Abstract
Background:
There is insufficient evidence regarding the management of hematochezia after colorectal endoscopic mucosal resection (EMR) without endoscopic hemostasis.
Objectives:
The aim of this study was to develop an observable score for hematochezia after colorectal EMR.
Design:
Retrospective study.
Methods:
This retrospective study included three hospitals in Japan. During the study period, colorectal EMR was performed in 3989 patients (11,414 lesions). Post-EMR hematochezia (delayed bleeding (DB)) was observed in 169 patients (512 lesions). Of these, 47 patients (150 lesions) were classified into the Hemostasis Group, comprising those who underwent endoscopic hemostasis. The remaining 122 (362 lesions) were classified into the non-hemostasis group, comprising those who underwent endoscopy without hemostasis, received preventive hemostasis, and did not undergo emergency endoscopy. Weighted points were assigned to predict observable cases following colorectal EMR through multivariate logistic regression analysis, enabling the development of a predictive model.
Results:
The prediction model comprised three variables (male gender, American Society of Anesthesiologists Physical Status 3, direct oral anticoagulant). According to the definition, the total score was categorized as lowly observable (2 or 3 points) and highly observable (0 or 1 points) for DB after colorectal EMR. As a result, the rates of observable cases for each risk category were 45.7% and 81.8%, respectively. The model showed good discrimination ability from the c-statistic (95% CI) of 0.71 (0.63–0.79).
Conclusion:
Although further prospective studies are necessary to validate the utility of the score, it may be useful in clinical practice.
Keywords
Introduction
According to 2020 data, mortality from colorectal cancer (CRC) ranks second among all cancers. 1 The detection and removal of adenoma lesions in the colon reduce the incidence and mortality of CRC by 53%.2,3
Among colorectal endoscopic resection (ER), endoscopic mucosal resection (EMR) is widely used worldwide. For example, according to the European Society of Gastrointestinal Endoscopy (ESGE) guidelines, EMR is generally recommended for non-submucosal (SM) invasive lesions ⩾20 mm or lesions suspected to have superficial SM invasion. 4 The American Society for Gastrointestinal Endoscopy (ASGE) guidelines recommend EMR for non-polypoid or suspected serrated lesions when the lesion diameter is 10–19 mm without SM invasion, non-SM invasive lesions ⩾20 mm, and lesions suspected to have a minimal or moderate risk of SM invasion. 5 According to the Japan Gastroenterological Endoscopy Society (JGES) guidelines, there are no specific size-based guidelines for EMR, 6 and EMR is also widely performed for intermediate-sized polyps measuring <20 mm as well.
Among the adverse events of EMR, delayed bleeding (DB) is a relatively frequent complication, ranging from 1.4% to 6.3%.7–11 The risk factors of DB after colorectal EMR include antithrombotic medications, polyp size, sessile serrated lesion, diabetes mellitus, pedunculated polyp, and involvement of the right-sided colon.9–15 In addition to EMR, direct oral anticoagulants (DOAC) and male gender are also considered risk factors for DB after colorectal endoscopic submucosal dissection (ESD) and other types of gastrointestinal (GI) bleeding, such as upper and lower GI bleeding.16–22 On the contrary, there is limited evidence to guide which cases of post-colorectal EMR bleeding (hematochezia), particularly in patients with intermediate-sized polyps, can be safely managed without endoscopic hemostasis. In daily clinical practice, appropriate assessment of cases that are amenable to follow-up helps avoid unnecessary invasion and excessive medical costs. Therefore, the development of an “observable score”—a clinical tool to identify patients who can be safely managed with observation alone, without the need for endoscopic evaluation and without compromising overall clinical condition—following hematochezia after colonic EMR is of critical importance. Notably, previous studies from Europe and the United States have predominantly focused on colorectal DB involving lesions ⩾20 mm,23–27 likely reflecting the focus of ESGE and ASGE guidelines on this lesion size for EMR. In contrast, EMR application to lesions <20 mm has been relatively limited in these guidelines. Furthermore, while colonoscopy is effective for managing DB, achieving hemostasis in approximately 1 out of every 4.5 procedures, 28 expectant management aimed at spontaneous resolution of bleeding is a valid alternative in selected cases. This underscores the clinical value of developing a simple, practical scoring model focused primarily on patient factors, which can guide clinicians in safely observing patients after hematochezia post-EMR and reduce unnecessary interventions.
Thus, the aim of this study was to develop an observable score for hematochezia after colorectal EMR.
