Abstract
Background:
It is not clear that antiviral therapy for hepatitis C virus (HCV) after recovery from curative treatment for hepatocellular carcinoma (HCC) has an effect on suppressing recurrence or improving survival rates.
Objectives:
We analyzed the impact of eradication by interferon (IFN)-free direct-acting antiviral (DAA) therapy on clinical outcomes of patients with HCV-associated HCC who underwent curative treatment.
Design:
This was a retrospective study.
Methods:
We retrospectively reviewed 109 consecutive patients with sustained virologic response with DAA therapy after HCC treatment and analyzed HCC recurrence and overall survival (OS). Among these patients are those with a history of HCC recurrence and curative HCC treatments administered as definitive HCC treatments prior to initiation of DAA therapy.
Results:
Among 109 patients, 64 received DAA therapy after curative treatment for HCC; the remaining 45 received ⩾2 subsequent treatments for HCC. Cumulative HCC recurrence rates at 1, 3, and 5 years were 23%, 47%, and 56%, respectively. Multivariate analysis identified predictive factors for suppression of HCC recurrence as tumor number (hazard ratio (HR) 2.293 for multiple; p = 0.006) and number of HCC treatments before DAA therapy (HR 2.928 for ⩾2; p = 0.001). Among 64 patients who received curative treatment for HCC, cumulative first HCC recurrence rates at 1, 3, and 5 years were 12%, 34%, and 44%, respectively, second recurrence rates were 11%, 28%, and 39%, and third recurrence rates were 0%, 22%, and 53%, respectively; recurrence tended to be suppressed until 3 years. Cumulative OS rates at 3 and 5 years were 87% and 75%, respectively. On multivariate analysis, tumor number (HR 2.452 for single; p = 0.026) was the only independent predictor of OS.
Conclusion:
DAA therapy after curative treatment for HCC suppresses HCC recurrence in the long term, but recurrence was higher in patients with a history of many HCC treatments.
Keywords
Introduction
Achieving a sustained virologic response (SVR) to antiviral therapy for chronic hepatitis C virus (HCV) infection improves patient survival rates 1 through a reduction in the risk of liver decompensation and hepatocellular carcinoma (HCC).2–4 Despite great advances in the treatment of HCC, however, the recurrence rate remains high, even after curative treatment, as a remaining unsolved problem.
Generally, even after curative hepatic resection for HCV-related HCC, the rate of intrahepatic tumor recurrence within 1 year is 20%–40%, increasing to about 80% by 5 years. 5 Intrahepatic recurrence of HCC may result from intrahepatic metastasis originating from the primary tumor or by multicentric carcinogenesis related to chronic HCV infection. Interferon (IFN)-based therapy has an adjunctive effect in suppressing HCC recurrence and improving prognosis after a complete cure of HCC.6,7 A similar reduction in HCC occurrence can be expected after HCV elimination by IFN-free direct-acting antiviral (DAA) therapy, and the carcinogenesis rate in patients who achieve an SVR with DAA therapy is about 70% lower than in patients who do not achieve an SVR. Therefore, SVR achievement is considered to lead to a reduction in HCC risk.1,8–11
In the current Japanese guidelines, it is not clear that antiviral therapy for chronic HCV infection after recovery from HCC has an effect on suppressing recurrence or improving survival rates, and it is not strongly recommended. Surely, DAA therapy in patients with HCV-related cirrhosis is effective in reducing the risk of HCC development. However, DAA therapy remains expensive; thus, treating all patients with HCV with DAA therapy may not have a significant impact on healthcare costs. In particular, the impact of SVR achievement with DAA therapy on HCC recurrence in patients with recurrent HCC (one or more recurrences) remains controversial.12–16
However, even in patients with different liver function backgrounds, maintaining hepatic reserve is necessary, because treatment options for HCC are significantly expanded if hepatic reserve is maintained. Specifically, it is expected to indirectly improve prognosis by maintaining and improving liver reserve function by achievement of HCV eradication. 17
Furthermore, we found previously that achievement of an SVR with DAA therapy after curative treatment for HCV-related HCC suppressed recurrence and improved overall survival (OS). 18
Currently, it is difficult to conduct RCTs of DAA therapy to prevent HCC recurrence after curative treatment for HCV-related HCC in Japan; thus, the effectiveness of DAA therapy for this indication remains unclear. No report has investigated the rate of multiple HCC recurrences after the achievement of an SVR with DAA therapy. We carried out a retrospective analysis to evaluate the impact of DAA therapy on recurrence rates and OS rates of patients with HCV-associated HCC, including those with HCC recurrence who underwent curative treatment.
