Abstract
Background:
Clinical trials have demonstrated the efficacy and safety of ustekinumab in Crohn’s disease (CD). However, more data are necessary on the effectiveness of ustekinumab in bio-naïve patients in real-life studies.
Objectives:
The aim of our study was to evaluate the effectiveness and safety of ustekinumab in patients with CD refractory or intolerant to conventional therapy and without previous exposure to biological drugs.
Design:
We performed a nationwide, observational, retrospective, multicentre study including patients with CD, in which ustekinumab was used as the first biological drug.
Methods:
The corticosteroid-free clinical and biological response and remission were analysed at weeks 16, 24, 52 and 72. Clinical remission was defined as Harvey–Bradshaw index ⩽ 4 and biological remission as a faecal calprotectin (FC) <250 mg/g and C-reactive protein (CRP) <5 mg/L. Moreover, the persistence of the treatment and any adverse events were assessed.
Results:
In all, 84 patients were included in the study, males and females were equally distributed, with a median age of 63 years [interquartile range (IQR): 51–75] and a median disease duration of 6.8 years [IQR: 3.6–17.0]. The majority (86.9%) of patients were treated with ustekinumab as monotherapy, without concomitant immunosuppressive medication. The proportion of patients in corticosteroid-free clinical remission or response at weeks 16, 24, 52 and 72 was 93.3% (56/60), 86.8% (46/53), 82.2% (37/45) and 71.4% (30/42), respectively. CRP returned to normal values in 47.6%, 43.2%, 50% and 52.4% of patients at weeks 16, 24, 52 and 72, respectively. Similarly, FC was normalized in 45.5%, 45.5%, 48.6% and 50% of patients at weeks 16, 24, 52 and 72, respectively. The cumulative probability of remaining on ustekinumab treatment was 84.8% (95% confidence interval: 73.3–91.6) after 72 weeks. Ustekinumab was discontinued in 10 patients (11.9%) within 72 weeks of follow-up. Reasons for discontinuing treatment were lack of response (n = 4), adverse events (n = 4) and death (n = 2). There were no discontinuations because of stable remission.
Conclusions:
Ustekinumab was effective and safe in Spanish bio-naïve CD patients, showing a quicker and more durable response than obtained in patients with previous biological treatment. In this cohort of bio-naïve patients starting on ustekinumab, the average age was high.
Plain language summary
Evidence on the use of ustekinumab in biological naïve real-world patients is scarce. Here, we present real-world data evaluating the effectiveness and safety of ustekinumab in 84 bio-naïve patients from 17 Spanish hospitals. We report high rates of both clinical and biological remission. Moreover, after 1 year, 90.4% of patients remained being treated with ustekinumab. The safety profile of ustekinumab in these patient population was favourable. In conclusion, our results show that in patients with CD, ustekinumab could be considered as first-line therapy.
Introduction
Ustekinumab is a fully human IgG1к monoclonal antibody that binds with high specificity to the cytokines interleukin (IL)−12 and IL-23. 1 It has been approved in Spain since 2016 for the treatment of moderate to severe active Crohn’s disease (CD) in adult patients who have had an insufficient response or loss of response (LOR) to antitumor necrosis factor (TNF) agents, who cannot tolerate conventional therapy or anti-TNF therapy, and for whom conventional therapy or anti-TNF therapy is contraindicated.2,3
The efficacy of ustekinumab was evaluated in two phase III induction trials (UNITI-1 and UNITI-2) and one phase III maintenance study (IM-UNITI) that included responders from UNITI-1/2. 4 All these studies demonstrated the efficacy and safety of ustekinumab for patients who were refractory to other biological treatments (UNITI-1), but also for patients that had failed conventional therapy (UNITI-2).
