Abstract
Pityriasis rubra pilaris is a rare inflammatory dermatosis characterized by orange-red confluent plaques, hyperkeratotic follicular papules, palmoplantar keratoderma, and, in some cases, erythroderma. The etiology of pityriasis rubra pilaris is unclear. This condition is often treated with oral retinoids and topical corticosteroids, and more recently, biological agents have become the mainstay of treatment. However, there is a paucity of high-quality evidence on the safety and effectiveness of these agents, and the disease often remains refractory to therapy. Herein, we present a case of pityriasis rubra pilaris with a favorable response to treatment with upadacitinib, a Janus kinase inhibitor, which has not been previously reported in the literature for the management of this condition.
Introduction
Pityriasis rubra pilaris (PRP) is a rare inflammatory papulosquamous disorder characterized by salmon-colored scaly confluent plaques with islands of sparing, hyperkeratotic follicular papules, and palmoplantar keratoderma. In some cases, it may progress to erythroderma. 1 The pathophysiology of PRP is poorly understood, but is thought to be related to the interleukin (IL) 23/Th17 axis. 2 PRP is commonly divided into six clinical subtypes: classical adult, atypical adult, classical juvenile, circumscribed juvenile, atypical juvenile, and HIV-associated, with the classical adult type accounting for half of all cases.3,4 PRP is a challenging disease to manage, and the refractory nature of this disease has significant impact on patient quality of life. Currently, there are no randomized clinical trials to support the use of any therapy for PRP. Oral retinoids are commonly used based on case reports and case series of favorable responses to isotretinoin, acitretin, and etretinate in PRP.5–7 However, systemic retinoids are associated with significant adverse effects, including teratogenicity, dyslipidemia, and ocular side effects. 1 Other conventional therapies include topical corticosteroids, cyclosporine, methotrexate, and phototherapy. 4 Biological agents such as tumor necrosis factor-α inhibitors, IL-17 and IL-23 inhibitors, are increasingly being employed as first-line systemic therapies for PRP.8,9 Data regarding their efficacy are limited to case reports and two single-arm trials supporting the use of anti-IL-17 agents.10,11
Upadacitinib is an oral selective Janus kinase 1 (JAK1) inhibitor indicated in the treatment of moderate-to-severe atopic dermatitis, psoriatic arthritis, rheumatoid arthritis, ankylosing spondylitis, and ulcerative colitis.12,13 There are no prior reports of its use in the management of PRP. Here, we describe a case of PRP with favorable response to upadacitinib.
Case report
A 26-year-old woman with no comorbidities was referred for management of long-standing PRP in 2015. Her family history was non-contributory. She was initially diagnosed with classical juvenile PRP at the age of 2 years and had tried several therapies throughout her youth, including topical steroids and calcineurin inhibitors, isotretinoin, and phototherapy, with limited success. At the time of assessment in 2015, she had not undergone any treatment for several years. She presented with confluent salmon-colored scaly patches and plaques on the face, extremities, and trunk, with islands of sparing on the antecubital and popliteal fossae. She also had nutmeg grater-like follicular papules on the dorsal hands and mild hyperkeratosis of the palms, consistent with her diagnosis of PRP. She opted for a topical corticosteroid, calcineurin inhibitor, and phototherapy at that time.
