Abstract
Background:
Inflammatory pouch disorders can be refractory to conventional therapy. Evidence on biological therapy from large cohorts is scarce.
Objective:
To explore the use and effectiveness of biological therapy for inflammatory pouch disorders.
Design:
A retrospective and multicentre study.
Methods:
We included patients diagnosed with pouchitis, Crohn’s Disease of the Pouch (CDP) or cuffitis and treated with biological therapy. Effectiveness was evaluated at 12 months based on normalisation of stool frequency, absence of pain, faecal urgency or fistula discharge (clinical remission), or as any improvement in these symptoms (clinical response). We also compared the effectiveness of a second biologic after anti-tumour necrosis factor (anti-TNF) failure using descriptive and comparative statistics.
Results:
In total, 145 patients were included; 62% were men and the median age was 54 (20–71) years. Overall, 212 lines of treatment were evaluated (95 infliximab, 69 adalimumab, 7 golimumab, 35 vedolizumab and 26 ustekinumab). At least a second line of treatment was received by 41% of patients. Overall, 66% had chronic pouchitis and 29% had CDP. Clinical remission rates at 12 months were 45%, 44%, 43%, 39% and 45% for infliximab, adalimumab, golimumab, vedolizumab and ustekinumab, respectively. No differences were found based on the type of disease. Vedolizumab was the only treatment to show better results as a first-line therapy (50% vs 33%, p < 0.05). Thirty-nine patients received a second therapy after anti-TNF failure. Second anti-TNF use had higher risks of failure (odds ratio (OR) 4.8, 95% confidence interval (CI) 2.3–19) and discontinuation (OR 5.52, 95% CI 1.94–25.5).
Conclusion:
Biological therapy is a cornerstone in the treatment of pouch disorders, demonstrating consistently high effectiveness. After anti-TNF failure, another mechanism of action should be employed.
Introduction
Proctocolectomy with ileal pouch-anal anastomosis remains the preferred reconstruction technique for patients with ulcerative colitis (UC),1,2 with pouchitis as its most common non-mechanical complication, affecting up to 30% of patients and increasing over.3,4 A Spanish study showed that pouchitis often becomes chronic, recurrent and antibiotic-refractory, requiring a comprehensive therapeutic approach.5,6 Crohn’s Disease of the Pouch (CDP) occurs in up to 15% of cases and may be misdiagnosed as chronic pouchitis.5,7
Therapeutic needs in these disorders are considerable, and real-world practice reflects wide use of various treatments despite limited high-quality evidence. Current guidelines suggest biological therapies such as anti-tumour necrosis factor (anti-TNF), vedolizumab or ustekinumab in antibiotic-refractory or -dependent pouchitis or after budesonide failure, while evidence for CDP remains limited.5,8–13 A meta-analysis reported clinical response rates of 25%–60% with biologics. 14 More recently, the first clinical trial with vedolizumab in chronic pouchitis showed superiority over placebo, 15 leading to its recommendation as first-line therapy, though comparative data are lacking.
Biological therapy failure is frequent, and evidence on sequential use or treatment positioning remains scarce. This is particularly relevant given that many patients eventually require pouch surgery or permanent ileostomy.5,16 The aim of our study was to evaluate the use, effectiveness and durability of biological therapies in inflammatory pouch disorders.
Methods
Study design and endpoints
The RESERVO study is a retrospective and multicentre nationwide study undertaken by the Young Members Group of the Spanish Working Group on Crohn’s Disease and Ulcerative Colitis (GETECCU).
For the present study, we enrolled patients aged 18 years of age and older who had undergone ileal pouch-anal anastomosis following a diagnosis of UC confirmed by histological examination of resection specimens between 1995 and 2020. Subsequent follow-up facilitated the identification of a subgroup of patients with an inflammatory pouch disorder, comprising pouchitis, CDP or cuffitis, in accordance with the diagnostic criteria established by the European Crohn’s and Colitis Organization, GETECCU and the International Ileal Pouch Consortium (IIPC).17–19 Diagnosis was further guided by medical criteria derived from clinical assessments, endoscopic findings, histopathological analyses and/or radiological evaluations.
