Abstract
Background:
Interventional endoscopic ultrasound (I-EUS), including EUS-guided biliary drainage, is now widely performed, but it becomes challenging if the visibility of devices is inadequate. A novel visibility enhancement mode, termed “Accent mode,” has recently become available for use with a fluoroscopic system.
Objective:
To compare the visibility of each device and pancreato-biliary ducts between Accent mode and Original mode during I-EUS.
Design:
A single-center non-randomized evaluation study.
Methods:
Patients who underwent I-EUS under Accent mode were prospectively enrolled. All evaluations were performed using recorded procedural videos. The visibility score was graded on a five-item scale, with evaluations performed by three experts and seven trainees.
Results:
Twenty patients (Accent group) and 24 patients (Original group) were enrolled. Mean guidewire visibility scores were significantly higher in the Accent group (4.95 and 4.95 in the expert and trainee observers, respectively) than in the Original group (2.53 and 2.32, respectively;
Conclusion:
In conclusion, Accent mode appears to reduce I-EUS procedure time. It is necessary to confirm these findings in a prospective, randomized, controlled trial.
Introduction
Interventional endoscopic ultrasound (I-EUS), including EUS-guided biliary drainage (BD), pancreatic duct drainage (PD), and gallbladder drainage (GBD), is now widely performed in patients for whom standard treatment techniques such as endoscopic retrograde cholangiopancreatography (ERCP) have been unsuccessful.1–4 I-EUS technique can be divided into several steps; that is, puncturing, guidewire deployment, tract dilation, and stent deployment. According to several meta-analyses, developments in devices and techniques have led to a reduction in the rate of adverse events from 23.3% to 13.7%,5,6 and various efforts aiming to improve the technical success rate have been reported.7–15 Despite these advances, the procedure remains challenging in the case of inadequate visibility on fluoroscopic imaging of the pancreato-biliary tract or of devices such as guidewires, dilation devices, or stents. To date, no study has attempted to improve the success rate by increasing visibility on fluoroscopic imaging.
A novel visibility enhancement mode, termed “Accent mode,” has recently become available for use with a fluoroscopic system (Astorex i9; Canon Medical Systems, Kanagawa, Japan) but has not yet been evaluated in clinical practice. This study aimed to compare the visibility of each device and the pancreato-biliary ducts between Accent mode and Original mode during I-EUS.
Patients and methods
Patients
Patients who underwent I-EUS under Accent mode between April and August 2024 were prospectively enrolled. As a historical control group, also enrolled were patients who underwent I-EUS under Conventional mode between January 2023 and March 2024. The inclusion criteria were as follows: (1) age >18 years, (2) unsuccessful ERCP, and (3) performance status 0–2. The exclusion criteria were as follows: (1) presence of ascites, (2) bleeding tendency, (3) use of a lumen-apposing metal stent, and (4) refusal to participate in the study. The study was conducted according to the tenets of the Declaration of Helsinki for biomedical research involving human subjects, and all patients provided written informed consent. A priori approval was given by the Human Research Committee of Osaka Medical and Pharmaceutical University (IRB No. 2024-080). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Techniques for I-EUS and devices
To exclude bias due to operator experience, EUS-HGS Endoscopic ultrasound was performed by one experienced endoscopist (T.O.) who has performed more than 700 I-EUS procedures. After inserting the echoendoscope (UCT260; Olympus, Tokyo, Japan or EG-740UT; Fujifilm, Tokyo, Japan) was inserted into the stomach or duodenum. After identifying the target lesion, such as an intrahepatic bile duct or pancreatic duct, evaluation using color Doppler was performed to avoid internal vessel injury. The target lesion was then punctured using a 19-G needle (EZ Shot 3 Plus; Olympus), after which a 0.025-inch guidewire (J-Wire; JMIT, Shiga, Japan; Capella; Japan Lifeline, Tokyo, Japan; Endoselector; Boston Scientific, Tokyo, Japan; or Fielder; Olympus) was inserted into the duct. Before self-expandable metal stent (SEMS) placement, tract dilation was performed using a 4-mm balloon catheter (REN biliary dilation catheter; KANEKA, Osaka, Japan) or drill dilator (Tornus ES; Asahi Intecc, Aichi, Japan). The SEMS (HANARO Benefit; M.I. Tech., Seoul, South Korea; Spring Stopper; TaeWoong Medical Co. Ltd, Seoul, South Korea) was then deployed from the duct to the stomach or duodenum using the intra-scope channel release technique, as described previously. 16 Post-procedural computed tomography (CT) was obtained the day after I-EUS to evaluate adverse events such as stent dislocation or migration. In our clinical practice, to prevent cholangitis or pancreatitis due to increased ductal pressure, the dose of contrast medium was around 5 ml per procedure.
