Abstract
Background:
The positioning of new biologic agents for the treatment of Crohn’s disease (CD) following failure of initial anti-tumor necrosis factor (anti-TNF) therapy remains a challenge in the real world.
Objectives:
This study aims to investigate the real-world outcomes associated with the sequential use of biologics in CD patients that newly initiate anti-TNFs, specifically comparing those that switch to another anti-TNF versus biologics with other modes of action.
Design:
Retrospective cohort study.
Methods:
We identified CD patients who newly began anti-TNF therapy between 1 October 2014 and 31 December 2018 using two German claims databases. Patients were classified as within-class switchers (WCS) if they switched to another anti-TNF or outside-class switchers (OCS) if they switched to vedolizumab (VDZ) or ustekinumab (UST). To compare WCS and OCS, baseline covariates were adjusted through inverse probability of treatment weighting (IPTW), and time-to-event analyses were performed using Cox Proportional Hazard regressions. Results from both databases were meta-analyzed using an inverse variance model.
Results:
Overall, 376 prevalent adult CD patients who initiated anti-TNFs and switched to another biologic were identified. After IPTW, there were 152 and 177 patients in the WCS and OCS group, respectively. WCS were more likely to receive prolonged corticosteroid therapy [hazard ratio (HR): 1.63, 95% confidence interval (CI): 1.17–2.27, p = 0.004], switch a second time to a different biologic (HR: 2.44, 95% CI: 1.63–3.66, p < 0.001), and discontinue treatment (HR: 1.71, 95% CI: 1.25–2.34, p = 0.001) than OCS.
Conclusion:
This study suggests that CD patients exhibit more favorable outcomes when switching outside the anti-TNF class to VDZ or UST after initial anti-TNF failure than switching to a second anti-TNF. With loss of response to anti-TNFs as a concern in the real world, comparative evidence from claims data assessing sequential use of biologics can help optimize treatment algorithms of patients after anti-TNF failure.
Introduction
Crohn’s disease (CD) is one of the most common types of inflammatory bowel diseases (IBDs), characterized by chronic inflammation of the gastrointestinal tract. 1 With an incidence of 6.6 per 100,000 and a prevalence of 100–200 per 100,000 individuals in Germany, CD can result in a considerably decreased quality of life, disabilities, and surgeries. 2
Due to its lifelong manifestation, therapy for CD aims to treat acute disease or induce clinical remission in the short term and to maintain response and remission in the long term. 3 According to the latest CD guidelines published by the German Society for Gastroenterology, Digestive and Metabolic Diseases, budesonide or steroids are recommended for the induction of clinical remission in patients with mild-to-moderate CD, whereas anti-tumor necrosis factor (anti-TNF) agents such as infliximab (IFX) and adalimumab (ADA) are recommended for induction and maintenance therapy in patients with moderate-to-severely active disease with inadequate response or intolerance to conventional therapy with steroids and/or immunosuppressants.4,5 Although the efficacy of anti-TNFs in inducing and maintaining remission in CD patients has been demonstrated in several clinical trials, treatment failure as a result of adverse drug reactions, lack of primary response, and secondary loss of response, defined as diminished or loss of response over time, remains a significant problem among patients receiving anti-TNFs in clinical practice.6–8 Approximately 10–30% of patients do not respond to the initial anti-TNF treatment during induction in real-world studies, whereas up to 40% of patients lose response within the first treatment year.9,10 In patients with an inadequate response to conventional or anti-TNF therapy, new treatment options are indicated, including the anti-p40 (anti-IL 12/23) antibody, ustekinumab (UST) or vedolizumab (VDZ), an agent targeting leukocyte trafficking. 5 Clinical trial data to support the direct positioning of UST compared to VDZ remain obsolete.
