Abstract
Background:
Endoscopic ultrasound-guided hepaticogastrostomy (EUS-HGS) is a biliary drainage technique performed in endoscopic centers worldwide.
Objectives:
No consensus exists on the different types of stents used. This study assessed the efficacy and safety profile of different EUS-HGS stents in malignant biliary obstruction.
Design:
This systematic review and meta-analysis followed the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (2020) guidelines.
Data sources and methods:
Three databases were searched for studies on HGS with plastic or metallic stents from inception to February 5, 2025. Primary outcomes were clinical success, overall adverse events with and without biliary obstruction, and recurrent biliary obstruction (RBO). A random-effects model was used to calculate pooled odds ratios (OR), hazard ratios (HR), and proportion rates with 95% confidence intervals (CIs).
Results:
A total of 25 studies were included. Adverse events excluding RBO were significantly less with plastic stents (OR: 2.20, CI: 1.26–3.84), while the risk of RBO was higher compared to metallic stents (HR: 1.60, CI: 1.21–2.12). There was no significant difference in clinical success between plastic and metallic stents (OR: 0.96, CI: 0.28–3.26). The pooled clinical success rates were 92% (CI: 87%–95%) for plastic stents, 89% (CI: 82%–94%) for partially covered self-expandable metal stents (PCSEMS), 96% (CI: 87%–99%) for fully covered SEMS, and 92% (CI: 86%–95%) for PCSEMS with anchoring.
Conclusion:
Metallic stents have a longer-term patency than plastic stents. Overall, adverse events were less common with plastic stents, and the clinical success was not different. Therefore, patients with short life expectancy may not benefit from metallic stent use in EUS-HGS.
Trial registration:
This study was registered with the following number. PROSPERO: CRD42024509412.
Keywords
Introduction
Endoscopic ultrasound-guided biliary drainage (EUS-BD) has consistently proven itself as not only a non-inferior but, in many cases, a superior alternative to well-established procedures that relieve biliary obstruction, such as endoscopic retrograde cholangiopancreatography and percutaneous biliary drainage. 1 Although stent types have been compared in the context of EUS-guided choledochoduodenostomy, EUS-HGS is one of the various EUS-BD techniques that have gained considerable momentum over the last decade; however, no consensus has emerged on the differences between stent types. 2
EUS-BD is typically reserved for a population group with low overall survival, such as patients with pancreatic cancer or cholangiocarcinoma. The type of stent used to relieve biliary obstruction can either assist or burden the patient; therefore, in deciding which stent type to use, clinicians must consider the risk of interrupted chemotherapy administration due to repeated re-interventions for stent dysfunction, as well as more serious consequences of stent deployment, such as cholangitis.
Conventional stents, such as straight or pigtail plastic stents, partially covered self-expanding metallic stents (PCSEMS), or fully covered self-expanding metallic stents (cSEMS), and braided or laser-cut stents have been used for EUS-HGS. The EUS-HGS stent is placed between the stomach and the B2 or B3 segments of the liver, which are closer to the stomach and more accessible via endoscope. As the stomach is a movable organ, novel stent types with anti-migratory properties have been created to mitigate migration risk.3,4
In this context, the present study aimed to assess which stent types are more appropriate in each given situation, taking into account recurrent biliary obstruction (RBO) and the adverse event profiles of individual stent types. It is hoped that the results of this study will support the emergence of a consensus on the advantages and disadvantages of different stents, thereby guiding clinical and interventional decision-making.
Methods
Protocol
Following the Cochrane Handbook, this systematic review and meta-analysis are reported based on the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines (Table S1). The study protocol was registered on PROSPERO (CRD42024509412).5–7
Eligibility criteria
The PICO framework (population, intervention, comparison, outcomes) was used to determine eligibility criteria. 8 Included were studies of patients with malignant biliary obstruction (MBO) who underwent EUS biliary drainage with HGS with plastic or metallic stents. Randomized controlled trials (RCTs), cohort studies, or case series with more than five patients were included. Primary outcomes were clinical success, overall adverse events with and without biliary obstruction, and RBO. Secondary outcomes were cholangitis, stent occlusion, stent migration, and hyperplasia.
Outcome definitions
Clinical success was defined as reducing bilirubin concentration to at least 40% of the pretreatment value within 4 weeks after the procedure. Overall, adverse events were defined as any undesired event occurring during and post-procedurally. RBO, as an adverse event, was included both with adverse events and separately. RBO for the Kaplan–Maier curves was defined as the time from stent deployment to a biliary reintervention due to stent dysfunction for any reason such as cholangitis with biliary tract dilation on imaging, stent migration (inwards or outwards) or stent occlusion, hyperplasia, or hemobilia.
Information sources
On February 5, 2025, a systematic search was conducted using the following medical databases:
Search strategy
During the systematic search, the following search key was used: ((“biliary” and “drainage”) OR “hepatogastrostomy” OR “hepaticogastrostomy”) AND ((“metal” OR “metallic” OR “plastic” OR “laser” OR “braided” OR “covered” OR “uncovered” OR “SEMS” OR “DPPS” OR “cSEMS” OR “pSEMS” OR “FCSEMS” OR “PCSEMS”) AND stent*; Table S2).
