Abstract
Background:
Digital single-operator cholangioscopy (DSOC) enhances biliary stricture diagnosis, but the collection of quality samples can be difficult due to the small diameter of the working channel.
Objectives:
A new DSOC system (EyeMAX™ 11Fr; Micro-Tech Endoscopy, Nanjing, China) with a 2.0-mm working channel, accommodating pediatric forceps (1.6 mm), has been introduced in France. This study reports on the first French experience.
Design:
A retrospective, multicenter observational study.
Methods:
The study on DSOC was conducted across five endoscopy units within the French Society of Private Hepato-Gastroenterology (SFHGL-CREGG). Satisfaction and procedural evaluations were recorded using a visual analog scale (VAS) and compared with the Spyglass™ DS II (Boston Scientific, Marlborough, MA, USA).
Results:
Among 28 patients, 71.5% had undergone prior endoscopic sphincterotomy. Indications were biliary strictures in 26 patients and lithiasis in 2. Strictures were located in the bile duct (41%) or hilum (59%). The mean satisfaction score was 8.69 out of 10, with the EyeMAX 11Fr rated as superior to the SpyGlass DS II in 86% of cases. The mean biopsy count was 8.52 in 13.19 min, with 100% procedural success. Pathology specimens were rich (24%), adequate (52%), low (12%), or absent (12%). Sensitivity for cholangiocarcinoma was 73.7% (14/19). Benign strictures regressed in all cases. Procedure-related complications included pain twice (7.1%), fever once (3.6%), infection once (3.6%), and pancreatitis twice (7.1%), all of which were resolved rapidly.
Conclusion:
This first study on the EyeMAX 11Fr DSOC highlights its ease of use, superior biopsy facilitation, and high user satisfaction, achieving 73.7% sensitivity for cholangiocarcinoma diagnosis.
Trial registration:
ClinicalTrials.gov ID: NCT06933576.
Plain language summary
This study looked at a new tool called the EyeMAX™ 11Fr, used during a procedure known as digital single-operator cholangioscopy (DSOC), which allows an endoscopist (a doctor specialized in digestive endoscopy) to directly visualize the inside of the bile ducts. The main goal was to determine whether this new device could improve the collection of tissue samples, a process that is often challenging with existing tools. Researchers reviewed cases from five medical centers in France where this new device was used in 28 patients, mainly to investigate bile duct narrowings (called strictures). The EyeMAX™ 11Fr features a slightly larger channel that accommodates improved biopsy forceps. Overall, endoscopists rated the device highly, preferring it in most cases over the SpyGlass™ system – until now, the only available DSOC platform. The procedures had a 100% success rate, and the tissue samples were generally of high quality. The device demonstrated good sensitivity (around 74%) in detecting bile duct cancer. A few minor complications occurred, such as pain or fever, but these were quickly managed. In summary, this early experience with the EyeMAX™ 11Fr in France suggests that it is easy to use, improves tissue sampling, and may enhance the diagnosis of biliary pathology.
Introduction
Cholangiocarcinoma is the most common cause of biliary stricture. 1 Cholangiocarcinoma cytopathological characterization must be determined before initiating antitumoral treatment such as chemotherapy and/or biliopancreatic surgical resection,2–5 due to the 5%–25% risk of benign disease in cases of indeterminate bile duct stenosis and the 3%–7% risk in cases of surgery for suspected malignant bile duct stenosis.1,6–8
In cases of mass syndrome, a cytopathological diagnosis is usually easily obtained via endoscopic ultrasound-guided fine needle biopsy (EUS-FNB).9,10 Where biliary stricture occurs without mass syndrome, cholangiocarcinoma is diagnosed through biliary brushing during endoscopic retrograde cholangio-pancreatography (ERCP) in only 30%–60% of cases.1,11,12 To obtain larger amounts of histological material and establish a diagnosis based on tissue fragments rather than cytology, cholangioscopy was developed, thereby overcoming the low sensitivity of biliary brushing for cholangiocarcinoma. Digital single-operator cholangioscopy (DSOC), such as the new-generation SpyGlass™ DS II cholangioscope (Boston Scientific, Marlborough, MA, USA), combined with biopsies, appears to be the most effective approach, offering a higher—though still imperfect—diagnostic sensitivity of 69%–74%.12,13 However, one of the challenges that makes DSOC less likely to be considered as a first-line approach is the complexity of the procedure, particularly in performing biopsies. Due to the small diameter of the working channel, only dedicated low-capacity forceps can be used, and the friction forces exerted with each pass of the forceps make the procedure challenging and time-consuming. The SpyGlass DS II was, for a long time, the only available DSOC system. Recently, however, another DSOC system has been developed, the EyeMAX™ 11Fr cholangioscope (Micro-Tech Endoscopy, Nanjing, China), which features a wider working channel, enabling the use of pediatric biopsy forceps with a theoretically higher capacity (Figure 1). 14

EyeMAX™ 11Fr DSOC, with a large (2.0 mm) working channel: Arrow.
