Abstract
Acute severe ulcerative colitis (ASUC) is a life-threatening medical emergency affecting over 20% of patients with ulcerative colitis (UC). Up to 40% of patients are refractory to intravenous corticosteroids (IVCS) and require rescue medical therapy or immediate colectomy. The potent Janus kinase (JAK) inhibitors, upadacitinib and tofacitinib, have proven efficacy in a randomised control trial setting for moderate-to-severe UC, but not ASUC. We describe a case series of sequential rescue therapy with JAK inhibitors following the failure of dose-intensified infliximab in corticosteroid-refractory ASUC. Six adult (>16 years old) patients received sequential rescue therapy with a JAK inhibitor (upadacitinib
Plain language summary
We studied six adults with acute severe ulcerative colitis (ASUC) who received JAK-inhibitors (upadacitinib or tofacitinib) during the same hospital admission after previous treatments (intravenous corticosteroids and high-dose infliximab) failed. Four out of six patients responded quickly to JAK-inhibitor therapy and were discharged, while two patients underwent colectomy due to ongoing severe disease, one of which was complicated by post-operative sepsis. Among the four patients who responded to JAK-inhibitors, all achieved steroid-free clinical remission, and three reached biochemical remission (Faecal Calprotectin <150 µg/mL and C-Reactive Protein <5 mg/L) within 90-days. No other adverse events occurred. These results suggest that JAK-inhibitors may be a promising option for stable ASUC patients not requiring immediate surgery. Further research is needed to confirm the safety and effectiveness of this treatment approach.
Introduction
Acute severe ulcerative colitis (ASUC) is a life-threatening medical emergency affecting over 20% of patients with ulcerative colitis (UC). 1 Initial management with intravenous corticosteroids (IVCS) is effective for most presentations. However, 30%–40% of patients fail to respond to IVCS and require rescue medical therapy or urgent colectomy.2,3 Infliximab or ciclosporin is considered first-line rescue therapy for IVCS-refractory ASUC and has comparable rates of clinical response, colectomy, adverse events and death.3,4 Unfortunately, despite improvements in the modern era, approximately 15% of patients are medically refractory and progress to colectomy, highlighting the need for other treatment options.4–8
Rescue therapy with dose-intensified infliximab is commonly used and may reduce colectomy rates further, 9 although observational evidence supporting this practice is equivocal and suggests this approach may only defer rather than avoid colectomy.10,11 More recently, an Australian multicentre randomised study (PREDICT-UC) found no difference in the day 7 clinical response between a standard (5 mg/kg) and intensified (10 mg/kg) initial infliximab dose. 12 However, subgroup analysis suggested that a higher initial infliximab dose may still provide short-term benefits for patients with lower serum albumin and higher inflammatory burden. Thus, there remains a potential role for dose-intensified infliximab in select patients.
Another approach used to reduce colectomy rates is sequential salvage therapy, where a second medical rescue therapy is initiated following inpatient failure of first-line treatment with infliximab or ciclosporin. Three studies using infliximab after ciclosporin, or ciclosporin after infliximab failure, have demonstrated effectiveness; however, safety concerns were raised due to the high rate of adverse events, including death.13–15 Identifying further medical salvage options has the potential to achieve remission and avoid colectomy in patients with refractory disease without indication for emergent surgery.
The novel Janus kinase (JAK) inhibitors, upadacitinib (JAK-1) and tofacitinib (JAK-1 and JAK-3), have proven efficacy for UC in a clinical trial setting for moderate-to-severe UC, but are not licensed for the treatment of ASUC.16,17 JAK inhibitors have theoretical advantages over current rescue therapy due to high oral availability, clearance unrelated to albumin concentration, rapid washout, low immunogenicity and long-term treatment durability.18–22 Recently, several case series have demonstrated the safety and efficacy of tofacitinib23–26 and upadacitinib27–29 as alternative rescue agents in IVCS-refractory infliximab-experienced patients. However, the safety of sequential JAK inhibition following successive failure of IVCS and dose-intensified infliximab within the same acute hospital admission is poorly defined, particularly for upadacitinib. Here we describe the novel use of JAK inhibitors as sequential rescue therapy following failure of dose-intensified infliximab in IVCS-refractory ASUC.