Selection process
Two independent review authors (P.P. and A.A.) performed the selection. After the removal of duplicates, published articles were selected first by title, then by abstract, and finally by full text. In case of disagreements, a third independent reviewer (M.O.) was also involved. Cohen’s kappa coefficient was calculated at both levels of selection to measure the inter-reviewer reliability. 9
Data collection process
Data were manually collected from the eligible articles into a predefined data table in an Excel spreadsheet (Office 365; Microsoft, Redmond, WA, USA) for analysis preparation. Two authors (P.P. and A.A.) collected the data independently and compared them to resolve disagreements.
Data items
The following data were extracted: first author, year of publication, countries, study periods, number of centers, number of patients, gender ratio, age, clinical success, adverse events, length of stents, and dilatation device.
Study risk of bias assessment and quality of evidence
Two authors (P.P. and A.A.) independently performed the risk of bias assessment, with disagreements resolved by a third independent reviewer (M.O.). The “Risk Of Bias In Non-randomized Studies—of Interventions” tool was used for studies included in the direct comparative meta-analysis, and the “methodological index for non-randomized studies” (MINORS) tool was used for studies included in the indirect single-arm studies. The Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) handbook was followed. The GRADEpro tool was used to assess the quality of evidence.10–12
Synthesis methods
Considerable between-study heterogeneity was assumed in all cases. A random-effects model was used to pool effect sizes. Pooled odds ratios (OR) based on raw data were calculated using the Mantel–Haenszel method with 95% confidence intervals (CI).
13
Small study publication bias was assessed by visual inspection of funnel plots and calculating modified Egger’s test
Two main kinds of analyses for pooling proportions were performed. Classical two-level meta-analyses were conducted for pooling when different studies were included. If more results were available in separate categories (subgroups) in the same study, a three-level model was used. For time-to-event data, the hazard ratio (HR) was used with a 95% CI as the primary measure of effect between the two groups of interest: Plastic as the experimental group and MS as the control group. The HR was calculated for the Plastic versus MS groups. The classical random-effects meta-analysis method and the IPD-based Cox hazard model with Gaussian random effects were used to calculate the pooled HRs. Additionally, as a secondary measure, the survival probabilities at specific time points were estimated and pooled using a three-level model (“multilevel”) to indirectly compare the survival rates in the groups. An estimation for distribution-free pooled survival curves was also performed using the method implemented by Pandey (“curve estimate”). The findings were summarized and presented in tables, on forest plots, Kaplan–Meier (KM) plots (using KM estimates), and estimated survival curves. 14
All statistical analyses were performed using R with the metaSchwarzer 2023, v6.2.1 package for basic meta-analysis calculations and plots, the metafor Viechtbauer 2023, v4.2.0 package for three-level models, the IPDfromKM v0.1.10 and metaSurvival v0.1.0 packages for summary survival curves, and the dmetar package for additional influential analysis calculations and plots.15–19
Additional details on calculations, data synthesis, publication bias assessment, and influential analyses are shown in the Synthesis Methods section in the Supplemental Material on pages 8 and 9.
Results
Search and selection
Of a total of 6465 records, 4812 were identified after duplicate removal. Of these, 4689 were excluded in the title and abstract selection phase. Of the 123 articles sought, 20 could not be retrieved, leaving 103 for full-text selection. Twenty-two were eligible for the analysis. A further 856 records were assessed from the reference and citation chasing. Fifty-two articles were evaluated, from which 3 were selected, resulting in a total of 25 articles for the final analysis. A detailed description of the screening and selection is provided in the PRISMA flowchart (Figure S1).
Basic characteristics of included studies
Two propensity-scored matched analyses, 19 cohort studies (of which 7 were prospective and 12 were retrospective), 1 RCT, and 3 conference abstracts were included. From these studies, plastic stents were inserted in 335 cases, PCSEMS in 327 cases, cSEMS in 77 instances, anchoring stents in 209 cases, and metallic laser-cut and metallic-braided stents in 39 and 197 cases. The main characteristics of the included studies are summarized in Table S3.