In 2016, she was started on ustekinumab, an IL-12/23 inhibitor, at standard dosing. She was eventually updosed to 90 mg every 8 weeks. She initially had significant improvement with much thinner, less erythematous plaques on her trunk and extremities, but still involving ~60% body surface area (BSA; Figure 1(a) and (b)). In late 2021, she presented with no further improvement. Her skin remained stable with ~60% BSA with thin pink plaques, and she wanted to try another agent to see whether her skin could be cleared further. She was switched to ixekizumab, an IL-17A inhibitor. On ixekizumab, she flared, and her skin was worse than prior to starting ustekinumab. She had thick red plaques over almost 80% of her body, making her skin feel tight. The drug was subsequently discontinued (Figures 2(a), 3(a), and 4(a)). In May 2022, she was started on a trial of off-label upadacitinib 15 mg/d. She was reassessed at day 14 of treatment and found to have a 25% improvement (Figures 2(b), 3(b), and 4(b)). The dose was increased to 30 mg/d at that time. By week 6 of treatment with upadacitinib, she had a 65% overall improvement in BSA involvement and thinner plaques in keeping with her initial response to ustekinumab (Figures 2(c) and 3(c)). She experienced headache on upadacitinib, but otherwise tolerated it very well with no serious side effects. She found her response to upadacitinib to be the same as her initial response to ustekinumab, but ultimately preferred to receive intermittent injections rather than continue oral therapy. In July 2022, she was started on guselkumab and her skin remains stable at a similar level to what she experienced on ustekinumab, which she is happy with.
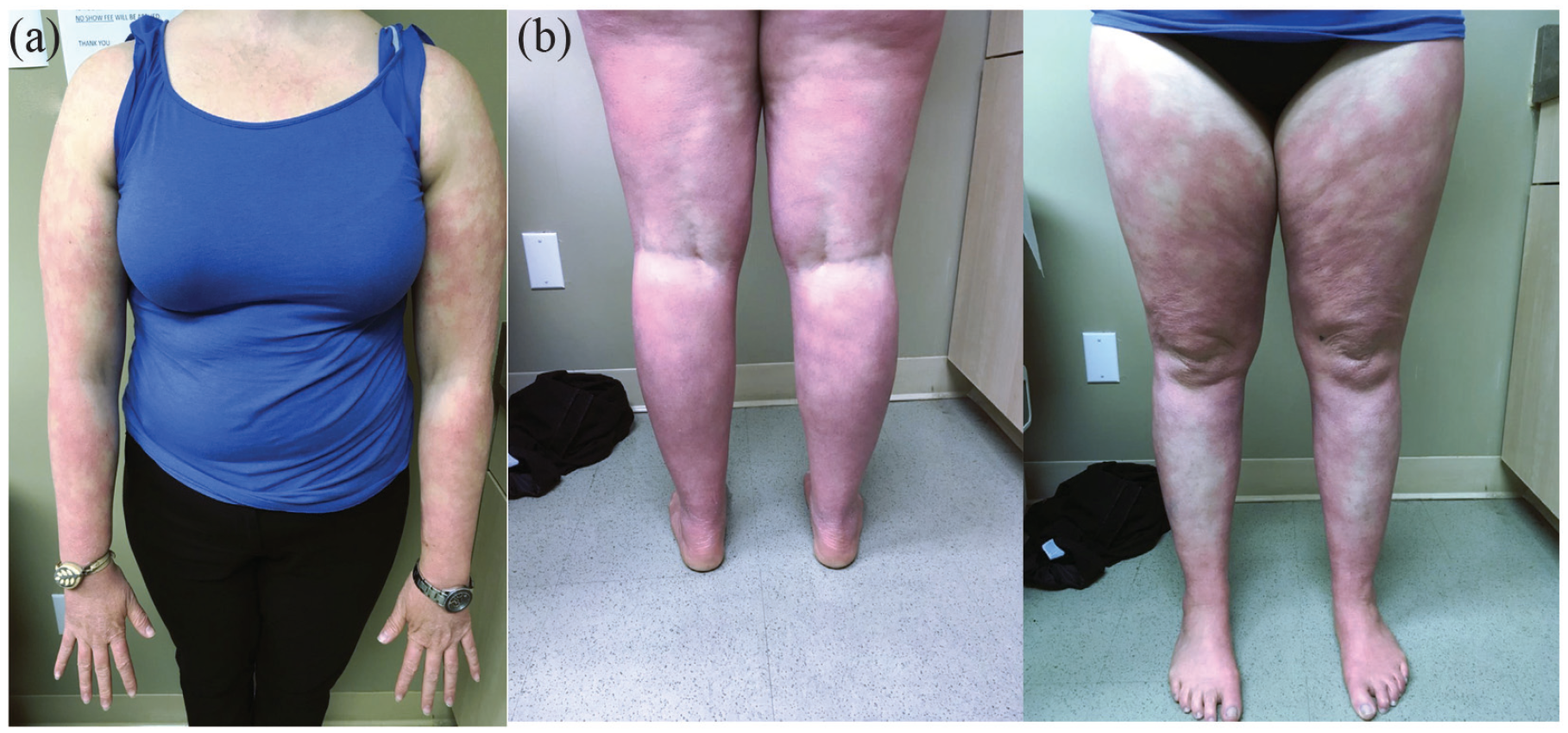
(a) Confluent pink patches on the upper extremities with islands of sparing over the antecubital fossae and (b) confluent pink patches on the lower extremities with islands of sparing over the popliteal fossae.
