Abstract
Background:
Selection of second-line therapy in Crohn’s disease (CD) patients after failure of first-line TNFα-inhibitor (TNFi) therapy to optimise outcomes remains challenging in real-world clinical practice.
Objectives:
This study aimed to provide real-world outcomes of CD patients who failed first-line TNFi therapy and switched to either another TNFi or to a biologic with a different mechanism of action. Patients were stratified as to whether they switched because of primary non-response or secondary loss of response to initial TNFi.
Design:
Retrospective cohort study.
Methods:
CD patients whose first biologic therapy was a TNFi and switched to another biologic therapy between 26 August 2015 and 31 March 2021 were identified in records held by the UK IBD Registry. Patients were enrolled at study sites, and data were validated by clinical teams. Patients were grouped as within-class switchers (WCS) if their second-line (index) biologic therapy was another TNFi, or out-of-class switchers (OCS) if their index therapy was vedolizumab or ustekinumab. Patients were followed up for at least 1 year. Time to drug discontinuation and outcomes at 1 year after index therapy start were analysed using Cox regression and binary logistic regression models, before and after baseline covariate adjustment through inverse probability of treatment weighting.
Results:
A total of 180 adult CD patients were included in the study. OCS were less likely to discontinue index therapy in both unweighted analysis (hazard ratio (HR): 0.64, 95% confidence interval (CI): 0.42–0.96, p = 0.03) and weighted analysis (HR: 0.58, 95% CI: 0.38–0.90, p = 0.01), and more likely to show index drug persistence at 1 year in both unweighted analysis (adjusted odds ratio (aOR): 3.66, 95% CI: 1.81–7.67, p < 0.001) and weighted analysis (aOR: 3.95, 95% CI: 2.04–7.89, p < 0.001). These findings were consistent across all secondary endpoints of steroid-free and/or surgery-free index drug survival at 1 year.
Conclusion:
Patients switching from a TNFi to either vedolizumab or ustekinumab exhibited significantly higher rates of drug persistence compared to those switching to another TNFi, particularly among those experiencing primary non-response to the initial TNFi.
Keywords
Introduction
Crohn’s disease (CD) is a chronic, relapsing, remitting inflammatory condition affecting the gastrointestinal tract and carries a high degree of morbidity with resultant impact on every aspect of patients’ lives. Therapeutic goals include not only treating acute relapses but also maintaining remission with the control of symptoms, restoration of quality of life, and ultimately healing of the gut mucosa.
Recent advances in elucidating the molecular mechanisms underlying its pathophysiology have expanded the number of available treatments for moderate-to-severe CD. 1 Anti-TNFα (TNFi) therapies, including infliximab and adalimumab, are well-established first-line agents for refractory cases not responding to conventional treatments2,3 and became available in the UK to treat CD in 1999 and 2009 respectively. Since 2015, several therapies with different modes of action, such as vedolizumab (anti-integrin), ustekinumab (anti-interleukin 12–23), risankizumab (anti-interleukin 23) and upadacitinib (Janus kinase (JAK) inhibitor), have emerged and been approved.
Persistence rates of these drugs remain relatively low, with nearly half of patients switching to a second biologic within a year, and two-thirds within three years. 4 The reasons for treatment failure are often categorised as primary non-response (PNR), that is, no initial benefit to the patient, usually after 3–6 months of treatment (suggesting a mechanistic treatment failure), or secondary loss of response where patients initially improve but then this benefit is lost (often due to anti-drug antibody formation as well as other less well understood mechanisms), or patients have an significant adverse event leading to discontinuation of therapy. The sequential use of biologic therapies has become central to preventing the consequences of uncontrolled disease, including fistulas, strictures, malnutrition and surgery. As more advanced therapies have become available, a consensus on the optimal sequence of biologic therapies in CD patients unresponsive or intolerant to first-line TNFi is yet to be established.2,5,6
‘Biologic Sequencing in Clinical Units’ (BISCUITS) is a multi-centre retrospective observational cohort study drawing from real-world data obtained from the UK IBD Registry (IBDR). The study aims to compare treatment outcomes among individuals with CD who transitioned from their initial TNFi treatment to either an alternative TNFi (within-class switch, WCS) or a biologic employing a different mechanism of action (out-of-class switch, OCS). The aim is to address the need for a deeper understanding of the outcomes in this patient population, lacking comparative data, gaining valuable insights into the optimisation of biologic sequencing strategies.
