Abstract
Extra-intestinal manifestations (EIMs) commonly occur in patients with inflammatory bowel diseases (IBD) and contribute significantly to morbidity and reduced quality of life. Their management remains challenging. Recently, the development of Janus Kinase (JAK) inhibitors has expanded the therapeutic options of luminal IBD, and three JAK inhibitors, tofacitinib, upadacitinib, and filgotinib, have been approved for IBD treatment, while a growing body of evidence suggests that JAK inhibitors may be a promising therapeutic option for the management of EIMs, particularly those affecting the joints and skin. In this comprehensive review, we aim to provide the available evidence concerning the impact of JAK inhibitors on EIMs treatment and analyze their underlying mechanisms of action.
Plain language summary
Inflammatory bowel disease (IBD) is a chronic condition that mainly affects the digestive system but can also cause problems in other parts of the body. These problems, called extra-intestinal manifestations (EIMs), may involve the joints, skin, eyes, or liver. They are common, difficult to treat, and often reduce quality of life as much as the bowel symptoms themselves. Recently, a new group of medicines known as Janus kinase (JAK) inhibitors has been introduced for the treatment of IBD. These drugs, such as tofacitinib, upadacitinib, and filgotinib, work inside immune cells by blocking signals that trigger inflammation. Unlike older treatments that target only one pathway, JAK inhibitors act on several inflammatory pathways at once. Evidence suggests that JAK inhibitors may not only control bowel inflammation but also improve EIMs, especially those affecting the joints and skin. There are also early signs of benefit for eye and liver problems, though more studies are needed to confirm this. As with all medicines, JAK inhibitors can have side effects, so their use requires regular monitoring by doctors to make sure the benefits outweigh the risks. Overall, JAK inhibitors represent a promising new treatment option for people with IBD. By reducing both gut symptoms and complications outside the gut, they may help patients achieve better overall health and quality of life.
Keywords
Introduction
Inflammatory bowel diseases (IBD) are a chronic immune-mediated gastrointestinal disorder comprising Crohn’s disease (CD) and ulcerative colitis (UC). Being a multisystemic disorder, IBD may affect various organs, leading to the development of extra-intestinal manifestations (EIMs) in more than one-third of IBD patients. 1 In both CD and UC, almost any organ may be affected. The most widely recognized EIMs involve peripheral and axial arthropathies, cutaneous manifestations, such as erythema nodosum and pyoderma gangrenosum, primary sclerosing cholangitis (PSC), and ocular manifestations such as episcleritis and uveitis. Other less typical but clinically relevant manifestations, including hidradenitis suppurativa (HS), vasculitis, alopecia areata (AA), or coexisting autoimmune conditions such as rheumatoid arthritis, have also been reported in association with IBD. 2 In addition, fatigue is frequently reported by IBD patients and significantly impacts daily functioning; however, it is not considered a classical EIM. 3 EIMs may cause significant morbidity and can affect the quality of life for patients with IBD. The relationship between IBD activity and EIMs varies, depending on the type of EIM. 4 Furthermore, the likelihood of developing EIMs increases with the disease duration and in patients who already have one EIM. 5 The pathogenesis of EIMs remains elusive, but it seems that they occur either as a result of an intestine-specific immune response in an extra-intestinal location or due to independent inflammatory occurrence triggered and/or sustained by genetic or environmental risk factors in the host or by IBD. 6
In recent years, the development of Janus Kinase (JAK) inhibitors has introduced a new class of effective therapies for treating IBD, offering more therapeutic options and frequently achieving induction and maintenance of remission in active luminal UC. The European Medicines Agency (EMA) has approved the JAK inhibitors tofacitinib, upadacitinib, and filgotinib for the treatment of moderate to severe UC, expanding the therapeutic armamentarium, which mainly consists of biological agents. 7 In addition, upadacitinib has been approved for the treatment of patients with CD, demonstrating long-term efficacy observed and a favorable safety profile. 8
JAK inhibitors are small molecules that inhibit the signal transduction activity of surface receptors for multiple cytokines. 9 The JAK-STAT (Signal Transducer and Activator of Transcription) pathway has been linked to the pathogenesis of certain inflammatory joints and cutaneous conditions. This pathway has also been found to be upregulated in cutaneous EIMs, suggesting its potential involvement in their development and progression.10–12 Understanding the role of the JAK-STAT pathway in intestinal and extra-intestinal inflammation could lead to targeted therapies and improved management of EIMs in patients with IBD. This article aims to provide a comprehensive overview of JAK inhibitors, including information on existing approved JAK inhibitors and those currently in clinical trials for managing EIMs in IBD.