Data from the literature on strategies to position biological agents are scarce. Guidance and consensus statements exist to help aid the decision-making process. However, there is a tendency to position all biologicals on the same level and to consider them as interchangeable in the European Crohn’s and Colitis Organisation Guidelines.5,6
In the most recent publication from the American Gastroenterology Association: ‘Technical Review, the Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn’s Disease’, the question of comparing the efficacy between all available biologicals is addressed. 7 The conclusion was that in biological-naïve patients with moderate to severely active CD, infliximab, adalimumab and ustekinumab are the most effective compared to vedolizumab and certolizumab pegol. Infliximab was considered the strongest for induction.
A large number of real-life studies have been published so far, confirming the efficacy and safety of ustekinumab in routine practice. However, the vast majority of patients included in these cohorts had previously failed one or several biologicals.8–19
It is important to continue to collect real-life data not only in bio-failure but also in bio-naïve patients, to compare response rate and safety in these two populations and to look for predictive factors of response.
Therefore, the aim of our study was to evaluate the short- and long-term effectiveness and safety of ustekinumab in patients with CD refractory, intolerant or contraindicated to conventional therapy (steroids and/or immunosuppressants) and without previous exposure to biological drugs.
Methods
Study design and patient population
This study was a nationwide, observational, retrospective, multicentre study (17 Spanish hospitals), whose aim was to assess the effectiveness, safety and persistence of ustekinumab in bio-naïve CD patients. Adult (⩾18 years) CD patients who started ustekinumab as the first biological between August 2017 and November 2021 were included. All patients signed the written informed consent before study initiation.
Patients were treated with at least two doses of ustekinumab, an initial intravenous (IV) induction dose of 6 mg/kg followed by a subcutaneous (SC) dose of 90 mg at week 8 and subsequent SC administration of 90 mg every 8 (q8w) or every 12 weeks (q12w) based on clinical judgement. Baseline demographic data, concomitant medication and Montreal classification details were collected at each hospital by review of electronic medical records. Data on clinical and objective markers of disease activity were assessed at baseline, and at weeks 16, 24, 52 and 72 (±4 weeks). Changes in ustekinumab dosing, concomitant medication, surgical intervention and adverse events were collected regardless of their timing.
The study was approved by the local ethics committee. Every medical record was analysed and details de-identified so that the identity of the patients could not be determined in any way, building an anonymous database.
Outcomes and definitions
The primary objective of this study was to determine the effectiveness and safety of ustekinumab in bio-naïve patients in routine practice, and to identify predictors of ustekinumab response. Effectiveness outcomes included were corticosteroid-free clinical remission and response, biochemical remission and response, and ustekinumab treatment persistence. In addition, predictors of corticosteroid-free clinical remission at week 52 were investigated.
Clinical effectiveness was only assessed in patients with active clinical disease at baseline [Harvey–Bradshaw index (HBI) > 4]. Clinical remission was defined as HBI ⩽ 4, and clinical response was defined as a decrease in HBI of at least 3 points from baseline without remission. Objective response and remission were evaluated using faecal calprotectin (FC) and C-reactive protein (CRP), FC and CRP normalization were considered when levels were <250 mg/kg and <5 mg/L, respectively. Patients who discontinued ustekinumab due to lack of effectiveness or adverse events were considered as treatment failure. Patients with insufficient follow-up or with no data for a specific visit were censored for that time point.
Treatment persistence with ustekinumab was also evaluated. The parameters analysed as predictors of clinical remission were gender, age at diagnosis, duration of disease, smoking habit, CD location, CD behaviour, perianal disease, extra-intestinal manifestations, baseline immunosuppressant and/or steroid use, prior intestinal resection, baseline HBI, baseline CRP and baseline FC.
Adverse events appearing during ustekinumab treatment were recorded to evaluate the safety profile of ustekinumab in real life.
Statistical analysis
Demographic data and baseline disease characteristics were reported for all patients. Categorical variables were presented as percentages and continuous variables were presented as mean with standard deviation or as median with interquartile range (IQR) depending on the normality of the underlying distribution. Normal distribution was determined using the Kolmogorov–Smirnov normality test. Statistical differences for HBI, FC and CRP were assessed versus baseline using the Wilcoxon rank-sum test. Overall drug persistence was assessed by Kaplan–Meier. Multivariate analysis was performed on variables with p < 0.2 on univariate analysis using backward stepwise logistic regression. A two-sided p value of 0.05 or less was considered statistically significant. All data analyses were performed using IBM SPSS Statistics for Windows, version 24.0. (IBM).
