed in the above section “Propensity Score Matching analysis”) was developed to identify the potential predictions of IBD progression by computing odds ratio (OR) and the corresponding 95% CIs. Statistical significance was accepted at p value < 0.05. All analyses were performed using Stata SE version 17.0 (StataCorp, College Station, TX, USA).
Results
A total of 14,349 patients with IBD diagnosis were identified; after excluding those who died in the 12 months after IBD diagnosis date or during follow-up, 13,475 patients were finally included in the analysis (Figure 2). As reported in Table 1, age at IBD-diagnosis date was 49.9 ± 18.2 years, and 53.9% of patients were male (and 46.1% of female). As for the comorbidity profile, the more frequent conditions were infections (52.4%) and hypertension (29.7%), followed by COPD (12.3%), diabetes (6.4%), cardiovascular events (4.1%), asthma (4.3%), and cancer (4.8%); previous IBD-related surgical interventions were reported for 9.0% of patients (Table 1).

Flowchart of the study population.
Demographic and clinical characteristics of IBD patients. Continuous variables were reported as mean ± SD, and categorical variables were expressed as numbers and percentages.
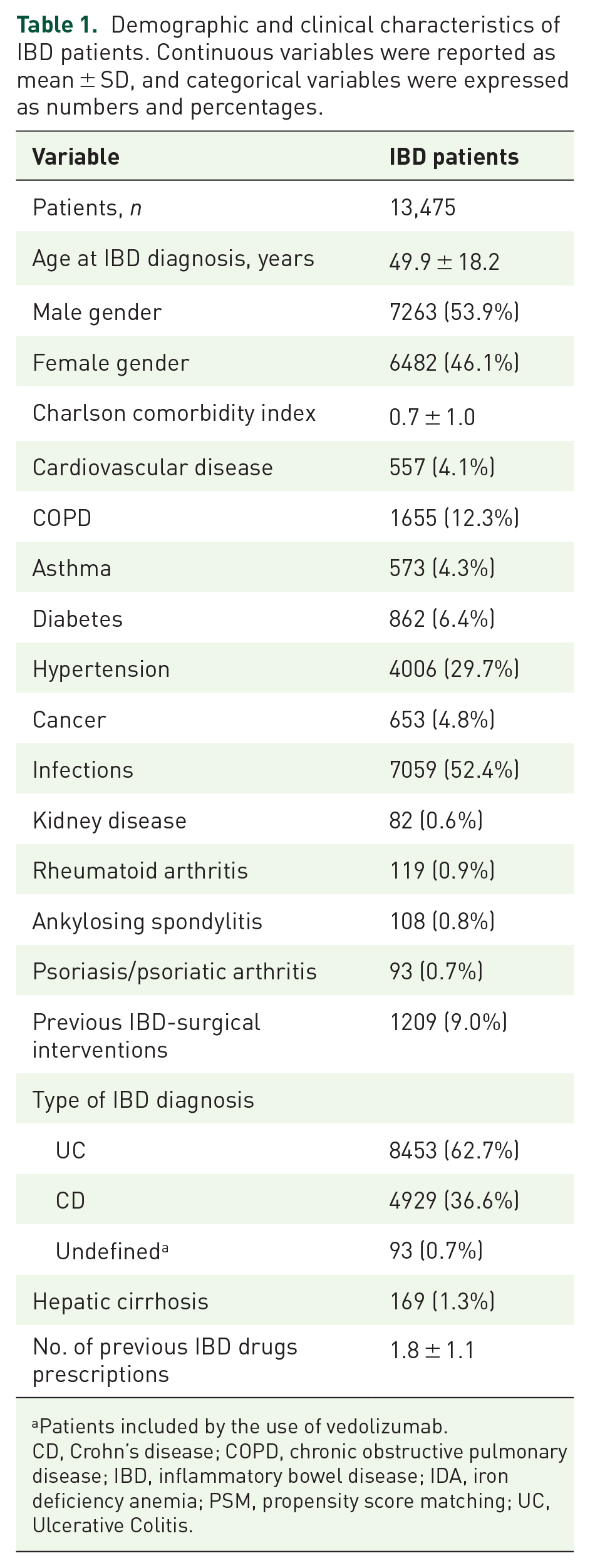
Patients included by the use of vedolizumab.