We included only those patients who received at least one biological therapy (infliximab, adalimumab, golimumab, vedolizumab and/or ustekinumab) for the treatment of these disorders and who had undergone at least the induction phase. We specifically excluded patients who received biological therapy for other conditions and patients who underwent other ileal pouch procedures (e.g. Koch pouch) or those with a permanent ileostomy.
We analysed the use (number and types) of biological therapies and their effectiveness at 12 months. Long-term durability was also evaluated. Differences in use and effectiveness were analysed according to the type of disorder and the number of therapeutic lines employed. As a secondary objective, we compared the strategy of the second biological treatment following failure of an anti-TNF drug (second anti-TNF vs non-anti-TNF). Patients and treatment lines were assessed separately.
Therapies used were recorded from the inflammatory pouch disorder diagnosis to the last clinical visit. We also recorded patients’ demographic characteristics and previous therapies. Pouchitis was classified according to recent guidelines,17–19 while CDP behaviour was established using the Montreal classification. 20 Perianal fistulas, strictures and penetrating CDP complications were considered and recorded if they appeared 6–12 months after stoma closure. 17
The need for surgical treatment was analysed as the need for an ileostomy, fistula treatment and/or pouch excision.11,12
All data were extracted from medical records.
Definitions
Effectiveness was evaluated using clinical definitions only. Clinical remission was defined as normal stool frequency without abdominal pain, rectal bleeding, faecal urgency or fistula drainage, and clinical response as improvement without remission; non-response indicated no change.12,21 Only patients not on concomitant steroids were considered responders. Effectiveness was assessed at 12 months, with early discontinuation due to intolerance, persistent activity, loss of response or adverse effects counted as non-response. Durability was the duration of active treatment. Antibiotic-dependent pouchitis required repeated antibiotic use (>3–4 relapses/year), and chronic refractory pouchitis showed no response after 2–4 weeks of antibiotics or other therapy. Therapy optimisation involved dose escalation or interval shortening, and combined a biological with an immunosuppressant. Pouch failure was defined as the need for permanent ileostomy with or without pouch excision due to treatment failure or intolerance.11,12
Statistical analysis
Categorical variables are presented as counts and percentages, and quantitative variables as mean ± standard deviation or median (interquartile range), depending on distribution. Categorical variables were compared using Chi-square tests, and quantitative variables with appropriate tests for normality. All analyses were conducted using the variables presented in the columns of the respective tables. This applies to all tables throughout the study. To analyse clinical outcomes according to the type of pouch disorder, adjusted odds ratios (ORs) were estimated using multivariable logistic regression, adjusting for sex, smoking status, age and time from diagnosis to treatment initiation. Therapy durability was evaluated by Kaplan–Meier analysis, with discontinuations right-censored, and first-line therapies compared using the log-rank test. Statistical significance was set at p < 0.05.
Results
Patients’ baseline characteristics
The cohort of the RESERVO study ultimately comprised 338 patients with inflammatory pouch disorder from 46 participating centres. Of these, 145 patients (43%) received at least one biological therapy for inflammatory pouch disorder treatment and were included (Figure 1). The main demographic and disease characteristics of the patients are provided in Table 1.