The reporting of this study conforms to the STROBE statement 20 (Supplemental Appendix 1).
Results
Baseline characteristics
Patients with CD who had received ustekinumab as the first biological agent after failure, intolerance or contraindication to conventional treatment (steroids and/or immunosuppressants) were included in the study. In all, 84 patients met the selection criteria, patients reaching each follow-up visit are depicted in Figure 1.

Flow chart of patients included in the study.
Males and females were equally distributed, with a median age of 63 years (IQR: 51–75) and a median disease duration of 6.8 years (IQR: 3.6–17.0). The majority (63.1%) of patients had ileal disease and an inflammatory disease phenotype (61.9%). At baseline, 18 patients (21.4%) experienced extra-intestinal manifestations, the most reported was arthralgia (Table 1). The majority (86.9%) of patients were treated with ustekinumab as monotherapy, without concomitant immunosuppressive medication (Table 1).
Demographic and clinical characteristics.

At inclusion.
Maximum extent until inclusion.
Available for 62 patients.
Available for 82 patients.
CRP, C-reactive protein; FC, faecal calprotectin; GI, gastrointestinal; HBI, Harvey–Bradshaw index; IQR, interquartile range.
At baseline, clinical disease activity (HBI > 4) was seen in 60/84 patients (71.4%) and the median HBI was 6 (IQR: 4–8), median CRP concentration at baseline was 5.1 mg/L (IQR: 1.5–13.0) and a median FC level of 852 mg/kg (IQR: 341–1800) (Table 1).
In the 24 patients who initially had an HBI ⩽ 4, ustekinumab was started because 13 patients had FC > 250, 5 patients had active perianal disease, 4 patients had extraintestinal manifestations and 2 patients had prior intestinal resections.
Clinical outcomes
Clinical remission without concomitant corticosteroids was calculated in all patients (n = 84) and reported separately for patients with HBI > 4 (n = 60) or HBI ⩽ 4 at baseline (n = 24). For those with clinical activity at baseline, the proportion of patients in corticosteroid-free clinical remission at weeks 16, 24, 52 and 72 was 80.0% (48/60), 77.4% (41/53), 80.0% (36/45), 71.4% and (30/42), respectively. Corticosteroid-free clinical response was calculated in patients not in clinical remission and was achieved by 13.3% (8/60) at week 16, 9.4% (5/53) at week 24, 2.2% (1/45) at week 52 and 0% (0/42) at week 72. The proportion of patients in corticosteroid-free clinical remission or response at weeks 16, 24, 52 and 72 was 93.3% (56/60), 86.8% (46/53), 82.2% (37/45), and 71.4% (30/42), respectively (Figure 2). Corticosteroid-free clinical remission was also evaluated among patients already in clinical remission (HBI ⩽ 4) at baseline (n = 24), all of which (100%) remained in corticosteroid-free clinical remission for the entire duration of the study.

Proportion of patients with CS-free clinical response and remission. Clinical response was defined as a decrease in HBI of at least 3 points from baseline without remission in patients not in clinical remission. Clinical remission was defined as HBI ⩽ 4. Numbers within bars indicate the percentage of patients in CS-free clinical remission or response.
A significant reduction in HBI score was observed for all patients, from a median HBI of 6 at baseline to a median HBI of 2 at weeks 16, 24, 52 and 72 (p < 0.001) (Figure 3(a)). In the 24 patients who initially had a HBI ⩽ 4, median baseline HBI was 3 and this continued to decline further after ustekinumab initiation (p < 0.05) (Figure 3(b)).