Case series
Patients who received sequential rescue therapy with a JAK inhibitor following the failure of IVCS and dose-intensified infliximab were identified from a prospectively maintained database at a single tertiary Australian (Queensland) Inflammatory Bowel Disease (IBD) centre. All patients included in this case series have given written consent to use their de-identified data as part of our ongoing Natural History of IBD study (HREC EC00278).
Our standard ASUC treatment protocol includes 100 mg IV hydrocortisone four times per day, venous thromboembolism (VTE) prophylaxis, flexible sigmoidoscopy within 48 h of admission and rescue therapy with dose-intensified infliximab (one or two doses at 10 mg/kg) following intravenous albumin infusion (with 20% human albumin solution to achieve serum albumin ⩾30 g/L).5,30,31 It has been our standard practice to co-administer albumin since 2016 in an attempt to reduce infliximab clearance which is associated with higher rates of colectomy. 32 We hypothesise that the additional albumin binds infliximab and slows down the distribution phase of the drug, potentially leading to a more prolonged tissue exposure.33,34 This practice is supported by findings from both observational series and interventional studies35,36
Following the failure of infliximab rescue therapy and detailed discussion between colorectal surgeons and IBD clinicians, carefully selected clinically stable patients were presented with the option of proceeding to colectomy as per standard of care, or, in the absence of any indication for emergent colectomy, a trial of an oral JAK inhibitor (upadacitinib 45 mg once daily (OD) or tofacitinib 10 mg three times per day (TDS)).37,38 All patients who received sequential rescue therapy with a JAK inhibitor were discharged on a 9-week oral prednisolone cascade (40 and 30 mg each for 1 week, 20 mg for 4 weeks, then 15, 10 and 5 mg each for a week) and did not receive further therapy with biologic agents or immunomodulators. It is our standard practice to continue oral mesalazines alongside biologic/small molecule therapies but to consider their withdrawal in patients who achieve deep remission.39–41 VTE chemoprophylaxis was discontinued on discharge except for the duration of induction (2 weeks) in the patient treated with tofacitinib. Sulfamethoxazole–trimethoprim (800–160 mg) was prescribed three times per week to reduce the risk of opportunistic infections until the daily prednisolone dose was <20 mg/day.
Co-primary outcomes were 90-day colectomy-free survival and inpatient clinical response. Secondary outcomes included 90-day steroid-free clinical remission, 90-day steroid-free biochemical remission and adverse events. ASUC was defined according to the Truelove and Witts Severity Index for UC 42 and IVCS non-response using the Day 3 Oxford criteria (C-reactive protein (CRP) greater than 45 mg/L, or 8 or more bowel motions per day) at 72 h. 43 Inpatient clinical response to dose-intensified infliximab or JAK-inhibitor rescue therapy was defined using the Day 7 Oxford criteria (<4 non-bloody stools per day), 48 h after the final infusion of dose-intensified infliximab or 72 h after JAK-inhibitor initiation. 43 An earlier time point was used for the assessment of JAK-inhibitor response due to the rapid onset of action of this small molecule therapy and to mitigate the risk of delayed colectomy given the off-label use of these agents outside of a clinical trial setting. Ninety-day clinical remission was defined by a Patient Reported Outcome (PRO-2) score <1, 44 and 90-day biochemical remission as Faecal Calprotectin (FCP) <150 µg/mL and CRP <5 mg/L, in the absence of continuing corticosteroids. Adverse events were defined as those requiring re-admission or delayed discharge from the hospital. Notably, patients with a normal CRP or a significant improvement in CRP following rescue infliximab may still be considered for sequential JAK-inhibitor therapy based solely on clinical criteria. This is particularly relevant for the 15%–20% of IBD patients who produce normal-range CRPs even during acute disease exacerbations. 45 The real-world delivery of ASUC treatment may differ from our protocol due to local access to rescue therapy in remote and regional IBD patients. We therefore recorded all treatment deviations to aid the interpretation of our findings.