Materials and methods
Patients
We retrospectively reviewed 109 consecutive patients with HCV-related HCC who achieved an SVR with DAA therapy after radical HCC treatment at Hiroshima University Hospital between January 2000 and December 2020. The study enrolled patients who satisfied the following criteria: (1) fulfilling the Milan criteria for a diagnosis of HCC (maximum of three <3-cm diameter nodules or a single <5-cm diameter nodule), (2) no TMN stage 4 disease, (3) chronic hepatitis or compensated Child-Pugh grade A cirrhosis, (4) history of therapeutic intervention for HCC with curative intent, (5) HCV RNA-positive before curative treatment for HCC, and (6) although it includes HCC recurrence (those treatments also include transcatheter arterial chemoembolization (TACE)), history of curative HCC treatment performed as radical HCC treatment before the start of DAA therapy. Curative treatment was defined as complete tumor eradication, as shown by the absence of residual tumor on computed tomography (CT) imaging or resection of all evident tumor tissue. Treatment for HCC included percutaneous radiofrequency ablation (RFA) and stereotactic body radiation therapy (SBRT). The selection criteria for SBRT, such as the number of tumors and tumor size, were selected based on the study by Yano et al. 19 Surgical treatment included hepatic resection and ablation during laparotomy. For curative HCC treatment, the hepatologist, hepatic surgeon, and radiologist after consultation selected the best method for a cure for each patient.
In this study, in addition to patients with primary HCC, patients with recurrent HCC (those treatments also included TACE) who received curative treatment for HCC immediately before the start of DAA therapy and then SVR with DAA therapy were included.
All participants gave written informed consent to participate in the study according to the protocol approved by the ethical committee of Hiroshima University Hospital, which conformed to the ethical guidelines of the Declaration of Helsinki (Approval No. E-873). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 20
Surveillance of HCC recurrence and follow-up
All patients underwent HCC surveillance by liver function testing, measurement of serum tumor markers, and imaging (ultrasonography and/or dynamic CT). After curative treatment for primary HCC, all patients underwent monthly testing for liver function and tumor markers, abdominal ultrasonography every 3 months, and/or dynamic CT every 6 months. Recurrence of HCC was defined as any new nodule on dynamic CT or magnetic resonance imaging. If HCC recurrence was suspected, hypovascular HCC with dynamic CT was confirmed histopathologically by examination of a specimen provided by fine-needle aspiration. We used the tumor-node-metastasis stage classification system of the Liver Cancer Study Group of Japan to stage HCC. 21 Patients with recurrent HCC were treated medically or surgically, with curative intent if possible.
Statistical analysis
Continuous variables are reported as median and were compared using the Mann–Whitney U test. Categorical variables were compared using the Chi-square or Fisher exact test, as appropriate. Recurrence and survival rates were estimated by the Kaplan–Meier method, and differences between groups were assessed by the log-rank test. Multivariate analysis was conducted by a Cox proportional hazards model using the stepwise selection of variables or two logistic regression analyses.
Statistical analysis was performed with SPSS software, version 23.0 (IBM, Armonk, NY, USA). A p value < 0.05 denoted statistical significance.
Results
Characteristics of enrolled patients
The clinical characteristics of the 109 patients are shown in Table 1. Among them, 64 patients received only one curative HCC treatment before the introduction of DAA therapy, and 45 patients received HCC treatment twice or more. There were 19 patients with 2 HCC treatments, 9 patients with 3 HCC treatments, and 17 patients with 4 or more HCC treatments. The median time to the start of DAA therapy after curative treatment for HCC was 13 months.
Patient clinical characteristics at the start of DAA therapy.