To determine whether the EyeMAX 11Fr provides a real improvement, we conducted a multicenter retrospective observational study on the initial use of the new DSOC system in France, in centers affiliated with the French Society of Private Hepato-Gastroenterology (SFHGL-CREGG). The primary endpoint was overall satisfaction with the EyeMAX 11Fr, assessed using a visual analog scale (VAS) and defined as better, equivalent, or worse compared with the SpyGlass DS II, habitually used by operators as it was the only available option. The secondary endpoints included procedural evaluations recorded using the VAS and compared with the SpyGlass DS II (rated as better, equivalent, or worse), the sensitivity for cholangiocarcinoma diagnosis, and the abundance of cellularity, assessed using a four-stage cellularity classification.
Patients and methods
This multicenter retrospective observational study was conducted from February 2024 to January 2025 across five expert tertiary endoscopy units. The work was sponsored by Paris-Bercy Clinic (Charenton-le-Pont, France) and conducted under the auspices of the French Society of Private Hepato-Gastroenterology (SFHGL-CREGG). Micro-Tech Endoscopy France made six units available free of charge per center, totaling 30 DSOC devices and the necessary forceps for biopsies. The DSOC procedures were performed consecutively in each center, and the satisfaction questionnaires were completed prospectively. Written informed consent for the ERCP procedures was obtained from all patients. The data were retrospectively collected by extraction from our medical patient management software, and via the satisfaction questionnaires systematically given to practitioners after each use. The study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 15
The study was approved (IRB_ADENE_20250302) by the Ethics Committee of the Adène group, France (IRB00013922, IORG0011759), and registered at ClinicalTrials.gov (NCT06933576).
All authors declare that they have access to the study data and have reviewed and approved the final manuscript.
Patients
The study included all consecutive patients who were referred to one of the participating expert tertiary endoscopy units for an ERCP for a bile duct stenosis. Patients with significant tissue mass that could easily be punctured by EUS-FNB were not included. In accordance with good practice guidelines, ERCP was contraindicated in cases of non-accessibility to the bile duct due to a history of Billroth II or Roux-en-Y reconstruction, coagulation disorders (such as partial thromboplastin time >42 s, prothrombin time (Quick value) <50%, or platelet count <50,000/mm³), or treatment with clopidogrel.