Methods
The IBDR is a not-for-profit organisation that holds records for 86,000 UK patients. A feasibility study was performed on the entire IBD Registry population. Through this process of cohort discovery, seven UK hospitals with the highest number of eligible patients were identified, which consented participants and backfilled and validated the relevant data. 7
Study population
Validated data from adult patients (⩾18 years old) with a confirmed diagnosis of CD and no prior alternative IBD diagnosis, whose first biologic was a TNFi and who switched to another biologic therapy (second-line) during the period 26 August 2015 (the date when vedolizumab was available for management of CD in the UK) to 31 March 2021 were included in the analysis, as depicted in Supplemental Figure 1. By definition, patients were biologic naïve at the point of first TNFi treatment.
Patients were categorised as either WCS if they changed (‘switched’) biologic treatment either to another TNFi (adalimumab (ADA), infliximab (IFX), certolizumab (CER)), or OCS if they changed (‘switched’) to another drug class (vedolizumab (VDZ) or ustekinumab (UST)). The index date was set as the date of first treatment change (first switch), and all patients were followed up for at least one year after first switch, or longer whenever possible, until the end of the study period (31 March 2022). Death during the study led to censoring.
Baseline demographic and clinical characteristics were collected, including smoking status, Montreal classification of disease, disease duration, prior CD-related surgery, name and duration of first-line TNFi, second-line biologic, and concomitant IBD medications at the time of switch.
Outcome measures
The primary outcome was time-to-treatment failure (TTF), described as the interval of time in days between initiation of second-line biologic and its cessation, which included discontinuation of therapy or switch to another biologic therapy. Secondary outcomes included treatment failure at 1 year and corticosteroid-free drug survival. Persistence at 1 year was a binary outcome observed at 1 year after starting the index second-line biologic, further defined in four subcategories: cessation of index drug within 12 months (as per the primary outcome), steroid-free survival (no cessation of index drug or record of oral corticosteroid within 12 months), surgery-free survival (no cessation of index drug or record of major IBD-related surgery within 12 months), and steroid- and surgery-free survival (no cessation of index drug, record of oral corticosteroid, or record of major IBD-related surgery within 12 months).
Statistical analysis
The main strata for comparison were WCS versus OCS. Patients within these alternative pathways were further stratified into subgroups based on their disease duration at the time they received their first-line TNFi, early (⩽2 years after diagnosis) versus later (>2 years after diagnosis) and by their ‘response’ to first-line TNFi: primary non-response (PNR, defined as ⩽6 months duration of drug) versus secondary loss of response (SLOR, defined as > 6 months duration of drug).
For the primary outcome, unadjusted Kaplan-Meier survival curves were generated to compare WCS and OCS groups, with further stratifications based on the subgrouping variables outlined above. Survival curves were truncated at 1500 days due to low numbers of patients at risk of event after this time. Time-to-event data were further analysed using Cox-proportional hazards models to adjust for potentially confounding covariates. The proportional hazards assumptions of the Cox regression models were ascertained by inspection of log-log plots and by using a formal hypothesis test based on Schoenfeld residuals. For secondary outcomes, binary logistic regression was used to generate adjusted odds ratios to test associations between second-line treatment choice (WCS as reference, OCS as comparator) and relevant one-year endpoints (binary dependent variable).
Imbalances in baseline covariates were adjusted for using inverse probability of treatment weighting (IPTW). The covariates likely to act as confounders were selected a priori through review of literature and expert clinical opinion and are described in Table 1: age at index, presence of perianal disease, early or later treatment with first-line TNFi, PNR or SLOR to first-line TNFi, steroid therapy at index, and immunomodulation therapy at index. Weights were stabilised and trimmed at 1% and 99% to avoid extreme values. Primary and secondary outcomes were re-analysed after IPTW using weighted Cox regression models and weighted logistic regression models respectively, adjusting for non-confounding baseline covariates.
Patient characteristics before and after inverse probability of treatment weighting.

Median (Interquartile Range); n (%).
Wilcoxon rank-sum test; Pearson’s Chi-squared test; Fisher’s exact test.
Wilcoxon rank-sum test for complex survey samples; chi-squared test with Rao & Scott’s second-order correction.
CD, Crohn’s disease; OCS, out-of-class switcher; PNR, primary non-response; SLOR, secondary loss of response; SMD, standardised mean difference; TNFi, TNFα-inhibitor; WCS, within-class switcher.