Categorical data are represented as numbers of patients, and continuous data are represented as median and range.
Final HCC before DAA therapy.
ALBI grade, albumin–bilirubin grade; DAA, direct-acting antiviral; FIB-4, fibrosis-4; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; PIVKA-II, protein induced by vitamin K absence or antagonist-II; RFA, percutaneous radiofrequency ablation; SBRT, stereotactic body radiation therapy.
HCC recurrence and risk factors for HCC recurrence
The median (range) observation period after the end of treatment for DAA therapy was 65 (3–79) months.
During the observation period, HCC recurred in 56 of 109 patients (54%). The respective cumulative HCC recurrence rates at 1, 3, and 5 years were 23%, 47%, and 56%, respectively (Figure 1). Univariate analysis identified the number of HCC treatments (⩾2, p = 0.001) and tumor number (multiple, p = 0.007) as significant risk factors associated with HCC recurrence. Multivariate analysis of the predictive value of each variable for suppression of HCC recurrence also identified the number of treatments (>2: hazard ratio (HR) 2.928; 95% CI 1.613–5.376; p = 0.001) and tumor number (multiple: HR 2.293; 95% CI 1.310–4.013; p = 0.006) as independent predictive factors (Table 2). The respective cumulative HCC recurrence rates at 1, 3, and 5 years were 16%, 35%, and 46% for patients with one HCC curative treatment and 33%, 64%, and 70% for patients with ⩾2 HCC treatments, respectively (p = 0.001). The respective cumulative HCC recurrence rates at 1, 3, and 5 years were 19%, 40%, and 50% for patients with single HCC tumors and 33%, 68%, and 75% for patients with multiple HCC tumors, respectively (p = 0.007; Figure 2). On the other hand, HCC recurrence in surgical resection and RFA, and SBRT, had no statistically significant contribution to HCC recurrence (p = 0.430) among these treatment modalities, respectively.
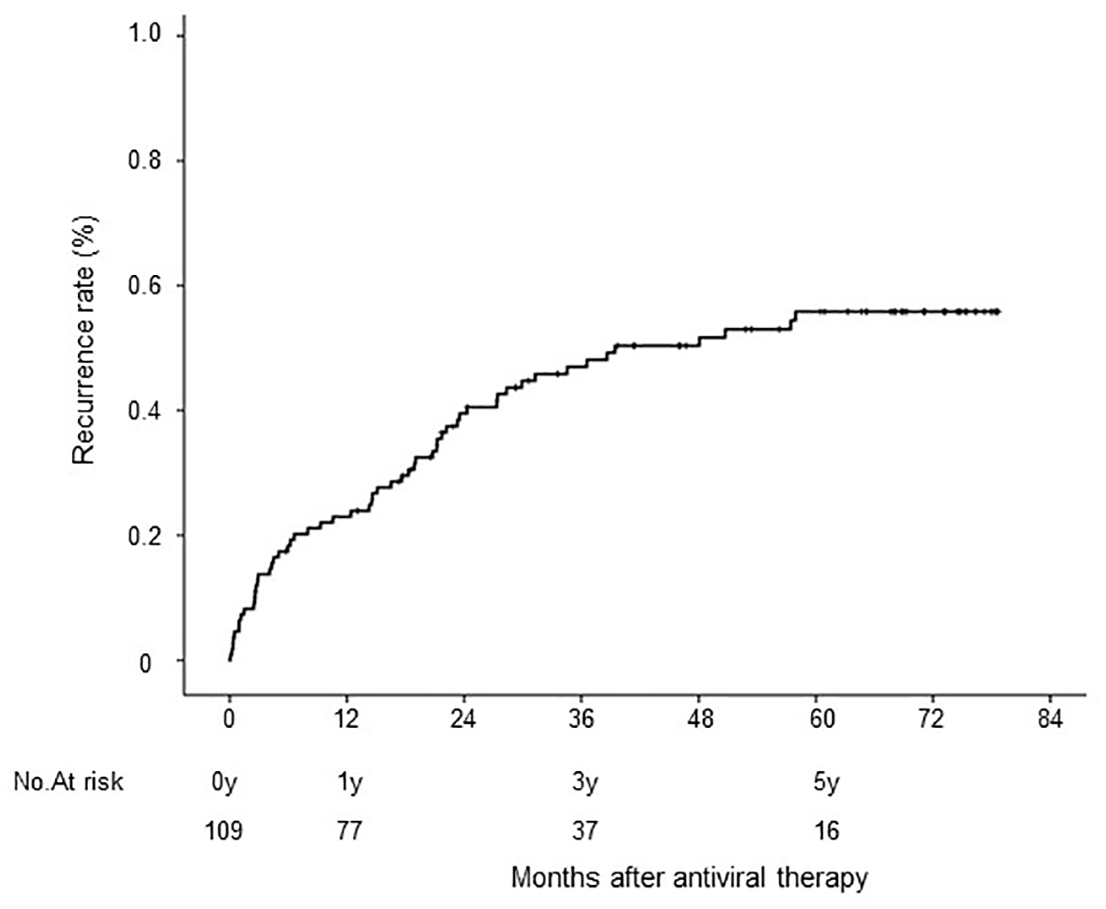
Cumulative rates of HCC recurrence among 109 patients.