Evaluation of visibility during I-EUS
Each procedure was evaluated using recorded procedural videos. Figure 1 shows Accent mode imaging (Supplemental Video 1; https://d.kuku.lu/px4hvcxw4). Needle visibility was evaluated immediately after duct puncture. After contrast medium injection, guidewire visibility was evaluated during guidewire manipulation (Figure 1(a)). Visibility of the dilation device was evaluated during tract dilation (Figure 1(b)), and that of the SEMS was evaluated during SEMS deployment (Figure 1(c)). Figure 2 shows Original mode imaging (Supplemental Video 2; https://d.kuku.lu/cyy6wuvw8). All steps were evaluated in the same way as for Accent mode. The visibility score was graded on a five-item scale (Excellent, Favorable, Good, Acceptable, and Poor), scored as follows. “Excellent,” the device was clearly identified during the entire procedure step; “Favorable,” the device was identifiable for 90% of the time during the procedure step; “Good,” the device could be identified for 80% of the time during the procedure step; “Acceptable,” the device was identifiable for 70% of the time during the procedure step; and “Poor,” visibility <70%. Evaluations were performed independently by three experts (S.U., A.O., and N.N.) and seven trainees (J.S., T.K., Y.U., M.T., N.H., J.N., and K.B.). “Expert” was defined as having performed at least 32 I-EUS procedures, as described previously. 17 In the video evaluations, each expert and trainee received a randomly selected video of the procedure and independently assessed the visibility score for each of the needle, guidewire, and dilation device. This step was repeated until all videos had been evaluated.

EUS-HGS under Accent mode. (a) After successful bile duct puncture, a 0.025-inch guidewire is inserted. (b) Tract dilation is performed using a 4-mm balloon catheter. (c) The stent delivery system is successfully inserted into the biliary tract.

EUS-HGS under original mode. (a) After successful bile duct puncture, a 0.025-inch guidewire is inserted. (b) Tract dilation is performed using a 4-mm balloon catheter. (c) The stent delivery system is successfully inserted into the biliary tract.
Outcome measurements and definitions
The primary outcome was the visibility score for each device. The secondary outcomes were technical success rate, procedure time, and adverse events associated with the procedures. Technical success was defined as successful stent deployment from the target site to the stomach or duodenum. Procedure time was measured from scope insertion to successful stent deployment. Peritonitis was diagnosed if fever, elevated inflammatory markers on blood examination, and abdominal pain were observed the day after I-EUS. On the post-procedural CT, peritonitis was defined as bile or pancreatic juice leakage or peritonitis observed around the stent. Intraoperative bleeding events were defined as puncture-site hematoma, continuous bleeding that required endoscopic and/or intravenous and/or surgical hemostasis around the puncture site, or bleeding on pancreatocholangiography. Postoperative bleeding events were defined as bleeding requiring blood transfusion, melena, hematemesis, bleeding confirmed on CT, or a decrease in hemoglobin ⩾2 g/dL. Adverse events were graded according to the severity grading system of the American Society for Gastrointestinal Endoscopy lexicon. 18 Because obesity can affect visibility on the fluoroscopic view, the body mass index (BMI) was calculated. As the presence of contrast medium can affect device visibility, the volume of contrast medium used during I-EUS was also recorded.
Descriptive statistics are presented as the mean ± standard deviation (SD) or median and range values for continuous variables and as frequencies for categorical variables. The two study arms were compared using analysis of variance for continuous factors, the Kruskal–Wallis test for number of events, and Pearson’s Chi-squared test or Fisher’s exact test for categorical factors. All data were statistically analyzed using SPSS version 13.0 statistical software (SPSS, Chicago, IL, USA).
Results
During the study period, 20 patients (median age, 74 years; 9 males, 11 females) were prospectively enrolled in the Accent group, and 24 patients (median age 77 years; 13 males, 11 females) were enrolled in the historical control group (Original group). Table 1 shows the patients’ characteristics. There was no significant difference in mean BMI between the Accent (23.3 ± 4.9 kg/m2) and Original groups (24.1 ± 4.1 kg/m2;
Patients’ characteristics.
