Abstract
Non-variceal upper gastrointestinal bleeding (NVUGIB) is a common type of upper gastrointestinal emergency with high risk of life-threatening conditions. Nowadays some standard guidelines for the treatments of NVUGIB have been established based on endoscopic therapies, including injection of glues or sclerosing agents, coiling, band ligation, and thermal therapies. Nevertheless, some patients are refractory to standard endoscopic treatments. We have noticed that endoscopic ultrasound (EUS)–guided treatments have been performed by some endoscopists in certain conditions. This review aims to evaluate the role of EUS-guided treatments in the management of NVUGIB. We performed a MEDLINE/PubMed search, and relevant studies were extracted and examined. According to the existing literature, the most common lesions were Dieulafoy’s lesion, pancreatic pseudoaneurysms, and gastrointestinal stromal tumors. EUS-guided treatments mainly include injection, mechanical therapies, and combined therapies, and seem to be a promising technique in the management of NVUGIB, especially for refractory bleeding.
Introduction
Non-variceal upper gastrointestinal bleeding (NVUGIB) is defined as any gastrointestinal bleeding caused by non-variceal diseases of the gastrointestinal tract above the ligament of Treitz, which includes the esophagus, stomach, duodenum, pancreatic and bile duct, and the anastomotic site after gastrojejunostomy. 1 Although the annual incidence decreased from 78 to 61 per 100,000 persons from 2001 to 2009, 30-day mortality remains up to 11%. 2 Peptic ulcer is the most common cause of NVUGIB, accounting for 20–67% of cases. 3 Erosive esophagitis, Dieulafoy’s lesion, Mallory–Weiss syndrome, gastric antral vascular ectasia, and neoplasms are other causes of NVUGIB.
The well-established treatments for NVUGIB include acid-suppressive drugs, endoscopic therapy (ET), and radiological and surgical approaches. 4 In recent years, the widespread use of ET has reduced the risk of rebleeding, surgical rate, and mortality in NVUGIB. 5 Besides, emerging novel endoscopic therapies have played important roles in achieving hemostasis, including over-the-scope-clips (OTSC), endoscopic suturing, coagulation grasper, and endoscopic ultrasound (EUS)–guided treatment.4,6–9 Nevertheless, refractory bleeding is still a tough condition for many endoscopists. Moreover, ET seems to be not effective enough in certain lesion. The rebleeding rates in patients who underwent ET for gastric cancer were up to 30–40%, 10 and high rebleeding rate was associated with a poor prognosis.11,12 In this case, EUS-guided treatment could serve as a salvage option for failed or not eligible standard therapy.
EUS has rapidly expanded from diagnostic modality to therapeutic technique in different directions because the emerging new accessories have broadened the use in recent years.13,14 It offers access to arterial and venous vasculature of abdomen and mediastinum with real-time image, which makes it possible to perform targeted therapeutic procedures to abnormal vessels. 15 Besides, Doppler ultrasound (US) endoscope can be used to detect the arterial blood flow and predict the risk of recurrent bleeding before and after hemostasis procedures. We notice that EUS has been great application and has positive clinical impact on the treatment of gastroesophageal varices.16,17
Nevertheless, researches about EUS-guided treatment in NVUGIB remain scarce, and the role in this direction still lacks the support of clinical comparative research, especially in cases of refractory gastrointestinal bleeding.18,19 This article aims to summarize current literature regarding EUS-guided interventions in NVUGIB as well as to discuss the specific procedures, the effectiveness, and possible clinical outcomes of EUS-guided management.
Methods
We performed a MEDLINE/PubMed search to identify the peer-reviewed original and review articles using keywords of EUS, NVUGIB, and hemostasis. The references of pertinent studies were manually searched to identify additional relevant studies. A total of 20 articles were found, including 15 case reports and 5 case series.20–39 The identifications of patients (including type of lesion, type of bleeding, and previous treatments), details of procedures (including specific type of treatment, type of needle), and main outcomes (including technical and clinical outcomes, and complications) for each study were reviewed. A total of 44 patients were induced and divided into different classifications according to the treatments.