CD, Crohn’s disease; COPD, chronic obstructive pulmonary disease; IBD, inflammatory bowel disease; IDA, iron deficiency anemia; PSM, propensity score matching; UC, Ulcerative Colitis.
Before PSM, from the target patient population, 11,722 (87%) patients were allocated to the IBD–non-IDA cohort and 1753 (13%) to the IBD–IDA cohort. As shown in Table 2, IBD–non-IDA patients were younger than IBD–IDA (mean age 49 versus 56 years), and generally displayed a less severe comorbidity profile (Charlson comorbidity index: 0.6 in IBD–non-IDA and 1.0 in IBD–IDA). During the characterization period, in IBD–non-IDA group, hypertension, diabetes, COPD, cancer, and infections were less common than in the IBD–IDA group (p < 0.001). Similarly, cardiovascular events showed a lower rate in IBD–non-IDA patients than in IBD–IDA (3.4% versus 9.0%, p < 0.001), and less patients without IDA had undergone a previous IBD surgery than those with IDA (8.1% versus 14.9%, p < 0.001) (Table 2).
Demographic and clinical characteristics in IBD–non-IDA and IBD–IDA cohorts, before PSM. Continuous variables were reported as mean ± SD, and categorical variables were expressed as numbers and percentages. Significances are highlighted in bold.

Patients included by the use of vedolizumab.
CD, Crohn’s disease; COPD, chronic obstructive pulmonary disease; IBD, inflammatory bowel disease; IDA, iron deficiency anemia; PSM, propensity score matching; UC, Ulcerative Colitis
During follow-up, IBD-related treatments were prescribed to 76.9% of IBD–non-IDA and to 74.7% of IBD–IDA patients (p < 0.05) [Figure 3(a)], but the proportion of patients prescribed at least two different concomitant drugs was higher in IBD–IDA cohort (IBD–non-IDA versus IBD–IDA: 44.8% versus 52.7%, p < 0.001) [Figure 3(b)].

Presence of IBD treatment during the follow-up period (a) and treatment combination regimens (b) during the follow-up period in IBD–non-IDA and IBD–IDA patients.
In Table 3, the baseline characteristics of patients after PSM analysis have been reported. Overall, 3506 IBD–non-IDA and 1753 IBD–IDA patients were included. After matching, no unbalance was observed among the two cohorts for all baseline variables, with a slight difference for the occurrence of kidney disease (SMD = 0.113) (Table 3).
Demographic and clinical characteristics in IBD–non-IDA and IBD–IDA cohorts, after PSM. Continuous variables were reported as mean ± SD, and categorical variables were expressed as numbers and percentages. Significances are highlighted in bold.

Patients included by the use of vedolizumab.
CD, Crohn’s disease; COPD, chronic obstructive pulmonary disease; IBD, inflammatory bowel disease; IDA, iron deficiency anemia; PSM, propensity score matching; SMD, Standardized Mean Difference; UC, Ulcerative Colitis.
Table 4 reports the events analyzed to estimate IBD progression. Before PSM, IBD–IDA patients showed higher rates of IBD-related surgical interventions (IBD–non-IDA versus IBD–IDA: 1.7% versus 3.0%, p < 0.001) and hospitalizations (IBD–non-IDA versus IBD–IDA: 5.9% versus 12.0%, p < 0.001). Overall, IBD progression was significantly higher in IBD–IDA (12.8%) than in IBD–non-IDA (6.5%) (p < 0.001) (Table 4). This significant trend was also evident among the two matched balanced cohorts: IBD progression (specifically IBD-related hospitalization) accounted more frequently in IBD–IDA patients, 12.8% (and 12.0%) with respect to 8.7% (and 7.7%) of IBD–non-IDA cohort (p < 0.001) (Table 4).