Univariate and multivariate analyses of factors associated with HCC recurrence.
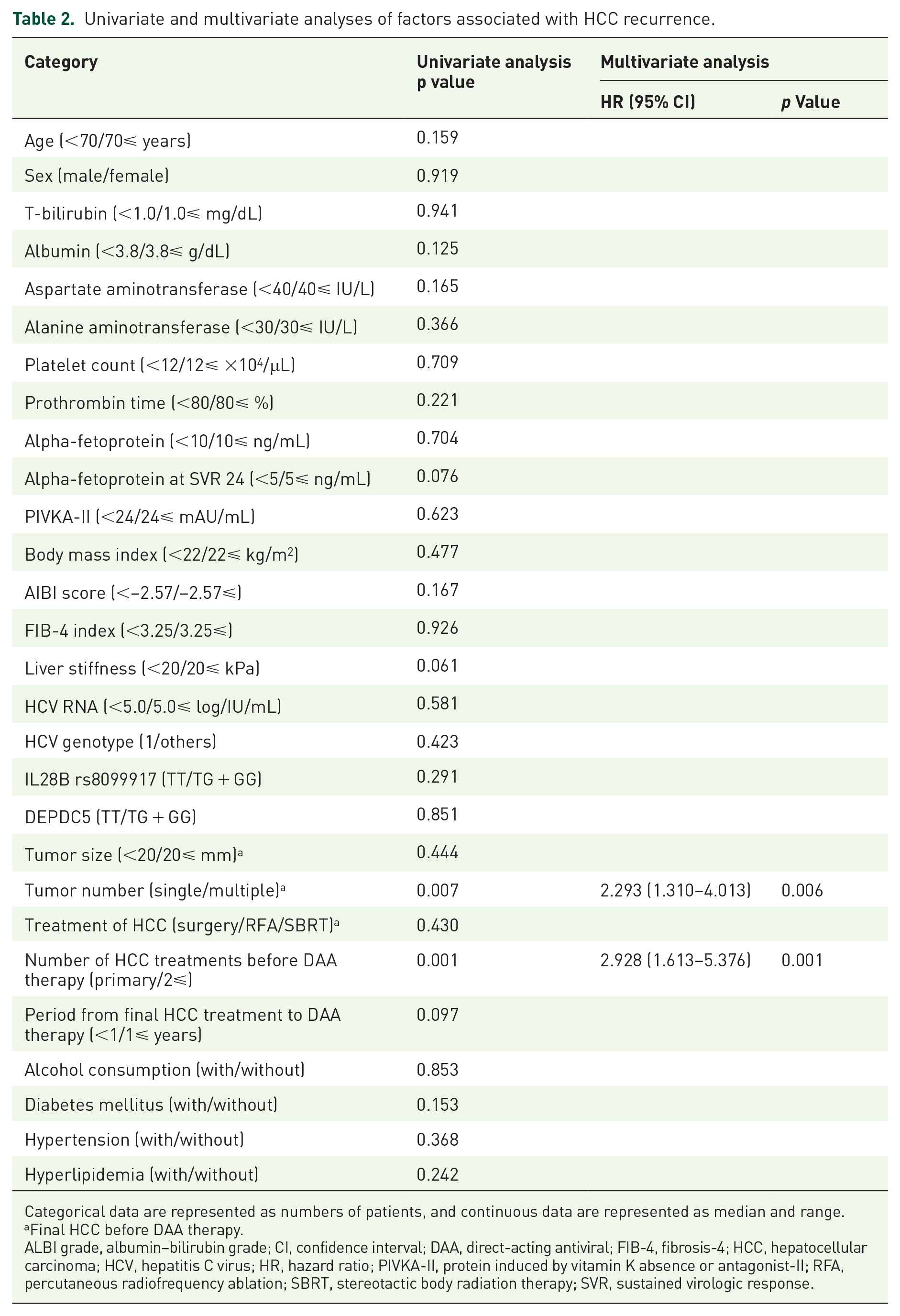
Categorical data are represented as numbers of patients, and continuous data are represented as median and range.