ERCP procedure
All the ERCP procedures included in the scope of the study were performed by endoscopic expert physicians, who annually perform more than 200 ERCPs each, and have already conducted over 100 cholangioscopies using the SpyGlass DS II system. The ERCP included in the scope of the study were the first procedures conducted in each participating center with the EyeMAX system. Patients were under general propofol-induced anesthesia, with antibiotic prophylaxis (2 g cefoxitin, administered intravenously, according to French guidelines 16 ) and lying supine or in the left lateral position. The following duodenoscopes were used: (i) the Olympus TJF-Q180 V and TJF-Q190 V (Olympus Europe Inc., Hamburg, Germany) with a working channel diameter of 4.2 mm, and (ii) the Fujinon ED-580XT (Fujifilm France (Medical Systems), Asnières, France) with a working channel diameter of 4.2 mm. After deep bile duct catheterization with a guidewire and biliary sphincterotomy, DSOC was performed using an over-the-wire technique with the EyeMAX CDS1001 (2200 mm length, 11Fr diameter, 2.0 mm working channel; Figure 1) from Micro-Tech Endoscopy (Nanjing, China), with the EyeMAX platform BS-W-100 featuring LED and HD via DVI (Figure 2). Two fixation systems were made available, one on a harness attached to the endoscopist’s torso and the other directly fixed on the duodenoscope. When performing biopsies, the forceps used was the EyeMAX Bite (BF16006, 2900 mm length, 1.8 mm diameter, 4.5 mm jaw opening, alligator type; Figure 3). Biliary balloon dilation of the stenosis and/or the papilla was recorded along with any additional sampling methods, such as biliary brushing or EUS-FNB, when applicable.

Comparison of the EyeMAX™ Bite (Micro-Tech Endoscopy, Nanjing, China; 1.8 mm diameter, alligator type): dedicated 2900 mm-pediatric forceps with serrated jaws (left) and pediatric forceps routinely used in digestive endoscopy (right).

Visualization of a cholangiocarcinomatous stricture using the EyeMAX™ 11Fr DSOC.
Histological preparations and analysis
The tissue fragments obtained were fixed in formalin prior to delivery to each cytopathology unit within 2 days (Figure 4). Quantification of the cellularity for each case was defined by the pathologist in the satisfaction questionnaire as: rich, adequate, or poor (Figure 5).

Macroscopic samples obtained from a cholangiocarcinomatous stricture using the EyeMAX™ Bite (1.8 mm diameter, alligator type) through the EyeMAX 11Fr DSOC. Close-up of the large fragment retrieved in the forceps at each biliary biopsy (left) and biopsy fragments observed in the formalin container (right).

Anatomopathological sampling performed using the EyeMAX™ 11Fr cholangioscopy with an EyeMAX Bite (1.8 mm diameter, alligator type).
Data collection
We collected patient characteristics (gender, age, American Society of Anesthesiologists score, and current acetylsalicylic acid treatment), biliary stricture location (main bile duct or hilar, with type according to the Bismuth-Corlette classification), and procedure characteristics (model used, harness/fixed on duodenoscope, total ERCP duration, cholangioscopy duration, success rate, number of biopsies performed, and biopsy duration), and recorded any post-procedure complications. Depending on the center and the other procedures performed, and according to the standard practices in each center, patients were discharged within 4 h of the endoscopic procedure or remained in the hospital for at least 24 h. Before discharge, they were examined to ensure there were no signs of perforation or bleeding or of acute pancreatitis, sepsis, or other complications. Any delayed adverse events within 1 month of the procedure reported by the patients and referring physicians were also recorded. Serious adverse events were defined as complications resulting in a new hospital admission or the extension of an existing hospital stay, or a significant disability or death. Adverse effects (AEs) and severe AEs due to ERCP were defined and graded according to the AGREE classification. 17 As recommended, repeat and/or alternative procedures were performed in patients where false-negative diagnoses were suspected.
Endpoint definitions
The primary endpoint was overall satisfaction with the EyeMAX 11Fr, evaluated using a VAS (ranging from 0—not at all satisfied—to 10—extremely satisfied) and compared with the device currently available in France, the SpyGlass DS II. Satisfaction was categorized as better, equivalent, or worse relative to the SpyGlass DS II.
The secondary endpoints included the evaluation of each step of the procedure using a VAS, as described below, and comparison with the SpyGlass DS II (EyeMAX 11Fr categorized as better, equivalent, or worse relative to the SpyGlass DS II). Additional endpoints included the sensitivity for cholangiocarcinoma diagnosis (biopsy positivity rate for malignancy in cases of cholangiocarcinoma) and the assessment of cellularity abundance, classified into four levels: rich, adequate, poor, or absent.