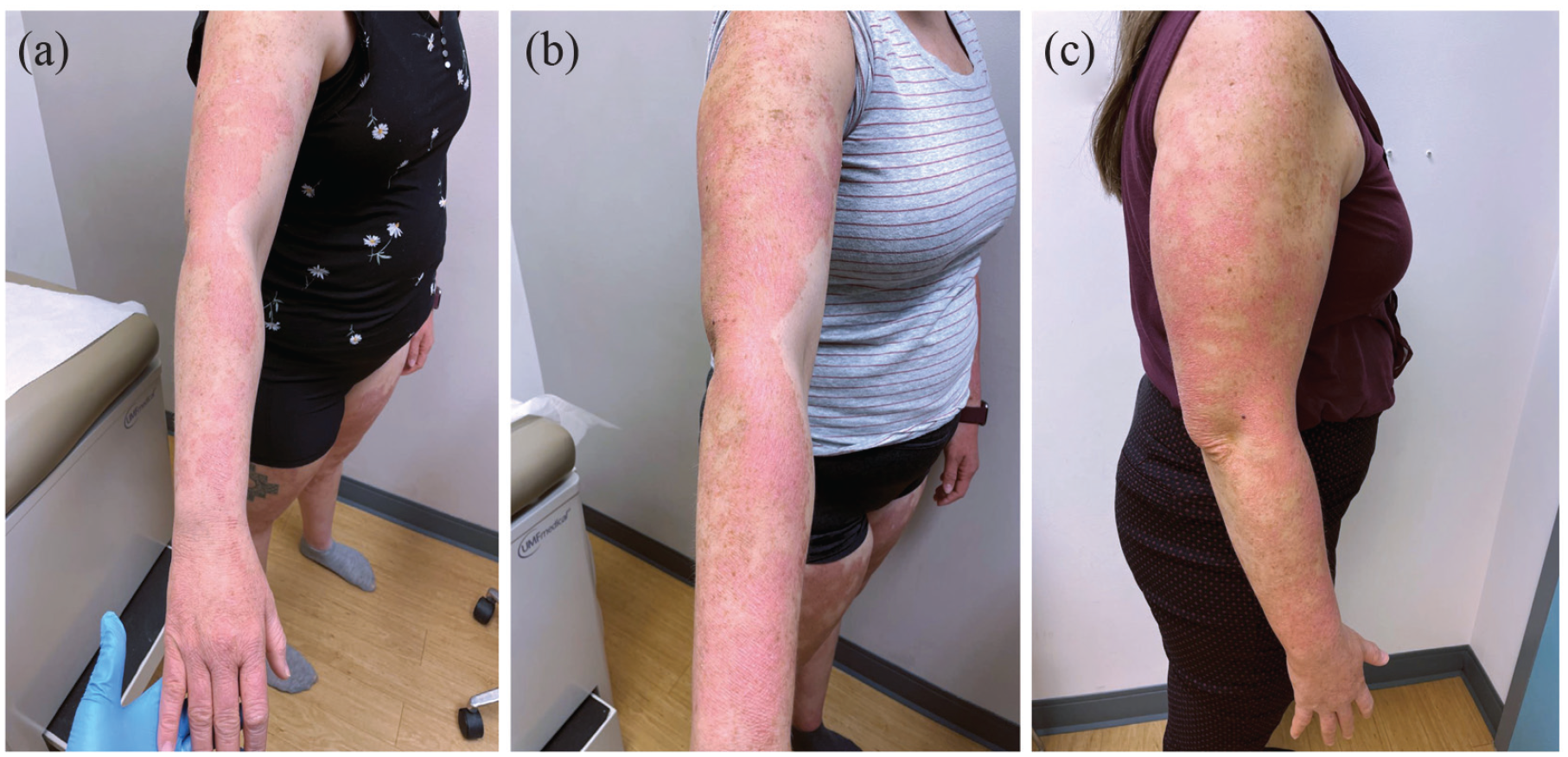
(a) Pink confluent scaly plaques on the upper extremities with islands of sparing, (b) improvement following 14 days of treatment with upadacitinib, and (c) improvement following 6 weeks of treatment with upadacitinib.