Materials and methods
Patients
This multicenter retrospective study was conducted at three hospitals in Japan. Consecutive patients who underwent colorectal EMR from January 2018 to December 2023 were extracted. Patients who underwent other ER procedures at the same time or who underwent EMR for SM tumors were excluded. Among these patients, the “DB group” included patients who experienced hematochezia after EMR. DB was evaluated up to 28 days after EMR according to our previous report. 29 Among the DB group, the “Hemostasis group (H group)” included patients who underwent endoscopic hemostasis (there was a post-EMR ulcer that showed exposed vessels or active bleeding). Conversely, the “Non-H group” included patients who had undergone endoscopy without hemostasis, undergone preventive hemostasis (there was a post-EMR ulcer that had no exposed vessels or active bleeding), and who had not undergone emergency endoscopy. Patient and lesion characteristics were extracted from the medical records of each hospital. This study was approved by the Medical Ethics Board of Chiba University Graduate School of Medicine (protocol number: 3593), the Medical Ethics Board of Seikei-kai Chiba Medical Center ((protocol number: D-2023-001), and the Medical Ethics Board of Chiba Municipal Aoba Hospital (protocol number: 20231101) and was conducted in accordance with the Declaration of Helsinki. This was a retrospective cohort study, and written informed consent was not obtained. Informed consent was waived by each participating hospital via the opt-out method. The medical ethics boards of the other collaborating institutions at their respective facilities also approved this study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Delayed bleeding
In this study, hematochezia was assessed based on patient reports, specifically when the toilet water appeared red due to blood or when visible blood was present in the stool. For hospitalized patients, nurses confirmed the presence of blood when necessary. In all facilities, when hematochezia was observed, patients were instructed to contact the attending physician (if it occurs after discharge or for outpatient EMR, mainly by phone). Additionally, patients were scheduled to return for an explanation of pathological results within 28 days after EMR, during which the presence of hematochezia was assessed. If hematochezia occurred, the attending physician decided to perform emergency endoscopy. The hematochezia details (duration, time of onset after EMR, and the number) were assessed. The day of EMR was set as day 0. The number of days of persistent hematochezia was counted until the day of hemostasis, if hemostasis was performed. Rebleeding after hemostasis was not counted because this study focused on whether hemostasis was required, rather than the number of hemostasis attempts made.
Procedures
During EMR, the localization, morphology, diameter of the lesions, and the number of EMR-treated lesions per patient were evaluated. The segment from the cecum to the transverse colon is referred to as the right colon, while the remaining part is considered the left colon. Is or Ip tumors were classified as polypoid, whereas others were considered non-polypoid according to the Paris classification. 30 For the EMR procedure, saline and/or 10% glycerol solution mixed with indigo carmine was injected into the submucosa. The lesions were resected with a snare until there were no visually detectable remnants. Prophylactic clipping after EMR was performed at the discretion of the endoscopist. For snaring, a 10–33 mm electrocautery snare (SnareMaster or SnareMaster Plus; Olympus Medical Systems, Tokyo, Japan. Captivator; Boston Scientific, USA. Dualoop; Medico’s Hirata Inc., Osaka, Japan. AGS polypectomy snare; AGS MedTech, Hangzhou, China) was used. No additional treatments were performed beyond snaring. Pre- and postoperative antithrombotic medications were administered according to the JGES guidelines.31,32
Endoscopic hemostasis at the time of DB
Endoscopists generally performed hemostasis when ulcers showed exposed vessels or active bleeding. Hemostasis was achieved in all cases using clips. The same type of clip (EZ Clip; Olympus Medical Systems) was used in all cases for hemostasis. No other hemostatic methods, such as SM injection or thermal coagulation, were performed.
Pathological evaluation of the resected specimen
All tissue specimens underwent histological analysis with standard hematoxylin-eosin staining and were independently evaluated by pathologists at their respective institutions. Each pathologist made histopathological diagnoses according to Japanese diagnostic standards. In this context, cancer refers to both intramucosal carcinoma and SM invasive cancer.
Factors considered related to the observation of DB after colorectal EMR
Factors potentially associated with DB after colorectal EMR were mainly identified based on existing literature on colorectal EMR.6,29,33–47 These included patient characteristics (age, sex, American Society of Anesthesiologists Physical Status (ASA-PS) classification, the number of patients with multiple EMR treated lesions), comorbidities (diabetes mellitus, liver cirrhosis, hemodialysis), medication history (antithrombotic therapy, nonsteroidal anti-inflammatory drugs (NSAIDs), steroids), use of prophylactic clips, and characteristics of hematochezia (duration, time of onset after EMR, and frequency).
Endpoint
The primary endpoint was to develop an observable score for DB after colorectal EMR.
Statistical methods
Since this was a retrospective study, no sample size calculation was performed in advance. However, the number of cases was confirmed to be sufficient for multivariate analysis. In the patient and lesion characteristic analysis, continuous variables were summarized as medians and compared using the Wilcoxon rank-sum test. Categorical variables were summarized in terms of n (%) and were compared using Fisher’s exact test. p-Values <0.05 were considered to be significantly different. Time of onset after EMR was unknown in two cases. Furthermore, the number of episodes of hematochezia was unknown in nine cases. We conducted both univariate and multivariate analyses by comparing the H group and the NH group among all EMRs performed across the three hospitals. The univariate analysis was performed for the number of cases in each category. In the univariate analysis, each covariate was dichotomized, p-values were calculated using Fisher’s exact test, and odd ratios were estimated using logistic regression. The multivariate analysis model included covariates with p-values of 0.1 or less. This study aims to predict the likelihood of directly observing hematochezia by analyzing factors present before bleeding occurs. Since blood transfusions in this study were administered after the occurrence of hematochezia, they were excluded from the univariate analysis. Furthermore, it is not possible to completely identify the lesion responsible for the bleeding because not all cases of DB underwent endoscopy. Accordingly, lesion factors such as location, morphology, diameter, etc. were not included in univariate analysis. Multivariate logistic regression analysis was performed on 167 cases with complete data, excluding two cases in which the time of onset after EMR was unknown. Both univariate and multivariate logistic regression analyses were performed to calculate odds ratios with the observable as the outcome. Multicollinearity between covariates was assessed using the variance inflation factor (VIF). For the covariates that resulted in p-values of 0.1 or less, we assigned weighted points proportional to the β regression coefficient values, following an approach similar to a previous study. 48 To ensure that higher scores correspond to higher risk (lower observable values), the regression coefficients of each covariate were multiplied by 2, rounded to the nearest integer, and used as the factor for the prediction score. The total prediction score was calculated as the sum of each factor. Based on the score, the receiver operating characteristics (ROC) curve and c-statistic were calculated to evaluate the overall discriminative ability of the model. Additionally, the cutoff value was determined based on the geometrically most appropriate point on the ROC curve, and sensitivity and specificity were computed. The goodness of fit of the model was evaluated using Nagelkerke R2. All statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Study flow
During the specified period, colorectal EMR was performed on 3989 patients with 11,414 lesions across three facilities. Among these, DB was identified in 169 patients with 512 lesions. The H group included 47 patients with 150 lesions while non-H group included 122 patients with 362 lesions (Figure 1).