Literature research
We performed an in-depth review of the literature in PubMed to identify articles about the role of JAK inhibitors in the management of EIMs of IBD, using the following search string (“JAK Inhibitors” OR “tofacitinib” OR “upadacitinib” OR “filgotinib”) AND (“inflammatory bowel disease” OR “extra-intestinal manifestations”) up to May 2025. Furthermore, the references of relevant papers were also reviewed. We extensively examined the abstracts of manuscripts and identified the most relevant articles for this narrative review.
JAK-STAT signaling pathway
The JAK-STAT pathway is a vital signaling cascade that transmits regulatory signals from cytokine receptors to the nucleus, which influences a variety of cellular processes. The JAK family, comprising non-receptor tyrosine protein kinases, including JAK1, JAK2, JAK3, and TYK2, 13 is activated when cytokines bind to their corresponding receptors and plays a significant role in signaling regulation (as shown in Figure 1). While other members are present in almost all tissues, JAK3 is specifically expressed in the bone marrow, lymphatic system, endothelial cells, and vascular smooth muscle cells. 11 In addition, there are seven members of the mammalian STAT family (STAT1, STAT2, STAT3, STAT4, STAT5A, STAT5B, and STAT6). Each member of the JAK family demonstrates specificity for distinct cytokines, resulting in the activation of different signal transduction pathways and the subsequent activation of specific STAT proteins. 14

Description of the JAK-STAT signaling pathway. (1) When a cytokine binds to its specific cell-surface receptor, the receptor dimerizes and activates the associated JAK. (2) When JAKs are activated, they phosphorylate certain residues in cytokine receptor chains, which, in turn, create docking sites for STATs. (3) Once docked, the activated JAKs phosphorylate the STATs, causing them to dissociate from the receptor chains. (4) The STATs then form dimers with each other and relocate to the cell nucleus. (5) Within the nucleus, these triggers initiate gene transcription, leading to the creation of proteins that assist in immune responses and inflammation.
The JAK-STAT pathway operates as follows: when a cytokine binds to its specific corresponding cell-surface receptor, the receptor dimerizes and activates the associated JAKs. These activated JAKs then phosphorylate specific residues in the cytokine receptor chains, creating docking sites for STATs. Once docked, the activated JAKs phosphorylate the STATs, causing them to dissociate from the receptor chains. The STATs then form dimers with each other and relocate to the cell nucleus. Inside the nucleus, they trigger gene transcription, resulting in the production of proteins that aid in immune responses and inflammation. This process creates a pro-inflammatory feedback loop, potentially worsening the inflammatory response.15,16
JAK inhibitors
Tofacitinib
Tofacitinib, an oral JAK inhibitor, has received approval for treating moderate to severe UC and acts by selectively inhibiting JAK1, JAK3, and to a lesser extent, JAK2, leading to the suppression of various pro-inflammatory cytokines’ signaling, including interleukin (IL)-2, -4, -7, -9, -10, 15, and -21, as well as interferon (IFN)-α or γ.17,18 Tofacitinib has also been approved for the treatment of rheumatoid arthritis, 19 ankylosing spondylitis, 20 psoriatic arthritis, 21 and polyarticular course juvenile idiopathic arthritis, 22 further supporting its use in managing EIMs.