Generally, there is a lack of clinical trials investigating the comparative effectiveness of different biologic agents in CD. 11 In addition, clinical trials are typically not aimed at generating evidence on drug efficacy in a longitudinal fashion, and therefore often do not address the consequences of sequential use of different biologics targeting the same molecular pathway. For instance, while the efficacy of VDZ and UST was investigated in mixed populations with significant numbers of anti-TNF naïve patients, more than 85% of patients treated with VDZ/UST in clinical practice have received prior anti-TNFs.12–15 Real-world observational studies have the potential to close this evidence gap by addressing comparisons which have not yet been explored in the randomized controlled trial setting.
With the absence of interventional clinical trials assessing the sequential use of biologics, this study aims to investigate the real-world treatment outcomes associated with TNF-cycling between patients initiating anti-TNFs and switching within class to a different anti-TNF and those switching outside the anti-TNF class to a different biologic mode of action after a first anti-TNF treatment failure.
Materials and methods
We conducted a retrospective claims data analysis using anonymized data from statutory health insurance funds in Germany, provided by AOK PLUS and GWQ ServicePlus AG and covering approximately 3.6 million and 4.2 million insured patients, respectively. The AOK PLUS dataset consists of all patients insured by the AOK PLUS sickness fund, covering the regions of Saxony/Thuringia, whereas the GWQ dataset consists of data from smaller German-wide sickness funds. Together, both databases are representative of the German healthcare system, covering approximately 11% of the population receiving statutory health insurance. As therapeutic agents prescribed to patients across all healthcare sectors are directly relevant for reimbursement purposes from health insurance companies, claims data offer a complete record of the patient’s treatment history.
Study population
Using the AOK PLUS and GWQ databases, patient information on demographics (age, gender), inpatient care (hospital admission/discharge, procedures), and outpatient care (GP/specialist visits, diagnoses, procedures, prescriptions) was retrieved. A full list of variables collected are found in Supplemental Tables 1 and 2. The study period began on 1 April 2014 and ended on 31 December 2019. As the base sample, all continuously insured adult patients (⩾18 years) with at least one inpatient or two outpatient confirmed diagnoses of CD (ICD-10-GM: K50.-) between 1 October 2014 and 31 December 2018 were included, allowing for a minimum 6-month baseline period and 1-year follow-up period (Supplemental Figure 1). Patients with an inpatient or outpatient diagnosis of ulcerative colitis (UC, ICD-10-GM: K51.-) after the first observed CD diagnosis and those receiving anti-TNF inhibitors in the six months prior to the first observed anti-TNF prescription were excluded. Given the timeframe of data availability, patients were considered anti-TNF naïve according to the absence of anti-TNF treatment in a period of six months before the first anti-TNF in the inclusion period. The final cohort consisted of patients that newly began treatment with anti-TNFs, ADA (ATC L04AB04) or IFX (ATC L04AB02) in the inclusion period after the first observable CD diagnosis and switched to a different biologic agent. Treatment switch was defined as a prescription of a biologic agent different from the index anti-TNF within 180 days of exhaustion of the supply or days covered for the index prescription, and no other prescription of the index anti-TNF until at least 180 days after supply exhaustion. Patients who did not switch to another biologic within the inclusion period were excluded from the final analytical population. Included patients were then classified as either within-class switchers (WCS), patients switching within the anti-TNF class to ADA or IFX, or outside-class switchers (OCS), patients switching to a medication outside the anti-TNF class to VDZ (ATC L04AA33) or UST (ATC L04AC05). The date of first treatment switch was set as the index date and patients were followed for at least one year after treatment switch or longer whenever possible, until the end of the study period on 31 December 2019. Patients were censored at the end of data availability or death.
