Abstract
Gastric metastasis from breast cancer has a high rate of misdiagnosis and missed diagnosis. Data of patients who had gastric metastasis from breast cancer were retrieved from our hospital between 2014 and 2020. The gastric metastasis from breast cancer incidence was 0.04% (5/14,169 cases of breast cancer). Four patients had invasive lobular carcinoma, and the other patient had invasive ductal carcinoma. The time from the initial diagnosis of breast cancer to the appearance of gastric metastasis ranged from 0 to 12 years. One patient’s endoscopic presentation was similar to mucosal-associated lymphoid tissue lymphoma and presented with gastric mucosal congestion and edema, widened wrinkles, mixed color fading, and redness. The initial pathological diagnosis of this patient was mucosal-associated lymphoid tissue lymphoma, and breast cancer was finally confirmed by immunohistochemistry. Hormonal receptors were highly expressed in four patients with primary and metastasis lesions and were negative in one patient. Human epidermal growth factor receptor 2 was negative in all patients. Mammaglobin and GATA3 were positive in all patients. In conclusion, the gastric metastasis of breast cancer incidence rate is low, and misdiagnosis can lead to insufficient or excessive treatment. Multiple biopsies and immunohistochemistry should be performed to diagnose gastric metastasis of breast cancer.
Background
Breast cancer is the most common malignant tumor in women. Although the 5-year survival rate of breast cancer has continued to increase in recent years, relapses still occur in 30% to 80% of patients. 1 The most common sites of distant metastasis are the lymph nodes, lungs, bone, liver, and brain. The gastrointestinal tract is a rare metastatic site of breast cancer, and it is often overlooked by clinicians. The incidence of gastric metastasis of breast cancer obtained by autopsy is 18%, which is much higher than that of clinical retrospective studies (0.1%–0.3%).2–5 This large discrepancy in the rate of gastric metastasis of breast cancer is a concern.
Breast cancer is one of the most common malignancies responsible for gastric metastasis, second only to malignant melanoma. 6 The actual incidence of gastric metastasis from breast cancer may be underestimated because of a potentially long interval between the primary diagnosis and occurrence of metastasis, atypical symptoms, and difficulty in diagnosis. Clinical symptoms in patients with gastric metastasis are often atypical. Some of these patients may be asymptomatic and only show non-specific gastrointestinal symptoms during an examination. There are also some unusual manifestations, such as gastric outlet obstruction and gastric perforation, in these patients.7–11 In addition, doctors’ lack of knowledge of gastric metastasis has led to a decrease in the accuracy of diagnosis of this type of metastasis.
In our study, we report five cases of gastric metastasis from breast cancer and present a review of the literature. This study aims to improve clinicians’ understanding of gastric metastasis of breast cancer and reduce the rate of misdiagnosis.
Case presentation
Patients
Data of five patients who had gastric metastasis from breast cancer were retrieved from the consultation files of The 4th Hospital of Hebei Medical University between 2014 and 2020. The patients’ clinical data are shown in Table 1. All patients were women and the average age at the diagnosis of gastric metastasis was 61.6 years (range: 52–69 years). All patients had invasive lobular carcinoma (ILC), except for Patient 4 who had invasive ductal carcinoma (IDC). The first four patients underwent surgery when they were initially diagnosed with breast cancer and received standardized adjuvant treatment. The last patient had abdominal discomfort as the first symptom and was diagnosed with stage IV breast cancer. Therefore, she did not have breast surgery. The incidence of gastric metastasis of breast cancer was 0.04% among 5/14,169 cases of breast cancer from 2014 to 2020.
Patients’ characteristics.

ILC, invasive lobular carcinoma; IDC, invasive ductal carcinoma; Chemo, chemotherapy; Radio, radiotherapy; Endo, endocrine therapy; MRM, modified radical mastectomy; BCS, breast conservation surgery; MALT, mucosal-associated lymphoid tissue; HIPEC, hyperthermic intraperitoneal chemotherapy; PD-L1, programmed death-ligand 1; –, not available.