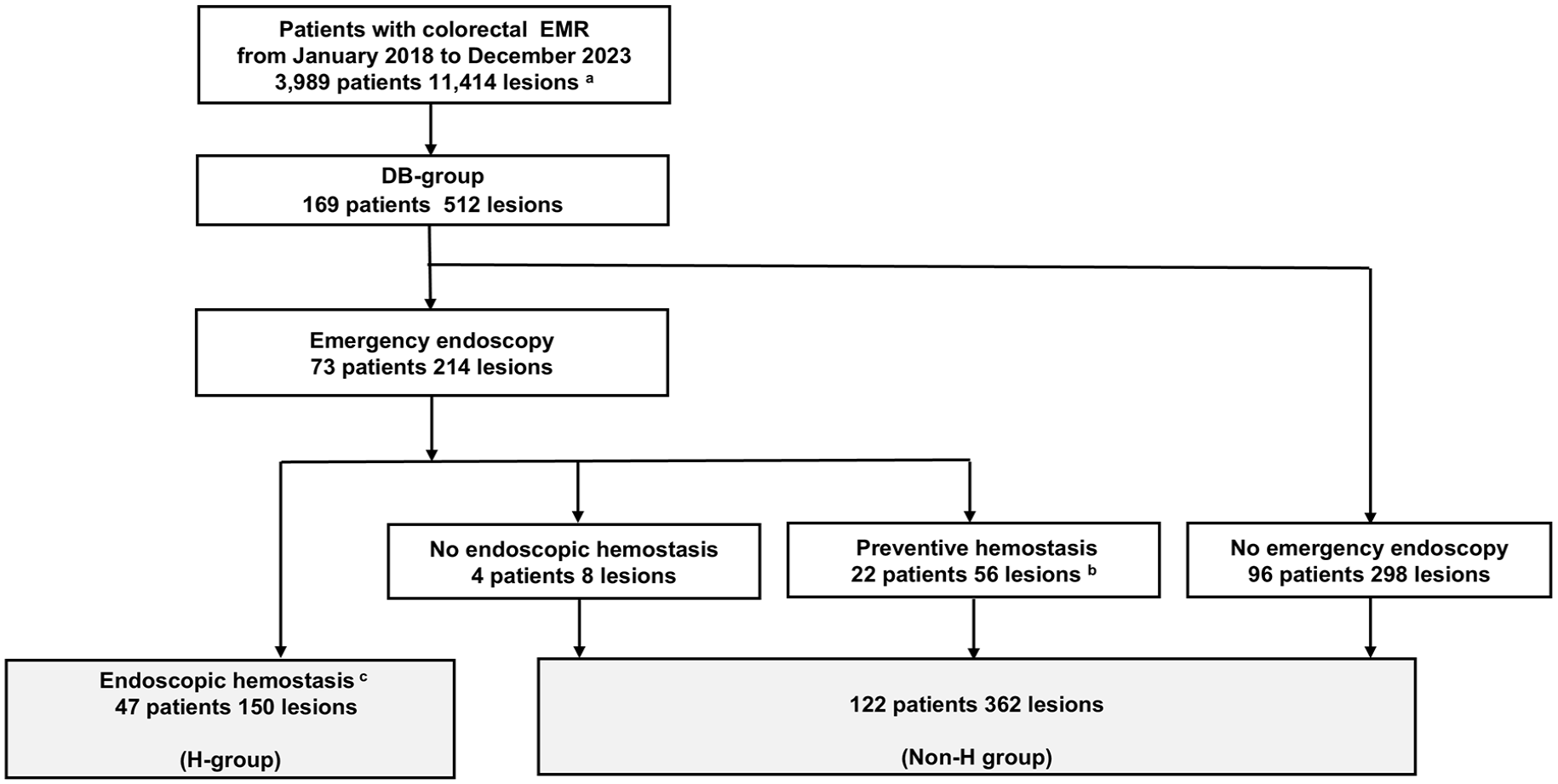
Study flow of the study.
Patient and lesion background
Since hematochezia never recurred after a gap of more than 1 day, all recurrences occurred either continuously or within 1 day. In most cases, prophylactic clips were used. In cases where clips were used, all lesions were clipped. Conversely, one patient in the non-H group did not receive a prophylactic clip, and the lesions in this patient were not clipped. Within the H group, two individuals required blood transfusions after being admitted post-EMR hematochezia, with one of them having hemodynamic instability. Significant differences were observed in several patient background factors (ASA-PS, hemodialysis, two antiplatelets, and hematochezia time of onset after EMR). As for lesion-related factors, the H group had a significantly higher prevalence of right colon involvement (Table 1).
Patient and lesion characteristics.

Aspirin, antiplatelets other than aspirin, DOAC, and warfarin are used as monotherapy.
In cases where clips were used, all lesions were clipped. Conversely, one patient in the non-H group did not receive a prophylactic clip, and all lesions in this patient were not clipped.