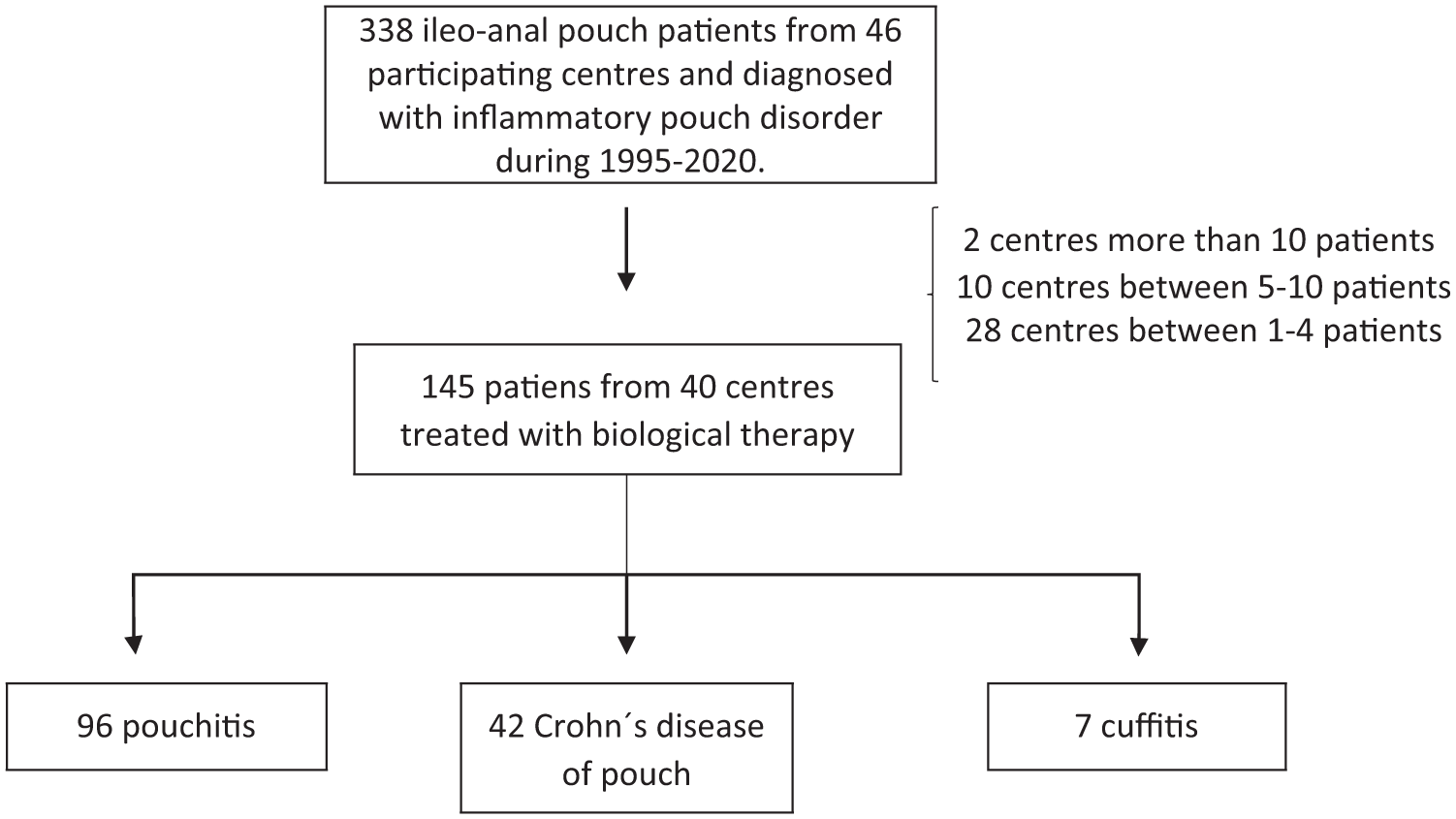
Flowchart of the included population.
Patient demographic and disease characteristics.

IQR, interquartile range.
Biological therapy characteristics
The therapeutic interventions included infliximab (95 cases), adalimumab (69 cases), golimumab (7 cases), vedolizumab (35 cases) and ustekinumab (26 cases). In addition, 60 patients (41%) and 21 patients (15%) received two or more than two sequential biological therapies, respectively. The median number of biological therapies per patient was 2 (1–4).