Imaging and endoscopy
The time from the initial diagnosis of breast cancer to the appearance of gastric metastasis ranged from 0 to 12 years (0 represents simultaneous diagnosis of breast cancer and gastric metastasis, Table 1). The symptoms of gastric metastasis, such as abdominal pain, vomiting, decreased appetite, and acid reflux, were non-specific. During the follow-up process, abdominal computed tomography in Patient 2 showed thickening of the gastric wall at the antrum, thickening of the peritoneum, and fluid accumulation in the abdominal cavity (Figure 1). A computed tomography scan of the other patients did not provide clear indications. The endoscopic findings varied among the patients. Patient 1 showed a rough mucosa at the junction of the gastric body and antrum. Patient 2 had diffuse infiltration of the entire gastric mucosa, and presented with a nodular appearance and stiffness. This condition caused stenosis and obstruction owing to edema and stiffness of the gastric antrum mucosa (Figure 2a). Patient 3’s lesion occurred at the junction of the gastric body and antrum, where mucosal congestion and erosion were found (Figure 2b). A congested nodule in the gastric body was found in Patient 4 (Figure 2c). Patient 5 had endoscopic findings similar to extranodal marginal zone lymphoma of mucosal-associated lymphoid tissue (MALT), with gastric mucosal congestion and edema, widened wrinkles, and scattered erosions all over the cardia, fundus, and gastric body (Figure 2d).

Abdominal computed tomography of Patient 2. (a) Thickening of the gastric wall at the antrum reaches 1.16 cm (red arrow). (b) Peritoneal thickened nodules (red arrow) can be seen and (c) Peritoneal thickening (red arrow) can be seen.
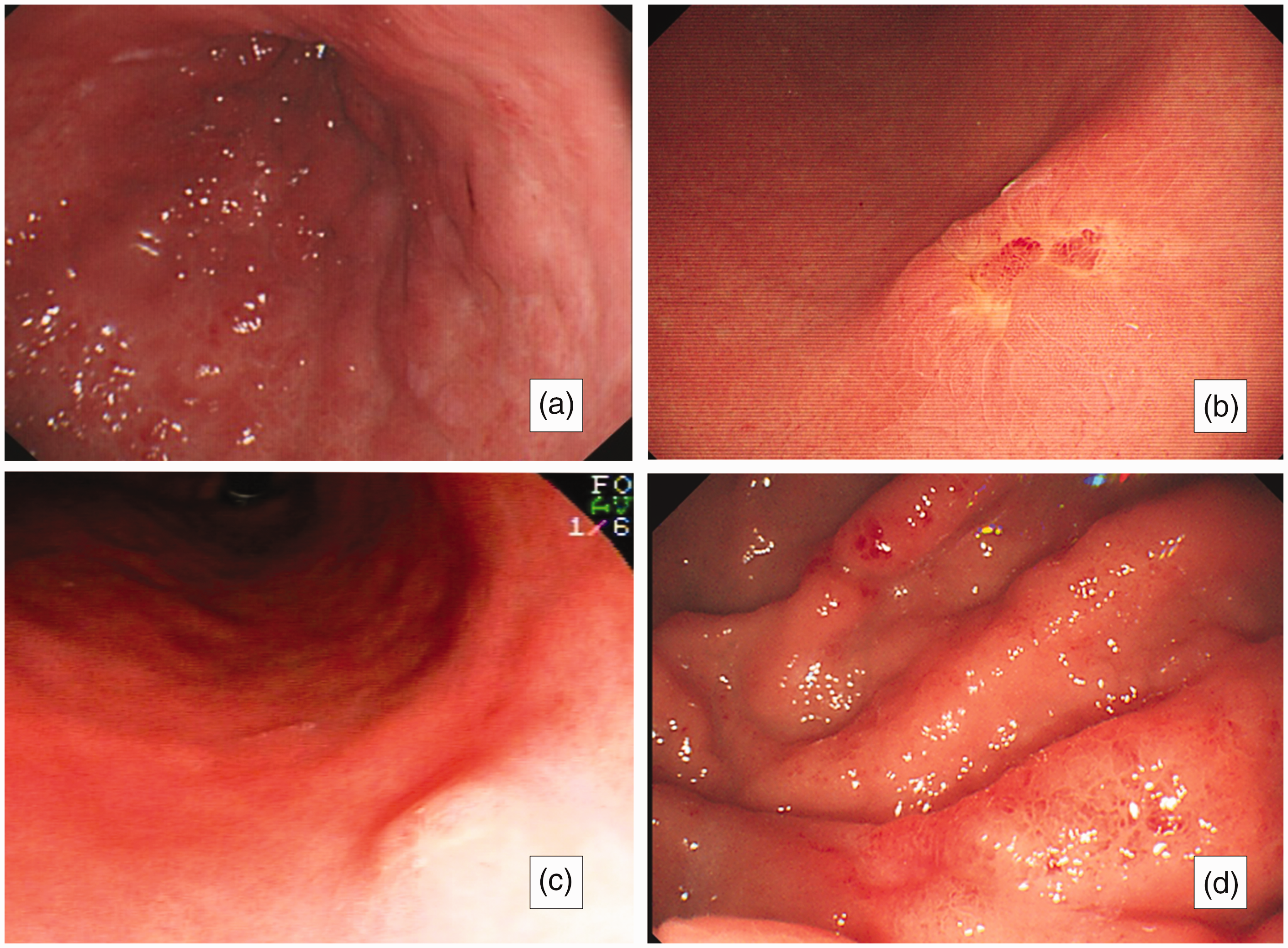
Endoscopic findings. (a) In Patient 2, the entire gastric mucosa is diffusely infiltrated with a nodular appearance and stiffness. (b) Mucosal congestion and erosion can be seen in Patient 3. (c) A congested nodule was found in Patient 4 and (d) Patient 5 shows endoscopic findings similar to those of mucosal-associated lymphoid tissue, with gastric mucosal congestion and edema, widened wrinkles, and scattered erosions.
Histology and immunohistochemistry