Results
Six adult (>16 years old) patients received sequential rescue therapy with a JAK inhibitor (5 upadacitinib, 1 tofacitinib) following successive failure of inpatient dose-intensified infliximab and IVCS (Figure 1).

CONSORT diagram depicting sequential management of ASUC in patients admitted between October 2023 and April 2024*.
Demographics
Median age on admission was 32 years (interquartile range (IQR) 23–39); five patients were male (Table 1). This presentation was the first diagnostic episode of UC for three patients; the remaining three patients had a diagnosis of UC established between 3 and 96 months. Two patients had previous admissions with ASUC (one requiring infliximab rescue therapy and one IVCS responsive).
Clinical, biochemical and endoscopic characteristics on admission with ASUC.

ROR. 30
Evidence of systemic toxicity defined as tachycardia (>90 beats/min) or fever (>37.8°C) as per Truelove and Witts Severity Index for UC. 39
Received tofacitinib; all other cases received Upadacitinib.
5ASA, mesalazine (oral); 6MP, 6-mercaptopurine; ASUC, acute severe ulcerative colitis; CRP, C-reactive protein; E2, left-sided ulcerative colitis; E3, pan-colitis; FCP, faecal calprotectin; Hb, haemoglobin; IBD, inflammatory bowel disease; IFX, infliximab; MES, Mayo endoscopic score; NP, not performed; Pred, oral prednisolone; ROR, risk of rescue score.
Patients with a known UC diagnosis had been prescribed oral mesalazine (
Disease assessment
All patients met Truelove and Witts criteria for ASUC on admission, with a median CRP of 100 mg/L (IQR 58–105) and median FCP of 3400 µg/g (IQR 910–4950; Table 1). Relevant to our previous work correlating ESR and CRP in ASUC, all six patients had a CRP >12 mg/L. 47
All patients underwent flexible sigmoidoscopy within 48 h of hospital admission: three patients had extensive disease (Montreal E3 – pancolitis) and the Mayo Endoscopic Score (MES) was 3 in all six cases. Where available, endoscopic images from the recto/sigmoid colon on admission are shown in Figure 2. Using the full Risk of Rescue (ROR) score, all patients (

Representative endoscopic images of sigmoid colon taken on admission flexible sigmoidoscopy (<48 h) for Cases #1, 3–6. Case #1 UCEIS score 6/Mayo 3, Case #3–6 UCEIS 7/Mayo 3. Images from Case #2 not available.
Rescue treatment
Following IVCS failure, patients progressed to rescue therapy with dose-intensified (10 mg/kg) infliximab between day 1 and day 6 of admission. Five of six patients received a second infusion of dose-intensified infliximab due to inadequate clinical and biochemical response 48 h after the first dose. One patient (Case #5) with prior infliximab-exposure, progressed directly to upadacitinib following inadequate clinical response to the first infusion of dose-intensified infliximab (point of care infliximab drug level at 72 h after infliximab infusion >20 µg/mL).
Sequential JAK-inhibitor treatment
Sequential rescue therapy with a JAK inhibitor (upadacitinib 45 mg OD; tofacitinib 10 mg TDS) was commenced following careful patient and multidisciplinary team consultation, a median of 2 days (IQR 2–2.75) after the final dose of infliximab. Clinical, biochemical and endoscopic characteristics at the commencement of sequential rescue therapy with JAK inhibitor are shown in Table 2, with evidence of systemic toxicity (tachycardia, HR >90 beats/min) in one patient (Case #3) but no megacolon on imaging.
Clinical, biochemical and endoscopic characteristics at the commencement of sequential rescue therapy with JAK inhibitor.