Evidence from clinical trials
Sandborn et al. 23 investigated the efficacy of tofacitinib for induction (OCTAVE Induction 1 and 2) and maintenance (OCTAVE Sustain) in the OCTAVE trials. Later, Rubin et al. performed a post hoc analysis to explore the impact of tofacitinib on different EIMs. They found that 27% of the patients in the induction trials and 9.0% of the maintenance trials suffered from EIMs. Rheumatological EIMs (peripheral arthritis, sacroiliitis, ankylosing spondylarthritis) were the most common: 11.2% (induction) and 3.6% (maintenance) of the patients had active peripheral arthritis, 1.0% (induction) and 0.7% (maintenance) had sacroiliitis, and 0.4 (induction) and none (maintenance) had ankylosing spondylitis. While treatment with tofacitinib had no effect on peripheral arthritis in comparison to placebo in the induction trials, 16.7% (5 mg BID (twice daily)) and 33.3% (10 mg BID) of tofacitinib-treated patients, but none of the placebo-treated patients experienced improvement after 52 weeks, suggesting a dose and time-dependent effect of tofacitinib treatment on peripheral arthritis. In the OCTAVE Induction studies 1 and 2, concerning oral ulcers/stomatitis, tofacitinib 10 mg twice daily improved symptoms in 3 out of 8 patients with ulcers/stomatitis, while 2 out of 16 given a placebo experienced worsening. New occurrences were recorded in five patients treated with tofacitinib (10 mg BID) and one patient receiving a placebo, corresponding to occurrence rates of 0.6% and 0.5%, respectively. 23 However, the low number of patients prevents drawing definite conclusions on the efficacy of treatment with tofacitinib regarding EIMs. In addition, the protocol design does not permit the differentiation of patients with active EIM from those with a non-active EIM. Furthermore, the pre-defined EIM list did not have a specific category for arthralgia, which is different from peripheral arthritis, 9 and IBD patients often experience joint pain (arthralgia) without inflammation, possibly due to adverse reactions to medical treatment or at the end of corticosteroid use. 24 The lack of a standardized approach for evaluating EIMs in UC clinical trials hinders data interpretation and therapy comparisons. 25 Finally, no imaging modalities were applied to confirm inflammation-related symptoms.
Real-world evidence
Another prospective cohort study was conducted in 29 UC patients with EIMs, out of which 17 had peripheral arthritis and axial damage. These patients received tofacitinib 20 mg/day for 8 weeks followed by a maintenance dose of 10 mg/day for 4 weeks. A reduction in EIM symptoms at 8 and 12 weeks was demonstrated in 55% (16/29) and 78% (14/18), respectively. 26 In a retrospective cohort study of 112 UC patients, with 35 patients having peripheral arthralgia, by week 24, 26 (74.2%) out of 35 patients reported improvement in arthralgia, and 14 (40%) patients reported resolution of joint symptoms. 27 A recent prospective observational study involving 103 patients with UC, 30 of whom had arthralgia, showed a 64% reduction in arthralgia symptoms following 16 weeks of tofacitinib treatment. Prevalence of arthralgia decreased from 29% at baseline to 11% at week 16. Tofacitinib was initiated at an induction dose of 10 mg twice daily for 8 weeks. However, at week 16, 62% of patients remained on the induction dose (10 mg BID) and only 38% had tapered to the maintenance dose of 5 mg twice daily. No specific data are available linking dosage adjustments directly to improvements in arthralgia. 28
In addition, several case reports have also described clinical improvement or the resolution of rheumatological EIMs, including spondyloarthropathy and peripheral arthropathy, in patients who received tofacitinib.29,30
Regarding dermatological manifestations, several case reports indicate that tofacitinib may be effective in treating pyoderma gangrenosum related to UC.31–33 Nevertheless, the lack of extensive studies limits the ability to draw definitive conclusions about its efficacy. Moreover, one patient with both UC and infliximab-refractory pyoderma gangrenosum was treated with a combination of infliximab and 10 mg tofacitinib twice daily, resulting in complete remission of both intestinal inflammation and cutaneous lesions. 34 In another study, three patients with CD who were treated with tofacitinib for 12 weeks experienced significant improvement in pyoderma gangrenosum. Two received 5 mg twice daily, while the third also started on 5 mg BID, but, due to only partial response, the dose was increased to 10 mg twice daily, leading to further improvement without the need for steroids. 35 Furthermore, there is also a reported case of erythema nodosum associated with UC that responded positively to tofacitinib treatment. 36
Regarding the effectiveness of tofacitinib for atypical dermatological symptoms associated with IBD, in a study, nine patients who had both AA and IBD (8 with UC, 1 with CD) were treated with tofacitinib. The use of tofacitinib resulted in remission of both AA and IBD in 85.7% of patients with active IBD. However, one patient did not respond to tofacitinib for controlling either UC or AA. 37 In addition, tofacitinib is effective in achieving clinical remission in a patient with UC, alopecia universalis, and atopic dermatitis. 38 Positive results were also observed in a patient with CD and concurrent dermatitis and vasculitis. 39