IBD progression during the follow-up period, before and after PSM. Variables were expressed as numbers and percentages. Significances are highlighted in bold.
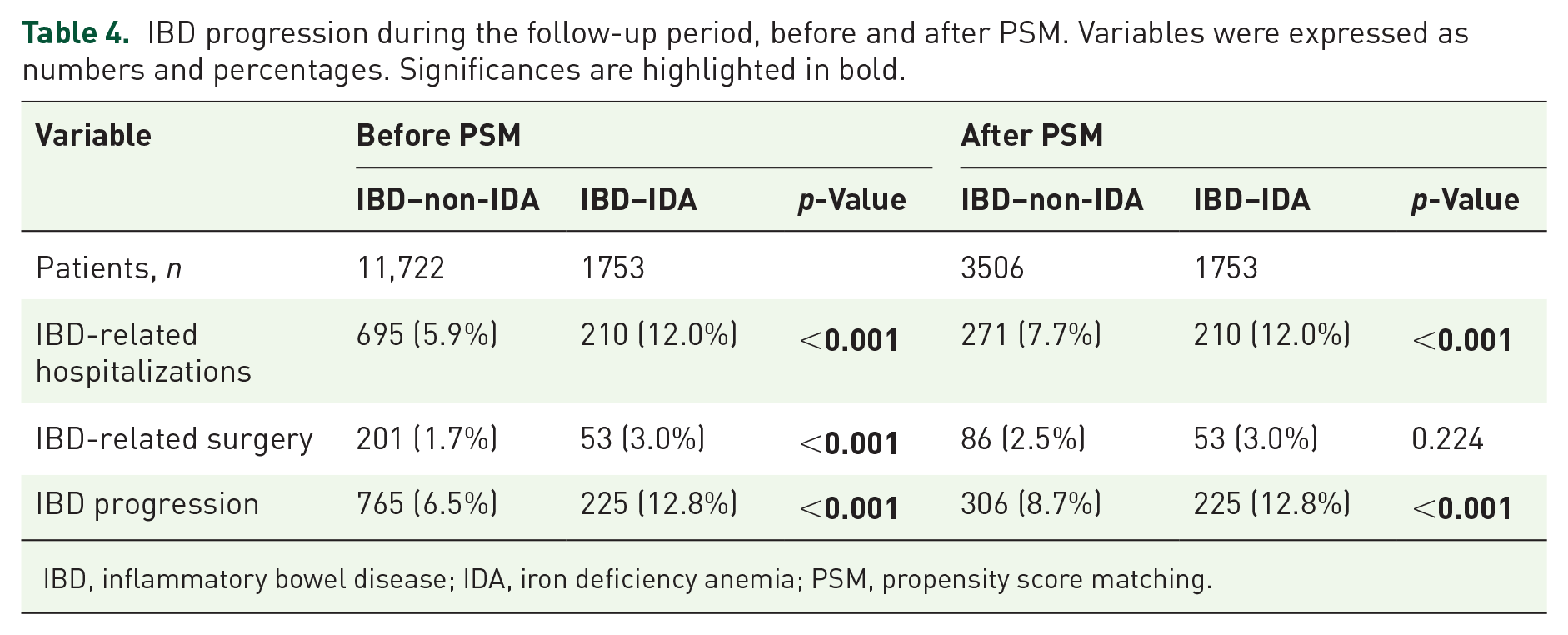
IBD, inflammatory bowel disease; IDA, iron deficiency anemia; PSM, propensity score matching.