The first biological therapy used was mainly an anti-TNF drug: infliximab (n = 88, 61%), adalimumab (n = 41, 28%), golimumab (n = 3, 2%), vedolizumab (n = 11, 8%) and ustekinumab (n = 2, 1%).
Pouchitis (n = 96, 66%), CDP (n = 42, 29%) and cuffitis (n = 7, 5%) were the primary indications. A total of 83 patients (57%) and 63 patients (43%) had prior exposure to steroids and immunosuppressants, respectively. The drug sequence and the remaining therapy characteristics are presented in Table 2.
Biological therapy use for inflammatory pouch disorders.

Percentages were calculated based on the number of patients receiving each biological therapy.
n = 47 patients.
n = 33 patients.
n = 3 patients.
n = 9 patients.
PDAI, pouchitis disease activity index.
Biological therapy effectiveness
A total of 212 therapies (91 infliximab, 63 adalimumab, 7 golimumab, 31 vedolizumab and 20 ustekinumab) were evaluated at 12 months. The clinical remission, response and non-response rates are presented in Figure 2.

Clinical effectiveness of the biological therapies at 12 months.
No overall differences in clinical remission were observed among patients with inflammatory pouch disorders, except for vedolizumab, which showed better clinical remission rates in pouchitis than CDP (adjusted OR 1.28, p = 0.043). Regarding the type of pouchitis (chronic refractory vs antibiotic-dependent), vedolizumab was the only drug that showed better results for antibiotic-dependent patients (adjusted OR 2.56, p = 0.015; Supplemental Material).
In the case of cuffitis (seven patients), the global rates of clinical remission, response and failure were 43%, 14% and 43%, respectively.
Similarly, no significant differences in clinical remission rates were found across different lines of treatment for each biological agent, except for vedolizumab. Notably, this drug demonstrated higher remission rates when used as a first-line treatment compared to subsequent lines (50% vs 33%, p = 0.05; Table 3).
Clinical remission of biological therapies at 12 months according to line. a

Fisher’s test.
Finally, we found no significant differences in clinical remission rates between monotherapy and combotherapy with either infliximab (43% vs 54%, p = 0.09) or adalimumab (41% vs 48%, p = 0.42).
Treatment durability
The median follow-up time under biological treatment was 15 months (range, 1–183) for infliximab, 13 months (range, 1–118) for adalimumab, 15 months (range, 1–42) for vedolizumab and 21 months (range, 2–63) for ustekinumab.
The overall durability of the different biological treatments is presented in Figure 3. No differences were found in the durability of infliximab, adalimumab or vedolizumab as first-line treatment (log-rank test, p = 0.723; Supplemental Material).

Drug durability for each therapy. (a) Infliximab. (b) Adalimumab. (c) Vedolizumab. (d) Ustekinumab.
A total of 95 therapies (45%) were intensified: 39% of infliximab therapies, 42% of adalimumab, 71% of golimumab, 49% of vedolizumab and 46% of ustekinumab. There was a higher rate of drug intensification in third or higher lines of therapy compared with first or second lines (66% vs 38%, p = 0.03). The incidence rates (% patient-years) of treatment withdrawal were 21% for infliximab, 29% for adalimumab, 27% for vedolizumab and 13% for ustekinumab. The reasons for treatment discontinuation were treatment failure (79%), adverse effects (16%) and sustained remission (5%). Withdrawal rates due to adverse effects were higher in patients treated with infliximab (24%) and golimumab (33%), although no significant differences were found among the therapies (p = 0.34).