Figure 3 shows gastric tissue samples stained with hematoxylin and eosin of two patients (Patients 2 and 5). Signet-ring cells were observed in Patient 2. The gastric tissue was infiltrated by non-cohesive small tumor cells with an occasional intracytoplasmic lumen arranged in linear cords. This appearance suggested diffuse gastric cancer. After the disease progressed, the diagnosis was confirmed by an ascites examination, and immunohistochemistry of the gastric biopsy tissue was rechecked to correct the diagnosis. In Patient 5, the tumor cells showed diffuse growth in the tissue, and the patient was diagnosed with extranodal marginal zone lymphoma of MALT combined with the endoscopic findings. This diagnosis was then corrected on the basis of the immunohistochemical findings.

Histological pathology. (a) Gastric tissue sample stained by hematoxylin and eosin in Patient 2 (20 ×). Signet-ring cells can be seen. The gastric tissue is infiltrated by non-cohesive small tumor cells with an occasional intracytoplasmic lumen arranged in linear cords and (b) Gastric tissue sample stained by hematoxylin and eosin in Patient 5 (20 ×). The tumor cells show diffuse growth.
The immunohistochemical results are shown in Table 2. Hormonal receptors were highly expressed in primary and metastatic lesions in four patients (Patients 1–3 and 5), and they were immunonegative in Patient 4. Human epidermal growth factor receptor 2 (HER-2) was negative in all patients. Gross cystic disease fluid protein-15 (GCDFP-15) was negative in Patients 1 and 2, but it has not been commonly used in recent years owing to its low sensitivity. In contrast, mammaglobin and GATA3 are mainly used to identify tumors of breast origin. Mammaglobin and GATA3 were positive in the tested tissue and thus assisted in the patients’ diagnosis.
Comparison of immunohistochemistry between primary and metastatic tissues.

ER, estrogen receptor; N, negative; PR, progesterone receptor; HER-2, human epidermal growth factor receptor 2; P, positive; GCDFP-15, gross cystic disease fluid protein-15; –, not available.
Treatment
Chemotherapy was used as the first-line treatment for all patients, and Patient 4 also had immunotherapy. Except for Patient 4, whose hormonal receptors were negative, endocrine therapy was used as a back-line treatment. Only two patients received local treatment for the metastasis site. Patient 1 underwent a radical total gastrectomy because she was misdiagnosed with primary gastric cancer, and Patient 3 received regular hyperthermic intraperitoneal chemotherapy in addition to systemic treatment.
