Abstract
Pyoderma gangrenosum (PG) is an immune-mediated skin condition that is commonly associated with inflammatory bowel diseases (IBD). Its diagnosis and management are often challenging due to the heterogeneity of the clinical presentation and the need for an individualized therapeutic approach. We present the case of a 19-year-old Caucasian male with Crohn’s disease, who received therapy with a biological agent and developed severe PG in the retroauricular region following mastoidectomy for mastoiditis, which was complicated by cerebral venous sinus thrombosis. The diagnosis of PG was made with the help of the PARACELSUS-score based on lesion characteristics, histology, the patient’s IBD history, and lack of response to antibiotics. Treatment with upadacitinib, a selective Janus kinase 1 inhibitor, was initiated after resolution of the previous thrombotic event. The patient responded rapidly to therapy, with a notable reduction of the local inflammatory activity and wound size, and ultimately complete healing. Anticoagulation therapy did not need to be continued during treatment. The patient has been on upadacitinib for 25 months to date without adverse events or recurrence of thrombosis. This case highlights upadacitinib as a promising and safe treatment option for PG, even in patients with Crohn’s disease and a history of thrombotic events after careful benefit–risk assessment.
Plain language summary
A 19-year-old man with Crohn’s disease developed a painful skin ulcer behind his ear after surgery for a severe ear infection and a blood clot in a brain vein. Standard treatments and antibiotics failed. Doctors diagnosed pyoderma gangrenosum, a rare immune-related skin disease. He was successfully treated with upadacitinib, a medication that controls immune inflammation. The wound healed completely, and no new clots occurred. This case suggests upadacitinib may be safe and effective for difficult cases of pyoderma gangrenosum.
Introduction
Inflammatory bowel disease (IBD) is a chronic, immune-mediated disorder of the gastrointestinal tract characterized by a relapsing-remitting course with varying phenotypes, mainly consisting of Crohn’s disease (CD) and ulcerative colitis (UC). IBD is frequently associated with extraintestinal manifestations, which can affect up to 47% of patients. It is noteworthy that up to 24% of these manifestations may occur before the onset of gastrointestinal symptoms. 1 Pyoderma gangrenosum (PG) is a rare skin condition characterized by progressively enlarging ulcerative lesions that may be triggered by minor trauma, known as the pathergy phenomenon. It typically develops as tender papules or sterile pustules, which rapidly progress to painful necrotic ulcers. These ulcers are distinguished by violaceous undermined edges and surrounding erythema and may also have a sterile discharge. 2 PG can be associated with various diseases, including hematological disorders, inflammatory arthritis, and connective tissue disease. With regard to the gastrointestinal tract, 18% of PG patients have been documented with concomitant IBD.3,4 It therefore represents the second most prevalent cutaneous manifestation of IBD, affecting approximately 0.5% to 2.6% of patients diagnosed with IBD. However, the development of PG seems to be independent from IBD activity, and PG has been found to be more frequently associated with UC than with CD.5,6
The diagnosis of PG is made with the PARACELSUS score or Delphi criteria.7,8 However, diagnosis remains challenging as other causes, depending on subtype and clinical presentation, have to be ruled out. Further, the response to immunosuppressive therapy is a key component of the diagnosis process, which can only be evaluated retrospectively. Misdiagnosis leads not only to delay in adequate treatment but can also induce worsening of the disease by surgical debridement, due to the described pathergy phenomenon.8,9
Here, we present the case of a patient with acute PG and underlying CD with a history of cerebral venous thrombosis, who was successfully treated with the selective Janus kinase (JAK) 1 inhibitor upadacitinib. This case indicates that the usage of selective JAK inhibitors should be evaluated in patients with PG. According to the available data, a history of thrombotic disease is not a contraindication for JAK inhibitors. They can be used under careful risk–benefit assessment. The reporting of this case report conforms to the CARE statement (CARE guidelines for case reports; Supplemental Material). 10
Case presentation
A 19-year-old Caucasian male had been diagnosed with CD at the age of 11. His disease phenotype was penetrating and stricturing upper gastrointestinal, ileocolonic, and perianal CD (Montreal classification A1 L3+L4 B3p). The disease course was steroid-dependent, and there was no history of smoking.