The various steps of the procedure evaluated using a VAS (ranging from 0—impossible—to 10—extremely easy) were as follows: introduction of the EyeMAX 11Fr into the duodenoscope’s working channel, exit of the device from the working channel, biliary cannulation through the papilla, progression through the bile duct, maneuverability, ease of introducing the biopsy forceps to the target, and ease of performing biopsies.
Other secondary endpoints included image quality (definition, lens cleaning quality, and brightness), assessed using a VAS (ranging from 0—very poor—to 10—exceptional), and the use of the harness model compared with the model fixed directly on the duodenoscope, evaluated by the question: “Does the harness facilitate handling? (Y/N).”
The diagnosis of cholangiocarcinoma was established through biliary cytopathological examination of biopsies obtained during DSOC (Figures 3–5), biliary brushing, EUS-FNB, or surgical resection, and/or based on tumor progression observed after more than 6 months of follow-up in cases where specimens were negative for malignancy.18,19
Statistical analysis
Quantitative variables were expressed as mean (SD: standard deviation) and/or median (IQR: interquartile range), while qualitative variables were expressed as numbers and percentages. Differences between centers were assessed by Fisher’s exact test for qualitative variables, and the Kruskal–Wallis test for quantitative variables. Alpha risk was set to 5% (α = 0.05). Statistical analysis was performed with EasyMedStat (version 3.37.1; www.easymedstat.com).
Results
A total of 28 patients (15 men, 13 women) with a mean age of 71.79 ± 10.5 years were enrolled in this study. Two EyeMAX devices were not used in one of the centers during the availability period due to a lack of patients admitted for DSOC. Of the 28 patients, 20 (71.5%) had a history of sphincterotomy. The procedure was performed during a scheduled anticoagulation therapy interruption for 2/28 patients (7.1%) and while on antiplatelet therapy for 5/28 patients (17.9%). The characteristics of the study population are summarized in Table 1.
Description of the study population.

ASA, American Society of Anesthesiologists; ERCP, endoscopic retrograde cholangiopancreatography; SD, standard deviation.
Demographic data, stricture location, and diagnosis are presented in Figure 6. The indication for cholangioscopy was biliary stricture in 26/28 patients (93%) and lithiasis or lithotripsy in 2/28 patients (7%). The location of the biliary stricture was the bile duct in 11/26 patients (42%), and hilar in 15/26 patients (58%), with 4 classified as stage I, 3 as stage II, 4 as stage III, and 2 as stage IV according to the Bismuth classification. The final diagnosis was cholangiocarcinoma in 19/28 patients (67.9%), while 9/28 patients (32.1%) had biliary stones or benign strictures, all of which regressed with a median follow-up of 10.02 months (range: 7.46–11.04).

Indication for cholangioscopy, and final diagnosis.
Biliary dilation was necessary in 2 patients with an immediate supra-ampullary stricture to allow cannulation of the bile duct through the papilla with the EyeMAX 11Fr.
Additional sampling methods were performed in some patients, including biliary brushing in 4/25 patients (16%) and EUS-FNB in 6/25 patients (24%). No additional sampling method was performed in 13/25 patients (52%). Diagnosis of cholangiocarcinoma was obtained through histopathological analysis of biliary biopsies via choledochoscopy in 14/19 cases (73.7%), and through the development of hepatic metastases and/or peritoneal carcinomatosis nodules during follow-up in 5/19 cases (26.3%). Additionally, biliary brushing did not lead to the diagnosis of cholangiocarcinoma in any of the cases where it was performed (0/4), while EUS-FNB did so in 2/6 cases (33%). However, these additional samples did not help recover the diagnosis in any of the falsely negative biliary biopsies via choledochoscopy.
Of the 14 patients with cholangiocarcinoma diagnosed through biopsies using the EyeMAX 11Fr, 8 had not undergone any prior ERCP, while 6 had negative endobiliary biopsies and/or biliary brushings during a previous ERCP. The five other patients with cholangiocarcinoma, in whom no cancerous cells were detected in the biopsies performed with the EyeMAX 11Fr, had all previously had negative samples during a prior ERCP.