Outcome measures
The primary outcomes of the study were corticosteroid-free drug survival, time to second treatment switch, and time to treatment discontinuation. Corticosteroid-free drug survival was determined as the time between the index date and start of prolonged corticosteroid therapy, defined by at least two prescriptions of corticosteroids within 12 months, including oral budesonide. Alternative definitions of at least three or at least four prescriptions of corticosteroids within 12 months were tested in sensitivity analyses. Time to second treatment switch was defined as the time between index date and date of a second treatment switch, otherwise initiation of another biologic agent. Finally, time to treatment discontinuation was defined as no prescriptions of a respective biologic agent 60 or 90 days (sensitivity analysis) after the presumed end of drug supply, determined by the quantity and strength of the prescription, assuming that the patient takes the defined daily dose as defined by the World Health Organization. In determining the presumed day supply, stockpiling was accounted for, and it was assumed that patients were covered with medication during any hospitalization periods. For time to treatment discontinuation, treatment switch was considered as a discontinuation event.
Statistical analysis
Due to database regulations and data accessibility, the GWQ and AOK PLUS cohorts were analyzed separately, and outcome results were meta-analyzed. Baseline characteristics were analyzed and reported using descriptive statistics, including frequency distributions for categorical variables and mean, standard deviation (SD) for continuous variables. Differences between WCS and OCS were tested using appropriate parametric or nonparametric tests including chi-squared tests and t-tests. To adjust for differences in baseline variables between WCS and OCS, inverse probability of treatment weighting (IPTW) was used. Propensity score weighting was utilized instead of matching due to smaller sample sizes among the study groups. Inverse probability weights were estimated using a logistic regression model with a binary indicator of whether the patient was WCS or OCS, using all relevant baseline covariates based on 12 months before the index date. The covariates included in the logistic regression in both databases are shown in Supplemental Tables 3 and 4, with respective c-statistics indicating appropriate model fit. To meet a common support assumption, we trimmed extreme values of the distribution. The quality of adjustment achieved was assessed by comparing weighted cohorts in terms of their baseline characteristics. Time-to-event analyses for primary and secondary outcomes were then conducted using inverse propensity score weighted Kaplan–Meier and Cox Proportional Hazard regression.
For meta-analyses of AOK PLUS and GWQ data, cumulative distributions were reported for categorical variables and combined mean and SD were reported for continuous variables with pooled p-values, respectively. Hazard ratios (HRs) of time-to-event outcomes from AOK PLUS and GWQ analyses were meta-analyzed using both a fixed-effect inverse variance model (Mantel-Haenszel) and random-effects inverse variance model with DerSimonian–Laird estimate of tau-squared (sensitivity). The Q statistic of I2 was used to determine statistical heterogeneity by describing the percent variation across the two analyses. An I2 value >50% implied significant statistical heterogeneity. Test p values of <0.05 implied statistical significance. All analyses were performed in STATA (Version 14.2).
Reporting
The reporting of this study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology statement for cohort studies (Supplemental Table 6). 16
Results
Patient selection
Between 1 October 2014 and 31 December 2018, 34,556 CD patients were identified (39.4% AOK PLUS and 60.6% GWQ). Of these, 8,060 (43.8% AOK PLUS and 56.2% GWQ) were excluded due to a double diagnosis of CD and UC. Overall, 25,339 prevalent adult CD patients (38.4% AOK PLUS and 61.6% GWQ) were included. The final analytical cohort comprised of 376 (112 AOK PLUS and 264 GWQ) continuously insured patients who newly started treatment with anti-TNFs and switched to a different biologic agent. Approximately 47.3% (178/376) of patients were classified as WCS, switching to either IFX (34 AOK PLUS and 52 GWQ) or ADA (19 AOK PLUS and 73 GWQ). Among the 198 total OCS, 119 patients switched to VDZ (55.9% in AOK PLUS and 61.9% GWQ), whereas 79 switched to UST (44.1% in AOK PLUS and 38.1% in GWQ). The detailed flow of patient selection is shown in Figure 1.

Patient selection flow chart.