POC and formal IFX levels were obtained 48 h after the first infusion of dose-intensified IFX.
Repeat flexible sigmoidoscopy 48 h after the second IFX infusion.
Evidence of systemic toxicity defined as tachycardia (>90 beats/min) or fever (>37.8°C) as per Truelove and Witts Severity Index for UC. 42
Received Tofacitinib. All other patients received upadacitinib.
First IFX dose at day 1 due to high-dose oral corticosteroid failure in the community.
First IFX dose at day 6 due to delayed transfer from remote and regional hospital.
CLIA, chemiluminescence immunoassay; CPR, C-reactive protein; Hb, haemoglobin; IFX, infliximab; JAK, Janus kinase; MES, Mayo Endoscopic Score; NP, not performed; POC, point of care assay.
Four out of six patients met the criteria for inpatient clinical response (<4 non-bloody stools per day) within 72 h of starting sequential JAK-inhibitor rescue therapy (Figure 3 and Table 3). Two patients (Cases #1 and #6) failed JAK-inhibitor treatment and underwent inpatient colectomy (both on day 15). JAK-inhibitor therapy was ceased on the decision to proceed to colectomy. Whilst one patient recovered well from emergent colectomy and was discharged on Day 23 (Case #1), the other patient (Case #6), developed a

Clinical and biochemical parameters at admission, Day 3 of admission, the commencement of JAK inhibitor, Day 3 post-JAK inhibitor, and Day 90 post-discharge.
Clinical and biochemical outcomes following sequential JAK-inhibitor rescue therapy.

Median (IQR) or % (
BD, twice daily; CRP, C-reactive protein; FCP, faecal calprotectin; IQR, interquartile range; JAK, Janus kinase; LOS, length of stay; OD, once daily; PRO-2, patient-reported outcome 2.
All four successful sequential JAK-inhibitor patients transitioned to maintenance dosing (upadacitinib 30 mg OD; tofacitinib 10 mg BD) and subsequently achieved corticosteroid-free clinical remission (PRO-2 < 1) at 90 days. For the three patients who remained on upadacitinib on discharge, the dose reduction followed a standard 8-week induction; for the one tofacitinib-treated patient the dose was reduced from 10 mg TDS to 10 mg twice daily (BD) after 14 days based on clinical and biochemical response. We note that whilst some centres administer a similar 2-week duration of the off-label high-dose regimen of tofacitinib (10 mg TDS), 25 others recommend a shorter course of nine doses before de-escalating to 10 mg BD.23,38
Three of four patients achieved biochemical remission (FCP <150 µg/mL
Endoscopic reassessment
Two patients (Cases #2 and #3) underwent repeat endoscopy within the follow-up period (37 and 17 weeks post-discharge), both had an MES of 0 and median Ulcerative Colitis Endoscopic Index of Severity of 0 (Figure 4).