A retrospective multicenter study of 43 patients with UC and PSC collected data on non-transplant patients with large-duct PSC receiving tofacitinib for at least 4 weeks, at the time of tofacitinib start, after approximately 3 and 12 months of follow-up, and at the last follow-up available. Tofacitinib treatment for a more extended period was associated with decreased serum alkaline phosphatase (ALP) levels, with a significant difference observed between those who continued treatment and those who discontinued. Improvement in IBD activity was also linked to a decrease in ALP levels. Tofacitinib was well-tolerated, and non-colitis-related adverse events led to drug discontinuation in only two patients. Mild to moderate adverse events occurred in 17% (7/41), but none required cessation. No serious events such as thrombosis, viral reactivation, or lymphoproliferative disease were reported. Notably, one case of bacterial cholangitis and one of severe Salmonella colitis were observed. 40 In another retrospective study of 58 patients with IBD (mostly UC) and PSC treatment with JAK inhibitors over a median of 32 weeks led to significant reductions in inflammation markers, with 30% and 13% of patients achieving normalization of ALP and gamma-glutamyl transferase (GGT) levels, respectively. Among these patients, 57% received tofacitinib, 26% upadacitinib, and 17% filgotinib. However, no sub-analysis was performed for each JAK inhibitor. 41 Furthermore, the effectiveness of tofacitinib treatment was investigated in a case series involving five patients with PSC. After 3–6 months, laboratory tests showed a decrease of 28% and 39% in total bilirubin and ALP levels, respectively, compared to their baseline levels. 42
Upadacitinib
Upadacitinib is an oral small molecule that selectively inhibits JAK1 by targeting its adenosine triphosphate binding pocket. In cellular assays, it has been proven that UPA has a significantly higher selectivity for JAK1 compared to JAK2 by a factor of 42, JAK3 by a factor of 133, and TYK2 by a factor of 194. 43 Upadacitinib has been approved by both the FDA and EMA for treating moderate to severe UC in adult patients. 44 In addition, upadacitinib received recent approval for treating adult patients with moderately to severely active CD who have not responded well or were intolerant to either conventional therapy or a biologic agent or have lost their response.8,45 Upadacitinib has also been approved for rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, non-radiographic axial spondyloarthritis, polyarticular juvenile idiopathic arthritis, and giant cell arteritis.46,47
Evidence from clinical trials
Concerning the efficacy of upadacitinib in treating EIMs, a post hoc analysis of data from two induction studies (U-ACHIEVE Induction and U-ACCOMPLISH) and a maintenance study (U-ACHIEVE Maintenance) 48 showed that upadacitinib may effectively improve EIMs in patients with UC. The most common EIMs at the beginning of the studies were anemia (14%), peripheral (10.7%), and axial (2.1%) arthropathy. Other EIMs affected less than 2% of the patients. The proportion of patients who experienced resolution of any EIM (40% vs 33.3%), arthropathy (54.7% vs 42.1%), or anemia (38.2% vs 32.6%) at week 8 was significantly higher in the upadacitinib 45 mg QD (once daily) group compared to placebo in the induction studies. Similar effects were also observed in the maintenance study. Resolution of any EIM at 52 weeks was experienced by 66% of those in the 30-mg upadacitinib group, compared with 42% in the 15-mg upadacitinib group and 24% in the placebo group, while only the higher dose demonstrated statistical significance (p < 0.001). 49
Real-world evidence
Upadacitinib has also demonstrated promising therapeutic potential in dermatological EIMs. There have been reports of successful use of upadacitinib in treating AA patients with coexisting CD.50,51 Moreover, upadacitinib appears to have promising results in treating pyoderma gangrenosum in patients with IBD. Case reports describe significant improvement in pyoderma gangrenosum lesions in patients with UC or CD after upadacitinib treatment, even in cases refractory to conventional therapies such as steroids or infliximab.52–54 In a recent case, a child with an unusual manifestation of CD in the penis and scrotum showed resistance to adalimumab and ustekinumab, but ultimately improved after treatment with the JAK inhibitor upadacitinib. 55 Furthermore, a recent case report documented the potential of upadacitinib in treating both UC and HS in pediatric patients. Despite the patient’s poor response to infliximab and ustekinumab, switching to upadacitinib monotherapy resulted in significant clinical improvement of both conditions within 3 months, allowing the discontinuation of other systemic therapies. 56
Filgotinib
Filgotinib has been approved for the treatment of UC in Europe, the UK, and Japan. The drug’s half-life is 6 h, and its active metabolite stays active for up to 27 h, contributing to its pharmacological effects. Filgotinib selectively inhibits JAK1-mediated signaling, with comparatively less inhibition of JAK2-dependent and JAK3-dependent pathways, making it an attractive therapeutic option for managing inflammatory conditions. 37 Although there is a paucity of data regarding its role in the management of EIMs in IBD, filgotinib is licensed for rheumatoid arthritis, 57 suggesting potential benefit in IBD-associated arthritis and highlighting the need for additional studies to determine its efficacy.