Baseline characteristics of patients prior to IPTW are shown in Supplemental Table 5. Prior to weighting, patients collectively had a mean age at index of 39.4 years for OCS (N = 198) and 36.6 years for WCS (N = 178), and approximately 63.1% of OCS and 60.1% of WCS were female (Supplemental Table 5). In the overall cohort, 45.4% of OCS and 42.7% of WCS received corticosteroids, approximately a third of patients received antibiotics (30.8% OCS and 34.3% WCS), and 18.2% of OCS and 20.2% of WCS received immunosuppressants during the 6-month baseline period. Furthermore, 11.1% of OCS and 10.1% of WCS had a history of cardiovascular disease. In the GWQ cohort, there were no significant differences in baseline characteristics between OCS and WCS, whereas significant differences were observed in age at index (39.6 versus 32.2 years, p < 0.001) and number of patients with a Charlson Comorbidity Index (CCI) score of 2 indicative of a mild comorbidity grade (13.6% versus 1.9%, p = 0.023; calculated as per Supplemental Table 2) in the AOK PLUS cohort among OCS and WCS (Supplemental Table 5).
After IPTW, the cohorts were restricted to 97 patients (55.7% OCS) from AOK PLUS and 232 patients (53.0% OCS) from GWQ. The propensity score weight distribution for both cohorts, stratified by WCS and OCS, is shown in Supplemental Figures 2 and 3. After weighting, characteristics of WCS and OCS were well balanced (Table 1). In the AOK PLUS cohort, the cohorts were better balanced after IPTW, with no statistically significant differences between OCS and WCS in CCI score of 2 (6.7% versus 1.1%, p = 0.068) and an improved distribution of age at index (38.8 versus 34.1 years, p = 0.012). Although age remained a significantly different factor among OCS and WCS in the AOK PLUS population, sensitivity analyses were conducted where age was controlled for within the weighted Cox regressions (double robust approach). Controlling for age did not affect the conclusions of this paper. In this study, CD patients are not shown separated by primary non-response and secondary loss of response due to small sample sizes in the primary non-response group given stratification via available real-world definitions, with primary response defined as treatment switch within four or six months (sensitivity) of initial anti-TNF initiation. However, additionally controlling for response did not affect the conclusions of this study.
Balance of covariates in AOK PLUS and GWQ cohorts before and after IPTW, stratified by OCS and WCS.

CCI, Charlson Comorbidity Index; CD, Crohn’s disease; GP, general practitioner; IPTW, inverse probability of treatment weighting; OCS, outside-class switchers; SD, standard deviation; UST, ustekinumab; VDZ, vedolizumab; WCS, within-class switchers.
Study outcomes
Corticosteroid-free drug survival
Altogether, 42.4% (75/177) of OCS (44.4% in AOK PLUS and 41.5% in GWQ) received at least two prescriptions of corticosteroids within 12 months, compared to 57.9% (88/152) of WCS (65.1% in AOK PLUS and 55.0% in GWQ). WCS were more likely to receive prolonged corticosteroid therapy than OCS both in the AOK PLUS [HR: 1.92, 95% confidence interval (CI): 1.03–3.59; Figure 2, panel a] and GWQ populations (HR: 1.53, 95% CI: 1.04–2.26; Figure 2, panel b). The risk of prolonged corticosteroid therapy in the two databases was 63% higher for WCS compared to OCS (pooled HR: 1.63, 95% CI: 1.17–2.27; Figure 2, panel c). In sensitivity analyses, prolonged corticosteroid therapy defined as at least three and four prescriptions of corticosteroids within 12 months after switch delivered qualitatively consistent results, while not statistically significant due to sample sizes (results not shown).

Time to corticosteroid-free drug survival. Kaplan–Meier curve and Cox Proportional Hazard models of time to corticosteroid-free drug survival for AOK PLUS (a) and GWQ (b) cohorts comparing WCS and OCS. Patients were censored at the end of data availability or death. HRs with 95% CI represent differences between the groups. Forest plots indicate the meta-analyzed results using a fixed-effects inverse-variance model (c).