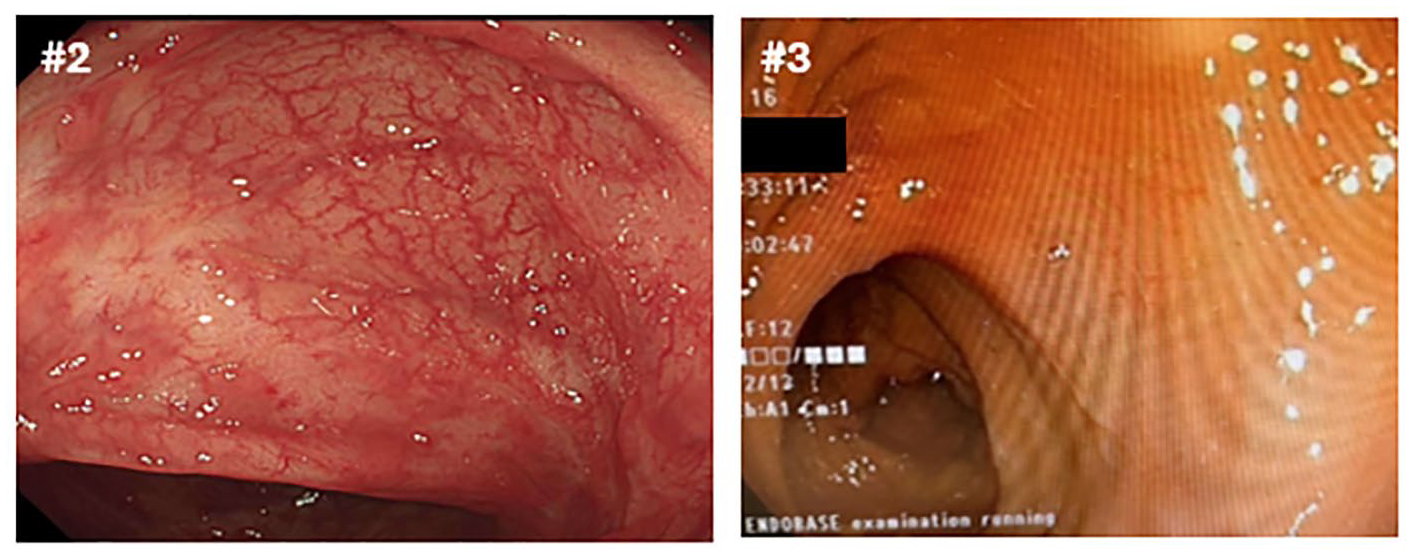
Representative endoscopic images of the sigmoid colon taken on follow-up endoscopy for Cases #2 and #3 (median 191 days following commencement of JAK inhibitor; 259 and 124 days post-commencement of JAK inhibitor, respectively), Median MES 0, UCEIS 0.
Discussion
This case series highlights the potential role of JAK inhibitors as sequential rescue therapy in ASUC refractory to IVCS and dose-intensified infliximab. Our experience may contribute to clinician confidence in justifying the safety of trialling sequential rescue therapy with a JAK inhibitor, amidst both the risk of deferring potentially life-saving surgery and patient concern surrounding colectomy. This treatment strategy, along with its inherent risks, differs subtly but significantly from the more commonly described loss of response to outpatient infliximab and subsequent JAK inhibitor ASUC rescue therapy.
In our small refractory cohort, we report that 33% (2/6) of patients underwent colectomy at 90 days. Our colectomy rate appears higher than the 14% (19/132) 90-day colectomy rate observed in a systematic review of tofacitinib rescue therapy in ASUC by Steenholdt et al. 24 Indeed, among the subset of 11 patients who underwent sequential IVCS followed by inpatient rescue treatment with infliximab and then third-line tofacitinib, as per our approach, we note that the colectomy rate was even lower at 9% (1/11). The literature on upadacitinib is less robust compared to tofacitinib. A systematic review by Damianos et al. 48 (2024) included 55 ASUC patients treated with upadacitinib and reported a 90-day colectomy rate of 16% (9/55). However, exploring the peer-reviewed data, only Zinger et al. 29 and Berinstein et al. 37 included patients who received sequential infliximab and upadacitinib therapy with a 90-day colectomy rate of 14% (1/7). Despite the limited number of JAK-inhibitor cases and the absence of matched cohort studies or randomised controlled trials, the available literature suggests that colectomy rates for sequential rescue treatment with upadacitinib or tofacitinib in ASUC may be slightly lower27,37,49 compared to the sequential use of infliximab and ciclosporin, or vice versa – where three studies reported a combined colectomy rate of 47% (71/152) for the latter approach.13–15
In our cohort, of the two patients who failed JAK-inhibitor rescue therapy and required urgent inpatient colectomy, one developed a post-operative complication with sepsis. This necessitated two laparoscopic washouts at 7 and 20 days post-colectomy, along with intravenous antibiotics and an extended hospital stay. This represents a significant adverse event and highlights the potential dangers of deferring colectomy and combining potent immunosuppressive therapies. In a retrospective study of over 2000 patients undergoing emergency colectomy for UC, Kaplan et al. 50 reported that the adjusted odds of morbidity among patients who underwent colectomy after 6 and 11 days was 2.1- and 2.9-fold higher than those who had surgery within 3 days of admission, highlighting the potential risks of deferring colectomy decisions.