Of the nine patients with benign disease, seven suffered from stenosis related to inflammatory cholangitis or post-surgical injury, and two suffered from biliary lithiasis.
AEs and severe AEs, according to the AGREE classification, were as follows 17 : two patients (7.1%) in the series suffered from procedure-related pain, one patient (3.6%) from fever, one patient (3.6%) from infectious complication, and two patients (7.1%) from acute pancreatitis (including one severe case requiring a 15-day hospital stay).
Endpoints
The mean satisfaction scores for the overall experience with the EyeMAX 11Fr and at each step of the procedure are reported in Table 2 and illustrated with boxplots in Figure 7. Concerning the primary endpoint, the mean overall satisfaction score for the EyeMAX 11Fr was 8.69 (SD 1.06), with the EyeMAX rated as better compared with the SpyGlass DS II in 85.7% of cases, in 10.7% of cases as equivalent, and in 3.6% of cases as worse, with no significant differences between centers.
Mean satisfaction scores: overall and at each step of the EyeMAX™ 11Fr DSOC procedure.

VAS (ranging from 0—not at all satisfied—to 10—extremely satisfied).
EyeMAX 11Fr DSOC compared with the device currently available in France, the SpyGlass DS II DSOC.
CBD, common bile duct; DSOC, digital single-operator cholangioscopy; VAS, Visual Analogic Scale.

Mean satisfaction scores: overall and at each step of the EyeMAX™ 11Fr DSOC procedure.
The secondary endpoints were as follows:
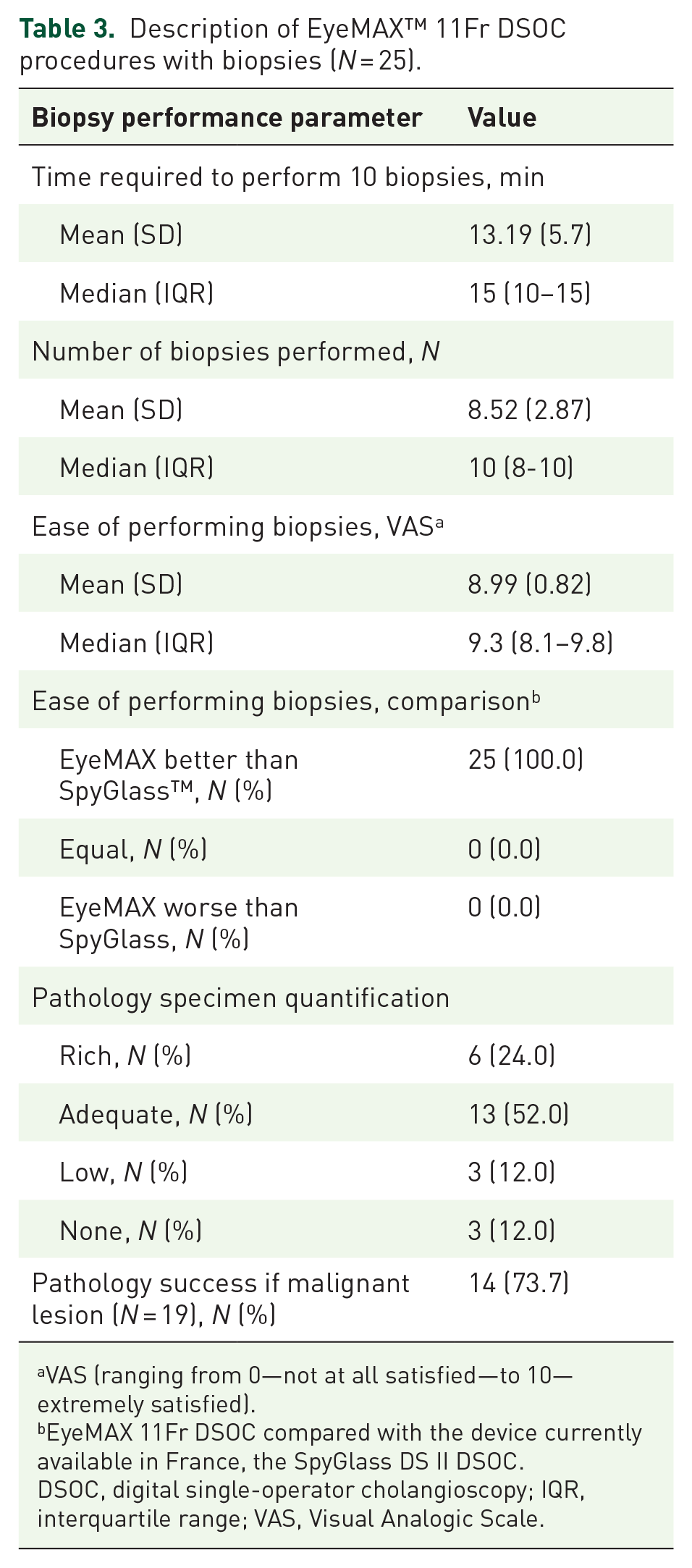
DSOC was feasible in all cases, and biopsies were consistently performed when indicated (25 patients). A breakdown of the EyeMAX 11Fr DSOC procedures with biopsies, limited to the 25 patients in whom biopsies were performed, is presented in Table 3. The mean number of biopsies was 8.52 (SD 2.87), with a mean procedure time of 13.19 min (SD 5.7) and a 100% success rate. Pathology specimens were classified as rich in 6/25 cases (24%), adequate in 13/25 cases (52%), low in 3/25 cases (12%), and absent in 3/25 cases (12%). Among the 19 cases of cholangiocarcinoma, pathology specimens were classified as rich or adequate in 13 of the 14 true positives (94%), and in 2 of the 5 false negatives (40%, p = 0.037).