Use and effectiveness of a second biological therapy after anti-TNF failure
Sixty patients received a second biological therapy. Of these, 39 (65%) had previously experienced anti-TNF drug failure (adalimumab or infliximab). The second treatment of choice was a second anti-TNF agent in 28 patients – 19 adalimumab, 7 infliximab and 2 golimumab – while 11 patients started a non-anti-TNF drug: 6 vedolizumab and 5 ustekinumab. In addition, 62% had chronic pouchitis and 38% had CDP. The mean duration of treatment was 21 ± 26 months. Due to the small number of patients who received non-anti-TNF therapies, they were analysed as a single group. No differences were found in baseline characteristics (age, smoking status, type of pouch disorder and mean duration of treatment) between the two subgroups.
Overall, 33% of the patients achieved remission and/or responded to treatment, 47% underwent treatment intensification and 72% discontinued the treatment due to failure. The anti-TNF treatment group had a lower remission/response rate (OR 0.16, 95% confidence interval (CI) 0.03–0.72, p = 0.022), a higher failure rate (OR 4.8, 95% CI 2.3–19, p = 0.041) and a higher drug withdrawal rate (OR 5.52, 95% CI 1.94–25.5, p = 0.044; Table 4).
Comparative analysis of second biological therapy after anti-TNF failure.
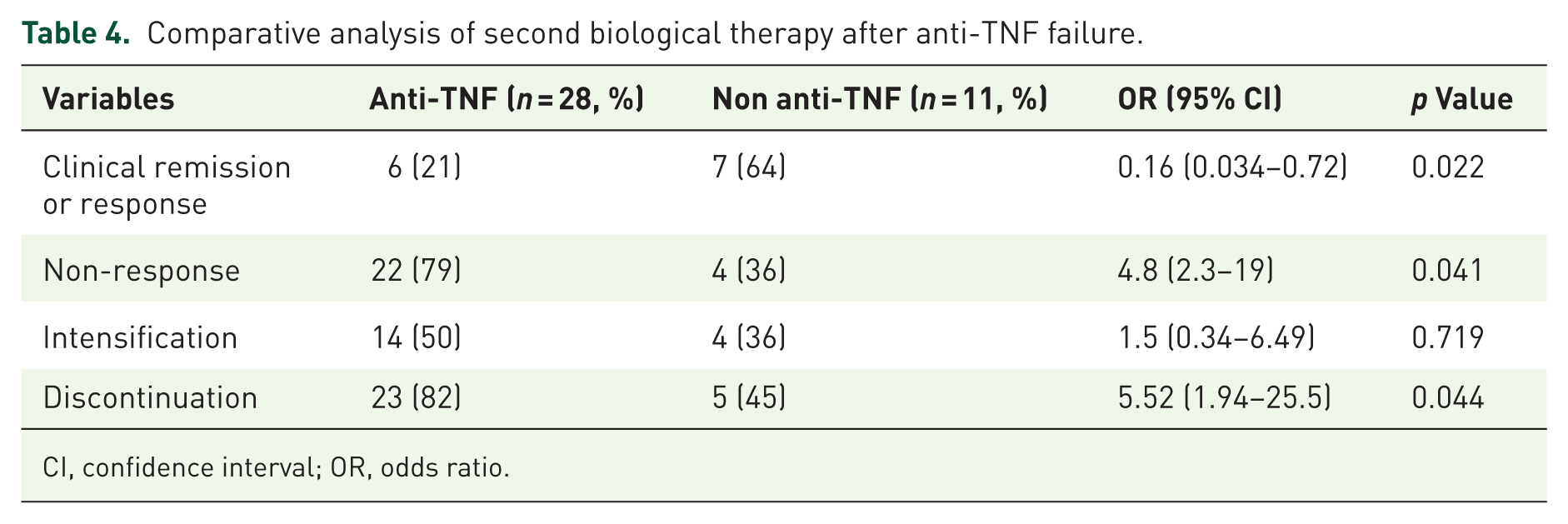
CI, confidence interval; OR, odds ratio.
Surgery treatment and pouch failure
Finally, 96 patients (66%) discontinued at least one biological therapy (mainly due to failure) and 41% used a second biological. Fifty-seven patients (39%) required surgical treatment (80% medical treatment failure, 18% others and 2% pouch adenocarcinoma) after a median time of 65 months (range 3–276).