Systemic treatment was initiated in March 2016 with the anti-Tumor Necrosis Factor (TNF) antibody infliximab. However, this therapy was discontinued in September 2016 due to the paradoxical occurrence of psoriasiform skin lesions on the trunk, extremities, palms, soles, and scalp. A skin biopsy in November 2016 revealed nonspecific chronic dermatitis, with no histological evidence of psoriasis vulgaris. In February 2017, the treatment was switched to the anti-TNF antibody adalimumab at a dose of 40 mg biweekly. Azathioprine was used as combination therapy with anti-TNF agents, resulting in clinical remission. There was no history of skin cancer, and regular dermatologic surveillance was recommended during immunosuppressive therapy.
Since June 2022, the patient suffered from right-sided otitis media, which progressed to chronic otitis media despite repeated antibiotic treatments. On October 21, 2022, the patient presented at our emergency department with a headache, blurred vision in his right eye, and suspected bilateral papilledema. Cranial magnetic resonance imaging revealed thrombosis of the right transverse sinus, sigmoid sinus, and jugular vein bulb, as well as mastoiditis, presumed to be septic in origin secondary to chronic otitis media (Figure 1). During the course of the day, the patient exhibited right-sided paralysis of the abducens and facial nerve, as well as latent paresis of his right arm. A mastoidectomy was performed on October 22, 2022, and the patient received antibiotic therapy initially with clindamycin, later with ceftriaxone and meropenem. Anticoagulation was also initiated with weight-adjusted enoxaparin, subsequently transitioned to dabigatran for 6 months, resulting in complete radiological resolution of the cerebral venous sinus thrombosis after 5 months. It should also be noted that, due to the development of eczematous skin lesions with scaling, suspected to represent adalimumab-associated psoriasis affecting the scalp, trunk, left arm and elbow, left knee, and lower leg, and following evaluation by our interdisciplinary inflammation board, therapy was switched to ustekinumab in January 2023.
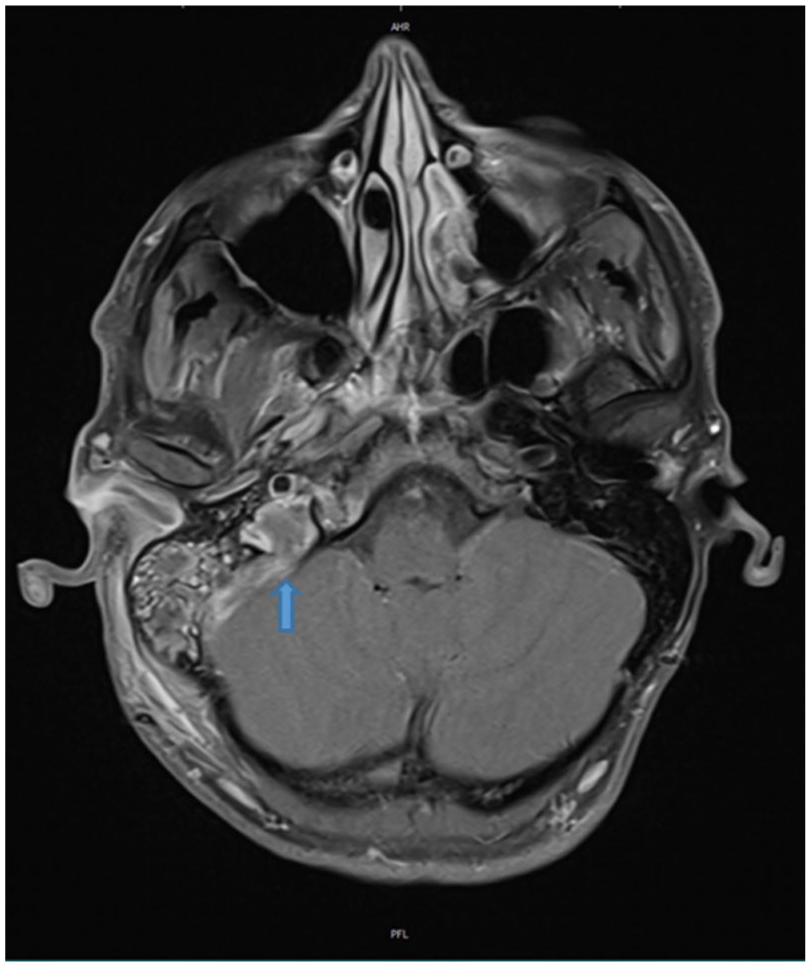
Magnetic resonance imaging assessment (axial post-contrast T1-weighted) at the time of presentation to the emergency department revealed thrombosis involving the right transverse and sigmoid sinuses (blue arrow) as well as the bulb of the right internal jugular vein. No evidence of venous infarction, edema, or hemorrhage was detected. Suspicion of right-sided mastoiditis without definitive imaging evidence of accompanying meningitis or encephalitis.