The cecum to the transverse colon is referred to as the right colon, whereas the remaining colon is the left colon.
Polypoid indicates the presence of Is or Ip polyps according to the Paris classification.
Cancer refers to both intramucosal carcinoma and submucosal invasive cancer.
The date of EMR was defined as day 0, and the days on which hematochezia began to appear is hematochezia time of onset. The analysis was conducted excluding the two cases with missing data.
The analysis was conducted excluding the nine cases with missing data.
ASA-PS, American Society of Anesthesiologists Physical Status classification; DOAC, direct oral anticoagulants; EMR, endoscopic mucosal resection; H group, hemostasis group; non-H group, non-hemostasis groupNSAIDs, nonsteroidal anti-inflammatory drugs.
Observational score for DB after colorectal EMR
We included six candidate predictors in the multivariate models. The VIFs of these six factors were satisfactory (Supplemental Table 1). The results of the univariate and multivariate analyses are presented in Table 2. In the multivariate model, three covariates were significant. Initially, each covariate was assigned a score of two points, resulting in equal scores across all covariates without weighting. Since using a single point for each covariate would not alter the overall score distribution, we chose this simplified scoring for ease of clinical application. Based on the adjusted β regression coefficients, integer scores were assigned to the selected predictors: one point for male gender; one point for ASA-PS3; one point for DOAC (Table 3). The observation score showed a c-statistic of 0.71 on the ROC curve, indicating a good overall discrimination ability (Supplemental Figure 1 and Supplemental Table 2). The cutoff point was determined as the point on the ROC curve closest to the (0, 1) coordinate, representing 100% sensitivity and 100% specificity. The resulting sensitivity was 82.5%, and specificity was 53.2%. Based on this cutoff, the total score was categorized as highly observable (0 or 1 point) or lowly observable (2 or 3 points) for DB after colorectal EMR. Thus, the rates of observable cases for each risk category were 81.8% and 45.7%, respectively (Table 4). The performance metrics of this predictive model were a Nagelkerke R² of 0.16, indicating fair performance (Supplemental Table 2).
Univariate and multivariate analyses of the predictive observable factors of DB.
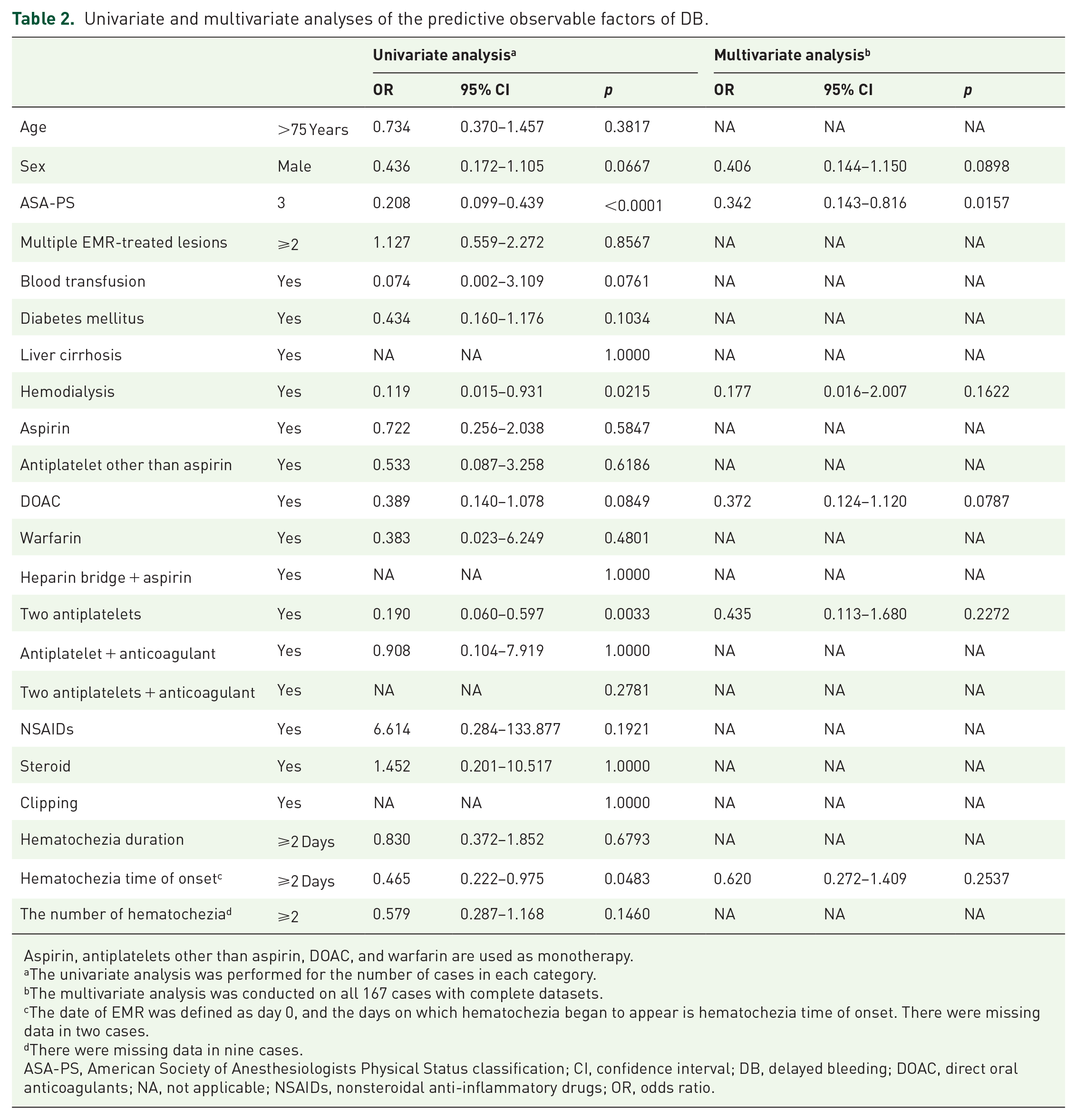
Aspirin, antiplatelets other than aspirin, DOAC, and warfarin are used as monotherapy.