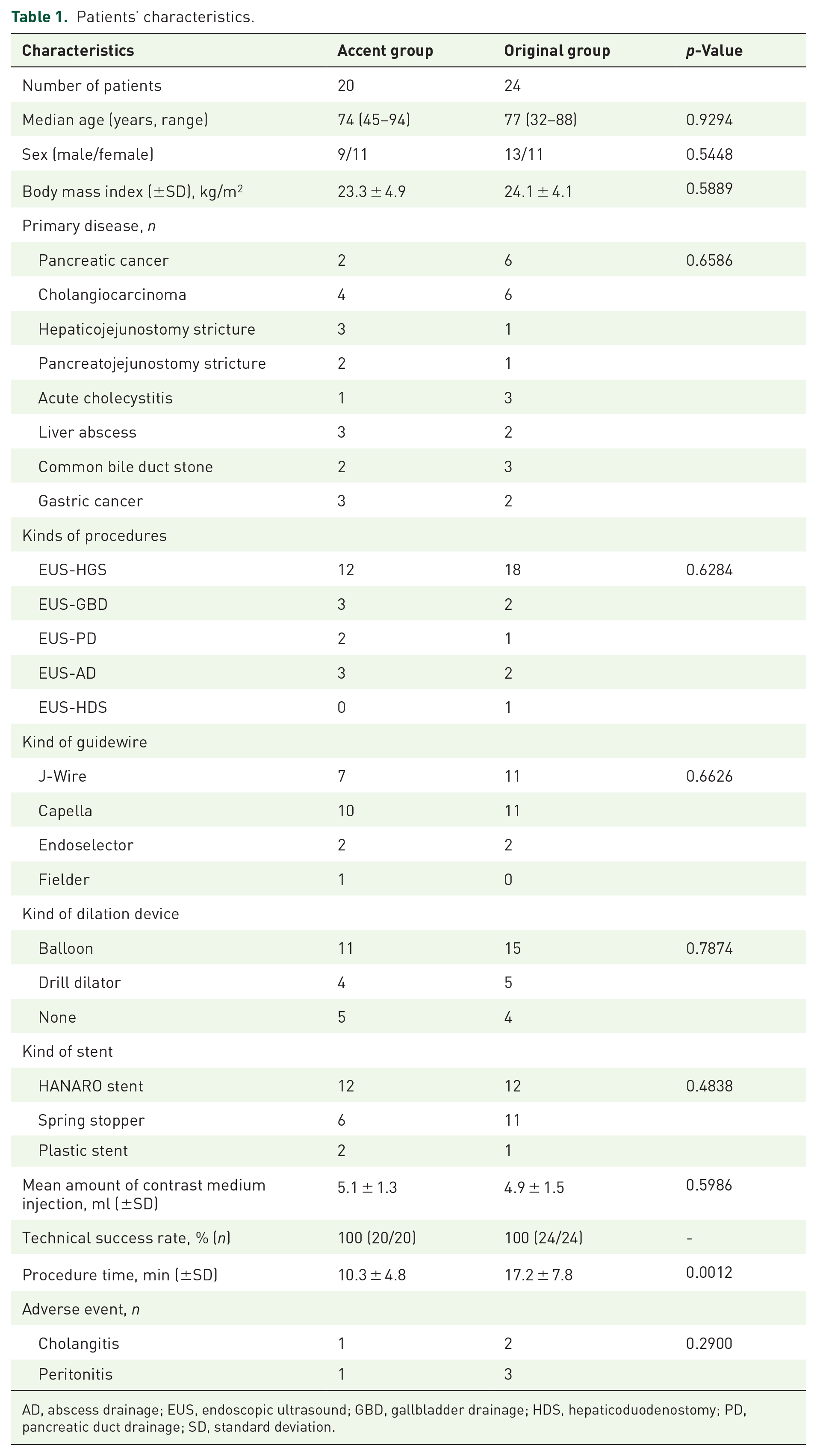
AD, abscess drainage; EUS, endoscopic ultrasound; GBD, gallbladder drainage; HDS, hepaticoduodenostomy; PD, pancreatic duct drainage; SD, standard deviation.
The visibility scores for all devices are shown in Table 2. There was no significant difference in needle visibility score between the Accent and Original groups for expert or trainee observers (
Visibility scores for each device.