Adverse events without RBO and individual patient data for RBO
Five studies included information on adverse events, without RBO.20–24 Four articles and their KM curves were used to collect information from individual patient data on the HR for RBO between plastic and metallic stents.21–24
The overall adverse events without RBO showed a statistically significant difference favoring plastic stents (OR: 2.20, CI: 1.26–3.84,
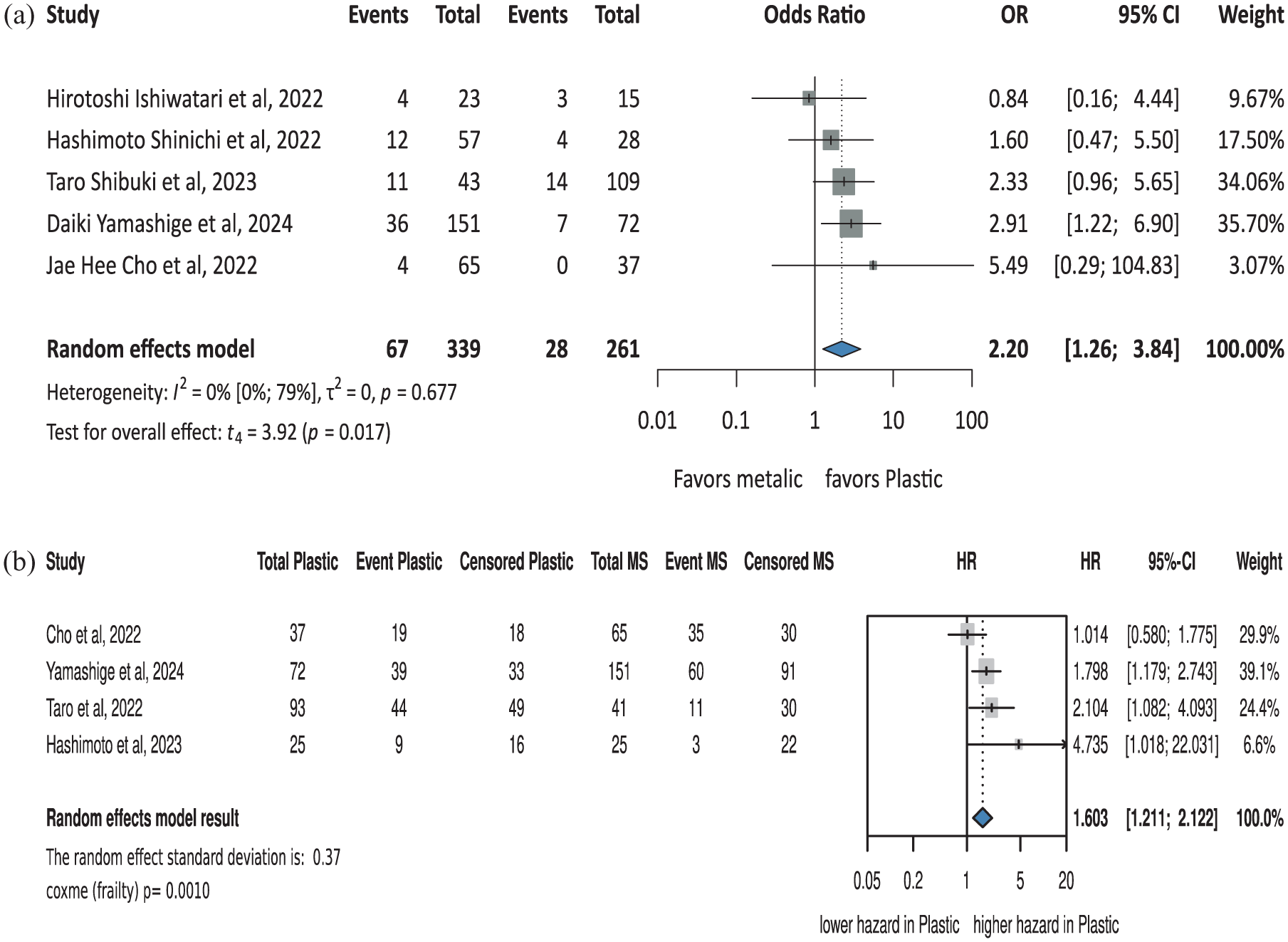
Summary figure of: (a) overall adverse events without RBO, (b) individual patient data for RBO.
Clinical success
Five comparative studies were included for clinical success.20–24 The results showed no statistically significant difference between plastic and metallic stents (OR: 0.96, CI: 0.28–3.26,
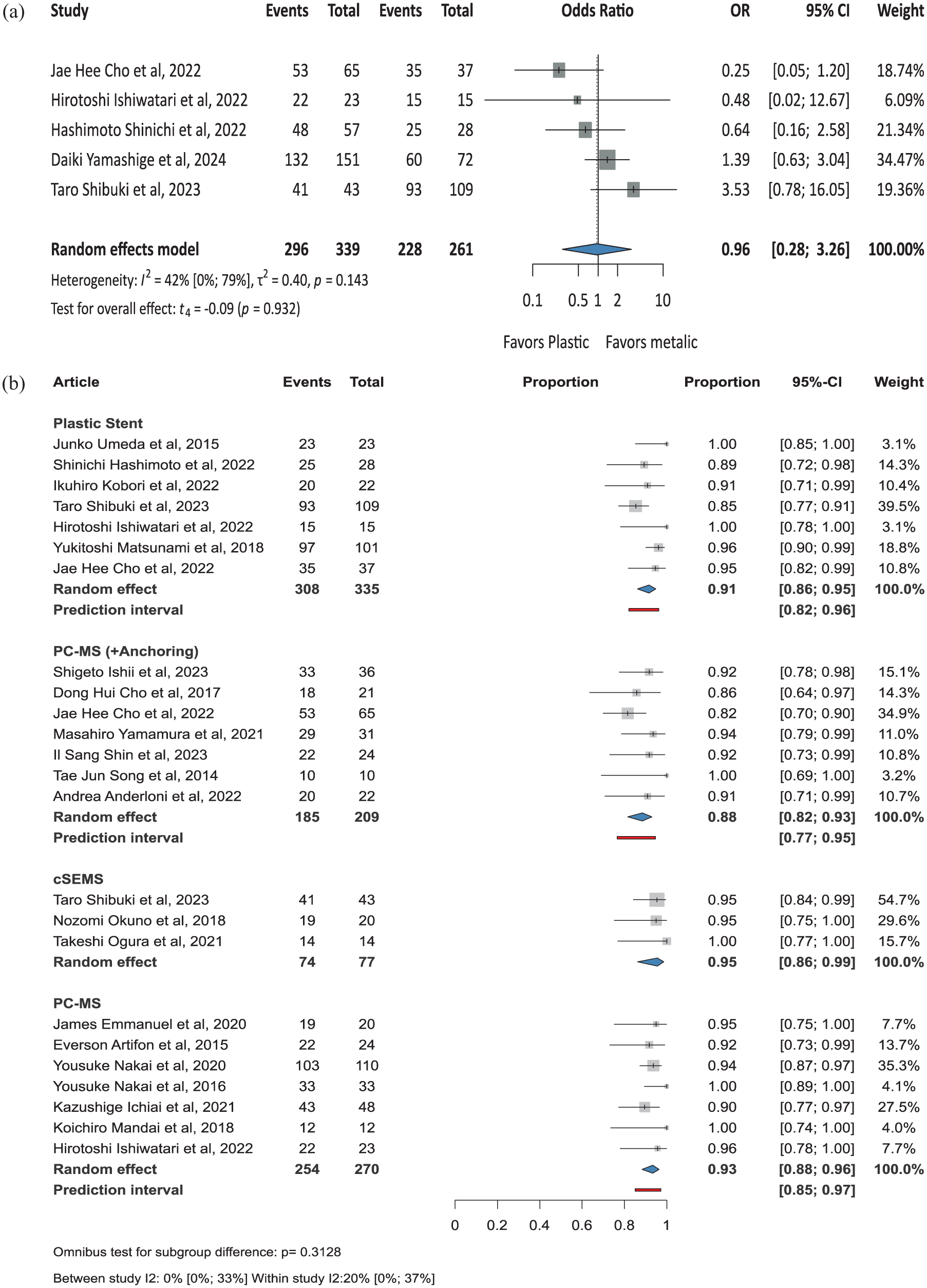
Summary figure of clinical success outcome. (a) Comparative studies. (b) Pooled single-arm studies for plastic and metallic stents.
Twenty-one studies were included for the pooled proportions of clinical success. Metallic stents were categorized based on their type; however, different plastic stents could not be pooled separately due to a lack of data. Overall, there was no statistically significant difference between the different stent types.
The pooled clinical success rate from seven studies was 91% (CI: 86%–95%) for plastic stents.21–23,25–28 For PCSEMS, the pooled rate from seven studies was 93% (CI: 88%–96%).27,29–34 PCSEMS with anchoring, based on seven studies, showed a pooled success rate of 88% (CI: 82%–93%).4,35–39 For cSEMS, the pooled clinical success proportion from three studies was 95% (CI: 86%–99%)22,40,41 (Figure 2(b)).
Pooled overall adverse events with and without RBO
Twenty-one studies were included; no difference was found between the subgroups in pooled adverse events with or without RBO.