Description of EyeMAX™ 11Fr DSOC procedures with biopsies (N = 25).
VAS (ranging from 0—not at all satisfied—to 10—extremely satisfied).
EyeMAX 11Fr DSOC compared with the device currently available in France, the SpyGlass DS II DSOC.
DSOC, digital single-operator cholangioscopy; IQR, interquartile range; VAS, Visual Analogic Scale.
Satisfaction at each step of the procedure was reported as better compared with the SpyGlass DS II: 57.1% for the simplicity of fixation, 39.3% for the introduction of the EyeMAX 11Fr into the duodenoscope’s working channel, 35.7% for the exit of the device from the working channel, 53.6% for biliary cannulation through the papilla, 64.3% for progression through the bile duct, 75.0% for maneuverability, 88.9% for the ease of introducing the biopsy forceps to the target, and 100% for the ease of performing biopsies.
Satisfaction with image definition quality, lens cleaning quality, and image brightness was also reported as better compared with the SpyGlass DS II in 82.1%, 53.6%, and 85.7% of cases, respectively.
Discussion
This multicenter observational study on consecutive patients scheduled for cholangioscopy, the first-ever published study specifically evaluating the EyeMAX 11Fr cholangioscope, demonstrated the ease of use of the device, its high satisfaction level at every step of the procedure, and its particular effectiveness in facilitating biopsies, which were easily performed in a short time. The ease of performing multiple large-volume sampling in a shorter time, promised by the EyeMAX 11Fr represents substantial progress in the field of cholangioscopy, and the device could henceforth be considered as a routine first-line approach for diagnosing biliary strictures. However, the 73.7% sensitivity for cholangiocarcinoma in our series remains suboptimal.