Median HBI with IQR over time. For all patients (n = 84) (a) and for patients with HBI ⩽ 4 at baseline (n = 24) (b). Comparisons between baseline and 16, 24, 52 and 72 weeks were performed using the Wilcoxon matched rank test.
At baseline, ustekinumab was given with concomitant corticosteroids in 22 patients (25.9%), 19 out of 22 (86.4%) were able to discontinue corticosteroid after a median time of 8 weeks (IQR: 7.8–10).
During the maintenance phase, no patient used additional corticosteroids and the 11 patients who were treated with concomitant immunosuppressive medication at baseline maintained it until the end.
Biochemical outcomes
Biochemical disease activity was assessed in patients with available CRP or FC data and with abnormal values at baseline. CRP values were altered at baseline in 42 out of 82 patients with available data (52.2%), in these patients, CRP returned to normal values in 47.6% (20/42), 43.2% (16/37), 50% (14/28) and 52.4% (11/21) of patients at weeks 16, 24, 52 and 72, respectively (Figure 4(a)). Similarly, FC values were altered in 51/62 (82.3%) patients with available data at baseline and were normalized in 45.5% (20/44), 45.5% (20/44), 48.6% (17/35) and 50% (13/26) of patients at weeks 16, 24, 52 and 72, respectively (Figure 4(b)).

Biochemical response in patients with available and altered baseline values. Proportion of patients with CRP normalization (<5 mg/L) (a) and with FC normalization (<250 mg/kg) (b).
Median CRP levels decreased significantly between baseline and weeks 16, 24, 52 and 72 in the 82 patients with available CRP levels at baseline. CRP levels dropped in the first 16 weeks of treatment and this reduction was maintained over time. The median CRP concentration at weeks 0, 16, 24, 52 and 72 was 5.1 mg/L (IQR: 1.5–12.6), 2.9 mg/L (IQR: 0.9–5.9), 2.0 mg/L (IQR: 0.86–6.6), 2.0 mg/L (IQR: 0.7–6.0) and 2.0 mg/L (IQR: 0.6–4.0), respectively (Figure 5(a)). A similar trend was observed for FC in the 62 patients with available data at baseline, FC was significantly reduced at all timepoints compared to baseline, median FC markedly dropped at week 16 and those levels were maintained for the rest of the follow-up period. The median FC level at weeks 0, 16, 24, 52 and 72 was 851.5 mg/kg (IQR: 341.3–1800.0), 223 mg/kg (IQR: 115.5–565.0), 231 mg/kg (IQR: 139.0–714.5), 233.5 mg/kg (IQR: 103.5–841.3) and 191.5 mg/kg (IQR: 83.25–698.2), respectively (Figure 5(b)).

Median CRP concentration with IQR over time (a). Median FC concentration with IQR over time (b). Comparisons between baseline and 16, 24, 52 and 72 weeks were performed using the Wilcoxon matched rank test.
Clinical factors associated with corticosteroid-free clinical remission
Predictors of clinical remission at weeks 16, 24 and 52 were investigated using univariate and multivariate analyses. In the multivariate analysis at week 16, no clinical factors were found to be associated with clinical remission (Supplemental Table 1). At week 24, male sex was associated with lower rates of clinical remission [odds ratio (OR): 0.13; 95% confidence interval (CI): 0.02–0.88] and younger age at onset (A2 in Montreal classification) associated with higher rates of clinical remission versus older age at onset (A3) (OR: 12.65; 95% CI: 1.22–131.62) (Supplemental Table 2). At week 52 only, disease location was found statistically significant in the multivariate analysis, with less probability of clinical remission in ileocolonic (L3) versus ileal (L1) involvement (OR: 0.05; 95% CI: 0.003–0.70) (Supplemental Table 3).
Ustekinumab drug survival
The cumulative probability of remaining on ustekinumab treatment was 98.8% (95% CI: 91.7–99.8) at week 16, 94.9% (95% CI: 86.9–98.0) at week 24, 90.4% (95% CI: 80.8–95.3) at week 52 and 84.8% (95% CI: 73.3–91.6) after 72 weeks (Figure 6). Ustekinumab was discontinued in 10 patients (11.9%) within 72 weeks of follow-up. Reasons for discontinuing treatment were lack of response (n = 4), adverse events (n = 4), and death (n = 2). There were no discontinuations because of stable remission.