The univariate analysis was performed for the number of cases in each category.
The multivariate analysis was conducted on all 167 cases with complete datasets.
The date of EMR was defined as day 0, and the days on which hematochezia began to appear is hematochezia time of onset. There were missing data in two cases.
There were missing data in nine cases.
ASA-PS, American Society of Anesthesiologists Physical Status classification; CI, confidence interval; DB, delayed bleeding; DOAC, direct oral anticoagulants; NA, not applicable; NSAIDs, nonsteroidal anti-inflammatory drugs; OR, odds ratio.
Multivariate logistic regression analysis of predictive observable factors for DB after colorectal EMR and scoring system.
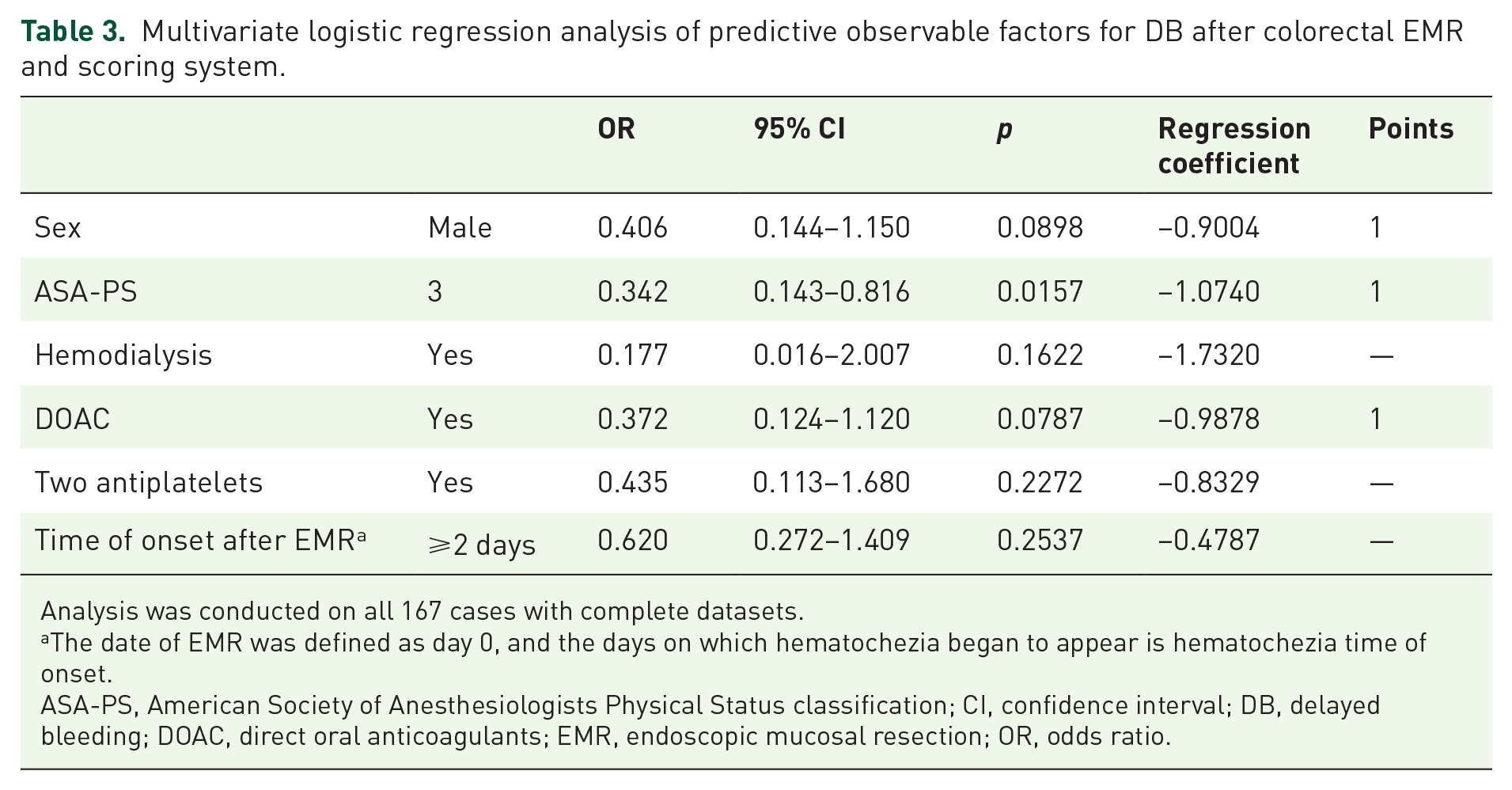
Analysis was conducted on all 167 cases with complete datasets.
The date of EMR was defined as day 0, and the days on which hematochezia began to appear is hematochezia time of onset.
ASA-PS, American Society of Anesthesiologists Physical Status classification; CI, confidence interval; DB, delayed bleeding; DOAC, direct oral anticoagulants; EMR, endoscopic mucosal resection; OR, odds ratio.