Surgery included defunctioning ileostomy (40 cases), pouch excision (30 cases) and fistula therapy (29 cases). The pouch failure rate was 24 % (35 cases). We found no significant differences in pouch failure between pouchitis and CDP (26% vs 23%, p = 0.15). Discontinuation of the first biological was associated with an increased risk of requiring surgery (OR 5.34, 95% CI 2.3–12.6, p < 0.001), but the use of a second biological did not significantly increase the risk (OR 1.81, 95% CI 0.9–3.7, p = 0.07). Patients who achieved clinical remission with a second biological exhibited a decreased risk of subsequent surgery (OR 0.66, 95% CI 0.21–0.86, p = 0.001).
Discussion
The RESERVO study is one of the longest-running cohorts of patients diagnosed with pouchitis and other pouch disorders. It has allowed for the extraction of highly relevant data regarding patients’ behaviour, needs and responses to several treatments.5,8
The main results of our study show that different biological therapies are effective for many patients with inflammatory pouch disorders in the medium and long term, regardless of the specific therapy used and even the therapeutic line employed. This is particularly interesting considering the high rate of refractoriness to previous treatments. Our initial data reveal that a significant number of patients require the use of biological therapies for the treatment of these disorders. Barnes et al. 9 reported the use of biological therapies in 60%–70% of patients diagnosed with antibiotic-refractory pouchitis or CDP, highlighting the importance of evaluating the efficacy and use of these treatments in these patients. Unfortunately, there are few studies with a high level of evidence and recommendations that would allow for the positioning of biological treatments in these conditions.10–13 Therefore, we believe that data from retrospective and real-life studies are valuable.
Infliximab has been the most widely used drug for pouchitis. A systematic review published in 2024 analysed infliximab outcomes in 14 studies and 247 patients, mostly with chronic refractory pouchitis. 14 The short-term response rate was 51% while the long-term remission rate (20 months on average) was 42%. These results are similar to those found in our series. Verstockt et al. 22 compared the effectiveness and durability of infliximab, adalimumab and vedolizumab in 33 patients diagnosed with chronic pouchitis and found higher rates of clinical remission and durability in the vedolizumab group over anti-TNF drugs. A notable finding was the high withdrawal rate of infliximab (74%), some of which was due to infusion reactions. This finding has been reported in some other studies, which identified infusion reactions as the main cause of withdrawal.23,24 Although this may be due to prior exposure to the drug (before colectomy), we do not have that information, and further studies are needed to confirm whether this is similar to the scenario of postsurgical Crohn’s recurrence. 25 Our results show similar remission and clinical response rates at 1 year for both therapies. We also found no differences in durability between these drugs in the first-line setting, which may be due to the lower rate of treatment withdrawal due to intolerance or adverse effects.
Adalimumab has also been used for these disorders. Although a clinical trial with 13 patients did not find differences from placebo, there are data in the literature supporting its use. 26 Barreiro-de Acosta et al. 27 published one of the first series in patients with pouchitis and previous failure to infliximab. The 1-year rate of clinical remission was 25%. Our cohort of patients treated with adalimumab is the largest to date and demonstrates that adalimumab offer good effectiveness and durability results (especially in CDP). Golimumab is a drug approved for UC, and data on pouchitis are very scarce (two cases). 28 The present study is the largest to address the effectiveness of this drug in pouchitis.