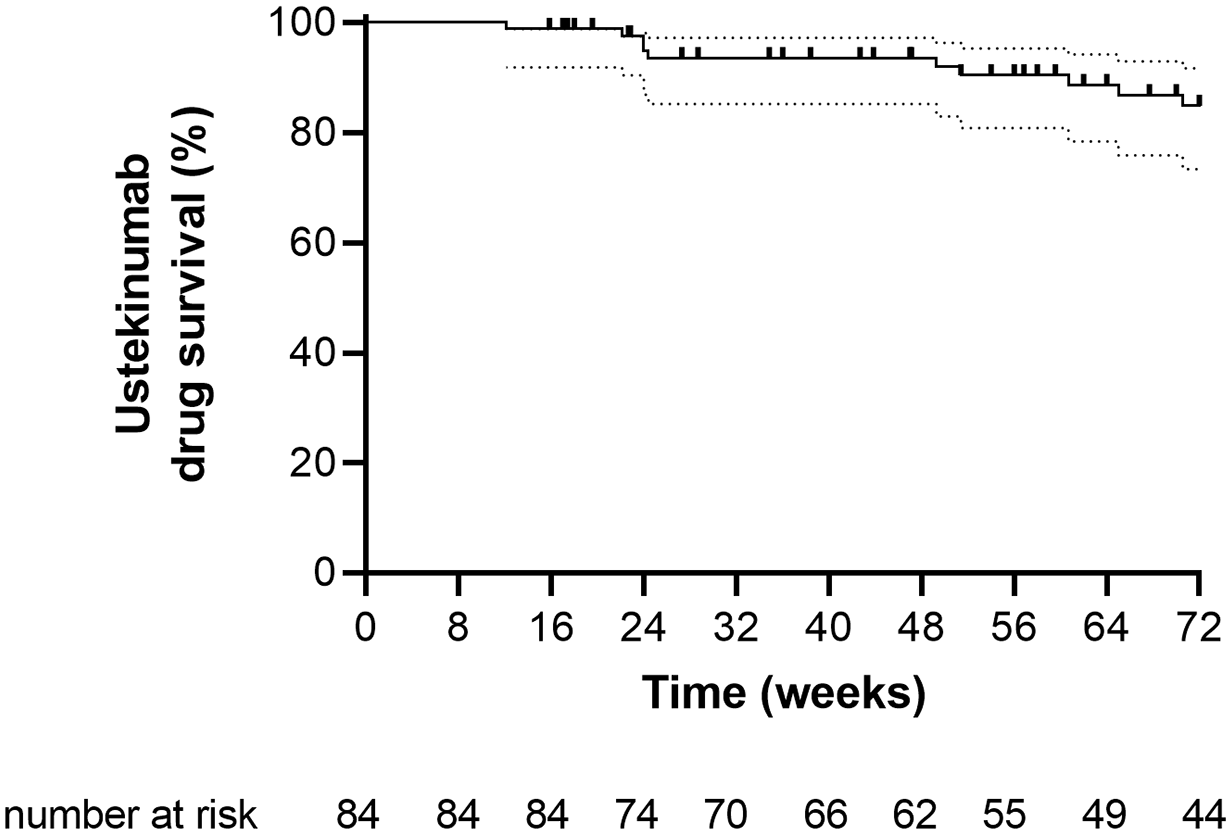
Probability of ustekinumab drug survival expressed as a Kaplan–Meier curve with 95% CI.