From seven studies, the pooled overall adverse event proportion without biliary obstruction was 12% (CI: 7%–19%) for plastic stents.21–23,25–28 From six studies, 15% (CI: 8%–24%) for PCSEMS.29–34 Three studies showed 20% (CI: 10%–37%) for cSEMS.22,40,41 From seven studies for PCSEMS with anchoring, the pooled adverse proportion was 12% (CI: 7%–21%).4,35–39 The rates were 8% (CI: 1%–27%) for metallic laser-cut stents and 10% (CI: 5%–17%) for metallic-braided stents; however, only one study was included for each 42 (Figure 3(a)).
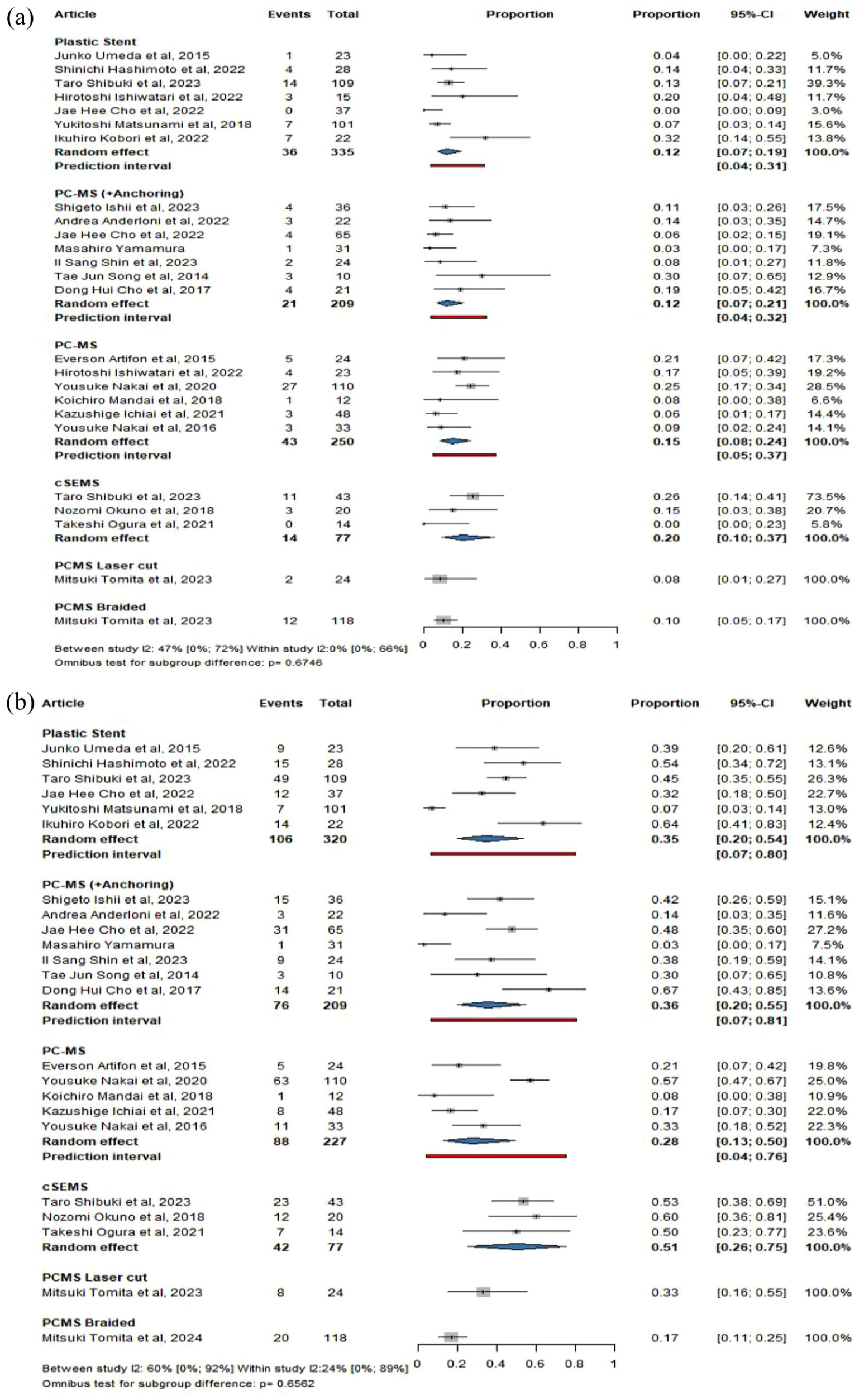
Summary figure of pooled overall adverse events. (a) Without RBO. (b) With RBO for plastic and different metallic stent types.
The pooled overall adverse event proportions with biliary obstruction were 35% (CI: 20%–54%) for plastic stents, 28% (CI: 13%–50%) for PCSEMS, and 51% (CI: 26%–75%) for cSEMS. For PCSEMS with anchoring, the pooled adverse event proportions were 36% (CI: 20%–55%). The proportions were 33% (CI: 16%–55%) for metallic laser-cut stents and 17% (CI: 11%–25%) for metallic-braided stents (Figure 3(b)).
For the pooled overall adverse events with and without RBO without subgrouping the results for plastic stents the pooled overall adverse event proportions with biliary obstruction were 37% (CI: 21%–56%) and 13% (CI: 8%–21%), respectively. For the metallic stents the pooled overall adverse event proportions with biliary obstruction were 34% (CI: 24%–46%) and 13% (CI: 10%–18%; Figure S2).
Cholangitis and stent occlusion