Final HCC before DAA therapy.
ALBI grade, albumin–bilirubin grade; CI, confidence interval; DAA, direct-acting antiviral; FIB-4, fibrosis-4; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HR, hazard ratio; PIVKA-II, protein induced by vitamin K absence or antagonist-II; RFA, percutaneous radiofrequency ablation; SBRT, stereotactic body radiation therapy; SVR, sustained virologic response.
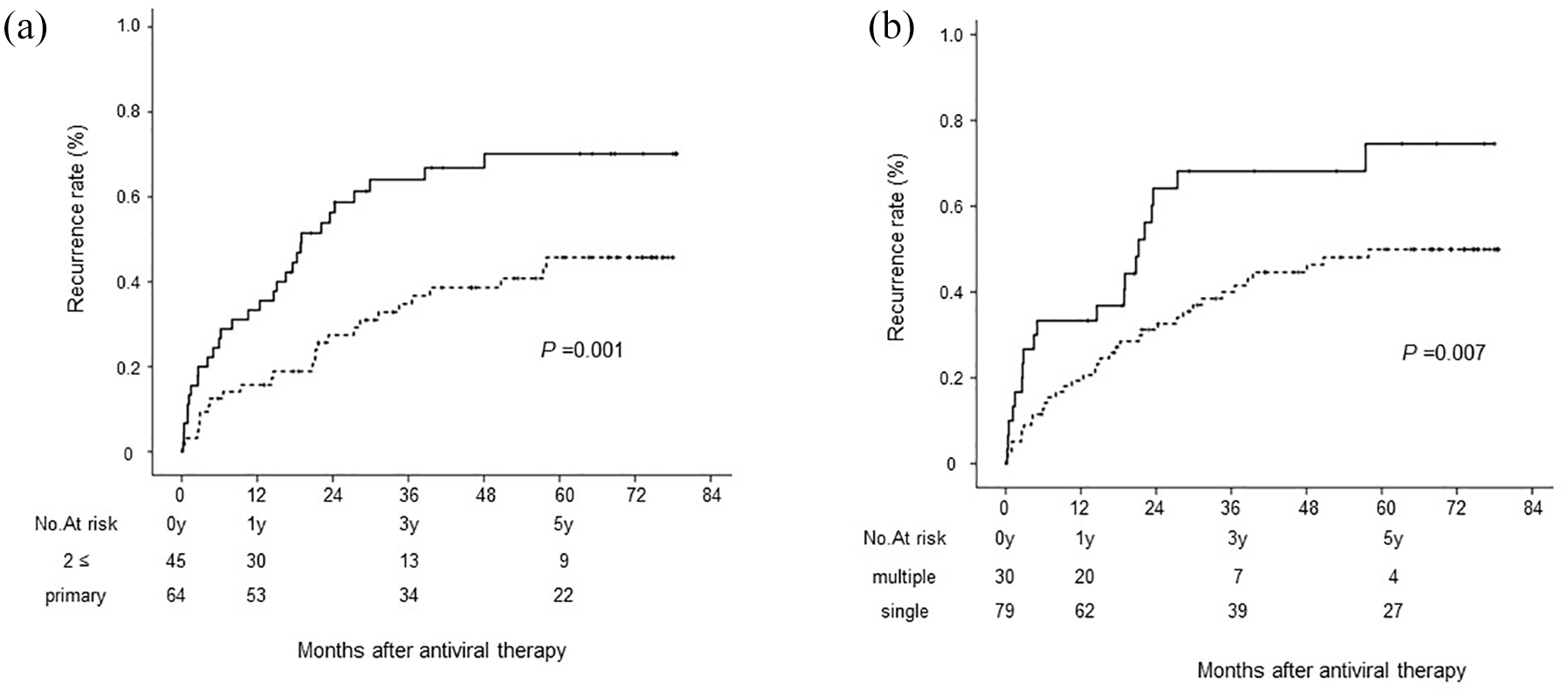
Cumulative rates of HCC recurrence (a) according to the number of HCC treatments before DAA therapy: >2 (solid line) and primary (dotted line). (b) According to the number of HCC tumors: multiple (solid line) and single (dotted line).