Time to second treatment switch
Specifically, 22.0% (39/177) of OCS (20.4% in AOK PLUS and 22.8% in GWQ) switched a second time to a different biologic agent compared to 42.8% (65/152) of WCS (44.2% in AOK PLUS and 42.2% in GWQ). In both databases, WCS were significantly more likely to switch a second time to another biologic after the first treatment switch than OCS (p = 0.025 AOK PLUS; p < 0.001 GWQ; Figure 3, panels a and b). Meta-analyzed, the overall HR for WCS in time to second treatment switch was 2.44 (95% CI: 1.63–3.66, p < 0.001; Figure 3, panel c). Out of 104 total patients that had a second treatment switch, 51.9% switched to UST (60.0% in AOK PLUS and 48.6% in GWQ), 28.8% to VDZ (23.3% in AOK PLUS and 31.2% GWQ), and 19.2% to anti-TNFs (16.7% in AOK PLUS and 20.3% in GWQ).
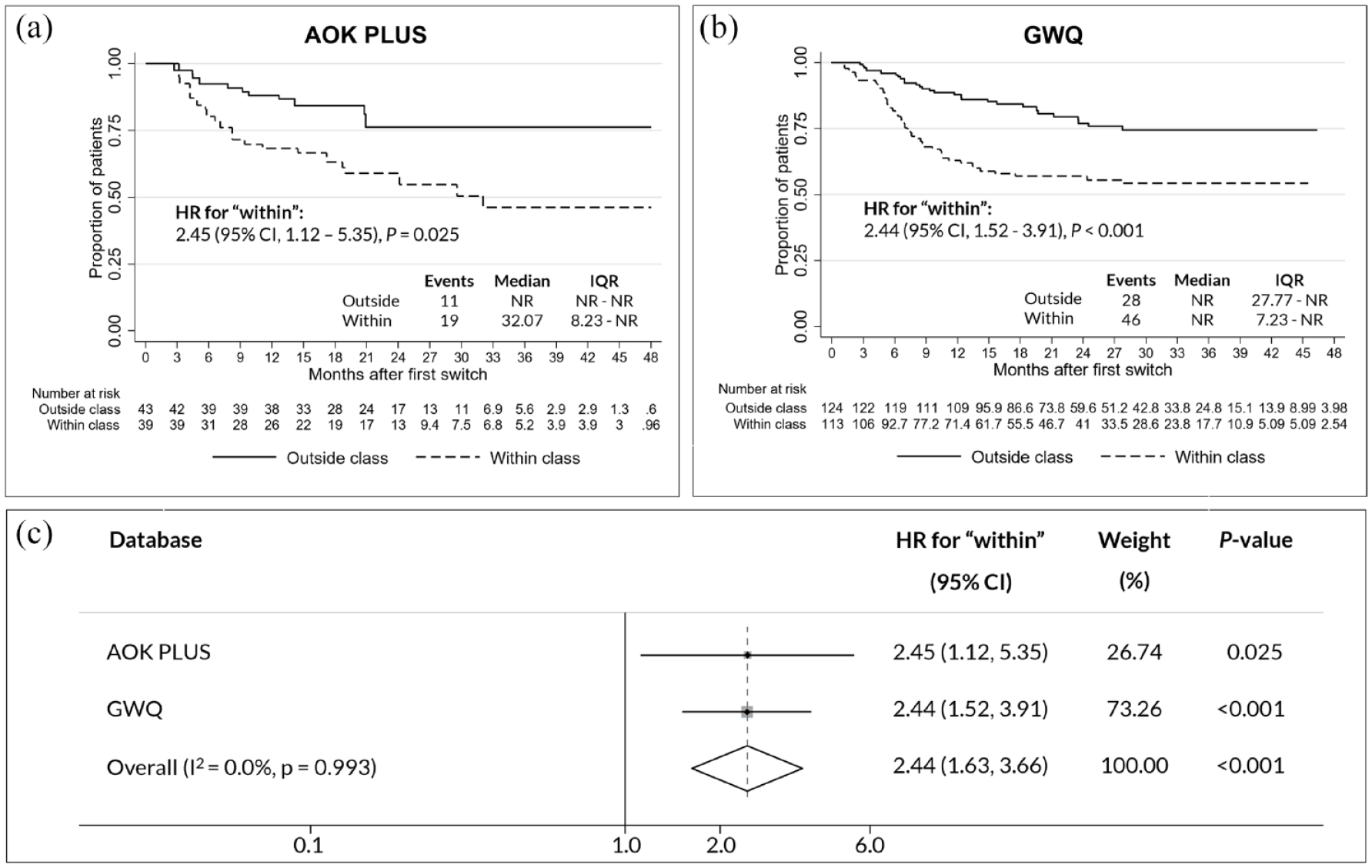
Time to second treatment switch. Kaplan–Meier curve and Cox Proportional Hazard models of time to second treatment switch for AOK PLUS (a) and GWQ (b) cohorts comparing WCS and OCS. Patients were censored at the end of data availability or death. HRs with 95% CI represent differences between the groups. Forest plots indicate the meta-analyzed results using a fixed-effects inverse-variance model (c).
Time to treatment discontinuation
Of OCS, 42.4% (38.9% in AOK PLUS and 43.9% in GWQ) discontinued their index treatment in contrast to 61.2% of WCS (60.5% in AOK PLUS and 61.5% in GWQ). WCS were more likely to discontinue the index treatment compared to OCS, with an overall HR of 1.71, 95% CI, 1.25–2.34 (p = 0.001; Figure 4, panel c). WCS showed a tendency of an 83% (AOK PLUS) and 67% (GWQ) increased likelihood to discontinue the index treatment compared to OCS, although likely due to lack of statistical power; the effect observed among the AOK PLUS population was not statistically significant (p = 0.052) (Figure 4, panels a and b). These results were confirmed in sensitivity analyses, whereby discontinuation was defined based on a 90 day gap after presumed end of drug supply (pooled HR for WCS: 1.68, 95% CI: 1.22–2.30, p = 0.001; Supplemental Figure 4).