Two months after initiation of ustekinumab, the patient underwent colonoscopy and upper gastrointestinal endoscopy. Colonoscopy revealed ileocolitis with a Simple Endoscopic Score for Crohn’s Disease (SES-CD) score of 4, while upper endoscopy demonstrated gastritis and duodenitis, as well as inflammatory lesions of the esophagus.
Postoperatively, the patient suffered from poor wound healing behind his right auricle, requiring multiple readmissions to the Department of Otorhinolaryngology and repeated antibiotic treatments. Clinically, he developed a retroauricular ulcer approximately 1 month after surgery, which progressively enlarged and was accompanied by widespread superficial erosions involving large parts of the neck and the outer ear canal; the lesion shown in Figure 2 represents the clinical appearance 6 months after mastoidectomy. He reported trismus and significant pain. Staphylococcus aureus and Staphylococcus intermedius were identified in swabs taken from the skin lesion. Despite multiple courses of antibiotics, the wound healing remained suboptimal. Differential diagnoses included impaired wound healing post-mastoidectomy and cutaneous extraintestinal manifestation of CD. A skin biopsy was obtained in January 2023. Histopathological examination revealed a dense neutrophilic infiltrate involving the dermis and subcutis, accompanied by areas of tissue necrosis, findings consistent with PG. Additional biopsies were obtained to rule out mycological and mycobacteriological causes. Based on the lack of response to antibiotic therapy, clinical presentation, the patient’s history of IBD, and histological findings, a diagnosis of PG was made. The calculated PARACELSUS score was 19, exceeding the cut-off value of 10 points, which indicates a high likelihood of PG.

Six months after mastoidectomy, the patient suffered from retroauricular ulceration with widespread erosions and superficial ulceration extending to the neck. There was a purulent discharge beneath the right auricle.
The case was discussed in our Interdisciplinary Inflammation Board (MDT) of the University Hospital Erlangen, including gastroenterology, dermatology, rheumatology, and otorhinolaryngology specialists. Following benefit–risk assessment, initiation of upadacitinib was recommended as a therapeutic option targeting both CD and PG. The diagnostic findings, therapeutic options, and potential risks were discussed in detail with the patient, who agreed with our proposed therapeutic procedure. Accordingly, ustekinumab was discontinued, and upadacitinib therapy was initiated in May 2023 at a dose of 45 mg/day for 12 weeks, followed by a dose reduction to 30 mg/day. After 5 months, the patient showed significant clinical improvement, with a substantial reduction of erythema and wound size (Figure 3). After 7 months, complete healing of PG was achieved. Along with clinical, endoscopic, and histological remission of CD, the upadacitinib dose was reduced to 15 mg/day after 14 months. Upadacitinib was well tolerated, without any adverse events during treatment. Therapeutic response to both CD and PG was sustained to the latest follow-up, 25 months after initiation of upadacitinib (Figures 4 and 5).