HCC recurrence for patients who underwent primary curative HCC treatment
In addition, we investigated the recurrence rate in patients who underwent primary curative HCC treatment. Among 109 patients, 64 received only one curative HCC treatment before the introduction of DAA therapy. First cumulative HCC recurrence rates at 1, 3, and 5 years were 12%, 34%, and 44%, respectively; second HCC recurrence rates were 11%, 28%, and 39%, respectively; and third HCC recurrence rates were 0%, 22%, and 53%, respectively. Recurrence rates tended to be suppressed, even in the long term (Figure 3).

Cumulative rates of HCC recurrence for patients who underwent primary curative HCC treatment. (a) First HCC recurrence. (b) Second HCC recurrence. (c) Third HCC recurrence.
Changes in hepatic spare ability and liver fibrosis markers with HCC recurrence
Regarding hepatic spare ability and liver fibrosis markers according to the presence or absence of HCC recurrence, pre-DAA therapy, the median ALBI score was −2.51 in the recurrence group and −2.63 in the non-recurrence group, showing no significant difference between groups. The median fibrosis-4 (FIB-4) index was 5.17 in the recurrence group and 4.36 in the non-recurrence group, also showing no significant difference between groups. In 56 patients with HCC recurrence, the median ALBI scores decreased significantly from −2.51 at pre-DAA therapy to −2.82 at HCC recurrence (p < 0.05; Figure 4(a)). The median FIB-4 index decreased significantly from 5.17 at pre-DAA therapy to 4.16 at HCC recurrence (p < 0.05; Figure 4(b)). These results indicate that both hepatic spare ability and liver fibrosis can improve following the achievement of SVR with DAA therapy, even in patients with HCC recurrence.

Changes in hepatic spare ability and liver fibrosis markers with HCC recurrence. (a) Changes in ALBI score at pre-DAA therapy and HCC recurrence. (b) Changes in FIB-4 index at pre-DAA therapy and HCC recurrence. In these box-and-whisker plots, lines within the boxes represent median values; the upper and lower lines of the boxes represent the 75th and 25th percentiles, respectively; and the upper and lower bars outside the boxes represent the 90th and 10th percentiles, respectively.
OS after curative HCC treatment and prognostic factors for OS
The respective cumulative OS rates at 1, 3, and 5 years were 97%, 87%, and 75%, respectively (Figure 5). Univariate analysis identified the following positive prognostic factors for OS: tumor number (single, p = 0.021) and tumor number at HCC recurrence (single, p = 0.049; Table 3). Multivariate analysis identified tumor number (single: HR 2.452; 95% CI 1.1134–6.966; p = 0.026) as the only independent predictor of OS. The respective cumulative OS rates at 1, 3, and 5 years were 99%, 88%, and 81% for patients with a single HCC tumor and 97%, 82%, and 52%, respectively, for patients with multiple HCC tumors. By contrast, the number of radical HCC treatments before DAA therapy introduction was also not associated with OS. The respective cumulative OS rates at 1, 3, and 5 years were 97%, 92%, and 80% for patients with only one HCC treatment and 100%, 80%, and 67%, respectively, for patients with ⩾2 HCC treatments (p = 0.134; Figure 6). Similar to recurrence rates, when comparing OS rates among surgical resection, RFA, and SBRT, no statistically significant contribution to OS was found among these treatment modalities, respectively (p = 0.743).