Maintenance regimen
All patients received the standard IV induction with 6 mg/kg ustekinumab and a first SC dose of 90 mg at week 8, during follow-up different dosing regimens were administered at the discretion of the treating physician. At the start of maintenance, 11 patients (13.1%) received 90 mg every 12 weeks (q12w), 72 patients (85.7%) 90 mg q8w and 1 patient (1.2%) 90 mg q4w. During follow-up, dose interval with 90 mg SC was reduced to q8w in 1 patient (1.2%), to q6w in 8 patients (9.5%) and to q4w in 13 patients (15.5%). In addition, one patient (1.2%) was escalated to maintenance with 130 mg IV q4w (Figure 7). In total, during the 72 weeks of follow-up, standard dosing was escalated in 23 patients (27.4%) after a median time of 24 weeks (IQR: 18–36).

Ustekinumab dosing schedule during maintenance. Shaded boxes represent dosing regimens different from approved standard dosing.
Effectiveness in patients who did not undergo dose escalation
Clinical remission without concomitant corticosteroids was also calculated in patients with baseline HBI > 4 and treated with the doses recommended in the summary of product characteristics (q8w and q12w). The proportion of patients in corticosteroid-free clinical remission under standard regimen at weeks 16, 24, 52 and 72 was 79.7% (47/59), 79.6% (39/49), 81.1% (30/37) and 76.5% (26/34), respectively.
Safety profile
The entire cohort of 84 patients was included in the safety analysis. During the 72-week follow-up period, 10 adverse events were reported in nine patients (10.7%), four of which led to treatment discontinuation, one uveitis recurrence, one infection, one infusion reaction and one adenocarcinoma of the ileum. The remaining six adverse events not requiring treatment discontinuation were headache, infection, arthralgia, dyspnoea, cutaneous rash and odynophagia.
Two patients died during follow-up. A 91-year-old female patient was treated with ustekinumab for 70 weeks and died of pneumonia secondary to COVID-19 infection. And a 63-year-old female patient, who was treated for 24 weeks and had multiple cardiovascular risk factors, died of acute mesenteric ischemia.
Ten patients who started treatment with ustekinumab had a history of neoplasms (two colon cancer, two breast cancer, two melanomas, one bladder cancer, one prostate cancer, one astrocytoma and one tongue cancer). No tumour recurrence was observed in any of them. Four patients who started treatment with ustekinumab had a history of congestive heart failure and no adverse events were observed.
During 72 weeks of follow-up, seven patients (8.2%) required surgery, three because of strictures, two for perianal disease, one for enterocutaneous fistula and one for adenocarcinoma of the ileum.
Discussion
We evaluated real-world effectiveness and safety of ustekinumab treatment for CD patients naïve to biological therapy in a retrospective multicentre study in Spain. To our knowledge, this is the real-world study with the highest number of patients published to date analysing the response rate in bio naïve CD patients treated with ustekinumab.
Our study is a multicentric one that included 84 bio-naïve patients with moderate to severe CD from 17 Spanish hospitals. Our results show that 80% of patients with clinical activity (HBI > 4) at baseline achieved steroid-free clinical remission at week 16 and 71.4% maintained it at week 72. Ustekinumab persistence was also high, with 84.8% of patients remaining on treatment after 72 weeks. These findings are showing high rates of remission and persistence could be considered positive and novel as no other study have reported these results. A large number or studies have reported effectiveness of ustekinumab in the real-world setting; however, these cohorts mainly include bio-experienced patients and in most cases, they did not report outcomes specifically in bio-naïve patients. In these refractory population, rates of clinical remission after 1 year range from 39% to 64%.9,11,14,18,19 In a Spanish study by Iborra et al., 14 from ENEIDA registry, a total of 463 patients were included, 96.5% of them had received prior biological therapy. At week 52, 64.4% had clinical remission; however, when analysing rates of clinical remission according to the number of previous anti-TNF agents, a trend towards better results in bio-naïve patients (n = 18) was observed. 83% of bio-naïve patients were in clinical remission at 1 year, whereas 79%, 68%, 63% and 50% of patients were in remission when exposed to 1, 2, 3 or 4 previous anti-TNFs, respectively.