The differences between the two cohorts were also evaluated in terms of healthcare resource consumption and costs during follow-up. Before PSM, a statistically significant increase in the mean annual number of all resources was observed in IBD–IDA patients: respectively, in IBD–non-IDA versus IBD–IDA cohorts, the mean number of drug prescriptions was 11.1 versus 16.0, that of hospitalization was 0.2 versus 0.5, and that of outpatient specialist service deliveries was 6.2 versus 9.7 [Figure 4(a)]. In parallel, mean annual healthcare costs were found to be significantly higher among IBD–IDA patients (p < 0.001): the overall mean annual cost was €2798 in IBD–non-IDA patients compared to €5317 in IBD–IDA ones [Figure 4(b)]. Among PSM-matched cohorts, the mean annual number of drug prescriptions (13.5 versus 16.0), hospitalizations (0.4 versus 0.5), and specialist service deliveries (7.4 versus 9.7) were significantly lower (p < 0.001) in IBD–non-IDA patients versus IBD–IDA cohorts [Figure 5(a)]. The mean annual total cost/patient was significantly higher in IBD–IDA patients versus the counterpart cohort (IBD–non-IDA versus IBD–IDA: €3046 versus €3693, p < 0.001), mostly accounting from higher hospitalization-related costs (€1462 versus €1020) and specialist services-related expenditures (€546 versus €452) [Figure 5(b)].

Mean annual number of healthcare resources (a) and related direct costs (b) during the follow-up period before PSM.

Mean annual number of healthcare resources (a) and related direct costs (b) during the follow-up period after PSM.
In the post-matched cohorts, predictors of IBD progression were identified by using a logistic model among all baseline variables. The age at diagnosis (OR 0.986, 95% CI: 0.979–0.992, p < 0.001), the presence of asthma (OR 0.586, 95% CI: 0.349–0.986, p < 0.05), ankylosing spondylitis (OR 2.385, 95% CI: 1.191–4.775, p < 0.05), previous IBD-surgical intervention (OR 1.361, 95% CI: 1.075–1.722, p = 0.01), and IBD severity (OR 1.741, 95% CI: 1.515–2.000, p < 0.001) were independent predictors of IBD progression. Additionally, the presence of IDA was positively correlated with IBD disease progression, as IBD–IDA patients could have a 1.59 increased risk of disease progression versus IBD–non-IDA cohort (OR 1.597, 95% CI: 1.329–1.928, p < 0.001) (Supplemental Table 4).
Discussion
In this real-world analysis conducted in a routine clinical setting, a co-diagnosis of IDA in IBD patients appears to be associated with an increased risk of disease progression and higher hospitalization rates, resulting in a substantial burden in terms of healthcare consumption and related costs. Although the role of treatment interventions for anemia with its possible clinical and economic benefits was not the focus of the present investigation, our findings seem to suggest that there is still room for improvement in the setting of IBD patients with IDA. The latest consensus European Crohn’s and Colitis Organisation established working groups to investigate the milestones to be considered for optimizing the management of iron deficiency and anemia in IBD: diagnosis, treatment, and prevention of IDA and of non-IDA. 11
Among included patients, IDA was detected in the year after IBD diagnosis in 13% of IBD patients, using as proxies the presence of iron preparation therapies, hospitalization(s) for IDA and/or at least one blood transfusion. In a review previously published by Gisbert and Gomollon, 29 the prevalence of anemia in IBD ranges from 16% in outpatients to 68% in hospitalized patients. However, compared with other IBD extraintestinal complications, anemia could be underestimated since it receives limited attention from both clinicians and researchers.30,31 Our data showed that IDA patients were characterized by an almost 60% increased risk of IBD disease progression. IBD-surgical intervention and IBD-related hospitalization during the follow-up were predictors of disease progression. These results are in line with a previous study reported by Koutroubakis et al. 17 , which stated that the persistent/recurrent anemia independently was correlated with hospitalizations, continued active disease, and IBD-related surgeries; thus, the presence of anemia could be considered to be associated with a disabling disease. Thus, greater efforts are needed to ameliorate the scenario of IDA management in IBD patients, also in view of the recent alarming data by Patel et al. 23 from a large retrospective US cohort study between 2011 and 2018, reporting that IBD patients spend on average 2 months each year in an anemic state. 23
In addition, a population-based IBD retrospective study among the Hungarian population reported that the presence of anemia was associated with medication use, the need for hospitalization and surgery in IBD patients. 32