Cumulative rates of overall survival among 109 patients.
Univariate and multivariate analyses of factors associated with prognostic influence on overall survival.

Categorical data are represented as numbers of patients, and continuous data are represented as median and range.
Final HCC before DAA therapy.
ALBI grade, albumin–bilirubin grade; CI, confidence interval; DAA, direct-acting antiviral; FIB-4, fibrosis-4; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HR, hazard ratio; PIVKA-II, protein induced by vitamin K absence or antagonist-II; RFA, percutaneous radiofrequency ablation; SBRT, stereotactic body radiation therapy; SVR, sustained virologic response.

Cumulative rates of overall survival (a) according to the number of HCC tumors: single (solid line) and multiple (dotted line), (b) according to number of HCC treatments before DAA therapy: primary (solid line) and ⩾2 (dotted line).
Discussion
The purpose of this retrospective study was to evaluate the impact of DAA therapy on HCC recurrence rates and OS rates of patients with HCV-associated HCC, including those with HCC recurrence who underwent curative treatment.
First, during the observation period, the respective cumulative HCC recurrence rates at 1, 3, and 5 years were 23%, 47%, and 56%, respectively. Multivariate analysis of the predictive value of each variable for HCC recurrence identified two or more treatments (HR 2.928; 95% CI 1.613–5.376; p = 0.001) and multiple tumors (HR 2.293; 95% CI 1.310–4.013; p = 0.006) as independent predictive factors. By contrast, the time from radical HCC treatment to DAA therapy introduction was not associated with HCC recurrence. Until now, there have been various discussions about the timing of the introduction of DAA therapy after HCC treatment; some reports stated that a long period from initial HCC treatment to DAA therapy introduction affects HCC recurrence,22–24 but it was not a risk factor in our analysis. In this study, the number of tumors and the frequency of HCC recurrence during treatment for HCC prior to antiviral therapy were reported as risk factors for HCC recurrence after curative treatment. Thus, our results were similar to those of previous reports.25–27
Therefore, while these subjects have a history of many treatments for HCC, including TACE, they have the opportunity to initiate DAA therapy and achieve an SVR. However, even if an SVR is achieved, the HCC recurrence rate is high for patients who have been treated for multiple HCC occurrences, and careful follow-up is necessary.
We also investigated the HCC recurrence rate in patients who underwent primary curative HCC treatment; 64 patients received only one curative HCC treatment before the introduction of DAA therapy. First cumulative HCC recurrence rates at 1, 3, and 5 years were 12%, 34%, and 44%, respectively; second HCC recurrence rates were 11%, 28%, and 39%, respectively; and third HCC recurrence rates were 0%, 22%, and 53%, respectively (Figure 5). This study suggested an effect of suppressing second recurrence. Although this result was a study of IFN therapy, it was similar to a report that examined HCC recurrence in the IFN group and non-IFN group after curative treatment for HCC and found that the IFN-treated group had an effect of suppressing second recurrence after curative treatment for HCC compared with patients without an SVR or non-IFN group patients. 28
However, the effect of suppressing the third recurrence after the 5th year recurrence has not yet been determined, and discussion is necessary. Of the 64 cases of primary HCC, 26 relapsed for the first time, and 9 of those cases had a second HCC recurrence. In addition, four of the nine cases with a second HCC recurrence experienced a third recurrence. All four of the patients who experienced a third recurrence were male, but no characteristics of the HCC at the time of primary HCC onset, such as progression of the stage of the primary HCC or marked increase in liver stiffness, were observed. Previously, several studies, reported an unexpectedly high HCC recurrence rate after DAA therapy in previously treated patients with HCV-associated HCC.29–31 However, similar to those reports, we believe that the HCC recurrence rate is not expected to be high.32,33
Results of our previous study indicated that both hepatic spare ability and liver fibrosis could improve following the achievement of an SVR with DAA therapy, even in patients with liver cirrhosis.34–36 Therefore, we analyzed whether hepatic spare ability and liver fibrosis markers were exacerbated in the 56 patients with HCC recurrence; the results indicated that both hepatic spare ability and liver fibrosis could improve following achievement of an SVR with DAA therapy, even in patients with HCC recurrence. If hepatic reserve does not deteriorate, the possibility of various treatment options expands, suggesting the possibility of more appropriate treatment.