Patient 4 was diagnosed with gastric and adrenal metastasis 1.5 years after breast cancer surgery, and died of brain metastasis 6 months later. The survival of Patient 1 is unknown, and the remaining patients are still under treatment. The follow-up time after the diagnosis of gastric metastasis ranged from 6 to 50 months. We have de-identified all patients’ details. The reporting of this study conforms to the STROBE guidelines. 12
Discussion and conclusion
Gastric metastasis from breast cancer has often been overlooked by doctors in the clinic. In our study, the incidence of gastric metastasis of breast cancer was 0.04%. Hong et al. reported an incidence of gastric metastasis of breast cancer (0.06%, 13/21,716 cases) similar to that in our center. 4 The actual incidence of gastric metastasis is higher than previously realized because it may be mistaken for other diagnoses (e.g., treatment side effects, inflammatory lesions, and primary tumors). In this report, we discuss the possible reasons for this discrepancy in the incidence of gastric metastasis and provide some feasible suggestions.
Clinical characteristics
Distant metastasis may be found after many years since the primary diagnosis of breast cancer. In the present study, the median time between the primary diagnosis of breast cancer and initial gastric metastasis was 36 months (range: 0–144 months). In one patient, gastric metastasis was found at the same time as the breast cancer diagnosis.
The symptoms of gastric metastasis are diverse. Some patients may be asymptomatic and gastric metastasis is only found in examinations, and some patients show non-specific gastrointestinal symptoms, presenting with anorexia, epigastric pain, indigestion, and nausea.7–8 Liang et al. reported that 76% of patients experienced abdominal pain, which is the most common symptom of gastric metastases. 9 In our study, all patients experienced varying degrees of abdominal pain. There are also some unusual presentations, such as gastric outlet obstruction and gastric perforation, which are pathologically confirmed as gastric metastasis from breast cancer.10,11
In our study, one patient was misdiagnosed as having primary gastric cancer and was treated for up to 1.5 years, and two other patients underwent abdominal surgical exploration before the correct diagnosis.
Endoscopic evaluation
Most breast cancer metastases are diagnosed through routine examinations. Therefore, choosing an appropriate inspection method is crucial. Usually, commonly used examination methods are ultrasound and computed tomography. If there is any suspicion of stomach disease, gastroscopy should be performed.
Endoscopy is non-specific and gastric metastasis is difficult to distinguish from primary tumors. Gastric metastasis is usually confined to the submucosa and muscular layer of the stomach, and endoscopic evaluation can only show discrete mucosal abnormalities, leading to a higher rate of a false negative biopsy. 7 In a series of endoscopic examinations from 1977 to 1997, 10/51 patients with gastric metastases were pathologically negative on a biopsy. 6 Deep biopsies should be performed using endoscopy or endoscopic ultrasound-guided biopsies if necessary. 10
In our study, the endoscopic findings were varied. Some patients (Patients 1, 3, and 4) only showed a nodule or abnormalities of part of the mucosa. However, in Patient 2, the entire gastric mucosa was diffusely infiltrated, and showed a nodular appearance and stiffness. Patient 5 had endoscopic findings similar to extranodal marginal zone lymphoma of MALT, which has not been previously reported.
Pathology and pathogenesis
ILC has higher potential to metastasize to the gastrointestinal tract than IDC.3,6,13 Studies have shown that a lack of E-cadherin leads to an irregular and aggressive morphology of ILC, resulting in a loss of adhesive properties and a gain in cell mobility, thereby improving invasiveness. 13 In addition, ILCs may produce a signet-ring morphology, which is easily confused with primary gastric carcinoma. 14
Taal et al. showed that 83% of patients with gastric metastasis had the lobular subtype. 5 The majority of gastric tumors were positive for hormonal receptors and HER-2-negative (estrogen receptor [ER], 94.0%; progesterone receptor [PR], 68.3%; HER-2, 5.9%). 10 In our study, only one patient had IDC, and immunohistochemistry was triple-negative (ER, PR, and HER-2). The other four patients had ILC, and were hormone receptor-positive and HER-2-negative.