Results
Injection therapy
EUS-guided cyanoacrylate injection
Overview. EUS-guided cyanoacrylate injection is an emerging endoscopic technology mostly used for variceal gastrointestinal bleeding. 16 Cyanoacrylate polymerizes when contacting with blood, then the vascular obstruction formed which can be confirmed by Doppler probe. The safety of cyanoacrylate is highly concerned because of the risk of systemic embolization, which can be reduced through the direct and low amount of glue injection under the guidance of EUS.40,41 In this setting, EUS-guided cyanoacrylate injection is superior to traditional endoscopic glue injection. In various cases, cyanoacrylate is mixed with lipiodol for the purpose of fluoroscopic monitoring and dilution of cyanoacrylate to prevent early hardening, which may cause adherence to endoscopy channel.42–44
Patients. For included cases, EUS-guided cyanoacrylate injection was performed in 16 cases (Table 1).25,31–33,35,36 The types of lesions included gastrointestinal stromal tumor (GIST; five cases), duodenal ulcer (two cases), Dieulafoy’s lesion (one case), pancreatic pseudoaneurysm (one case), splenic artery pseudoaneurysm (one case), gastroduodenal artery pseudoaneurysm (one case), left gastric artery pseudoaneurysm (one case), gastroduodenal artery bleeding due to a pancreatic tumor (one case), arterial anomaly of the gastric fundus (one case), duodenal metastasis (one case), and esophageal cancer (one case). There were 10 patients (62.5%) who had endured endoscopic hemostasis before the EUS-guided cyanoacrylate injection. The other six patients (37.5%) received EUS-guided treatments for the first time due to the limitations of anatomical reasons and poor conditions of patients. It is worth mentioning that Gonzalez et al. 36 reported they performed EUS-guided cyanoacrylate injection on a patient who had arterial leak during EUS examination because of pancreatic pseudoaneurysm.
Summary of the studies reported on EUS-guided cyanoacrylate injection..

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; GISTs, gastrointestinal stromal tumors; IR, interventional radiology; ml, milliliters; NA, not applicable.
Needles and volumes. When injecting cyanoacrylate glue, the mostly chosen puncture points were the targeted vessels followed by direct injection. With regard to the selection of needles, the sizes of the needles used in these cases were 19G in seven cases (43.75%), 22G in eight cases (50%), and unspecified in one case (6.25%). And for the injection dose, as the risk of embolization increases with the injected volume, endoscopists always inject cyanoacrylate glue with the necessary minimum dose. 32 Volume injected in the induced cases varies from 1 to 7 ml, and the mean volume is 2.9 ml (except for unspecified 2 ampoules in one case). At the end of the procedure, Doppler was used to monitor the blood flow and ensure the disappearance of Doppler signal which indicates the end point of treatment. Levy et al. 31 reported that for a GIST patient, the end point of injection was the complete filling of the GIST with the injectate because the specific bleeding vessel is hard to clearly discern.
Prognoses and complications. Much attention should be paid to the possible complications, mainly distant thrombosis. Indeed, pulmonary glue embolisms have been reported in patients with varices treated by EUS-guided cyanoacrylate injection.41,45 Systemic embolization and splenic infarction have also been observed after endoscopic cyanoacrylate injection.46,47 The bleeding was stopped and the EUS-guided procedure was thought to be successful in all included cases. The clinical outcomes of all these 16 patients (100%) were satisfactory with no further bleeding, and no adverse events or complications were observed. Only one patient of gastroduodenal artery pseudoaneurysm, reported by Gonzalez et al., 35 suffered from a rebleeding 9 months later resulted from a new vascular lesion which was treated with radiological embolization. The median follow-up time was 6 months (interquartile range = 4.5–13).
EUS-guided sclerosing agent injection
Overview. EUS-guided injection therapies of other medicaments, similar to EUS-guided cyanoacrylate injection, have been applied to achieve hemostasis. The most widely used injections are sclerosing agents which include absolute alcohol and polidocanol. Once sclerosing agents are injected, vascular endothelial cells are damaged, which leads to the adhesion and aggregation of platelets and further thrombosis.
Patients. We induced 10 cases, in which 11 EUS-guided procedures were included (Table 2).20,21,31,32,35,38 The source of bleeding mainly focuses on Dieulafoy’s lesion (five cases). Other pathogenesis includes pancreatic pseudoaneurysm (two cases), duodenal metastasis (one case), periampullary tumor (one case), and GIST (one case). In these cases, three patients (30%) of Dieulafoy’s lesions had only undergone upper gastrointestinal endoscopy before EUS was performed. 38 For all other cases (70%) induced, patients had received endoscopic hemostasis or angiographies before the EUS-guided procedures.