Plastic stents had zero events for cholangitis in 257 cases, with a pooled proportion of 1% (CI: 0%–5%). PCSEMS with anchoring also had zero events in 60 cases, yielding a proportion of 3% (CI: 0%–16%). For cSEMS, zero events were reported in 69 cases, with a proportion of 2% (CI: 0%–14%). One event was observed in 192 cases among PCSEMS, resulting in a proportion of 1% (CI: 0%–5%). Two events were reported in 39 instances for metallic laser-cut stents, 5% (CI: 1%–23%), and 19 events in 197 cases for metallic-braided stents (10%, CI: 5%–19%). The difference was statistically significant between the groups (
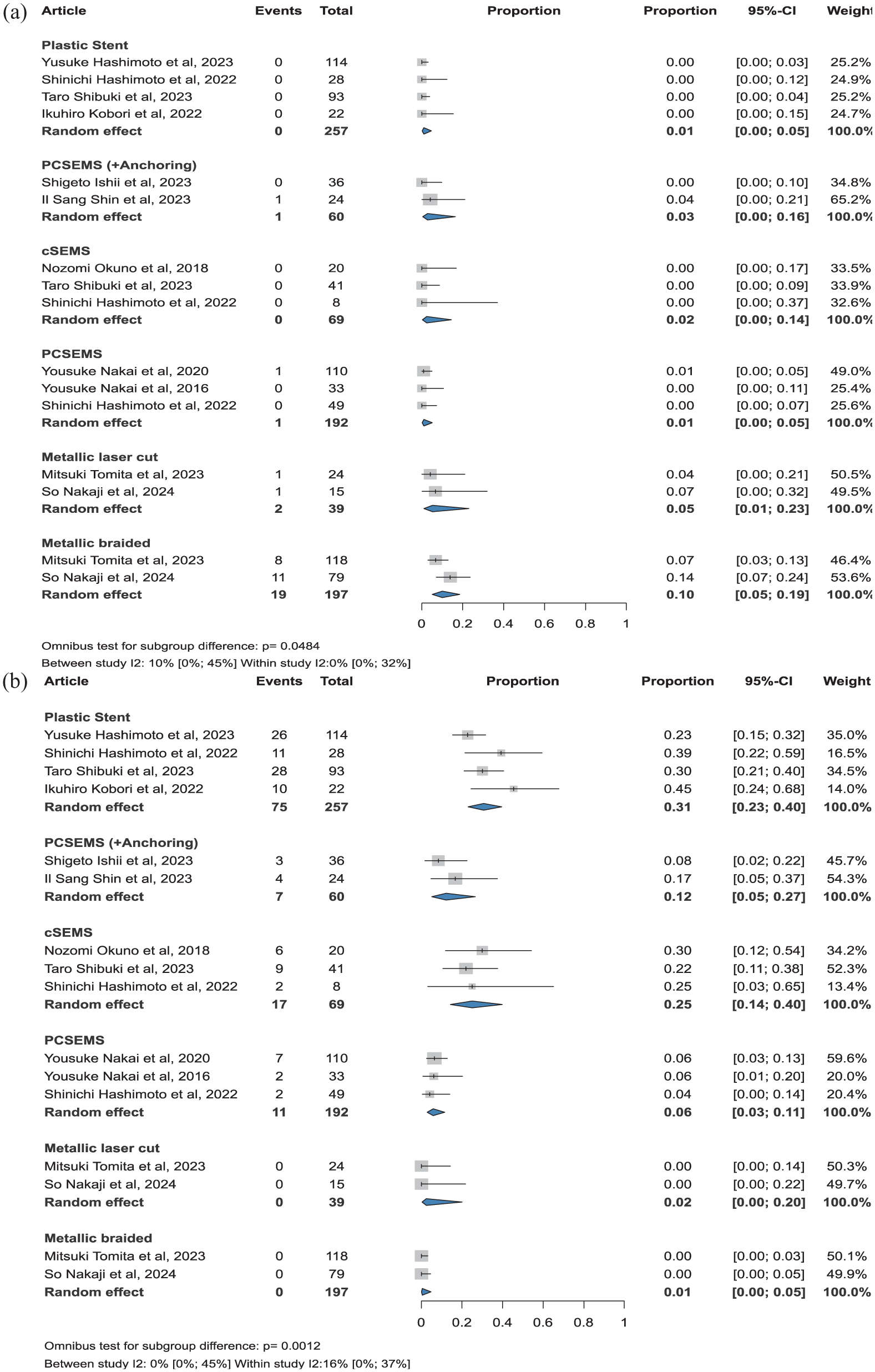
Summary figure of: (a) RBO caused by cholangitis, (b) RBO caused by stent occlusion for plastic and different metallic stent types.
Stent occlusion was observed in 75 of 257 cases with plastic stents, yielding a pooled proportion of 31% (CI: 23%–40%). For cSEMS, 17 events were reported in 69 cases, corresponding to 25% (CI: 14%–40%). For PCSEMS with anchoring, 7 events occurred in 60 cases, resulting in a proportion of 12% (CI: 5%–27%). In PCSEMS, 11 events were noted among 192 cases, 6% (CI: 3%–11%). No events were reported with metallic laser-cut or metallic-braided stents, with a pooled proportion of 2% (CI: 0%–20%) and 1% (CI: 0%–5%), respectively. The difference was statistically significant between the groups (
Stent migration and stent hyperplasia
For stent migration, 5 events were reported in 257 cases with plastic stents, yielding a pooled proportion of 2% (CI: 1%–6%). PCSEMS with anchoring had 0 events in 60 cases, with a proportion of 2% (CI: 0%–14%). For cSEMS, 6 events were observed in 69 cases, corresponding to a proportion of 17% (CI: 7%–35%). Among PCSEMS, 2 events occurred in 192 cases, 2% (CI: 1%–8%). No events were reported for metallic laser-cut (39 cases) and metallic-braided stents (197 cases), with pooled 2% (CI: 0%–20%) and 1% (CI: 0%–5%), respectively. The difference was statistically significant (
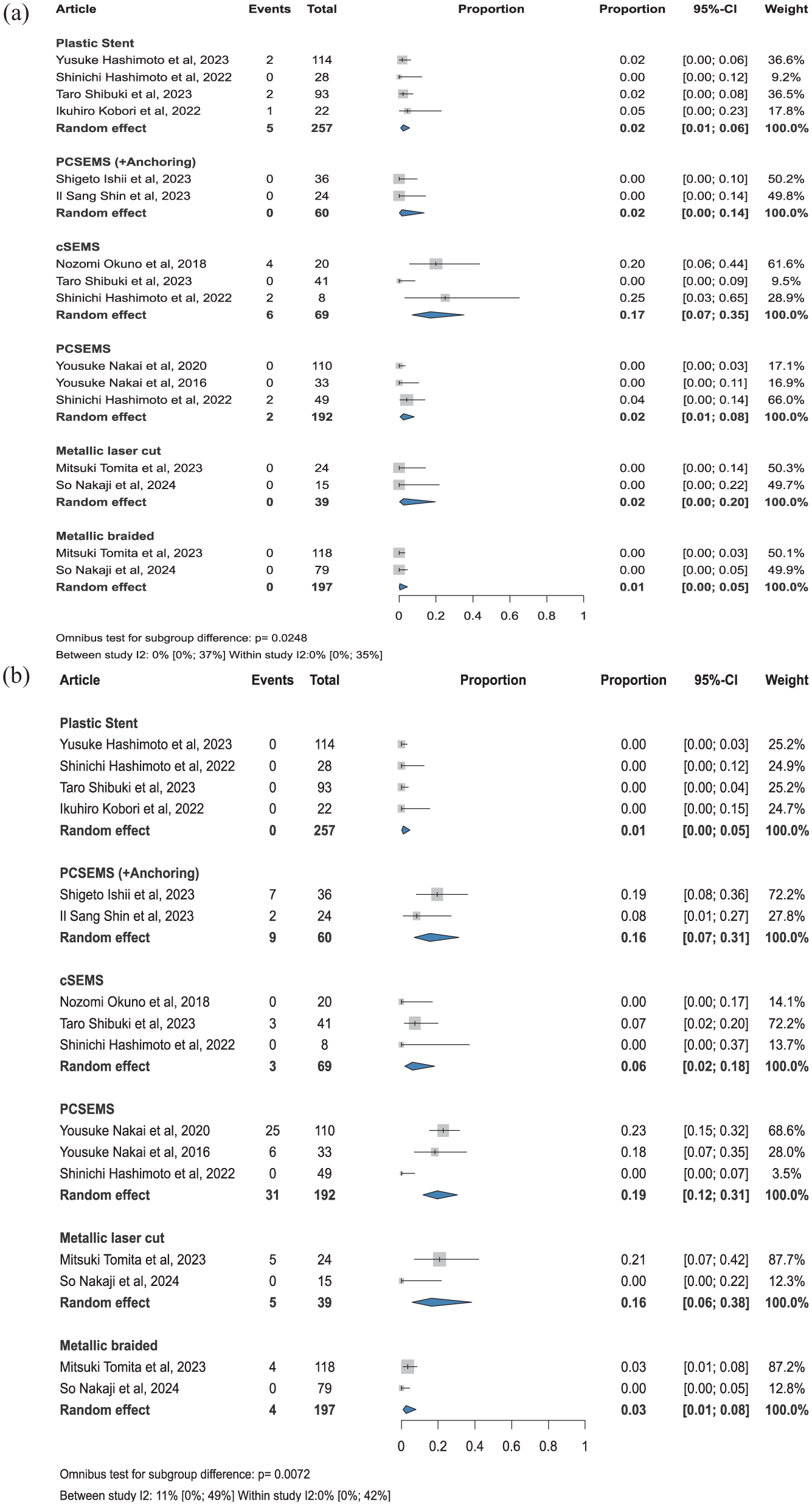