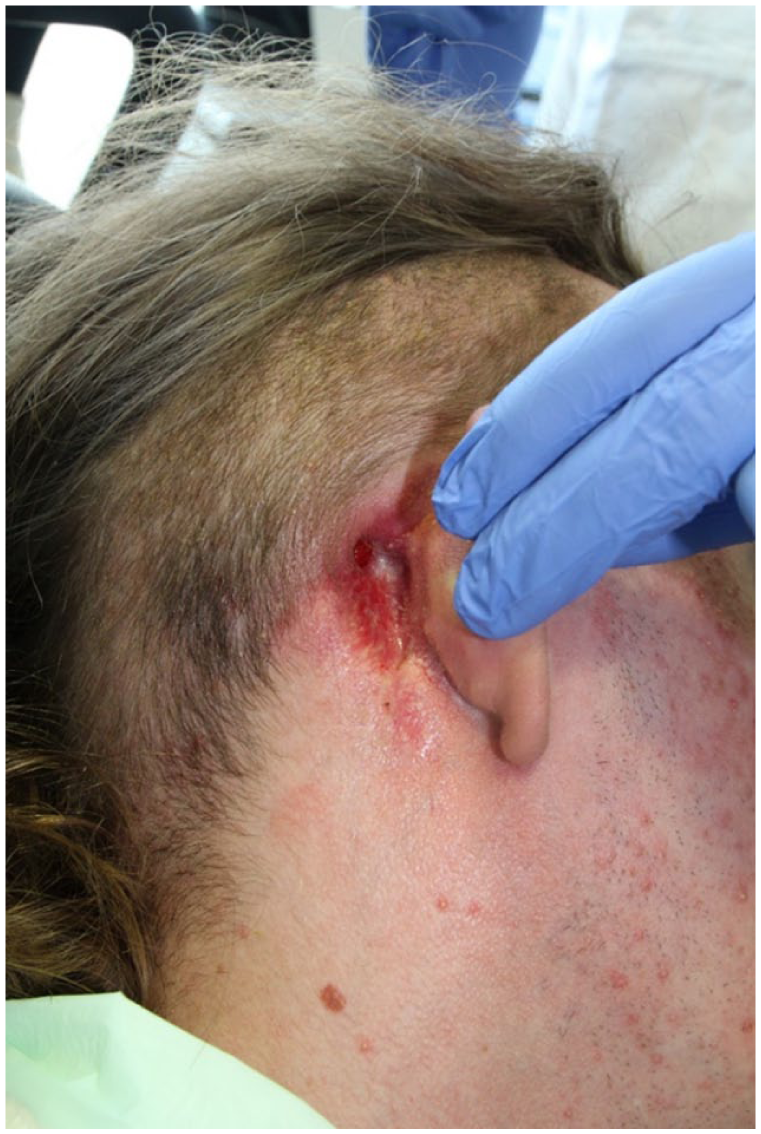
Five months after starting upadacitinib therapy, clinical improvement of the retroauricular lesion was observed, with decreased erythema and signs of healing compared to the initial presentation.