Summary of the studies reported on EUS-guided sclerosing agent injection..

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; GIST, gastrointestinal stromal tumor; IR, interventional radiology; ml, milliliters; NA, not applicable.
Needles and volumes. As for the choice of injection site, the sclerosing agents were injected into or adjacent to the targeted vessels. In a study reported by Levy et al., 31 the needle was punctured directly into the pseudoaneurysm followed by advancing to the feeding vessel and injection when treating a patient of pancreatic pseudoaneurysm . Valdivielso Cortazar et al. 20 reported that the needle was placed in the middle of the lesion of a GIST and alcohol was injected in the middle of the lesion. The sizes of the needles used in these cases were 19G in one case (two procedures, 18.2%), 20G in one case (9.1%), 22G in four cases (36.4%), 23G in three cases (27.3%), and 25G in one case (9.1%). With regard to the selection of dose, the injected volume of ethanol varies from 0.2 to 7.5 ml and the mean volume is 3.7 ml (six cases). The volume of polidocanol for single use is 4 ml (one case, two procedures). Furthermore, epinephrine has the effect of constricting blood vessels. 32 Fockens et al. 38 reported that they used polidocanol 1% or a combination of polidocanol 1% and epinephrine 1:10,000 to treat three patients with Dieulafoy’s lesions. However, the exact volume was unspecified.
Prognoses and complications. Some complications had been reported after endoscopic procedures with sclerosing agents.48,49 In several comparative studies about the treatment of variceal bleeding, alcohol or polidocanol injection is associated to relatively higher risk of complication, higher mortality, and lower eradication rates, when compared with cyanoacrylate injection.50,51 The clinical outcomes of induced cases were favorable in seven cases (70%) for no further bleeding was observed during the follow-up period. Nevertheless, three patients (30%) experienced recurrent bleeding after the EUS-guided procedure. In the study of Fockens et al., 38 one patient rebled 5 months later and successfully received sclerotherapy followed by a wedge resection. Gonzalez et al. 35 reported a patient exhibited rebleeding 3 months later which was successfully treated by a second injection of polidocanol. In the study reported by Sharma et al., 21 the patient rebled on Day 6 after the EUS-guided injection, and hemostasis was achieved through another EUS-guided coil deployment. In general, the rebleeding rate after EUS-guided sclerosing agent injection is 30% in our included cases. The median follow-up time was 18 months (interquartile range = 10–29.5). It seems that the safety of injection of sclerosing agents requires more investigation in the future.
EUS-guided other hemostatic agent injection
Hyaluronate. Hyaluronate has a strong water retention effect and its aqueous solution is a highly viscous agent that can increase the pressure within the lesion and fill the vessels. 52 According to a clinical trial reported by Cho et al., 53 the hemostatic rate of endoscopic sodium hyaluronate injection in peptic ulcer bleeding is 96.2%. It serves as an excellent agent for endoscopic injection therapy. In the study reported by Law et al., 32 two patients of gastric GIST suffered from resistant bleeding. EUS-guided hyaluronate injection was performed with 22G aspiration needle. To minimize the risk of embolization, hyaluronate was injected next to the targeted vessels; 1 ml of hyaluronate was injected to one patient, and another patient underwent twice injection of hyaluronate, 3 and 4 ml, respectively, to achieve complete cessation. No complications and further bleeding were reported in these two cases. The median follow-up time was 40 and 45 months (Table 3).
Summary of the studies reported on EUS-guided other hemostatic agent injection.

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; GISTs, gastrointestinal stromal tumors; IU, international units; ml, milliliters; NA, not applicable.
Thrombin. Thrombin plays an important role in the final pathway of the coagulation cascade. It hydrolyzes the fibrinogen to produce strong fibrin clot and activates Factor XIII, which crosslinks adjacent fibrin molecules to stabilize the fibrin clot. 54 Thanks to the few complications and side effects, thrombin has been used to treat visceral pseudoaneurysms through percutaneous US- or computed-tomography-guided injection54,55 and EUS-guided intravascular injection. Indeed, EUS-guided injection is superior to percutaneous injection because it allows safe, minimally invasive access to visceral pseudoaneurysms.