The respective cumulative OS rates at 1, 3, and 5 years were 97%, 87%, and 75%, respectively (Figure 5). Multivariate analysis identified tumor number (single: HR 2.452; 95% CI 1.134–6.966; p = 0.026) as the only independent predictor of OS. The respective cumulative OS rates at 1, 3, and 5 years were 99%, 88%, and 81% for patients with a single HCC tumor and 97%, 82%, and 52%, respectively, for patients with multiple HCC tumors (p = 0.026). Of the 30 patients with multiple HCC tumors before DAA therapy, 14 (47%) had multiple HCC tumors at the time of recurrence after the achievement of an SVR, and of the 79 patients with a single HCC tumor before DAA therapy, 18 (23%) had multiple HCC tumors at the time of recurrence. Regarding OS after the achievement of an SVR, the tumor number at HCC recurrence was not a significant factor in multivariate analysis. Furthermore, the number of HCC treatments before DAA therapy introduction was not associated with OS. The respective cumulative OS rates at 1, 3, and 5 years were 97%, 92%, and 80% for patients with only one HCC treatment and 100%, 80%, and 67%, respectively, for patients with two or more HCC treatments (p = 0.134; Figure 6(b)). Based on this, we believe that the prognosis is not always poor, even if HCC has been treated multiple times, and that it is necessary to administer DAA therapy promptly after determining the timing of complete cure with HCC treatment. Furthermore, in the case of multiple HCC recurrences, we believe that it is necessary to combine treatments that can be curative.
Results of previous studies have suggested that the risk of HCC is correlated with age, sex, and stage of liver fibrosis after chronic HCV infection. 37 Therefore, in addition to the above, it is important to screen for HCC recurrence while considering tumor factors such as the number of tumors at the time of HCC treatment before the introduction of DAA therapy and the number of recurrences of HCC. In any case, much evidence has accumulated regarding HCC recurrence after DAA therapy, and although it is a controversial issue, it has been shown that there is no disadvantage to administering DAA therapy after HCC treatment.38–40 However, few reports have addressed the effect of suppressing multiple recurrences, so we believe that the findings of the present study are useful. It is highly expected that the prognosis will be good if an SVR is acquired with DAA therapy after curative treatment of HCC.
This study has limitations. First, although this clinical study had a long observation period, it was conducted at a single center and the number of cases was small. And, it is difficult to conduct RCTs of DAA therapy to prevent HCC recurrence after curative treatment for HCV-related HCC in Japan. Therefore, this study has to be a retrospective cohort study.
Conclusion
We found that an SVR obtained after curative treatment for HCV-associated HCC suppressed recurrence and improved OS. In addition, while the recurrence rate was higher in patients with HCC with a history of many treatments, DAA therapy after curative treatment for HCC tended to suppress HCC recurrence in the long term.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251324094 – Supplemental material for Impact of viral eradication by direct-acting antivirals on clinical outcomes after curative treatment for hepatitis C virus-associated hepatocellular carcinoma
Supplemental material, sj-docx-1-tag-10.1177_17562848251324094 for Impact of viral eradication by direct-acting antivirals on clinical outcomes after curative treatment for hepatitis C virus-associated hepatocellular carcinoma by Yuko Nagaoki, Kenji Yamaoka, Yasutoshi Fujii, Shinsuke Uchikawa, Hatsue Fujino, Atsushi Ono, Eisuke Murakami, Tomokazu Kawaoka, Daiki Miki, Hiroshi Aikata, Clair Nelson Hayes, Masataka Tsuge and Shiro Oka in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