Summary figure of: (a) RBO caused by stent migration and (b) RBO caused by stent hyperplasia for plastic and different metallic stent types.
For plastic stents, hyperplasia was not observed in any of the 257 cases, with a pooled proportion of 1% (CI: 0%–5%). In PCSEMS with anchoring, 9 events were reported in 60 cases 16% (CI: 7%–31%), while PCSEMS had 31 events in 192 cases (19%, CI: 12%–31%). For cSEMS, 3 events were observed in 69 cases, 6% (CI: 2%–18%). PCSEMS had 31 events in 192 cases, 19% (CI: 12%–31%). Among metallic stents, 5 events were reported in 39 cases with laser-cut stents, 16% (CI: 6%–38%), and 4 events in 197 cases with braided stents, 3% (CI: 1%–8%). The difference was statistically significant (
Hemobilia
Hemobilia was observed in 0 cases out of 254 patients with plastic stents, with a proportion of 1% (CI: 0%–5%). For PCSEMS with anchoring, there was 1 event in 60 cases, 2% (CI: 1%–14%). For cSEMS, 0 events were observed in 69 cases at a proportion of 2% (CI: 0%–14%). For PCSEMS, 1 event in 192 cases was observed, 2% (CI: 0%–5%). For metallic laser-cut stents, 0 events in 39 cases were observed at 2% (CI: 0%–20%). For metallic-braided stents, 0 events in 197 cases were observed at 1% (CI: 0%–5%; Figure S3).
Risk of bias assessment and quality of evidence
Overall, no significant bias was observed; results of the risk of bias assessment are summarized in Table S5 and Figure S4. According to the GRADE system, the certainty of evidence was considered low for clinical success and overall adverse events (Table S4).
Publication bias and heterogeneity