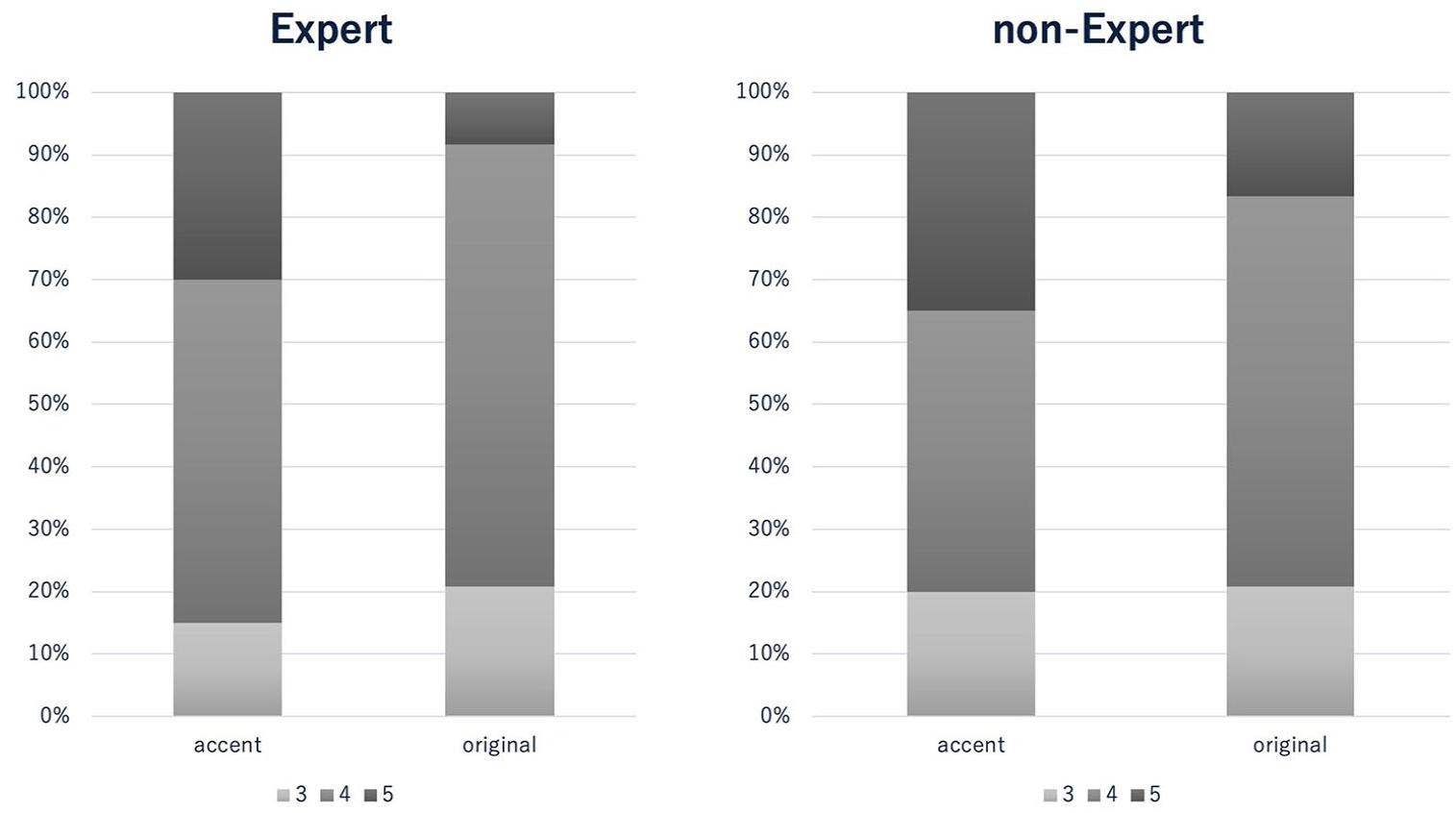
There is no significant difference in needle visibility score between the Accent and Original groups by the expert and trainee observers (

Mean guidewire visibility scores are significantly higher in the Accent group (4.95 and 4.95 in the expert and trainee observers, respectively) than in the Original group (2.53 and 2.32, respectively;

For the dilation device, visibility scores are significantly higher in the Accent group (4.47 and 4.58 in the expert and trainee observers, respectively) than in the Original group (2.68 and 2.53, respectively;

Stent visibility scores were significantly higher in the Accent group (4.16 and 4.32 in the expert and trainee observers, respectively) than in the Original group (2.89 and 2.68, respectively;
Discussion
Numerous studies have reported efforts to improve the success rate of I-EUS procedures, particularly EUS-BD. However, these have focused mainly on optimizing techniques or devices. The present study may be the first to approach improvement from the perspective of evaluating visibility during fluoroscopy. Needles, guidewires, dilation devices, and stents are utilized in the various steps of I-EUS, and the visibility of each affects the success of the procedure. Among these devices, needles are highly radio-opaque, and it is not necessary to improve their visibility under fluoroscopy. Indeed, in the present study, needle visibility was the same in the Accent and Original groups. However, the visibility of other devices showed significant differences between the two groups.