The management of patients with biliary cholangiocarcinoma relies on therapeutic options such as surgery and/or chemotherapy, for which an anatomopathological diagnosis is typically required. 5 Biliary brushing is often the initial diagnostic step due to its simplicity and rapid execution during ERCP. However, despite the availability of more aggressive brushes with stiffer bristles designed to obtain more abundant cytological material 20 compared with standard brushes, tissue samples obtained through cholangioscopy-guided biopsies should provide more frequent and reliable diagnoses of cholangiocarcinoma. Moreover, additional cytopathological examinations (such as immunohistochemistry and molecular biology for mismatch repair testing, microsatellite instability, etc.), easily performed on such biopsy samples, are increasingly required by oncologists for subsequent treatment. Nevertheless, despite obtaining abundant material (rich or adequate in more than three-quarters of cases), the sensitivity for cholangiocarcinoma in our series does not exceed 80%, highlighting the difficulty of making a pathological diagnosis in certain forms of cholangiocarcinoma, in which rare tumor cells are highlighted within an abundant fibrous stroma.21,22 In this study, the failure to diagnose cholangiocarcinoma via EyeMAX-guided biopsies was significantly associated with insufficient cytopathological material (none or low) compared with successful biopsy-based diagnoses (60% vs 7%, respectively; p = 0.037), highlighting the challenge of obtaining high-quality samples in such cases. The difficulty of consistently making a pathological diagnosis of cholangiocarcinoma should encourage us to employ all possible sampling methods, such as biliary brushing and even EUS-FNB, especially in locations where biopsies under cholangioscopy are difficult to perform (immediate supra-ampullary location and intrahepatic sites). To our knowledge, only two studies in the literature have compared the sensitivity of biliary brushing and targeted biopsies under cholangioscopy for cholangiocarcinoma.23,24 The first study, a retrospective study of 92 patients, reported a higher sensitivity of 71% for cholangioscopy compared with 45% for biliary brushing (p = 0.03). 23 The second, the only published randomized controlled trial (RCT), involved 61 patients and demonstrated a significantly higher sensitivity for targeted biopsies compared with biliary brushing, with 68.2% sensitivity versus a surprisingly very low 21.4%, respectively (p < 0.01). 24 In both studies, brushing was performed using a standard brush rather than a more aggressive and productive brush. 20 Additionally, in the latter study, the difference between the two arms was also attributed to an unbalanced proportion of benign versus malignant strictures, with nearly 50% benign strictures in the biliary brushing arm. 24 While these data highlight the diagnostic superiority of targeted biopsies under cholangioscopy compared with brushing, cholangioscopy has not yet been established as a routine first-line diagnostic tool. This is partly due to the limitations of these studies, but primarily due to the complexity of cholangioscopy, including the length of the procedure, the difficulty of scheduling it within a busy endoscopy program, and the technical challenges of performing biopsies, as a result of the friction of the forceps within the working channel.
Overall satisfaction with the EyeMAX 11Fr in our study was excellent, with the new device deemed superior to the one previously available in France (SpyGlass DS II) in 85.7% of cases. While satisfaction was reported at all stages of the cholangioscopy procedure, the most striking factor is undoubtedly the impact on the performance of biopsies, with the EyeMAX 11Fr making the biopsy process easier and faster due to the use of higher-capacity (pediatric) forceps. By facilitating cholangioscopy procedures that are easy to plan within the endoscopy program, and thereby allowing for the performance of numerous large biopsy specimens in a very short procedure time, the EyeMAX 11Fr thus represents a promising advancement in the field of cholangioscopy, making DSOC a viable first-line diagnostic option for indeterminate bile duct strictures. While this pilot study, based on the initial uses of the EyeMAX 11Fr, does not yet warrant the immediate replacement of the only device currently available, the fact that the new device facilitates the acquisition of larger and more numerous biopsy samples within a short procedural time is promising. These advantages should prompt the initiation of a comparative study, with a primary objective focused on diagnostic sensitivity for cholangiocarcinoma and the quantity of histological material obtained to support accurate diagnosis.