(a) Red confluent scaly plaques on the lower extremities with islands of sparing, (b) improvement following 14 days of treatment with upadacitinib, and (c) near-complete resolution following 6 weeks of treatment with upadacitinib.

(a) Erythema and xerosis of dorsal hand with nutmeg grater-like papules and (b) improvement following 14 days of treatment with upadacitinib.
Discussion
This report provides support for the novel use of upadacitinib in PRP. Here, we illustrate the efficacy of upadacitinib in achieving a partial clinical response similar to that experienced with ustekinumab and guselkumab, in a case of PRP previously treated with conventional therapies, including topical corticosteroids, isotretinoin, phototherapy, and biological agents. Furthermore, this clinical benefit was achieved rapidly with minimal side effects.
The use of JAK inhibitors has not been investigated in the management of PRP. However, there is increasing evidence, including randomized, double-blinded, and placebo-controlled clinical trials, supporting the use of JAK inhibitors in other inflammatory dermatoses such as atopic dermatitis and psoriasis,14–16 which share many clinical and histological features with PRP. Pro-inflammatory cytokines involved in the pathogenesis of psoriasis and atopic dermatitis, such as IL-12/IL-23 and IL-4/IL-13/IL-31, respectively, act via JAK/STAT signaling pathways. The inhibition of JAK/STAT pathways by agents such as upadacitinib thereby suppresses intracellular cytokine response.17,18 The IL-23/IL-17 axis may be implicated in the pathogenesis of PRP. 2 In light of this, there may be a promising therapeutic role for these agents. Moreover, multiple case studies have reported positive outcomes with the use of IL-17A inhibitors such as ixekizumab and secukinumab, and IL-23 inhibitors such as ustekinumab and guselkumab, in PRP.8,9 In this case, the patient did not have a favorable response to ixekizumab, which may be related to insufficient blockade of cytokine release due to its specific targeting of IL-17A. Conversely, ustekinumab, which targets IL-23 and IL-12, may have been more effective due to its inhibition of additional cytokine release. Thus, the upstream inhibition of cytokine signaling pathways represents a biologically plausible mechanism for successful treatment of PRP using JAK inhibitors. Although upadacitinib is more selective for JAK1 inhibition, which is not involved in the IL-23 axis, 18 it stands to reason that inhibition of JAK1 has positive effects on modulating the immune response involved in the pathogenesis of PRP. Future studies are needed to investigate the effectiveness of agents with increased JAK2 selectivity, as they may play a more direct role in the IL-23/IL-17 axis.
To our knowledge, this is the first report of treatment of PRP with upadacitinib. This patient had a favorable response similar to what she achieved with ustekinumab and guselkumab. This case offers JAK inhibitors such as upadacitinib as a potentially promising alternative therapeutic approach for PRP, given the successful response in a patient previously trialed on conventional therapies. Furthermore, studies are required to ascertain their safety and effectiveness, to improve patient satisfaction and quality of life.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patient provided informed consent for publication of the case report and images.