Data were missing in two variables, ‘Presence of perianal disease’ (3.9%) and ‘Current smoker’ (5.6%). As binary variables, a ‘No’ response was imputed in the absence of a value, and the risk of bias was deemed to be minimal. Statistical analysis was performed using R (version 4.3.0, 2023-04-21, R Foundation for Statistical Computing, Vienna, Austria).
Reporting
The reporting of this study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology statement for cohort studies (Supplemental Table 1). 8
Results
A total of 83,658 adult patients with inflammatory bowel disease were identified from the IBDR. Of these, 44,837 patients had been excluded due to a current or prior co-diagnosis with ulcerative colitis (UC). After applying the exclusion criteria of no biologic therapy initiated or no switch to a new mode of action within the study duration, a final feasibility cohort of 2168 CD patients has been established. From this feasibility cohort, 180 patients were recruited into the study, as shown in the detailed flow of patients from Figure 1.
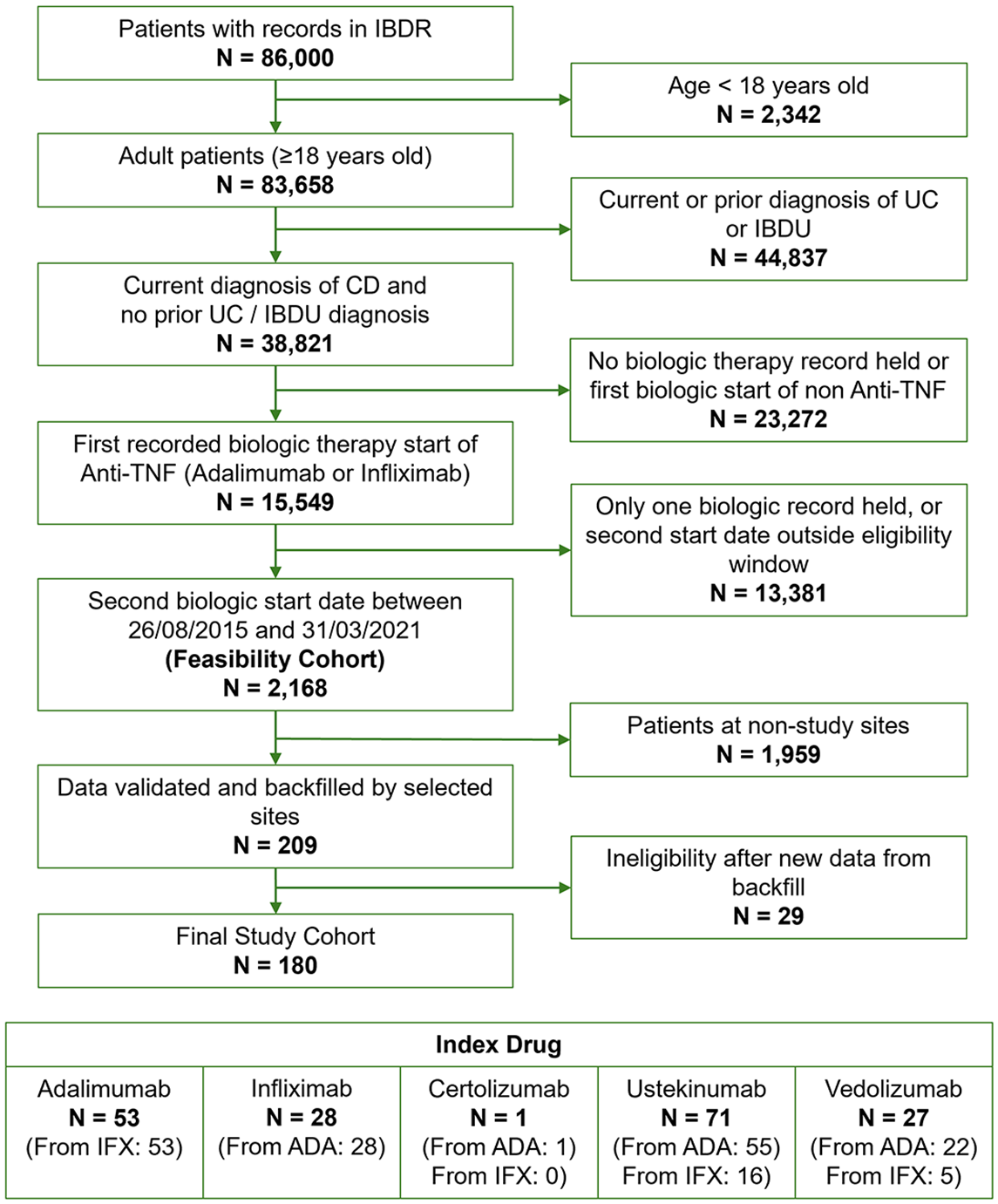
CONSORT diagram showing patient selection.