The analysis of healthcare resource consumption showed a higher number of annual hospitalizations and a higher number of specialistic service deliveries in anemic IBD patients; this is translated into a statistically significant increase in direct costs covered by the INHS in IBD–IDA patients compared to IBD patients without IDA, both in unbalanced and balanced cohorts. Although data on the economic burden of IBD patients with IDA are limited among the Italian population, two retrospective studies in the US population have reported results in line with our findings. Blaney et al. 33 showed that IBD patients with anemia had significantly more hospitalizations, increased length of stay, more emergency department, gastroenterology, and primary care provider visits, and higher costs compared to patients with IBD without anemia. Patients with more severe anemia had more healthcare resource utilization and incurred even higher total costs. These data are congruent with those reported by the Crohn’s & Colitis Foundation initiative: the presence of anemia, among other comorbidities, was defined as a cost-driver in IBD since it was associated with higher resource utilization, particularly hospitalization, and patients with the previous diagnosis of anemia had costs that were 8% higher compared with patients without anemia. 34 Based on this evidence and the data emerging from the present analysis, IDA might be plausibly associated with a more severe IBD course and negative repercussions on cost sustainability by the INHS. In view of that, further studies are needed to evaluate the possible beneficial role of therapeutic interventions to correct anemia (i.e., iron supplementation, erythropoietin)35,36 on the clinical and economic burden of IDA in IBD patients. Moreover, the benefits of anemia correction on quality of life have been well assessed and appear to be independent of the disease activity status. 11
The present analysis results should be interpreted by considering some limitations related to its retrospective nature and to the use of anonymized data derived from administrative databases. Region/Local Health Unit’s administrative databases have progressively improved the quality of the collected data. However, some information may be lacking, and therefore, patients with missing data were excluded from the analysis. In addition, there were incomplete or absent clinical data on IBD phenotypes according to Montreal classification and its updates,37,38 as well as on disease severity grades based on biomarkers and test laboratory assessment. In the PSM analysis, the presence of some confounding factors that can affect disease progression (i.e., smoking habit and ethnicity) was not included since they were not available in the database, thus representing a limitation in the analysis. Thus, a proxy of disease severity score (based on the presence of previous IBD treatments and hospitalizations) was used to balance the two cohorts. Since comorbidities and IDA were evaluated based using a proxy of diagnosis, there might be incomplete capture of these variables among patients, and primary care data could not be collected by administrative databases used in the present analysis. Therefore, in the cost and healthcare utilization analysis, a limitation could be that we cannot account for visits and expenses outside the INHS and did not account for indirect costs such as lost wages and transportation to and from healthcare services. Despite these limitations, our analysis provides a description of the plausible associations between anemia and IBD disease progression and utilization and cost of care in a setting of real clinical Italian practice.
In conclusion, our data obtained from the real-world clinical Italian practice indicate a probable correlation between anemia occurrence and a more demanding challenge in IBD setting. The anemic state could be considered a surrogate biomarker of disease severity and, therefore, of IBD disease progression, in terms of hospitalization, surgeries, and recurrence to treatments. Together with the high impact of anemia on the economic burden of IBD, these findings suggest that the appropriate therapeutic management of anemic IBD patients should be properly considered.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231177153 – Supplemental material for Iron deficiency anemia impacts disease progression and healthcare resource consumption in patients with inflammatory bowel disease: a real-world evidence study
Supplemental material, sj-docx-1-tag-10.1177_17562848231177153 for Iron deficiency anemia impacts disease progression and healthcare resource consumption in patients with inflammatory bowel disease: a real-world evidence study by Gionata Fiorino, Jean-Frederic Colombel, Kostas Katsanos, Fermín Mearin, Jürgen Stein, Margherita Andretta, Stefania Antonacci, Loredana Arenare, Rita Citraro, Stefania Dell’Orco, Luca Degli Esposti, Antonio Ramirez de Arellano Serna, Neige Morin and Ioannis E. Koutroubakis in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