Vedolizumab is an anti-integrin drug that has shown good results for patients with inflammatory bowel diseases. 29 Several studies have evaluated the short-term effectiveness of the drug in patients with chronic antibiotic-refractory pouchitis.14,30 Our results even exceed those reported to date, 31 which could be attributed to the fact that one-third of our patients received vedolizumab as a first-line treatment and some patients were treated for antibiotic-dependent pouchitis, where we have seen better results. The EARNEST study was the first clinical trial to demonstrate the short-term efficacy of a biological therapy in patients with chronic recurrent pouchitis. 15 In that study, 70% of patients had not received any biological drug after colectomy, and all received antibiotics at the beginning of the study. A post hoc analysis showed that vedolizumab was superior to placebo regardless of antibiotic use. Our results indicate that vedolizumab has long-term effectiveness and achieves better outcomes as first-line therapy and in antibiotic-dependent patients. However, we did not find statistically significant differences from other biological treatments. Currently, the American guidelines include the use of vedolizumab, as it is approved for use in chronic pouchitis, and the IIPC guidelines consider that vedolizumab should be the biological of choice to induce remission in patients with chronic antibiotic-refractory pouchitis.12,13
The available evidence on the use of ustekinumab is scarce, and in our series, this drug was the least commonly used.32–34 However, the effectiveness and durability data are also favourable regarding the use of this drug, even in advanced lines. Weaver et al. 35 evaluated the effectiveness of ustekinumab in 47 patients with CDP refractory to other biological treatments: the clinical remission rate was 36% at 1 year. More studies, particularly on pouchitis, are needed to position this drug.
Another important finding is that we were able to analyse the effectiveness of biological therapies in different disorders. CDP is an infrequent entity with a greater need for biological treatment and a higher rate of pouch failure compared with other pouch pathologies.4,5,7,14,36–38 A systematic review published by Huguet et al. 21 found higher clinical remission rates after induction in patients treated with anti-TNF agents compared to those with pouchitis. Our results did not indicate differences among the different pathologies, except for vedolizumab. Regardless of these considerations, we believe that it is important to emphasise the effectiveness of biological therapy in CDP, even in complex phenotypes, as our series found no significant differences in the need for surgery between the two subgroups, highlighting the need to consider biologics in these patients.
Currently, there are no data on the need for biological treatment in cuffitis and its effectiveness. In our series, seven patients required biologics due to refractoriness to mesalazine and topical corticosteroids. None of these patients had associated pouchitis, and CDP was ruled out in all cases. The results suggest that biological therapy could also be an option in patients who are refractory to standard treatment.
A relevant point of our study is that we were able to analyse the effectiveness of different therapeutic options in subsequent lines. Many of the studies that evaluated the use of adalimumab, vedolizumab or ustekinumab used them in the second or third line but none compared the different possible strategies. Although we were only able to analyse a limited number of patients in this setting, we believe that the results are valuable. Similar to other pathologies, the use of a different mechanism of action after anti-TNF failure should be considered.39–41 We believe that the second treatment should be carefully considered, and we must identify the most effective option because patients who achieved remission with the second treatment were more likely to avoid surgery (18% vs 82%). Nevertheless, 24% of the series experienced pouch failure, which necessitated surgery and a permanent ileostomy. These numbers appear to be higher than those of other studies.4,16,36–38 This can be attributed to many factors, such as a high refractoriness to previous treatments, the prolonged follow-up, the experience of each centre with biological treatment and, above all, the previous lower availability of treatments (more than half of the surgeries were performed before 2012, data not shown). The study conducted by Ricardo et al. 36 found that pouch failure was associated with factors such as the presence of CDP or the use of biologics prior to colectomy. We were unable to analyse the latter factor in our series, although the surgical requirements for CDP were similar to those for pouchitis. New therapies, such as small molecules, are being evaluated with promising results. 42
Our study has several strengths. It is a multicentre study comprising a large number of patients with a very rare pathology that is sometimes only managed in referral centres. This has allowed for the evaluation of a large number of treatments and treatment sequences, which will aid in their positioning. We were also able to evaluate long-term effectiveness, which is interesting because these are chronic pathologies with recurrent symptoms that diminish quality of life. The use of different treatments allowed us to analyse the differences between them and even evaluate sequencing strategies.