Time to treatment discontinuation. Kaplan–Meier curve and Cox Proportional Hazard models of time to treatment discontinuation for AOK PLUS (a) and GWQ (b) cohorts comparing WCS and OCS. Patients were censored at the end of data availability or death. HRs with 95% CI represent differences between the groups. Forest plots indicate the meta-analyzed results using a fixed-effects inverse-variance model (c).
Discussion
The positioning of different biologic agents in the treatment algorithms of CD patients is a challenging process that requires head-to-head comparative evidence. This study aimed to assess the real-world outcomes of CD patients in Germany associated with the sequential use of different biologics (biologic cycling), specifically comparing patients that initiate anti-TNFs and switch within the anti-TNF class to another anti-TNF (WCS) or those that switch outside-class to VDZ or UST (OCS). Our data indicate that the real-world outcomes of CD patients may improve if patients switch outside the anti-TNF class to VDZ or UST rather than a second anti-TNF after initial anti-TNF failure.
As interventional studies directly comparing the use of different biologics after a first anti-TNF treatment failure in CD are not available, the evidence is limited to observational studies in the real world. Several real-world evidence studies have been conducted to determine the comparative effectiveness of initiating therapy with a different anti-TNF or another mode of action.17–19 The results from these studies are difficult to contextualize, as the target populations included mixed population of patients with and without prior anti-TNFs. In UC, a few real-world studies comparing VDZ and anti-TNFs suggest that VDZ may have a superior effect after initial anti-TNF failure compared to a second anti-TNF, whereas a systematic review and network meta-analysis comparing various biologics found that UST and tofacitinib performed best in patients with prior anti-TNF exposure.20–22 Evidence comparing similar populations in CD is limited, highlighting the significance of this comparative study using claims data from a large cohort of patients treated in the real-world setting.