Other JAK inhibitors
Other JAK inhibitors, such as peficitinib, izencitinib, and deucravacitinib, have also been evaluated in clinical trials for IBD. 58 However, further trial programs were discontinued after failing to demonstrate sufficient efficacy,59–61 and at present, no active trials are ongoing in UC or CD.
Discussion
EIMs remain a major issue for IBD patients, causing a significant impact on their quality of life. They may occur before or after the onset (or diagnosis) of IBD. Up to 26% of patients experience their first EIM before being diagnosed with IBD, with a median time of 5 months before diagnosis. By contrast, 74% of patients experience their first EIM manifestation with a median time of 92 months after IBD diagnosis. 62 Development of JAK inhibitors has introduced potent therapeutic options for IBD, changing the approach to the treatment of severe cases and their associated complications. 47 In recent times, mounting evidence highlights the effectiveness of JAK inhibitors in managing EIMs 33 (Tables 1 and 2). The mechanism underlying how JAK inhibitors improve or resolve EIMs remains incompletely comprehended. JAK inhibitors are known to suppress various cytokines that play a critical role in the pathogenesis of EIMs and immune-mediated disorders.
Extra-intestinal manifestations with potential positive effects of JAK inhibitors.

JAK, Janus kinase.
Key studies about the effectiveness of JAK inhibitors in EIMs.

AA, alopecia areata; AIH, autoimmune hepatitis; ALP, alkaline phosphatase; BID, twice daily; CD, Crohn’s disease; EIMs, extra-intestinal manifestations; IBD, inflammatory bowel disease; IQR, interquartile range; JAKi, Janus kinase inhibitor; PBO, Placebo; PSC, primary sclerosing cholangitis; QD, once daily; RCT, randomized clinical trial; UC, ulcerative colitis; UPA, upadacitinib.