Distribution of risk scores and percentages of observable cases.
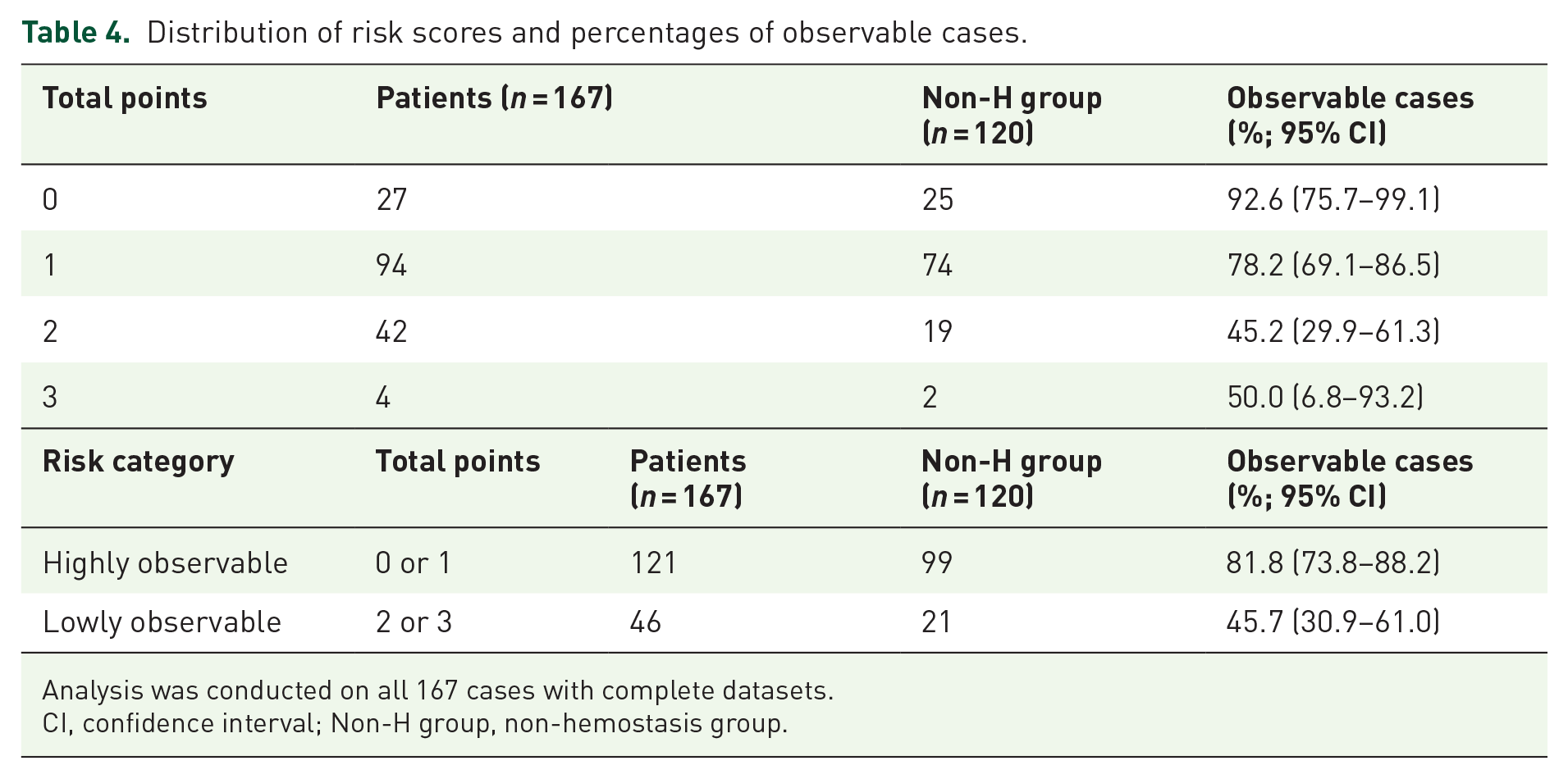
Analysis was conducted on all 167 cases with complete datasets.
CI, confidence interval; Non-H group, non-hemostasis group.
Case presentation
A typical example of the H group is shown in Figure 2. A man in his 70s underwent EMR for one lesion. Three days after the EMR, he required endoscopic hemostasis with clipping for bleeding polyps in the ascending colon. According to the scoring system, this case received one point for an ASA-PS score of 3, and one point for male gender, totaling two points (lowly observable). Typical examples of the non-H group are shown in Figure 3. A woman in her seventies underwent EMR for two lesions. One day after EMR she had hematochezia. Emergency endoscopy was performed; however, hemostasis was not performed for both polyps because there was no bleeding. This patient did not score on any scale, resulting in a total score of 0 (highly observable).