Some other mechanisms of gastric metastasis have also been discussed in the literature. A study on prostate cancer showed a relationship between inflammation and tumorigenesis, and showed that inflammation induced chemokines and relative receptors, which may be involved in attracting tumor cells. 15 Heliobacter pylori infection can cause chronic active gastritis, increasing the expression of chemokines. There is a hypothesis that the chronic inflammation caused by H. pylori attracts tumor cells to the stomach, 16 but further research needs to be conducted to confirm this possibility.
Immunohistochemistry
As mentioned above, relying solely on clinical symptoms, endoscopy, and pathological biopsies is insufficient to distinguish primary and metastatic gastric tumors. In our cases, three of five patients were initially pathologically misdiagnosed as gastric carcinoma, and then breast-derived gastric metastasis was confirmed by immunohistochemistry. Therefore, we strongly recommended that all biopsy tissues under endoscopy should be taken to have immunohistochemistry performed in similar patients.
The determination of hormone expression is necessary in diagnosing breast cancer. The majority of gastric metastasis from breast cancer is positive for ER and PR (80%), but they are also positive in 32% (ER) and 12% (PR) of patients with primary gastric carcinoma.2,17 Lobular breast cancer is usually negative for HER-2 expression. Therefore, HER-2 can be used as a reference indicator to help distinguish primary gastric cancer from metastatic cancer.
CK20 is particularly positive in gastrointestinal cancer, pancreatic cancer, and transitional cell carcinoma, but it has not been discovered in breast cancer. 18 In contrast, mammaglobin, GATA3, and GCDFP-15 are breast-specific antigens. GATA3 shows high sensitivity to lymph node metastasis and distant metastasis, and is superior to mammaglobin and GCDFP-15 in identifying primary and metastatic breast cancer 19 Research has shown that, when combined with hepatocyte nuclear factor 4 alpha, the sensitivity of the ER-PR-GCDFP-15 panel can be increased from 52% to 100.0%. 20
In our study, hormonal receptors were highly expressed in four patients with primary and metastatic lesions, and negative in one patient. HER-2 was negative in all patients. Mammaglobin and GATA3 were all positive in the tested tissue, thus assisting the patients’ diagnosis.
Treatment
Based on immunohistochemistry of primary and metastatic lesions, systemic therapy (e.g., chemotherapy, endocrine therapy, targeted therapy, and immunotherapy) is the main treatment for most metastatic cases. Additionally, the individual’s overall medical condition and previous treatment should be considered.
Surgical removal of the stomach has no significant effect on survival according to various published studies.5,21 However, some studies showed that, in selected patients with metastasis only to the gastrointestinal tract, surgical intervention tended to have an advantage in their prognosis.3,22 Surgery may also play an important role in palliative care to relieve symptoms.2,11 In some cases, when bleeding, obstruction, or perforation occurs, surgery or endoscopic interventional treatment should be performed. In our study, all patients received systemic therapy as the frontline treatment after diagnosis. On this basis, the third patient received hyperthermic intraperitoneal chemotherapy at the same time because of multiple metastasis of the abdominal cavity.
In summary, gastric metastasis from breast cancer has a high rate of misdiagnosis and missed diagnosis. The clinical symptoms of patients with gastric metastasis are often atypical. Some of these patients may not have symptoms and only show non-specific gastrointestinal symptoms during an examination. Multiple biopsies and immunohistochemistry of suspicious areas may improve the diagnostic accuracy of gastric metastasis from breast cancer. In addition, the awareness of diagnosis and treatment by clinicians should be improved, and a multi-disciplinary team should be recommended to reduce improper treatment caused by misdiagnosis.
Footnotes
Acknowledgements
We thank all professionals who treated the patients and helped formulate the final diagnoses.
Availability of data and materials
All data generated or analyzed are included within the article.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Informed consent for treatment was obtained from all individual participants included in the study. All procedures were supervised and approved by the Ethics Committee of the Fourth Hospital of Hebei Medical University (ID: 2021YK148). Written informed consent was obtained from the patients for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor of this journal.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