No significant small study publication bias was observed in the funnel plots (Figures S5 and S6). Although for most outcomes, the heterogeneity was considered low. Heterogeneity for overall adverse events could be explained by the addition of antegrade stenting together with HGS, without noting this in the studies, which can lead to differences in adverse events, as well as time-to-stent dysfunction. Other possible reasons for the high between-study heterogeneity and low within-study heterogeneity are the length and diameter of the stents, different follow-up times, and the different experience levels of the operators, which can be contributing factors to adverse events.
Discussion
The analysis showed no significant difference in clinical success between plastic and metallic stents; however, overall adverse events were more frequent for metallic stents. Another clinically relevant difference between metallic and plastic stents is their different patency.
For the comparative studies, metallic stents were associated with significantly higher odds of adverse events (OR: 2.20, CI: 1.26–3.84). At the same time, when examining pooled adverse events across all studies, including single-arm cohorts, no consistent difference was observed between the various stent types. Since the between-study heterogeneity was high, this discrepancy reflects unmeasured confounding, such as different follow-up times, different procedural techniques, such as absence or presence of dilation, different stent lengths, operator’s experience and selection bias between studies, especially for RBO as an adverse event. Interestingly, it appears that for the different causes of RBO, there is no significant heterogeneity between the different studies. This leads us to believe that although adverse events are subject to various factors apart from stent type, once the stent is deployed, encountering the different causes of RBO is homogeneous across different centers and patients.