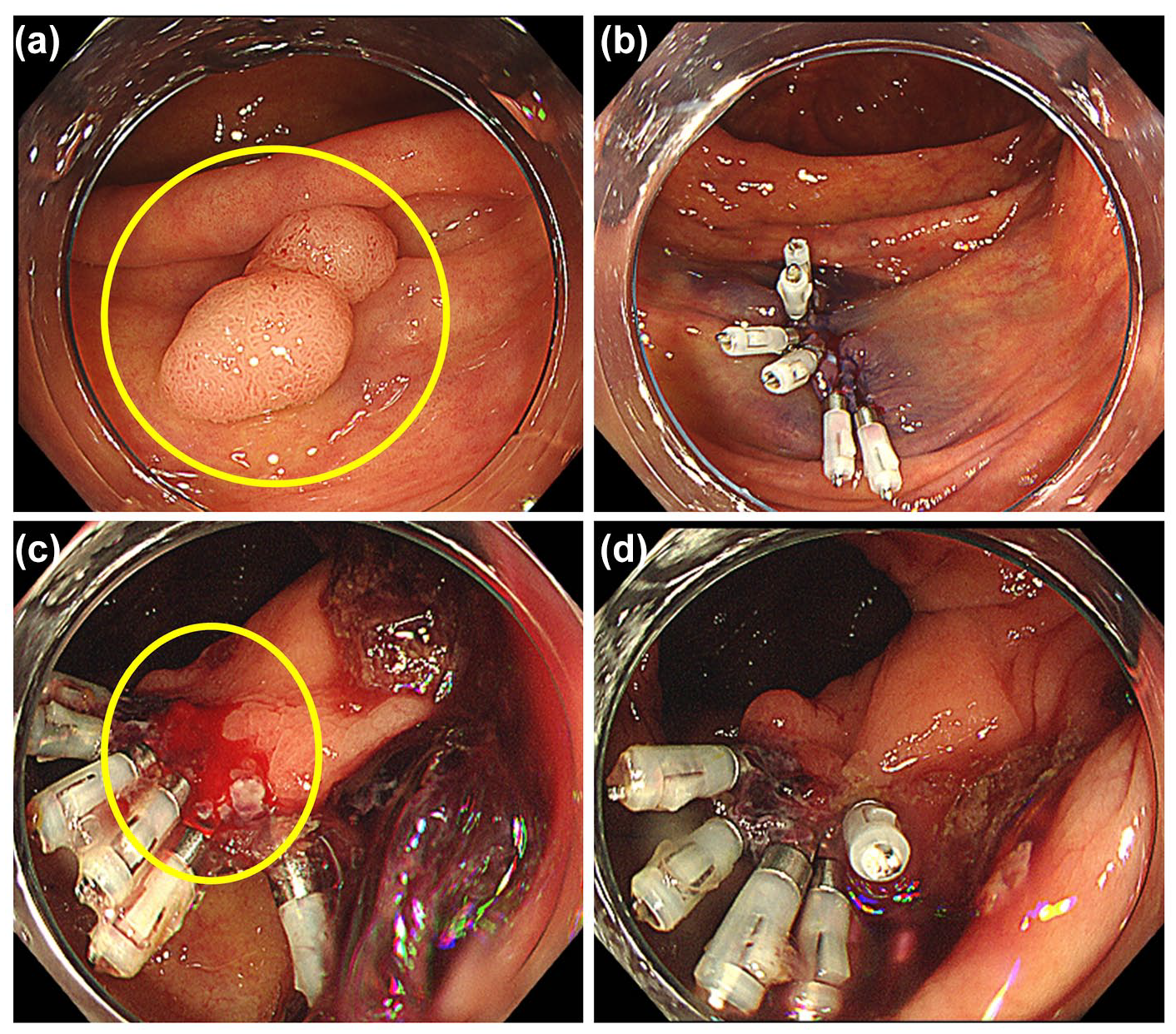
Case presentation of H group (male patient in his seventies). (a) 10 mm polyp in the ascending colon (yellow circle surrounds the lesion). (b) Post-EMR wound was clipped. Pathological result was adenoma. (c) He had a hematochezia 3 days after EMR. Emergency endoscopy and additional clip was performed (yellow circle surrounds the bleeding point). (d) Bleeding was stopped with additional clip.

Case presentation of non-H group (female patient in her seventies). (a) 7 mm polyp in the descending colon (yellow circle surrounds the lesion). (b) Post-EMR wound of descending colon was clipped. Pathological result was adenoma. (c) She had a hematochezia 1 day after EMR. Emergency endoscopy was performed but hemostasis was not performed for the descending colon polyp as there was no bleeding. (d) 7 mm in the sigmoid colon (yellow circle surrounds the lesion). (e) Post-EMR wound of sigmoid colon polyp was clipped. Pathological result was adenoma. (f) Hemostasis was not performed for the sigmoid colon polyp neither.
Discussion
This study establishes an observational score. It is a simple scoring model that focuses primarily on patient factors rather than lesion factors. While the predictive model demonstrated good performance based on its c-statistic, its relatively low specificity and a Nagelkerke R² of 0.16 may undermine its utility in clinical decision-making and thus should be interpreted with caution. The Birmingham Score has been reported to associate admission hemoglobin levels and male gender with adverse outcomes of lower gastrointestinal bleeding (LGIB). These adverse outcomes include blood transfusion, endoscopic intervention, computed tomography angiography, surgical intervention, rebleeding, and mortality. 49 In fact, mortality from LGIB bleeding was reported to be as high as 3.4%. 50 A comparison of these data and the few cases of blood transfusion in the H group (2/47, 4.3%) suggests that colorectal DB after EMR may generally be milder than typical LGIB. Conversely, there was a case in which emergency endoscopy was required, such as those with hemodynamic instability. In such cases where significant bleeding is clearly suspected, prompt intervention is necessary.