This study compared corticosteroid-free drug survival, time to treatment discontinuation, and second treatment switch of patients switching within-class or outside-class based on outpatient prescriptions coded in German claims data. Due to the regulations of the AOK PLUS and GWQ databases used in this study, the two datasets could not be combined and analyzed at patient level. Therefore, the populations were analyzed separately, and results were meta-analyzed using a fixed effects inverse variance model due to overall low heterogeneity between studies. No differences were observed when testing a random effects model to pool outcomes, indicating that the observed effects were robust. Overall, we observed that patients initiating anti-TNFs and switching to a second anti-TNF (WCS) were associated with inferior outcomes when compared to patients switching outside class to VDZ or UST (OCS) in corticosteroid-free drug survival (HR for WCS: 1.63, 95% CI: 1.17–2.27, p = 0.004), time to second treatment switch (HR: 2.44, 95% CI: 1.63–3.66, p < 0.001), and treatment discontinuation (HR: 1.71, 95% CI: 1.25–2.34, p = 0.001) (Figures 2–4).
Corticosteroid-free drug survival is an important endpoint for the treatment of CD patients in clinical practice. In this study, the endpoint was based on a recurrent use of steroids (prolonged corticosteroid and/or budesonide therapy), defined as the prescription of at least two corticosteroids within 12 months after anti-TNF treatment in an attempt to achieve disease control. 23 Alternative definitions requiring at least three and four corticosteroid prescriptions within 12 months were tested in sensitivity analyses. Although not statistically significant due to lower sample sizes, the results of sensitivity analyses were qualitatively consistent, with WCS showing a higher tendency to receive prolonged corticosteroid therapy than OCS. Furthermore, the definitions for treatment switch and discontinuation were based on prior literature.24,25 Our results showed that WCS were more likely to discontinue treatment than OCS whether treatment discontinuation was defined based on 60 or 90 days after presumed end of drug supply (60 days: p = 0.001, 90 days: p = 0.001; Figure 4). Overall, the initiation of prolonged corticosteroid therapy, second treatment switch, and treatment discontinuation (including second treatment switch) serve as an indication for failure of the second anti-TNF or outside class biologic.
In contrast to our findings, a Swedish registry-based study on IBD patients exposed to one prior anti-TNF concluded that the clinical effectiveness and safety of switching to a second anti-TNF was similar to switching to VDZ in CD patients (drug survival: 74% anti-TNFs versus 73% VDZ), with overall no significant differences in drug survival between various switching strategies (81% IFX to ADA, 75% IFX to VDZ, 71% ADA to IFX, and 71% ADA to VDZ, p = 0.64). 26 However, it is important to note that this study did not consider patients switching to UST, and the lack of significant differences between the individual switching strategies may have been due to low statistical power. The importance of investigating switching to UST as part of an OCS comparator group can be highlighted by three observational studies comparing VDZ and UST among patients with prior anti-TNF failure, which showed that UST was associated with superior effectiveness, and higher rates of clinical remission and treatment persistence than VDZ.27–29 As opposed to our study population, these studies also included mixed populations of patients that received prior treatment with one or two anti-TNFs.
Overall, our findings indicate that the clinical efficacy of patients that are treated with and discontinue an initial anti-TNF in clinical practice based on a proxy of corticosteroid-free drug survival, second treatment switch, or treatment discontinuation may improve if patients receive either VDZ or UST as a second-line biologic instead of a second anti-TNF. The results of this study highlight the need for comparative evidence in a controlled setting to address the sequential use of biologics after initial anti-TNF therapy failure to further guide treatment of CD patients in clinical practice.