Patients who switched within class were more likely to be on concomitant immunosuppressant therapy (p = 0.006) and less likely to be on corticosteroids (p = 0.075) at initiation of the index drug on unweighted analysis, as indicated in Table 1.
Time-to-treatment failure
Unadjusted analysis showed that OCS were less likely to discontinue index treatment compared to WCS (hazard ratio (HR): 0.64, 95% confidence interval (CI): 0.43–0.96, p = 0.03), as indicated in Figure 2. When adjusting for age at index, early or later treatment with first-line TNFi, PNR or SLOR to first-line TNFi and immunomodulation therapy at index, findings continue to indicate that OCS were less likely to discontinue index treatment compared to WCS (HR: 0.58, 95% CI: 0.38–0.90, p = 0.02), as indicated in Supplemental Table 2.

Unadjusted Kaplan-Meier survival curves and log-rank test p-values for time to index treatment discontinuation of within-class switchers versus out-of-class switchers.
After IPTW, weighted Cox-proportional hazards analysis indicated that OCS were less likely to discontinue index treatment compared to WCS (HR: 0.58, 95% CI: 0.38–0.90, p = 0.01) when adjusting for current smoking status and prior major surgery, as shown in Supplemental Table 2.
Subgroup analysis of time to discontinuation of patients with early (⩽2 years from diagnosis) or later (>2 years from diagnosis) initiations of initial TNFi is shown in Figure 3. There was no statistically significant difference in time to discontinuation overall (p = 0.13) nor when stratified for WCS (p = 0.82) or OCS (p = 0.09); however, in patients who initiated TNFi later, OCS showed a statistically significant difference in TTF with a lower probability of failure, as shown in Figure 3(e) (HR: 0.55, 95% CI: 0.33–0.93, p = 0.03).

Unadjusted Kaplan-Meier survival curves and Cox-proportional hazards models for time to index treatment discontinuation of (a) patients with Early versus Later initial TNFi exposure overall, (b) patients with Early versus Later initial TNFi exposure of WCS, (c) Early versus Later initial TNFi exposure of OCS, (d) WCS versus OCS for patients with Early initial TNFi exposure and (e) WCS versus OCS for patients with Later initial TNFi exposure.