We report no additional adverse events over a median follow-up of 173 days among the four patients successfully treated with a JAK inhibitor, although we recognise a significant selection bias, as sequential JAK-inhibitor rescue therapy was limited to carefully chosen patients who were physiologically stable at the start of treatment. In comparison, in a systematic review of 148 patients receiving rescue tofacitinib, notable adverse events included infections in 9% (
JAK inhibitors have theoretical superiority over protein-bound rescue agents, with preserved pharmacokinetics amidst fluctuating acute phase reactants and rapid washout if colectomy is warranted.51–53 Sequential JAK inhibition following anti-TNF-alpha (anti-TNFα) blockade may yield synergistic efficacy to overcome the inflammatory burden of fulminant ASUC by targeting alternative non-TNFα-mediated pathways, that may be present in as many as 25% of patients with ASUC.12,54 Berinstein et al. 23 found the colectomy rates among anti-TNFα naïve (22%) and exposed (25%) patients receiving tofacitinib for ASUC are similar, despite the latter cohort representing a more treatment refractory phenotype, further suggesting the importance of non-TNFα inflammatory pathways.
A future approach among patients with a partial response to rescue infliximab may be dual-targeted therapy (DTT), whereby a JAK inhibitor is commenced alongside infliximab.25,55,56 Gilmore et al.
25
described a case series involving five patients with prior ASUC and partial responses to intensified infliximab dosing after a median treatment duration of 8 months who were then treated with DTT using infliximab and tofacitinib for at least 90 days. All five patients avoided colectomy, with four achieving both clinical and biochemical responses. No VTE or cardiovascular events were reported; however, one patient developed herpes zoster, which was successfully treated with oral valaciclovir. Additional reports of opportunistic infections, including
Our case series is limited by cohort size and follow-up. With a longer duration of follow-up, we would expect the number of colectomies to increase. In the systematic review of tofacitinib for ASUC by Steenholdt et al, the authors found that colectomy-free survival reduced from 85% at 30 days to 69% at 180 days. 24 However, deferring colectomy may not necessarily indicate treatment failure; rather, downstaging the inflammatory burden with a JAK inhibitor might facilitate an elective rather than an emergent colectomy, mitigating the impact of potent immunosuppression on the risk of infection and anastomosis breakdown. Additionally, this delay allow patients time to adjust to the idea of surgery, which may still represent an advantageous outcome.
Despite the small number of patients, this case series is of interest given the use of a dose-intensified and in most instances, dose-accelerated infliximab rescue treatment, combined with drug-level monitoring to distinguish between pharmacokinetic and pharmacodynamic failure. Whilst the recently published PREDICT-UC ASUC study found no difference in Day 7 clinical response rates between patients receiving first doses of 10 and 5 mg/kg infliximab, there appeared to be a subgroup of patients with greater baseline inflammation (CRP >50 mg/L) and lower albumin (<25 g/L) who benefited from the higher 10 mg/kg first dose, suggesting that higher doses of infliximab remain important for patients with the highest levels of inflammatory burden. 12
While recent case series have highlighted the safety and efficacy of JAK inhibitors as rescue therapy in infliximab-experienced patients,23–29,49,57 the optimal positioning and sequencing of JAK inhibitors within the ASUC treatment hierarchy remains unclear. Exploring alternative strategies for medical salvage therapy in ASUC remains a challenge due to the severity of the disease phenotype, the limited number of efficacious agents, and the significant morbidity and mortality associated with unchecked systemic inflammation. To further reduce colectomy rates and shorten hospital stays, one paradigm-shifting approach to ASUC treatment might include initiating day-1 rescue therapy in select patients predicted to fail corticosteroids using a tool such as the RoR score. 30 Future prospective and randomised studies are required to confirm the safety and efficacy of JAK inhibitors as first- and second-line rescue therapy or as part of a novel combination of advanced therapies, in medically refractory ASUC.