Pivotal studies demonstrated higher rates of clinical remission in conventional therapy-failure patients (UNITI-2 study) compared with bio-failure patients (UNITI-1 study). After induction, the rates of clinical remission with ustekinumab 6 mg/kg at week 8 were 37.8% versus 57.9% in UNITI-1 and UNITI-2, respectively. This difference was also observed in the maintenance study IM-UNITI, patients treated with ustekinumab q8w from the UNITI-1 population showed remission rates of 41.1%, whereas those from UNITI-2 achieved remission in 62.5% of the cases. 4
Three real-world studies have reported effectiveness specifically in bio-naïve patients treated with ustekinumab. A study from Brazil by Parra et al. 21 reported effectiveness of ustekinumab in 245 patients, most (86.5%) had been previously exposed to biologicals and 33 were bio-naïve. The proportion of bio-naïve patients in clinical remission at weeks 8, 24 and 56 were 36.4%, 54.5% and 39.4%, respectively. In this study, no significant differences in either clinical or biological remission were noted between bio-naïve and bio-experienced patients. Consistent with this finding, a recent Belgian study by Monin et al. 22 evaluated the short- and long-term response to ustekinumab in both bio-naïve (n = 35) and bio-failure (n = 113) CD patients and found that previous biological failure did not influence the initial response or treatment persistence; however, they noted a trend towards better biological response in bio-naïve versus bio-failure patients. Conversely, a study from Canada by Sedano et al. 23 found higher rates of clinical remission and endoscopic healing in bio-naïve (n = 48) versus bio-failure (n = 181) patients. Rates of clinical remission at 3 and 6 months for bio-naïve patients were 79.4% and 71.4%, respectively. Rates of remission in this study were much higher than those reported in the Brazilian study by Parra et al. 21 and very similar to the results reported in our study.
In our study, 35.7% of patients needed dose escalation and regained clinical remission. This is similar to other studies. Yang et al. 24 demonstrated that primary responders with CD experienced LOR to ustekinumab at a risk of 21% per person-year and required dose escalation at a risk of 25% per person-year and 28% of secondary non-responders with CD may benefit from dose escalation.
Older age is an important indication for ustekinumab as first-line biological due to its favourable safety profile and its efficacy as monotherapy. In our study, the median age of the patients was 63 years. It is also important to note that in Spain ustekinumab is not frequently used as first-line biological, due to reimbursement policy. Therefore, it is frequently positioned after treatment with an anti-TNF biosimilar and only as a first line in certain patients (elderly patients or patients with comorbidities that prevent the use of an anti-TNF drugs). This is one of the reasons why the average age of our patients is high. Based on its safety profile, ustekinumab is considered as first-line treatment in fragile patients. In a recent published study, 25 elderly patients, older than 65 years (n = 39), had predominantly moderate disease, and compared to non-elderly, elderly patients were less likely to achieve complete clinical remission (28.2% versus 52.6%, p = 0.01). These results are clearly different to ours, perhaps, because our population is bio-naïve. In another Spanish study from the ENEIDA registry by Casas-Deza et al. 26 published in 2022 and including a considerable number of patients, the effectiveness and safety of ustekinumab were assessed in elderly (>60 years, n = 212) versus young (<60 years, n = 436) patients. Every elderly patient was matched with two young controls under 60 years of age, according to anti-TNF use and smoking habit; however, comorbidities and rate of complicated disease (structuring or fistulizing) were more common in elderly patients. Rates of steroid-free remission were found to be similar in young versus elderly patients at 1 year (57.8% versus 51.1%, p = 0.21). Moreover, there was no difference in the rate of adverse effects.