We induced three patients of visceral pseudoaneurysm who underwent EUS-guided thrombin injection without hemostasis treatments before (Table 3). Roach et al. 29 succeeded in injecting 1 ml of solution containing 500 IU of thrombin to treat a patient of pancreatic pseudoaneurysm. Lameris et al. 39 used 7 ml thrombin for the patient of splenic artery pseudoaneurysm. They both punctured the pseudoaneurysm with a 22G needle and color Doppler confirmed the obliteration of the lesion. In the study of Sharma et al., 28 they tried EUS-guided coil embolization initially, but follow-up EUS showed high flow in the pseudoaneurysm, in which case on Day 3 a further 8 ml of thrombin was injected through a 22G needle and color Doppler showed complete obliteration of the pseudoaneurysm. The clinical outcomes of all three patients (100%) were satisfactory. The median follow-up time was 10 months (range = 0.5–10.5). EUS-guided thrombin injection seems to be a safe and effective therapy for visceral pseudoaneurysm.
Mechanical therapy
EUS-guided coiling
Overview. EUS-guided coiling is performed by the insertion of coils into the vessels. Targeted vessel is identified endoscopically and then an FNA (fine needle aspiration) needle is preloaded with a coil whose diameter is approximately 1.25 times the diameter of the targeted vessel. The needle is advanced slowly into the vessel, then the stylet is withdrawn to allow the coil to deploy. The proximal portion of the coil is anchored to the non-vascular tissue close to the vessel. 32 Platelets adhere to the fibrillary structures on the coil, which induces the following thrombosis and obliteration. 56 The decreased or absence of blood flow is confirmed by Doppler US endoscope after the coil is deployed.
Patients. We induced six cases including six procedures of EUS-guided coiling (Table 4).21,26–28,32,34 The type of lesions refers to ulcer after Roux-en-Y gastric bypass (RYGB; one case), pancreatic pseudoaneurysm (one case), gastroduodenal artery pseudoaneurysm (one case), periampullary tumor (one case), portoduodenal fistula (one case), and post-ERCP (endoscopic retrograde cholangiopancreatography) bleeding (one case). Except for one patient of gastroduodenal artery pseudoaneurysm, the other five patients (83.3%) had undergone hemostasis therapies before EUS-guided procedures.
Summary of the studies reported on EUS-guided coiling..

ERCP, endoscopic retrograde cholangiopancreatography; EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; ml, milliliters; NA, not applicable; RYGB, Roux-en-Y gastric bypass.
Needles and sizes. The size of the coil ranges from 4 to 10 mm depending on the diameter of the targeted vessel. Interestingly, Sharma et al. 21 reported that they used a transducer to measure the diameter of targeted vessel, which decided the selection of the size of the coil required. The sizes of the needles used were 19G in one case (16.7%), 22G in four cases (66.7%), and unspecified in one case (16.7%). The reason why 22G FNA needle accommodating 0.018-inch coils is preferable to 19G needle accommodating 0.035-inch coils is that the former one is easier to use and relates to less risk of bleeding. 32
Prognoses and complications. All the EUS-guided procedures were technically successful, and the clinical outcomes were favorable in five cases (83.3%). The patient in the case report of Sharma et al. 28 showed high flow in the pseudoaneurysm 1 day after coil deployment, which has been stated in the former chapter. No complication related to EUS-guided coiling was observed although there are concerns regarding the risk of coil migration and subsequent thrombosis. The median follow-up time was 4.6 months (range = 0.5–15). Fluoroscopy and anchor technique may be useful to ensure the safety of EUS-guided coiling. Although EUS-guided coiling seems to be a safe and effective alternative therapy for refractory non-variceal bleeding, more clinical comparative investigation is required.
EUS-guided band ligation
Endoscopic band ligation is a well-established therapeutic modality in the treatment of bleeding varices 57 and has been used to manage other non-variceal gastrointestinal bleeding. 58 EUS-guided band ligation is an emerging therapy for hemostasis, especially for Dieulafoy’s lesion, because EUS allows precise imaging of structures in the gastrointestinal tract, which is helpful for accurate delivery of therapy and evaluation of vessel obliteration.
We induced three cases of recurrent bleeding due to Dieulafoy’s lesions (Table 5). Before bands were deployed, EUS was used to confirm and mark the targeted vessels. EUS was also performed after the procedure to assess the efficacy of band ligation. The patient in the case report of Folvik et al. 37 had no further bleeding and complication. Law et al. 32 reported a patient suffered from a clinically mild bleeding requiring no transfusion 38 months after the first EUS-guided band ligation and no subsequent bleeding after the second EUS treatment. The other patient received EUS-guided band ligation combined with injection of 4 ml of alcohol and had no rebleeding. In general, complete hemostasis was achieved in two of three patients (66.7%), but ultimate clinical outcomes were all favorable. The median follow-up time was 43 months (range = 11–60).