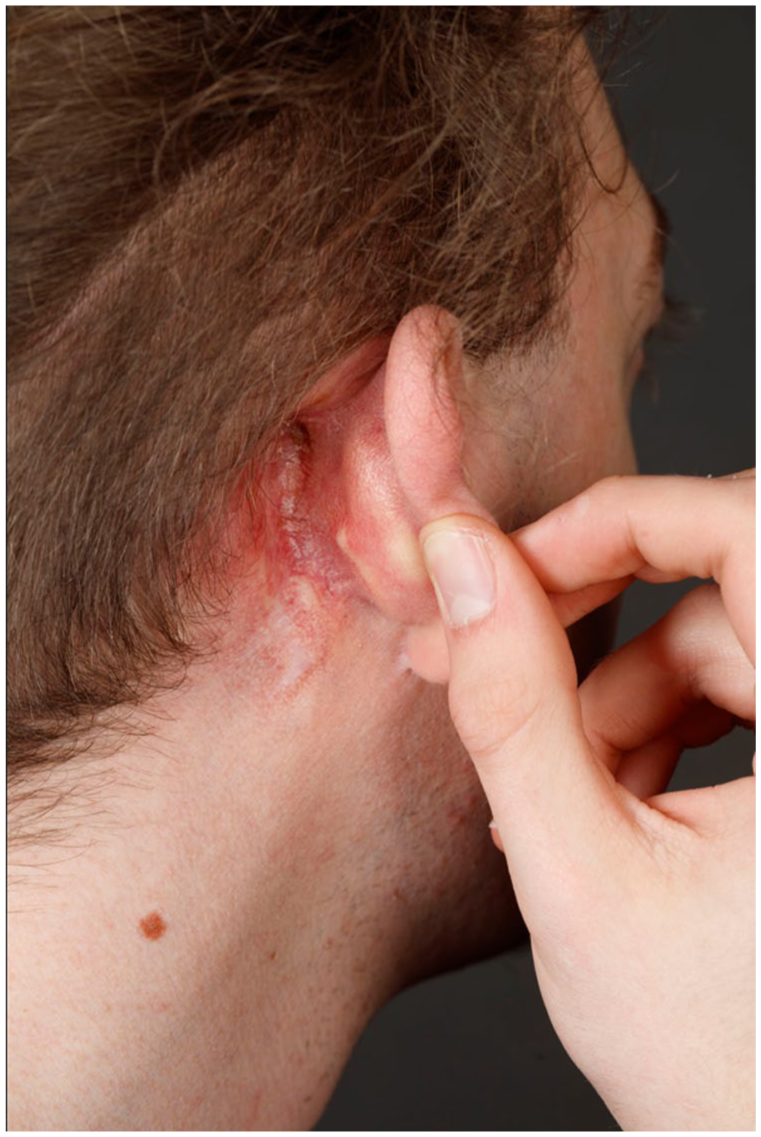
Seventeen months after initiation of upadacitinib therapy, the retroauricular lesion shows marked improvement, being completely epithelialized with minimal residual erythema and evidence of scar formation.

Clinical timeline of otologic disease, CVST, development of PG, and subsequent therapeutic interventions.
Discussion
The exact pathophysiology of PG remains unclear; however, the existing data suggest a multifactorial and complex pathogenesis. A growing body of evidence indicates that genetic predisposition and abnormal activation of the innate immune system create an environment conducive to the development of PG. In PG lesions, aberrant activation of neutrophils and T-cells is present, along with inflammatory mediators such as IL-1β, IL-8, IL-17, IL-23, and TNF-α.11,12 TNF-α promotes the expression of adhesion molecules on endothelial cells, aiding neutrophil migration to inflamed areas, a hallmark of PG autoinflammation. 13 Recent findings suggest that interleukin-17E (IL-25) plays a role in neutrophil-driven PG, activating macrophages and inducing chemokines such as CXCL1, CXCL10, and CCL20, which promote neutrophil recruitment. 14 Other chemotactic factors such as CCL3, CCL5, IL-8, and IL-16 are also found to be upregulated.15,16 In addition, trauma can trigger or exacerbate PG through pathergy, releasing autoantigens and IL-36 by damaged keratinocytes and upregulating the expressions of IL-8 and IL-17. 17 Overall, these findings suggest that PG is an autoinflammatory disorder. In our case, the combination of CD-related immune dysregulation and the tissue trauma induced by mastoidectomy likely contributed synergistically to the development of PG.