A recent systematic review and meta-analysis demonstrated that the proportion of patients with improvement in joint manifestations was comparable between JAK inhibitor subgroups, 47% for tofacitinib (95% CI, 4%–95%; I2 = 94%, 95% CI, 87%–98%) and 48% for upadacitinib (95% CI, 32%–65%; I2 = 43%, 95% CI, 0%–83%). 63 These findings may be explained by their overlapping mechanisms of action. More specifically, tofacitinib blocks common γ-chain cytokines, including IL-2, IL-4, IL-15, and IL-21, efficiently. By inhibiting both JAK1 and JAK2, tofacitinib also constrains signaling via IFN-γ, IL-6, and, to a lesser extent, IL-12 and IL-23. Because of these broader actions, tofacitinib impairs the differentiation of CD4+ T helper cells, including Th1 and Th2, and restricts the formation of pathogenic Th17 cells. 64 On the other hand, upadacitinib shows increased selectivity for JAK1 compared with JAK2, JAK3, and TYK2. JAK1 is involved in the signaling of γ-chain receptor cytokines, such as IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21, as well as pro-inflammatory cytokines like IL-6 and IFN. 65
IL-15 has been identified as a critical cytokine in promoting wound repair and regeneration through stimulation of keratinocyte proliferation, and it is upregulated in patients with pyoderma gangrenosum. 66 The efficacy of JAK inhibitors for the treatment of AA might be attributed to interference with IL-2/21, which are overexpressed in those patients as it suggested by a genome-wide association study. 67 Moreover, JAK1-mediated signaling appears to play a crucial role in synovial inflammation. Synovial fluid T cells from patients with psoriatic arthritis showed an enhanced phosphorylation of JAK1, and STAT3/STAT1 was detected in synovial fluid T cells from patients with psoriatic arthritis. This could contribute to an IL-6-dependent expansion of Th17 T effector cells in inflamed joints. 68 Furthermore, in spondyloarthritis, immune cells like dendritic cells, T cells, type 3 innate lymphoid cells, and neutrophils invade the tissue, leading to the release of many inflammatory mediators, such as IFN-γ, IL-6, IL-12, IL-17, IL-23, and TNF, and subsequent activation of signaling pathways, including JAK–STAT pathways. These cytokines perpetuate inflammation and interact with different cells, like fibroblasts and monocytes/macrophages, resulting in disease symptoms and complications. Targeting these effector cytokines with JAK inhibitors can help to resolve arthritis, cartilage damage, and spine and joint damage in spondyloarthritis.69–71
Regarding the potential effectiveness of JAK inhibitors in treating pyoderma gangrenosum, these agents inhibit the JAK-STAT signaling pathway and reduce the levels of pro-inflammatory cytokines, including IL-6, IL-17, and IL-23. In the context of pyoderma gangrenosum, these cytokines are often overproduced, contributing to chronic inflammation, immune dysregulation, and subsequent tissue damage. By inhibiting JAK enzymes, JAK inhibitors can help to restore normal cytokine production, thus reducing inflammation and supporting the healing of affected tissues.72,73
Ocular manifestations are among the most common EIMs. 74 Episcleritis generally parallels intestinal activity, whereas uveitis can occur independently and lead to sight-threatening complications such as cataracts, glaucoma, or permanent vision loss if left untreated. 75 Currently, ongoing studies investigate the use of JAK inhibitors for non-infectious ocular inflammation.76,77 However, there is a lack of data on the effectiveness of JAK inhibitors in treating ocular manifestations of IBD.
After reviewing the available evidence, out of all the JAK inhibitors that have been approved for IBD treatment, tofacitinib has the most extensive evidence supporting its efficacy in treating a broad spectrum of EIMs. Upadacitinib has also demonstrated promising results. From the available data, we conclude that JAK inhibitors, specifically tofacitinib, could be a potential treatment for dermatological manifestations of IBD, including pyoderma gangrenosum and AA. In addition, JAK inhibitors seem to positively impact rheumatological symptoms of IBD, including peripheral and axial arthropathy. When selecting a JAK inhibitor, it is crucial to personalize the choice by taking into account the potential presence of other autoimmune diseases. It is worth noting that JAK inhibitors have received approval for numerous immune-mediated disorders (Box 1) (Table 3). 78
Current non-inflammatory bowel disease approved indications for JAK inhibitors.