Summary of the studies reported on EUS-guided band ligation.
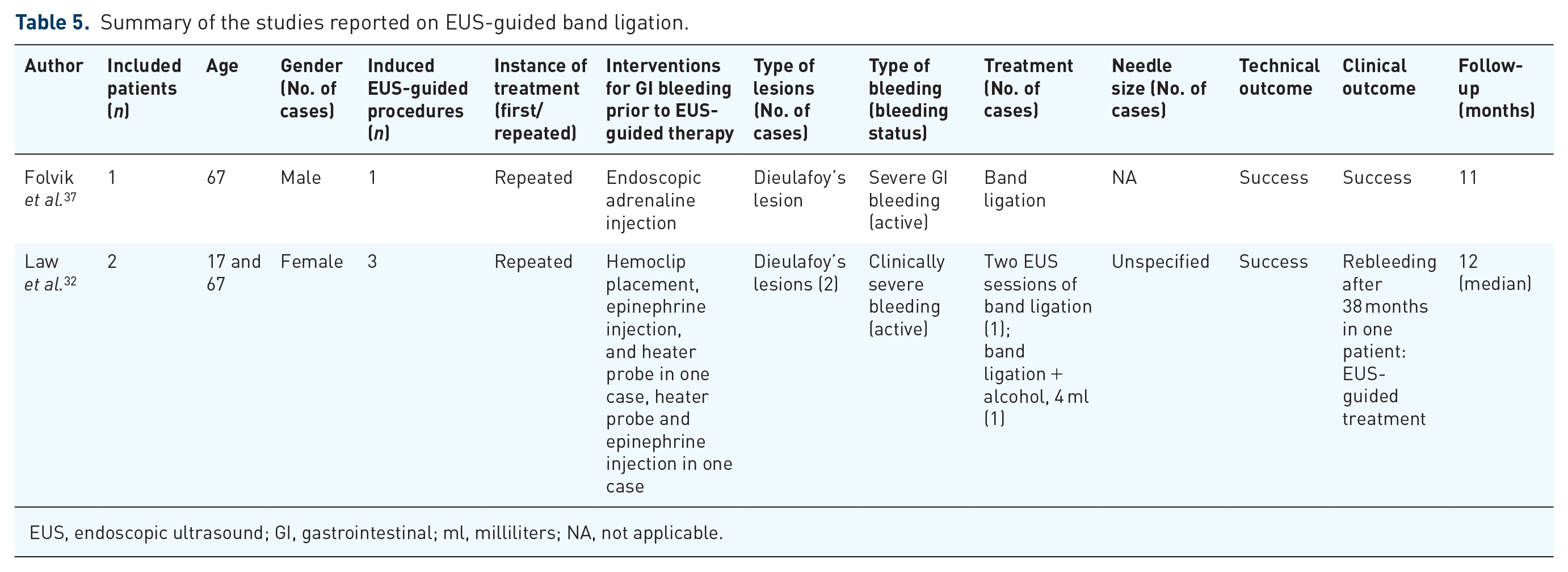
EUS, endoscopic ultrasound; GI, gastrointestinal; ml, milliliters; NA, not applicable.
Combined therapy
Combined EUS-guided coiling and cyanoacrylate injection
When cyanoacrylate is combined with coil, the glue immediately adhered to the synthetic fibers. This kind of adherence is firm, which indicates that the deployment of a coil can work as a scaffold to retain cyanoacrylate at the site of inserted coil. Besides, both coiling and cyanoacrylate injection contribute to promote hemostasis. 59 This combined therapy seems to acquire higher safety and effectiveness because the risk of embolization is minimized and better theoretically eradication rate is expected. Combined EUS-guided coiling and cyanoacrylate injection have been used in patients with gastric varices and lead to favorable clinical outcomes. 42
We induced two cases of bleeding GISTs reported by Romero-Castro et al. 23 in a case series (Table 6). The two patients had not received previous hemostasis therapy. EUS-guided coil deployments with a 19G FNA needle were performed, followed by injection of cyanoacrylate. The targeted vessels were the feeding arteries. Coils are both 6 mm and the volume of cyanoacrylate is 2 ml. It is worth mentioning that this combined therapy managed to achieve hemostasis in a case of spurting bleeding after initial EUS-guided puncture. It seems that EUS-guided therapy could have the additional advantage to solve adverse event during the EUS-guided procedure. The clinical outcome in both patients was uneventful and the follow-up time was 1 month. More clinical researches are needed to identify the effect of combined EUS-guided coiling and cyanoacrylate in the management of NVUGIB.