When PG is suspected, clinicians must consider a broad differential diagnosis, as many diseases can mimic the appearance of PG. These include various autoimmune and vasculitic diseases, such as polyarteritis nodosa, Behçet’s disease, and systemic lupus erythematosus. Vascular disorders, including venous insufficiency and arterial micro-occlusions, may also result in chronic, painful ulcers. Hematologic conditions, such as cryoglobulinemia and sickle cell disease, can further complicate the clinical presentation. 18 Furthermore, infectious etiologies, including fungal infections such as cryptococcosis, in addition to mycobacterial, bacterial, viral, and parasitic infections—must be considered early in the diagnostic process. 19 Neoplastic diseases such as metastatic skin tumors, cutaneous lymphomas, or primary skin cancers may also mimic PG. 20 The diagnosis was similarly complicated in our case. The lesion was initially suspected to be a complication of mastoiditis or impaired wound healing following surgery, particularly given the recent mastoidectomy. Furthermore, the lesion was located in the retroauricular region, which is an atypical site for PG development. The lower legs are most commonly affected, although PG can occur on any part of the body, such as the thighs, abdomen, and breast. 21 Moreover, recurrent infections further complicated the diagnostic process, with the differential diagnosis also including chronic infection. In addition, the patient was treated with biologics, initially the TNF-α inhibitor adalimumab and subsequently with ustekinumab, an IL-12/IL-23 inhibitor. Current evidence supports the efficacy of adalimumab and ustekinumab in the treatment of PG, even in steroid-refractory cases, with a significant proportion of patients achieving remission.22,23 Importantly, histopathology was helpful in our case, as it showed findings that were consistent with PG. Histopathological features of PG include a dense neutrophilic infiltrate (“sea of neutrophils”), which is often associated with leukocytoclasia and visible nuclear debris but not with vasculitis or granuloma formation. However, CD-associated lesions may occasionally contain granulomatous foci.24,25
Managing PG in patients with CD requires a multidisciplinary approach involving both gastroenterologists and dermatologists. Treatment aims to reduce inflammation, modulate immune activity, and promote wound healing. For mild cases, topical corticosteroids and calcineurin inhibitors such as tacrolimus are commonly used and are generally well tolerated. 26 Supportive wound care, including non-traumatic cleansing and maintenance of a moist environment, is crucial for minimizing the risk of infection and supporting the healing process. 27 For more extensive and severe cases, systemic corticosteroids may be considered as first-line therapy with a response rate of 40%–50%. 28 Cyclosporine has shown similar efficacy with a different safety profile. 29 Besides prednisolone and cyclosporine, the best evidence is available for the anti-TNF-inhibitor infliximab.30,31 Adalimumab has shown promising results in an open-label trial. 32 Ustekinumab, 33 IL-17 inhibitors, 34 anakinra, 35 or rituximab 36 have shown remission in case reports or case series. The choice of the treatment depends on the severity of the disease, comorbidities, and previous response, especially in patients with underlying diseases. In the present case, systemic corticosteroids were not administered due to diagnostic uncertainty between PG and delayed postoperative wound healing following mastoidectomy in the setting of recurrent local infections. Cyclosporine was considered suboptimal given the patient’s concomitant CD, for which it has no approved indication and limited therapeutic benefit. 37 In contrast, upadacitinib may offer a treatment option that could target both PG and CD simultaneously.
In recent years, JAK inhibitors appear to represent an emerging therapeutic option in the management of refractory PG. 38 Upadacitinib, a selective JAK1 inhibitor, has been approved for the treatment of numerous immune-mediated diseases, including IBD and rheumatoid arthritis.39,40 Although data on the efficacy of upadacitinib in treating PG remain limited, recent case reports and case series suggest that upadacitinib may be a promising treatment option, even in cases refractory to conventional therapies such as steroids or infliximab.41,42 Recent research indicates that the JAK-STAT pathway is implicated in the inflammation associated with PG. A study with 31 patients with PG demonstrated that JAK1, JAK2, JAK3, tyrosine kinase 2 (TYK2), STAT1, STAT3, STAT4, STAT5, and STAT6 are overexpressed in PG skin biopsies compared with both psoriasis and healthy subjects. 43 Upadacitinib may reduce pro-inflammatory cytokines like IL-6, IL-17, and IL-23, which are often overproduced in PG, by targeting the JAK/STAT signaling pathway. 34