Figure 4 examines the time to discontinuation for either PNR or SLOR to initial TNFi. For WCS patients, unsurprisingly those with SLOR to their initial TNFi had a lower probability of subsequent TNFi failure compared to those with PNR (HR: 0.25, 95% CI: 0.14–0.46, p < 0.001), whilst there was a clear benefit to OCS in patients with PNR to their initial TNFi (HR: 0.25, 95% CI: 0.12–0.51, p < 0.001).
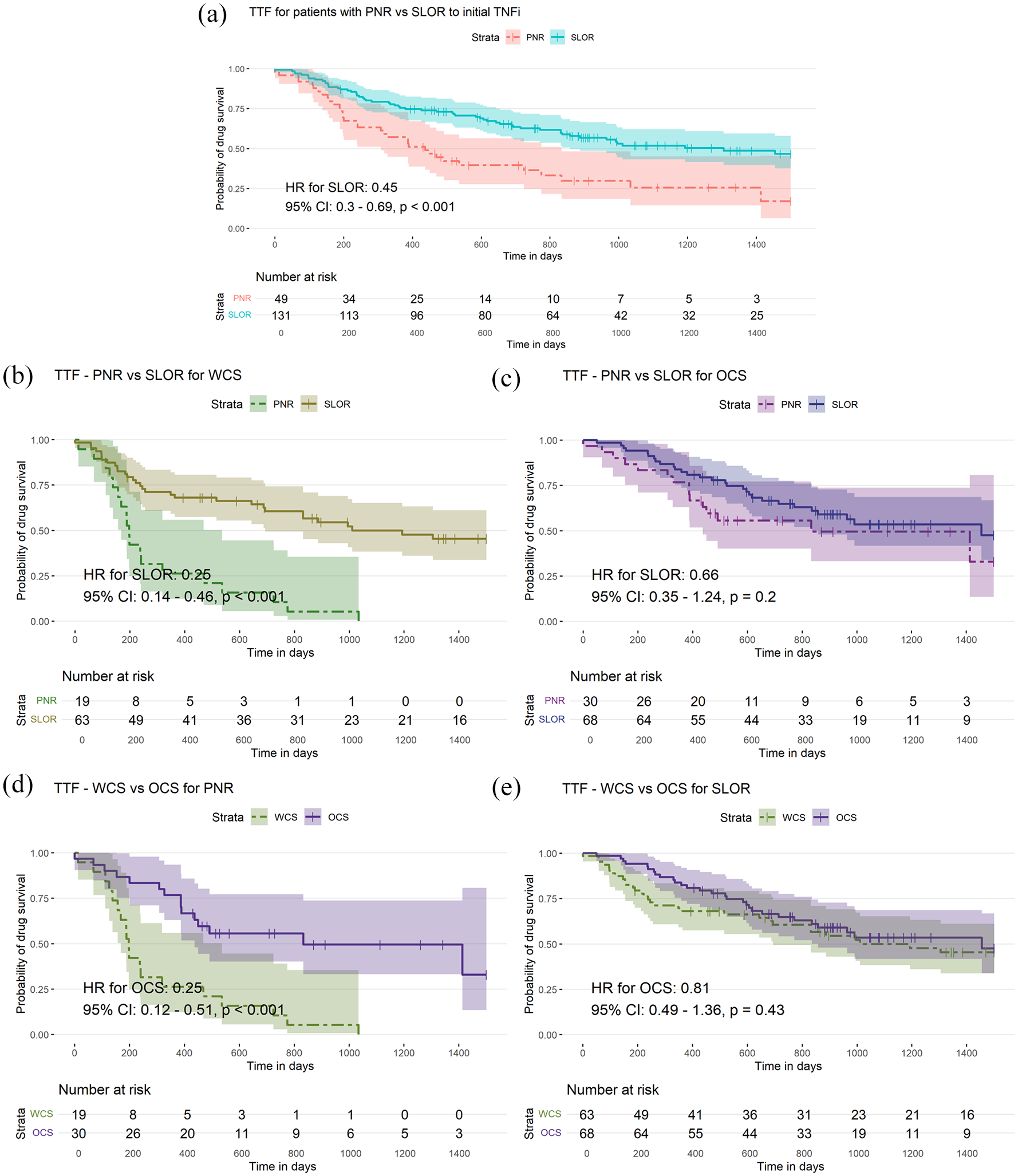
Unadjusted Kaplan-Meier survival curves and Cox-proportional hazards models for time to index treatment discontinuation of (a) patients with PNR versus SLOR to initial TNFi overall, (b) WCS with PNR versus SLOR to initial TNFi, (c) OCS with PNR versus SLOR to initial TNFi, (d) WCS versus OCS with PNR to initial TNFi and (e) WCS versus OCS with SLOR to initial TNFi.
Treatment outcomes at 1 year
Drug persistence at 1 year for patients in the study is illustrated in Figure 5. Binary logistic regression adjusting for age at index, early or later treatment with first-line TNFi, PNR or SLOR to first-line TNFi and immunomodulation therapy at index indicated OCS were more likely to show drug survival at 1 year (adjusted odds ratio (aOR): 3.66, 95% CI: 1.81–7.67, p < 0.001, Supplemental Figure 2).

Flow diagram of index drug persistence at 1 year.
For other study outcomes such as steroid-free drug survival at 1 year (aOR: 2.10, 95% CI: 1.10–4.10, p = 0.026), surgery-free drug survival at 1 year (aOR = 3.18, 95% CI: 1.61–6.47, p = 0.001), and steroid- and surgery-free drug survival at 1 year (aOR = 2.01, 95% CI: 1.06–3.89, p = 0.035), OCS continued to show improved drug survival at 1 year, as shown in Supplemental Figure 2.
Weighted binary logistic regression adjusting for current smoking status and prior major surgery indicated OCS were more likely to show drug survival at 1 year (aOR: 3.95, 95% CI: 2.04–7.89, p < 0.001), steroid-free drug survival at 1 year (aOR: 2.34, 95% CI: 1.27–4.36, p = 0.007), surgery-free drug survival at 1 year (aOR = 3.47, 95% CI: 1.82–6.78, p < 0.001) and steroid- and surgery-free drug survival at 1 year (aOR = 2.21, 95% CI: 1.21–4.11, p = 0.01), as indicated in Supplemental Figure 3.