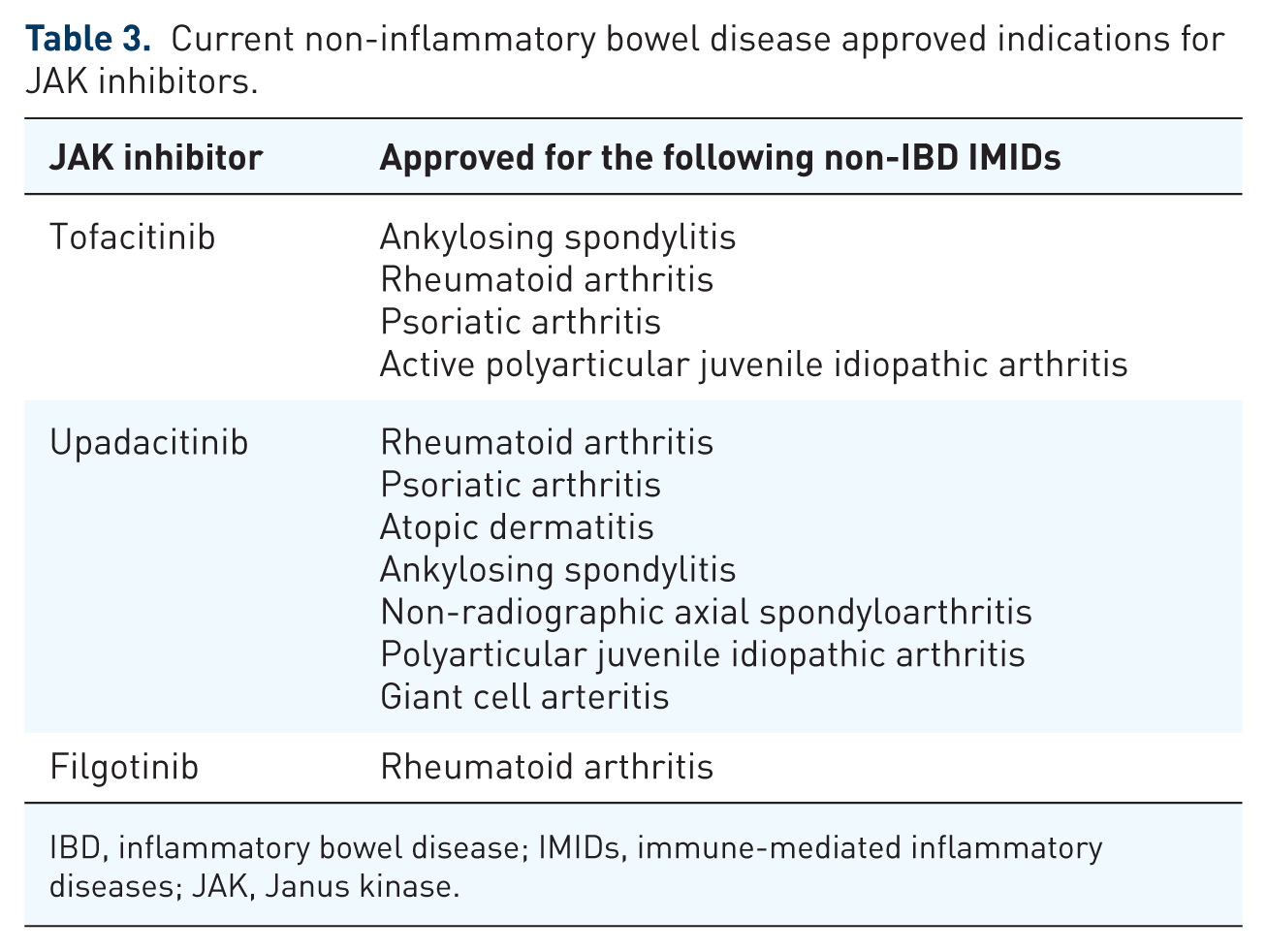
IBD, inflammatory bowel disease; IMIDs, immune-mediated inflammatory diseases; JAK, Janus kinase.
When to consider a JAK inhibitor for EIMs in inflammatory bowel disease.

EIMs, extra-intestinal manifestations; IBD, inflammatory bowel disease; JAK, Janus kinase.
In addition, patients with a history of malignancy or cardiovascular risk factors should receive JAK inhibitors with caution, due to the potential higher risk of cardiovascular events and malignancy relapse.79,80 However, recent data from studies in IBD patients suggest that JAK inhibitor use is not associated with an increased risk for cardiovascular events. 81 Regarding safety in the treatment of EIMs, available evidence suggests that JAK inhibitors are generally well-tolerated in this population. Adverse events are typically mild to moderate and align with those reported in luminal IBD treatment. Nevertheless, long-term data on safety in EIM-specific populations remain limited, and future prospective trials are needed to better define the safety profile of JAK inhibitors in the management of EIMs.
Despite the encouraging results, JAK inhibitors are still mostly used off-label for EIMs. The majority of the available evidence originates from post hoc analyses, observational studies, and case reports, making it challenging to draw robust conclusions. Therefore, prospective clinical trials that focus specifically on EIMs need to confirm the effectiveness and safety of JAK inhibitors and improve EIM treatment strategies in clinical practice.