Study reported on combined EUS-guided coiling and cyanoacrylate injection.

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; GISTs, gastrointestinal stromal tumors; ml, milliliters; NA, not applicable.
Combined EUS-guided coil/clip deployment and epinephrine injection
There are two cases where EUS-guided coil/clip deployment combined with epinephrine injection was performed (Table 7). In the case series of Satyavada et al., 22 EUS was used in the diagnosis and treatment of an obscure gastrointestinal bleeding which turned out due to Dieulafoy’s lesion. EUS-guided therapy was performed with the injection of 1:10,000 epinephrine and the deployment of four hemoclips. Marya et al. 24 attempted to perform complicated EUS-guided procedures on a patient of recurrent bleeding due to duodenal ulcer with RYGB. The patient had received surgery before but had rebleeding. Under EUS guidance, lumen-apposing metal stent (LAMS) was used to access the targeted bleeding ulcer followed by injection of dilute epinephrine and deployment of two hemoclips. A fully covered metal stent (FCMS) was additionally placed for tamponade. After the procedure, an interventional angiography was performed but failed, and the patient had persistent bleeding. Second EUS-guided procedure was performed with injection of coils and a mixture of gelfoam and epinephrine into the targeted artery by a 22G needle.
Summary of the studies reported on combined EUS-guided coil/clip deployment and epinephrine injection.

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; NA, not applicable.
These two patients had no further bleeding. A previous randomized trial compared epinephrine injection alone versus dual therapy (epinephrine injection plus other therapy). The results showed that dual therapy significantly reduced the rate of recurrent bleeding, surgery, and mortality. 60 Combined EUS-guided injection therapy and mechanical therapy may provide a strategy for complicated non-variceal gastrointestinal bleeding.
EUS-guided other combined therapies
Riberio et al. 30 reported a patient of recurrent episodes of bleeding due to Dieulafoy’s lesion. Under the guidance of EUS, thermal contact therapy was performed and followed by injection of 2.5 ml of absolute alcohol because the thermal therapy did not result in complete cessation. The target area was the bleeding artery and alcohol was injected along the course of the vessel. Repeated Doppler image confirmed the eradication of arterial flow. Law et al. 32 performed EUS-guided injection of 10 ml of epinephrine and snare ligature with polypectomy on a patient of duodenal Brunner’s gland hamartoma who had rebleeding after endoscopic treatments. Prior attempt to resect the duodenal mass failed due to the characteristic of the lesion, friable and bled easily. EUS-guided injection eradicated the blood flow, which promoted the success of resection. Both the patients had no further bleeding, and the follow-up time was 1 and 9 months, respectively (Table 8).
Summary of the studies reported on EUS-guided other combined therapies.

EUS, endoscopic ultrasound; G, gauge; GI, gastrointestinal; ml, milliliters.
Conclusion
EUS-guided treatments are gaining popularity in the diagnostic and therapeutic fields. Thanks to its capacity to offer direct access to targeted lesions that are difficult to explore, treat lesions under visualizing guidance, detect the specific blood flow, and check the therapeutic effectiveness, EUS-guided therapies have been proved to be powerful tool in the treatment of various conditions. As mentioned in this review, EUS-guided treatment is a technique with promising therapeutic potential for NVUGIB, especially in cases where standard endoscopic treatments failed, as it has shown effectiveness with high safety profile in most cases. Meanwhile, concerns regarding the role of EUS-guided hemostasis for both variceal and non-variceal upper gastrointestinal bleeding still exist, especially in case of active bleeding at the time of the treatment. Overall, we believe the application of EUS-guided therapies in NVUGIB will expand with the further development of EUS technology. However, the literature regarding this field is scarce and limits to case report and case series. Large randomized clinical controlled trials seem to be warranted.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the National Natural Science Foundation of China (No. 81770637, No. 81800467).