First, guidewire visibility is crucial during its manipulation for deployment at the appropriate site. According to a national survey conducted in Spain of EUS-BD and PD performed by non-experts, 19 manipulation of the guidewire inside the duct was a critical limiting step, accounting for 28 of 41 failures. Therefore, improving the technical success rate of guidewire manipulation will have a clinical impact on I-EUS. Various methods have been reported for improving the technical success rate of guidewire insertion and manipulation, including our previous report describing the clinical usefulness of a liver impaction technique. 15 Of the total of 166 patients enrolled in that study, initial successful guidewire insertion without using the liver impaction technique was obtained in 108 patients (65.1%). All 166 patients underwent EUS-HGS, but guidewire insertion or manipulation initially failed in 58 patients. The reasons for failed guidewire insertion included guidewire advancement into the periphery of the bile duct in 32 patients (55.2%) and into a not-of-interest bile duct branch in 26 patients (44.8%). However, the use of the liver impaction technique improved the technical success rate of guidewire insertion from 65.1% to 95.8%. Matsubara et al. 20 described techniques to manipulate guidewires toward the hepatic hilar region using an uneven ERCP catheter or rotatable sphincterotome to redirect the guidewire. In another study that reported initial experience with a steerable access needle during EUS-BD, 21 both needle puncture and selective wire advancement in the intended direction were successful in 100% of cases (22/22). Although these techniques might be useful, guidewire visibility under EUS can be challenging if the bile duct is not very dilated. In this situation, guidewire visibility might be important because the procedures are performed under fluoroscopic guidance.
Second, optimal visibility of the dilation device may help prevent inadequate tract dilation. To date, various dilation devices have been used. Honjo et al.
22
conducted a comparison study between an ultra-tapered mechanical dilator, which had favorable visibility under fluoroscopy in Original mode (
Third, SEMS visibility may play an important role in deployment to the appropriate duct site, especially during EUS-HGS. To prevent bile duct branch obstruction, a partially covered SEMS (PCSEMS) with a stent delivery system diameter of 8.5 Fr is usually selected in Japan.
10
The uncovered site of the PCSEMS helps prevent focal cholangitis that can occur due to obstruction by the covered site. However, to insert a stent with an 8.5-Fr stent delivery system into the biliary tract, balloon dilation is needed.9,23 In a comparison of dilation by a balloon catheter (balloon dilation group,
Taken together, these findings indicate that clear visibility of devices during I-EUS facilitates safe performance and improved technical success. In the present study, the visibility of all devices (excluding needles) was significantly improved when Accent mode was used. Accordingly, procedure time was shorter in Accent mode than in Original mode, with no significant difference in terms of technical success or the rate of adverse events. The review article by Fukuma et al. 25 mentioned a similar system, a dedicated X-ray fluoroscopic apparatus (Fujifilm) for pancreatobiliary endoscopy that incorporates a novel enhancement mode. They termed this mode “wire-optimized weighted processing” (WOW) due to its ability to improve guidewire visibility. Although WOW is similar to our Accent mode, we also demonstrated improvements in visibility for other devices, as well as the guidewire. To the best of our knowledge, the present study may be the first to compare device visibility in clinical practice between original and enhanced modes.
There are several limitations in this study. First, as the study was a non-randomized, controlled trial, it is necessary to confirm our results in a prospective randomized controlled trial. Second, various procedures were included. Third, although Accent mode may be helpful for non-experts, all of the procedures in the present study were performed by experts. A further multicenter randomized study that includes experts and non-experts is required. Nevertheless, Accent mode may also be helpful in expert hands, as its superior visibility enables I-EUS to be performed smoothly. Fourth, the method used to evaluate the visibility score might not be adequate. Although the evaluations were performed independently, we used an evaluation method that might not be suitable for all devices in current use, and our results might not apply to other endoscopists. Accordingly, in future studies, it will be necessary to establish a standard scoring system for the evaluation of all such devices. Finally, compared with the Original mode, procedure time was shorter in Accent mode, and therefore, radiation dose might be lower in Accent mode. However, we could not evaluate radiation dose in this study. This important point should be evaluated in further studies.
Conclusion
In conclusion, Accent mode appears to increase the visibility of devices and reduce procedure time during I-EUS. A prospective, randomized, controlled trial that includes experts and non-experts is necessary to confirm these results.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