The factors identified in this study are presented next. In a prospective cohort study using polyps ⩾20 mm, interventions for hemostasis were associated with hourly or more frequent hematochezia, ASA grade 2 or higher, and blood transfusion. 24 In this study, where the median tumor diameter was smaller, ASA-PS 3 was identified as a risk factor of unsuitability for observation. Patients with poor overall condition may require hemostatic intervention even for colorectal EMR of small polyps. Our study did not include patients with an ASA score of ⩾4, which was considered reasonable, as EMR is rarely performed in such cases. Male gender has been identified as a risk factor for DB in therapeutic colonoscopy and gastric ESD.16,17 Although the reason why male gender was identified as a factor remains unclear, this study may be the first to identify male gender as a risk factor for unsuitability for observation after DB of colorectal EMR.
Regarding antithrombotic agents, previous reports have indicated that the use of DOAC is associated with a higher incidence of DB after colorectal EMR compared with the use of warfarin and antiplatelet agents. 15 In this study, DOAC was the only antithrombotic agents identified as contributing to non-observation factors, indicating that patients receiving DOAC may require endoscopic hemostasis in the event of DB. Conversely, the risk of Gl associated with DOAC administration showed varying results compared with that of warfarin administration. For instance, studies have shown that dabigatran, edoxaban, and rivaroxaban increase the risk of non-procedure-related GI bleeding compared to warfarin.18–20 In contrast, apixaban was reported not to increase the risk of GI bleeding. 21 A recent study on colorectal ESD reported that dabigatran was associated with a higher incidence of post procedural bleeding compared to other DOAC, 22 with a rate of 18.26%. In our study, the number of patients taking DOAC was small; thus, we did not analyze the risk according to type. Therefore, assessing the type-specific risk in patients receiving DOAC remains a challenge.
In univariate analysis, earlier onset of hematochezia was identified as a contributing factor for observation, although this association did not maintain statistical significance in the multivariable analysis. This trend aligns with findings from studies on colorectal ESD.29,44 As noted in our previous report, 29 early hematochezia typically results from intraoperative bleeding. This timing corresponds with the normal colonic transit time of 19–40 h in healthy adults, 51 during which stools are completely cleared from the digestive tract following EMR. However, severe hematochezia requiring hemostasis may develop later due to various factors, including stool irritation, intestinal peristalsis, or exposure of blood vessels from expanding post-EMR ulcers. 52
This study has several limitations. First, the timing of emergency endoscopy is not standardized. Second, in cases with multiple lesions, it is not possible to completely identify which lesion is responsible for the bleeding because not all cases of DB have undergone endoscopy. Accordingly, the evaluation of lesion factors such as location, morphology, lesion size, and histological type was not incorporated into the multivariate analysis, and their impact remains unclear. We recognize that this is a significant limitation and that lesion-level analysis should be addressed in future prospective studies. Third, caution should be taken because only a few factors clearly indicate risks, such as the use of heparin bridge. Fourth, the lack of internal validation raises concerns about possible overfitting and the reproducibility of the model. As such, there is a risk of overfitting, and the reproducibility and generalizability of the model should be interpreted with caution. Future studies with prospective design and internal (or external) validation are warranted. Fifth, management of antithrombotic medications was performed according to the JGES guidelines. However, even within these guidelines, there is variability in the duration of drug discontinuation and bridging therapy, which are not fully standardized. Sixth, since this was a retrospective study, even though some criteria for prophylactic hemostasis may have been predefined, their application could have been inconsistent in actual clinical practice.
Conclusion
This study established an observable score for bleeding after colorectal EMR using retrospective data from multiple institutions. Although further prospective studies are necessary to validate its true utility, this score may be useful in clinical practice.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251356112 – Supplemental material for Observable score for bleeding after colorectal endoscopic mucosal resection
Supplemental material, sj-docx-1-tag-10.1177_17562848251356112 for Observable score for bleeding after colorectal endoscopic mucosal resection by Hayato Nakazawa, Kenichiro Okimoto, Tomoaki Matsumura, Masayuki Yokoyama, Tsubasa Ishikawa, Yoshihiro Fukuda, Yoshio Kitsukawa, Akane Kurosugi, Michiko Sonoda, Tatsuya Kaneko, Yuki Ohta, Takashi Taida, Keisuke Matsusaka, Jun-ichiro Ikeda and Jun Kato in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251356112 – Supplemental material for Observable score for bleeding after colorectal endoscopic mucosal resection
Supplemental material, sj-docx-2-tag-10.1177_17562848251356112 for Observable score for bleeding after colorectal endoscopic mucosal resection by Hayato Nakazawa, Kenichiro Okimoto, Tomoaki Matsumura, Masayuki Yokoyama, Tsubasa Ishikawa, Yoshihiro Fukuda, Yoshio Kitsukawa, Akane Kurosugi, Michiko Sonoda, Tatsuya Kaneko, Yuki Ohta, Takashi Taida, Keisuke Matsusaka, Jun-ichiro Ikeda and Jun Kato in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-3-tag-10.1177_17562848251356112 – Supplemental material for Observable score for bleeding after colorectal endoscopic mucosal resection
Supplemental material, sj-tif-3-tag-10.1177_17562848251356112 for Observable score for bleeding after colorectal endoscopic mucosal resection by Hayato Nakazawa, Kenichiro Okimoto, Tomoaki Matsumura, Masayuki Yokoyama, Tsubasa Ishikawa, Yoshihiro Fukuda, Yoshio Kitsukawa, Akane Kurosugi, Michiko Sonoda, Tatsuya Kaneko, Yuki Ohta, Takashi Taida, Keisuke Matsusaka, Jun-ichiro Ikeda and Jun Kato in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Author’s note
We confirm that all figures in the submitted manuscript are original and created by the authors. No material requiring copyright permission has been reused.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