Metallic stents have a lower hazard for RBO, which is attributed to their higher radial force, allowing for a firmer grip of the organs’ parenchyma. However, as Yamashige et al. 24 have noted, there is no significant difference in biliary obstruction between the two stent types until the first 100 days. This result is consistent across all studies that compare stent patency using KM plots.21–24 For this reason, plastic stents could be used for periampullary or resectable pancreatic tumors as a bridge to surgery or in uncertain cases awaiting a definitive diagnosis from biopsy results or further imaging, for which metallic stents could act as artifacts.
Moreover, although dedicated PCSEMS stents with anchoring have been created for the EUS-HGS procedure, our analysis did not show any significant difference between them and PCSEMS without such anchoring. Although in theory, anchoring hooks provide a firmer attachment between the stent and organs, factors such as stent length, intragastric positioning (at least 3 cm), and distance of attachment from liver parenchyma are already associated with reduced migration and higher stent patency. 43 Still, standardized prospective studies are required to better compare the effects of anchoring stents relative to other stents in EUS-HGS.
Lastly, although metallic stents appear to have a worse overall adverse events profile, examining the individual causes of RBO, it is possible to tailor the use of different stent types for each individual patient. More specifically, it appears that metallic-braided stents carry the highest risk of RBO, 10% (CI: 5%–19%) due to cholangitis, compared to other stent types. For this reason, the use of these stents should be avoided in patients with a history of cholangitis. At the same time, in case of RBO, stent migration appears to be the highest in cSEMS, at 17% (CI: 7%–35%). Since the HGS tract is a fistulous tract between the stomach and liver, it is prone to constant movements due to respiration and peristalsis. While this can be beneficial in preventing bile leakage, the lack of strong attachments makes it more prone to the continuous movement of the tract, which can lead more easily to migration. Since the covered portion of the covered metallic stents does not allow for tissue ingrowth, the attachment to the organs’ parenchyma is decreased. Stent occlusion appears to be more frequent in plastic stents, 31% (CI: 23%–40%), cSEMS, 25% (CI: 14%–40%), and PCSEMS with anchoring, 12% (CI: 5%–27%), over metallic laser-cut, braided, or PCSEMS without anchoring.
Strengths and limitations
This meta-analysis is the first to comprehensively compare different stent types for EUS-HGS, although with limitations such as the inclusion of observational studies. We assessed the differences between metallic stent types; however, we were unable to examine the various types of plastic stents, such as straight, single, or double pigtails, due to a lack of data. Overall, our analysis did not exhibit significant heterogeneity, except for the case of adverse events from non-comparative studies. Different centers might use different protocols, devices, stent lengths, and follow-up times, and ways of reporting events, which can lead to varying adverse event outcomes as well as differences arising from the level of obstruction between distal and hilar obstruction that was not separated between the articles. For that reason, it is essential to consider confounding factors that may influence the results and are not always explicitly mentioned in the literature. Moreover, the combination of EUS-HGS with antegrade stenting is frequently used, but it is often not clearly separated in articles that we have encountered. Additionally, the incidence of cholangitis as a cause of RBO might appear low due to different reporting in the included studies. Ranging from adverse events (not causing RBO) or hyperplasia and stent migration being end-points for RBO while they can later develop into cholangitis, leading to events such as sepsis or death.
Clinical and research implications
The stent choice is a balance between avoiding adverse events causing RBO, such as cholangitis, stent migration, and stent occlusion, and achieving longer-term stent patency, for which metallic stents appear to be superior to plastic ones. These detailed results are useful for endoscopists to be aware of, enabling patients to benefit from evidence-based care.44,45 Our study showed that for patients with advanced malignancy with a low life expectancy, plastic stents may be sufficient for palliative care and symptomatic relief. For patients early in their diagnosis or those with an expected higher life expectancy, metallic stents should be favored, although the evidence might be limited; attention should be paid, especially for a history of cholangitis, where metallic-braided stents might carry a higher risk, as well as reintervention removability issues due to hyperplasia.
Future research should focus on the use of different plastic stent types and increase reporting on the parallel use of antegrade stenting to ensure it does not act as a confounder in cases of time-to-stent dysfunction and other events. The effects of different stent lengths, dilation techniques, and the experience of endoscopists should be explored further in relation to adverse events. Another important route is the comparison between anchoring and non-anchoring stents, as well as more studies focusing on directly comparing different types of metallic stents, such as PCSEMS and cSEMS, with each other.
Conclusion
The stent choice is a delicate balance between adverse events favoring plastic stents and the longer-term stent patency, for which metallic stents appear to excel, but may be influenced by unmeasured procedural differences. Clinicians and interventionists, respectively, are advised that when choosing the stent type for an EUS-HGS procedure, patient history—including past cholangitis events, where metallic-braided stents should be avoided—and future treatment steps, including time to surgery or chemotherapy initiation, should be taken into account.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261430516 – Supplemental material for Plastic versus metallic stents for endoscopic ultrasound-guided hepaticogastrostomy in malignant biliary obstruction: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848261430516 for Plastic versus metallic stents for endoscopic ultrasound-guided hepaticogastrostomy in malignant biliary obstruction: a systematic review and meta-analysis by Panagiotis Paraskevopoulos, Mahmoud Obeidat, Adib Abed, Ádám Zolcsák, Péter Jenő Hegyi, Péter Hegyi and Bálint Erőss in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