However, this study also has limitations such as the retrospective design, the lack of a short-term treatment evaluation and the absence of an objective efficacy assessment through endoscopy. 43 Therefore, we have been unable to detect the primary failure rate. Nonetheless, we believe that long-term evaluation provides more robust data on effectiveness than short-term evaluation. We could not consider endoscopic evaluation as it was conducted (baseline and follow-up) in a small percentage of first-line patients (37% of cases). Endoscopic evaluation could not be considered, as baseline and follow-up endoscopy were performed in only a small proportion of patients (37% of cases). Consequently, activity indices incorporating endoscopic parameters, such as the modified PDAI, were not applied. Moreover, none of these activity indices has been formally validated. Recently, an expert consensus group applied a RAND/UCLA methodology to support clinicians and researchers in assessing pouchitis activity using clinical parameters, such as stool frequency and faecal urgency, in combination with endoscopic and histological findings. 44 However, most studies have evaluated treatment responses through clinical assessment only,13,20 and there are no recommendations for endoscopic response as a therapeutic goal in these patients, mainly because there is no validated definition of response/remission. We also understand that, in patients who have experienced the failure of more than one treatment, the goals remain symptom control or quality of life. Finally, the retrospective nature of the study, the inclusion of different inflammatory disorders and use of treatments in different lines did not allow us to make direct comparisons among the drugs. In addition, there are treatments such as golimumab and ustekinumab, which we have been able to explore only to a limited extent due to the small number of patients.
In conclusion, the use of biological therapies in inflammatory pouch disorders is extensive. Infliximab was the most commonly used treatment, but all drugs show long-term effectiveness and persistence, supporting their use, even after the failure of other biological therapies. Vedolizumab could be positioned as a first-line therapy for pouchitis, especially for antibiotic-dependent cases, based on the available evidence, recent guidelines and our data. However, we must not forget that the failure of biological treatments and the need for surgery remain considerable, and further studies are needed to indicate and position their use.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261422373 – Supplemental material for Biological therapies for inflammatory pouch disorders: insights and outcomes from the RESERVO study of GETECCU
Supplemental material, sj-docx-1-tag-10.1177_17562848261422373 for Biological therapies for inflammatory pouch disorders: insights and outcomes from the RESERVO study of GETECCU by Francisco Mesonero, Yamile Zabana, Agnès Fernández-Clotet, Eduardo Leo-Carnerero, Berta Caballol, Andrea Núñez, María José García, Federico Bertoletti, Guillermo Bastida, Gerard Suris, Begoña Casis, Rocío Ferreiro-Iglesias, Margalida Calafat, Itxaso Jiménez, José Miranda-Bautista, Luis Javier Lamuela, Ingrid Fajardo, Leyanira Torrealba, Rodrigo Nájera, Rosa María Sáiz-Chumillas, Irene González-Partida, Miren Vicuña, Natalia García-Morales, Ana Gutiérrez, Alicia López-García, José Manuel Benítez, Cristina Rubín de Célix, Coral Tejido, Eduard Brunet-Mas, Alejandro Hernandez-Camba, Cristina Suarez Ferrer, Iago Rodríguez-Lago, Marta Piqueras, Andrés Castaño, Laura Ramos, Ana Sobrino, María Carmen Rodríguez-Grau, Alfonso Elosua, Miguel A. Montoro-Huguet, Ruth Baltar, José María Huguet, Benito Hermida, Antonio Caballero-Mateos, Luis Sánchez-Guillén, Abdel Bouhmidi, Ramón Pajares, Iria Baston-Rey, Antonio López-Sanromán, Agustín Albillos and Manuel Barreiro-de Acosta in Therapeutic Advances in Gastroenterology
Footnotes
Appendix
Acknowledgements
The authors thank the Spanish Working Group on Crohn’s Disease and Ulcerative Colitis (GETECCU) for sponsoring this study.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