Safety is one of the main attributes of ustekinumab. In our study, only 10 adverse events were reported during follow-up, and only four of them led to treatment discontinuation. Of note, 10 patients (13%) had a previous history of malignancy, and none of them showed tumour recurrence. Our results are in agreement with those of other studies, even when including a younger population.9–19,21–23 In a recent published study, ustekinumab was associated with reduced risk of infections compared to anti-TNF biologicals in inflammatory bowel disease. 27 In the IM-UNITI study and long-term extension (LTE), 28 adverse event rates (per 100 patient-years) from maintenance week 0 through the final visit generally were similar in the placebo and combined ustekinumab groups for all adverse events (440.3 versus 327.6), serious adverse events (19.3 versus 17.5), infections (99.8 versus 93.8) and serious infections (3.9 versus 3.4). It is remarkable that in these studies, the population is younger than in our study and without history of malignancy.
This multicentre real-life study has several strengths. First, all patients in our cohort had not been previously exposed to any biological agent. Furthermore, most patients had long-standing disease (median duration, 10 years) and only 50% never had smoked. Second, the effectiveness and safety of ustekinumab were evaluated until week 72, unlike in other real-life studies, which evaluated patients for shorter periods of time.
The study also has some limitations that should be considered when interpreting the findings. The retrospective design of the study could have led to overestimation of the positive response rate and underestimation of AEs. Endoscopic or magnetic resonance data were not available for the assessment of mucosal healing or improvement. FC data were only available for a limited number of patients. Effectiveness on extraintestinal manifestations or perianal disease was not assessed. Finally, the median age of our cohort was high and may be not representative of other age groups.
In summary, our findings confirm the effectiveness, a quite high persistence rate and a good safety profile of ustekinumab in Spanish bio-naïve CD patients. Consequently, ustekinumab could be considered as a first-line treatment for patients with moderate to severe CD. However, prospective, and head-to-head studies are needed to be able to establish sequencing and positioning of available therapies.
Supplemental Material
sj-docx-2-tag-10.1177_17562848231153560 – Supplemental material for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study
Supplemental material, sj-docx-2-tag-10.1177_17562848231153560 for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study by Valdés Delgado Teresa, Olmedo Martín Rául, Iborra Marisa, Herrera de Guisé Claudia, Fuentes-Valenzuela Esteban, Melcarne Luigi, Martín-Rodríguez Mª Mar, Kolle Casso Lilyan, De Castro Parga Luisa, Ponferrada Díaz Ángel, Vicente Lidón Raquel, Manceñido Marcos Noemí, Velayos Jiménez Benito, Lázaro Sáez Marta, López Cauce Beatriz, Mesonero Gismero Francisco, Gilabert Álvarez Pau and Argüelles-Arias Federico in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848231153560 – Supplemental material for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study
Supplemental material, sj-docx-3-tag-10.1177_17562848231153560 for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study by Valdés Delgado Teresa, Olmedo Martín Rául, Iborra Marisa, Herrera de Guisé Claudia, Fuentes-Valenzuela Esteban, Melcarne Luigi, Martín-Rodríguez Mª Mar, Kolle Casso Lilyan, De Castro Parga Luisa, Ponferrada Díaz Ángel, Vicente Lidón Raquel, Manceñido Marcos Noemí, Velayos Jiménez Benito, Lázaro Sáez Marta, López Cauce Beatriz, Mesonero Gismero Francisco, Gilabert Álvarez Pau and Argüelles-Arias Federico in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-1-tag-10.1177_17562848231153560 – Supplemental material for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study
Supplemental material, sj-jpg-1-tag-10.1177_17562848231153560 for Effectiveness and safety of ustekinumab in bio-naïve Crohn’s disease patients: a multicentre observational retrospective study by Valdés Delgado Teresa, Olmedo Martín Rául, Iborra Marisa, Herrera de Guisé Claudia, Fuentes-Valenzuela Esteban, Melcarne Luigi, Martín-Rodríguez Mª Mar, Kolle Casso Lilyan, De Castro Parga Luisa, Ponferrada Díaz Ángel, Vicente Lidón Raquel, Manceñido Marcos Noemí, Velayos Jiménez Benito, Lázaro Sáez Marta, López Cauce Beatriz, Mesonero Gismero Francisco, Gilabert Álvarez Pau and Argüelles-Arias Federico in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