The initiation of upadacitinib in the presented case was complicated by the patient’s history of cerebral venous sinus thrombosis, as JAK inhibitors have been subject to “black-boxed warnings” due to an increased risk of thromboembolic events, reported in patients with pan-JAK inhibitor tofacitinib. The post-marketing ORAL Surveillance study compared tofacitinib with TNF inhibitors in patients with rheumatoid arthritis, who were aged 50 years and over and had at least one additional cardiovascular risk factor. Over a median follow-up period of 4 years, the incidence of major adverse cardiovascular events was higher in the combined tofacitinib groups (3.4%) than in the TNF inhibitor group (2.5%). 44 These findings have raised important safety considerations for all JAK inhibitors in populations with elevated cardiovascular risk. A recent meta-analysis including 42 studies and over 800,000 adult patients with immune-mediated diseases reported a modest but statistically significant increase in venous thromboembolism risk among JAK inhibitor users compared with TNF antagonists [pooled Hazard ratio (HR) 1.26; 95% CI 1.03–1.54], while no significant differences were observed in serious infections, malignancies, or major adverse cardiovascular events. However, the magnitude of risk differed significantly depending on the type of immune-mediated inflammatory disease, with rheumatoid arthritis being associated with a higher risk than IBD. In patients with rheumatoid arthritis, JAK inhibitors were associated with a 31.0% higher risk of VTE compared with TNF antagonists (HR, 1.31 (95% CI, 1.07–1.60)), with moderate heterogeneity; in contrast, in patients with IBD, there was no significant difference in the risk of venous thromboembolism (HR, 0.70 (95% CI, 0.05–10.49)). 45 A propensity-matched cohort of 10,174 IBD patients (1050 receiving JAK inhibitors and 9124 receiving TNF inhibitors) with a median age of 61 years found no significant difference in major adverse cardiovascular events between those receiving JAK inhibitors and TNF inhibitors over 12 months (adjusted odds ratio 0.92; 95% CI: 0.41–2.03). 46 Similarly, a Bayesian network meta-analysis of 26 randomized IBD trials involving 10,537 patients reported no statistically significant increase in major adverse cardiovascular events and venous thromboembolism events, although higher doses, such as tofacitinib 5 mg twice daily and upadacitinib 30 mg once daily, suggested a possible signal that requires further investigation. 47 Furthermore, despite shared targets, selective JAK1 inhibitors such as upadacitinib cannot be equated with pan-JAK inhibitors. In a recent meta-analysis of data from randomized clinical trials involving 9547 patients, upadacitinib showed no increased risk for venous thromboembolic events or major cardiovascular events. 48 Taken together, the available evidence indicates that while JAK inhibitors as a class may be associated with an increased risk of thromboembolic events—particularly in older patients and those with multiple cardiovascular risk factors—this risk is heterogeneous, appears to be driven predominantly by tofacitinib, and is not consistently observed with selective JAK1 inhibitors such as upadacitinib. Further high-quality evidence and long-term pharmacovigilance data are needed to determine whether upadacitinib should be removed from the FDA’s class-wide black box warning. In this present case, the thrombosis was attributed to chronic otitis media, which presented as a transient risk factor. Following complete resolution and after careful interdisciplinary risk–benefit assessment, initiation of upadacitinib was considered justified and was not associated with recurrent thromboembolic events during long-term follow-up.
Conclusion
The presented case highlights the effectiveness of upadacitinib in treating PG, as well as its safe use in a patient with a recent history of cerebral venous sinus thrombosis. Despite concerns about the risk of thrombosis, the treatment went smoothly with clear clinical improvement. Further studies and real-world data are needed to better define the role of upadacitinib in the management of PG and to clarify the risk of thromboembolic events in this cohort.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261424321 – Supplemental material for Successful treatment of severe retroauricular pyoderma gangrenosum with upadacitinib in a patient with Crohn’s disease and a history of cerebral venous sinus thrombosis
Supplemental material, sj-docx-1-tag-10.1177_17562848261424321 for Successful treatment of severe retroauricular pyoderma gangrenosum with upadacitinib in a patient with Crohn’s disease and a history of cerebral venous sinus thrombosis by Fotios Fousekis, Moritz Ronicke, Lukas Sollfrank, Jürgen Rech, Carola Berking, Markus F. Neurath, Michael Sticherling and Raja Atreya in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