Discussion
The management of CD often requires changes to medical therapy due to PNR or SLOR to initial TNFi therapy. Conducting head-to-head comparative clinical trials to establish robust evidence for drug sequencing poses challenges, primarily due to the extended follow-up required to assess outcomes, but also due to the increasing number of treatment options available. The applicability of evidence generated from closely monitored clinical trials with strict inclusion and exclusion criteria, as well as patient selection bias from trial recruitment, is challenging. Consequently, there are currently no randomised head-to-head studies comparing outcomes of treatment following TNFi failure. 3 This retrospective cohort study, relying on validated real-world data from the IBDR, assesses the TTF in various biologic sequencing pathways, mirroring clinical practice conditions over an extended time period.
Our data showed that patients undergoing OCS exhibited significantly higher rates of drug persistence compared to WCS, particularly among those experiencing PNR to the initial TNFi. However, there is no significant difference in drug persistence between OCS and WCS for patients with SLOR. Consistent with prior cohort studies, no appreciable distinction in drug persistence is observed between ustekinumab and vedolizumab,9,10 nor between adalimumab and infliximab, 11 as indicated in Supplemental Figures 4 and 5.
Selection of a second-line therapy is dependent on many factors such as patient choice, ease of administration, patient demographics, use of immunomodulators, drug acquisition costs, age and comorbidities. These findings suggest that opting for OCS in patients with PNR may result in lower rates of treatment failure, irrespective of whether ustekinumab or vedolizumab is chosen as second-line treatment. On the other hand, in the setting of SLOR, within- and out-of-class switches appear equally effective.
This study used a novel pathway for the identification and recruitment of eligible patients. The study is constrained by a relatively small sample size, potentially limiting the robustness of our findings. However, other similar studies conducted in recent years had comparable patient numbers included for their analyses, with similar results to our study.12 –15 In addition, larger cohort studies have demonstrated consistent outcomes,16 –18 although their greater patient numbers further highlight this limitation. The study was also potentially limited by the possibility of selection bias in the choice of index biologic, as patients who switched within class were more likely to be on concomitant immunosuppressant therapy and have perianal disease, and less likely to be on corticosteroids, than those who switched out of class. While IPTW was used in weighted analysis to simulate a randomised comparison by appropriately adjusting for confounding, the limitation of this method is the assumption of ‘no unmeasured confounders’. The likelihood of this assumption being true is reduced due to a lack of information on patients’ Charlson Comorbidity Index score, history of IBD-unrelated disease or number of hospitalisations before the inclusion period of the study. Although data on biologics is expected to be complete as they are exclusively prescribed by secondary care, prescriptions for steroids from primary care and emergency departments may not be captured, resulting in uncertainty regarding concomitant steroid use. While the contrast in drug persistence of OCS patients with PNR and SLOR appears to differ from prior findings, 6 direct comparisons between studies are problematic due to divergent definitions of stratification of treatment failure, highlighting the need for standardised classification. The absence of data items such as anti-drug antibody levels, due to the required use of the pre-existing data submission framework of the IBDR, may have impacted the interpretability of SLOR switches in real-world settings. Moreover, the pragmatic definition of PNR and SLOR based solely on treatment duration is likely an oversimplification, not fully capturing the complexities of underlying immunogenic and pharmacodynamic changes influencing clinical status, as well as variation in the timing of clinical assessment of response to therapy. However, all data were collected at the point of submission from clinical notes/electronic patient records, and 10% of cases were validated by the local principal investigator, suggesting that the primary outcome (TTF) was robust.
Conclusion
In this real-world study of second-line biologic therapy in CD, patients who switched from a TNFi to a biologic with a different mechanism of action (vedolizumab or ustekinumab) exhibited significantly higher rates of drug persistence compared to those who switched to another TNFi, particularly among those with PNR to the initial TNFi. While these findings highlight the potential benefits of out-of-class switching in optimising long-term treatment outcomes, further research is needed to validate these results and refine treatment sequencing strategies in CD management.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-docx-1-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-2-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-tiff-2-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-3-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-tiff-3-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-4-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-tiff-4-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-5-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-tiff-5-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tiff-6-tag-10.1177_17562848251352446 – Supplemental material for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study
Supplemental material, sj-tiff-6-tag-10.1177_17562848251352446 for Real-world outcomes from the UK IBD Registry on second-line biologic therapy following anti-TNF exposure in Crohn’s disease: results from the BISCUITS study by Frederick Taylor, Giorgio Bartalucci, Catherine Gleave, Liz Dobson, Keith Bodger, Susanna Dodd, Stuart Bloom, Alun Passey, Riikka Nissinen, Daniel Wirth, Andreas Duva, Jennifer Lee and J. R. Fraser Cummings in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