With almost 86% of the German population insured through statutory health insurance and uniform healthcare regulations and policies across Germany, this study made use of two claims databases providing a representative population of patients across Germany and allowed for the identification of treated CD patients free of study site or selection bias. Despite this, there were several limitations to this study. Although IPTW was used to control for a range of patient characteristics, this study was non-randomized and claims data based. Therefore, potential unobserved confounders may still exist, including disease severity indicators, disease duration, and prescribed dosages for medications, which are not available in claims data. In addition, the reason for discontinuation or switch as a result of primary non-response or secondary loss of response is an important distinction for determining the choice of a second biologic agent. As the reason for discontinuation or switch is not available in claims data, we used a proxy to control for our results by grouping patients as having primary non-response if treatment switch occurred within four or six months, and as secondary loss of response if switching occurred after four or six months. As previously mentioned, CD patients in this study are not shown separated by primary non-response and secondary loss of response as this resulted in a small group of primary non-responders based on real-world definitions, indicating that our results may be driven by secondary loss of response as a result of ineffective treatment. Nevertheless, when stratifying the analyses using an indicator for primary non-response or secondary loss of response in sensitiviy analyses, the results were consistent with our main study conclusions. This is in contrast to the German guidelines for the treatment of CD, which suggest a second anti-TNF in the event of secondary loss of response among patients with anti-TNF antibodies, given that patients initially responded to the first anti-TNF and potentially had a diminished or loss of response due to lower anti-TNF concentrations as a result of antibodies targeted against the drug. 4 This phenomenon can be confirmed via Therapeutic Drug Monitoring, which is not available in German claims data.
Furthermore, both the AOK PLUS and GWQ datasets consist of data from routine medical practice and may therefore exhibit a degree of missing data and coding error with regard to outpatient diagnoses. Despite this, the data are considered to be of high quality and valid.30–32 In addition, the data do not reflect all potential medical interventions endured by a patient, potentially excluding holistic or other interventions used in clinical practice which do not qualify for medical claims. Specific to the selection criteria, we included patients without prior anti-TNFs in a 6-month baseline period. If patients had anti-TNFs before this 6-month period, they would have been included as anti-TNF initiators despite potential prior anti-TNF therapy. Notably, while the study does not describe concomitant treatment with immunosuppressants, which is a factor that may influence treatment effectiveness, the overall use of concomitant immunosuppressants was low among the overall population, and relatively balanced among WCS and OCS in both datasets, indicating that concomitant use of immunosuppressants among the two groups likely does not account for the significance in outcomes observed. Furthermore, we acknowledge that discontinuation analyses were based on presumed days of supply and could not adjust for dosing assumptions based on body weight, adding uncertainty on prescription gaps. However, when stratifying results for time to discontinuation between patients who had switched to ADA versus IFX, we did not find any significant differences, suggesting gaps for IFX were likely not significantly under- or over-estimated.
Conclusions
In conclusion, our data suggest that in clinical practice, CD patients that are treated with one anti-TNF and switch outside the anti-TNF class to VDZ or UST exhibit superior corticosteroid-free drug survival, and are less likely to switch biologics a second time and discontinue treatment than patients who initiate a second anti-TNF. With loss of response to anti-TNFs as a concern in the real world, comparative controlled clinical studies investigating the sequential use of biological agents in patients failing a first anti-TNF therapy are needed to guide recommendations and optimize treatment algorithms in CD patients.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221130554 – Supplemental material for Real-world outcomes associated with switching to anti-TNFs versus other biologics in Crohn’s Disease patients: A retrospective analysis using German claims data
Supplemental material, sj-docx-1-tag-10.1177_17562848221130554 for Real-world outcomes associated with switching to anti-TNFs versus other biologics in Crohn’s Disease patients: A retrospective analysis using German claims data by Evi Zhuleku, Beatriz Antolin-Fontes, Andras Borsi, Riikka Nissinen, Ivana Bravatà, Jennifer Norma Barthelmes, Manuela Le Bars, Jennifer Lee, Alun Passey, Ulf Maywald, Barthold Deiters, Bernd Bokemeyer, Thomas Wilke and Marco Ghiani in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