Conclusion
There is a promising role for JAK inhibitors as sequential rescue therapy following the failure of dose-intensified infliximab in select patients with corticosteroid-refractory ASUC.
Footnotes
Appendix A
Appendix B
CARE 2016 checklist
| Topic | Item | Checklist | Line/page |
|---|---|---|---|
| Title | 1 | The words ‘case report’ should be in the title along with the area of focus | 6–7/1 |
| Key words | 2 | Four to seven key words – include ‘case report’ as one of the key words | 62/2 |
| Abstract | 3a | Background: what does this case report add to the medical literature? | 26–30/2 |
| Abstract | 3b | Case summary: chief complaint, diagnoses, interventions and outcomes | 31–47/2 |
| Abstract | 3c | Conclusion: what is the main ‘take-away’ lesson from this case? | 48–49/2 |
| Introduction | 4 | The current standard of care and contributions of this case – with references (1–2 paragraphs | 65–97/3 |
| Timeline | 5 | Information from this case report organised into a timeline (table or figure) | Figure 1/19 |
| Patient information | 6a | De-identified demographic and other patient or client specific information | 152–163/6 |
| Patient information | 6b | Chief complaint – what prompted this visit? _______ | 148–150/6 |
| Patient information | 6c | Relevant history including past interventions and outcomes | 152–163/6 |
| Physical exam | 7 | Relevant physical examination findings | 165–173/6 |
| Diagnostic assessment | 8a | Evaluations such as surveys, laboratory testing, imaging, etc. | 165–173/6 |
| Diagnostic assessment | 8b | Diagnostic reasoning including other diagnoses considered and challenges | 165–173/6 |
| Diagnostic assessment | 8c | Consider tables or figures linking assessment, diagnoses and interventions | Table 1/16 |
| Diagnostic assessment | 8d | Prognostic characteristics where applicable | 172–173/6 |
| Interventions | 9a | Types such as life-style recommendations, treatments, medications, surgery | 175–205/6–7 |
| Interventions | 9b | Intervention administration such as dosage, frequency and duration | 183–189/6–7 |
| Interventions | 9c | Note changes in intervention with explanation | 201–205/7 |
| Interventions | 9d | Other concurrent interventions | 117–126/4 |
| Follow up and outcomes | 10a | Clinician assessment (and patient or client assessed outcomes when appropriate) | 201–205/7 |
| Follow up and outcomes | 10b | Important follow-up diagnostic evaluations | 215–217/7 |
| Follow up and outcomes | 10c | Assessment of intervention adherence and tolerability, including adverse events | 191–217/7 |
| Discussion | 11a | Strengths and limitations in your approach to this case | 219–240/8 |
| Discussion | 11b | Specify how this case report informs practice or CPG | 219–225/8 |
| Discussion | 11c | How does this case report suggest a testable hypothesis? | 296–305/10 |
| Discussion | 11d | Conclusions and rationale | 219–225/8 |
| Discussion | 11e | Conclusions and rationale | 307–309/10 |
| Patient perspective | 12 | When appropriate include the assessment of the patient or client on this episode of care | NA |
| Informed consent | 13 | Informed consent from the person who is the subject of this case report is required by most journals | 103–105/4 |
| Additional information | 14 | Acknowledgement section; competing interests; IRB approval when required | 312–331/11 |
CPG, clinical practice guidelines.
Acknowledgements
We acknowledge and thank the patients and staff of the Royal Brisbane and Women’s Hospital Department of Gastroenterology and Hepatology for their invaluable contributions to this work.
Author’s note
The authors believe the manuscript meets the criteria for a publishable case series (Appendix A)The authors have read and completed the 2016 CARE checklist (![]() ).
).