This study has several strengths. First, it is the first to specifically evaluate this model of cholangioscope with a large working channel (EyeMAX, 11Fr diameter, 2.0 mm working channel). To date, only case reports25–28 and one retrospective study combining data from different models (9Fr and 11Fr) across multiple indications have been published on the EyeMAX 11Fr cholangioscope. 14 In the latter study, fewer than a dozen patients were ultimately explored for suspected bile duct neoplastic lesions using the EyeMAX 11Fr, and no evaluation of procedural satisfaction was conducted. 14 Our study is, therefore, the first-ever user satisfaction study conducted, as well as the first to directly compare the only cholangioscope available until now, the 10.5Fr diameter SpyGlass DS II. Although maneuverability challenges might have been expected with the larger EyeMAX 11Fr model, this was not observed in our study. Moreover, the anticipated benefit of improved biopsy feasibility—enabled by the large 2.0 mm working channel—was confirmed, with the EyeMAX 11Fr rated as superior to the SpyGlass DS II (1.2 mm working channel) in 100% of cases. Second, this study is a multicenter one, conducted in five expert private units, located throughout France, in contrast to the only previously published study on the initial use of the EyeMAX 9Fr and 11Fr, which was monocentric. 14 This not only allowed us to compare the perspectives of different operators but also to demonstrate the consistency of their feedback. The multicenter nature of the study and the inclusion of experienced operators across France significantly strengthen the generalizability of our findings, which is particularly important given the novelty of the device evaluated.
This study has some limitations. First, it is a retrospective study. Despite not being prospective, it is consecutive and includes all EyeMAX 11Fr DSOC procedures performed in every unit. Second, this study is a satisfactory study, and the comparison between the two DSOC systems is based solely on the recollections of users regarding their habitual device. Although it could be argued that such a satisfaction evaluation is inherently subjective, the high satisfaction reported in this study is nonetheless supported by the very high success rate in performing around 10 biopsies and the notably short time required to complete them, especially when compared with the time usually needed with the Spyglass system. Third, there is undeniably a novelty bias in this type of study that favors the new device, as practitioners are pleased to use innovative equipment.29,30 However, the likely overrating of the new device due to the novelty effect should, at least partly, be counterbalanced by the negative impact of the mandatory habit change required from the endoscopist. The design of this study did not permit the implementation of any blinding procedures for the comparison. Fourth, the abundance quantification in this study was evaluated by a home-made four-stage classification due to the lack of a reference classification. The lack of consensus surrounding this classification is all the more relevant in a non-comparative study such as ours, as opposed to comparative studies, where this limitation is mitigated by the fact that the classification applies equally to both techniques being evaluated. Despite the concerns regarding this non-standardized, easy-to-use classification, which is a subjective semi-quantitative evaluation, it demonstrated particularly remarkable results, unusual in the exploration of bile duct stenosis, with more than 75% of samples considered rich or adequate. In the absence of a standardized classification system, computer-assisted quantification of the surface area of pathological samples could be considered to reduce subjective bias, particularly in prospective comparative studies. Due to these limitations, the results of this study must therefore be applied cautiously in routine practice and should be confirmed by a comparative RCT study.
In conclusion, this first-ever study on the EyeMAX 11Fr (Micro-Tech Endoscopy) highlighted its ease of use, high satisfaction at every step of the procedure, and its particular effectiveness in facilitating the rapid and easy performance of numerous large biopsies, suggesting the new device represents a significant advancement in the field of cholangioscopy, which could, at last, make the routine use of DSOC as a first-line approach for diagnosing biliary strictures possible. However, this potential needs to be confirmed by further randomized comparative studies before any change in clinical practice can be recommended.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251360117 – Supplemental material for Contribution of the EyeMAX™ 11Fr (Micro-Tech) digital single-operator cholangioscope with a wide working channel: a multicenter pilot study on the first French experiences
Supplemental material, sj-docx-1-tag-10.1177_17562848251360117 for Contribution of the EyeMAX™ 11Fr (Micro-Tech) digital single-operator cholangioscope with a wide working channel: a multicenter pilot study on the first French experiences by David Karsenti, Adrien Sportes, Sarah Leblanc, Stéphane Béchet, Jonathan Derman, Isaac Fassler and Bertrand Brieau in Therapeutic Advances in Gastroenterology
Footnotes
Appendix
Acknowledgements
We thank the SFHGL-CREGG (French Society of Private Hepato-Gastroenterology) for its role in coordinating the study. We also gratefully acknowledge the contribution of the nursing and administrative staff involved in data collection across the five centers where the endoscopic procedures were performed.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
