Abstract
Background:
Endoscopic submucosal dissection (ESD) is a representative treatment modality for early gastric neoplasms. However, the learning curve for beginners performing ESD using a multibending endoscope has not been introduced.
Objective:
This study aimed to evaluate the learning curves of operators undergoing intensive training using a multibending endoscope.
Design:
This was a retrospective single center study.
Methods:
We retrospectively analyzed data of over 1500 consecutive gastric ESDs performed by two operators using a multibending endoscope. A change-point analysis with 50 cases of moving average speeds was used to determine the new target resection speed. Cumulative sum (CUSUM) analysis was used to identify the cases required for proficiency in ESD. Risk-adjusted CUSUM (RA-CUSUM) analysis was performed for each operator after adjusting for confounding factors influencing the resection speed.
Results:
In total, 1491 cases were enrolled, with early gastric cancer accounting for 43.2% (n = 644). Overall, the en bloc resection, R0 resection, and curability rates were 97.7%, 96.0%, and 92.3%, respectively. The mean resection speed was 19.8 cm2/h. Because both operators surpassed the commonly used benchmark resection speed of 9 cm2/h in the first 50-case block, we established a new target benchmark of 17.9 cm2/h in the change-point analysis. CUSUM analysis indicated that performing 166 cases overall was required to achieve the benchmark, with the 2 operators needing 153 and 69 cases to meet this target speed after RA-CUSUM analysis.
Conclusion:
Using a multibending endoscope for gastric ESD can help beginners achieve safe and excellent outcomes. These findings will serve as a useful guide for beginners attempting to use a multibending endoscope.
Keywords
Introduction
Endoscopic submucosal dissection (ESD) is the first-choice modality for the treatment of early gastric neoplasms, as it enables stomach preservation and complete pathologic evaluation through single-piece resection with adequate margins. 1 Its application extends beyond gastric lesions, being a viable treatment option for early neoplasms of the gastrointestinal (GI) tract, including the esophagus, duodenum, and colorectum.2 –5 The stomach is the first organ selected for ESD training because of easy endoscope manipulation and its walls are thicker than those of other GI organs. Nevertheless, training in ESD remains challenging for endoscopists because of the risk of complications, primarily perforation, and concerns regarding difficult tumor location.
Asian studies have indicated that performing a minimum of 30–75 cases is necessary to achieve early proficiency in gastric ESD,6 –9 and a recent study reported that 165 cases are required to reach a plateau in the learning curve. 10 Another recent Western study reported that 150 cases are required to achieve a resection speed of >9 cm2/h. 11 All these studies performed learning curve analyses using a single-channel therapeutic endoscope. In contrast to a conventional single-bending endoscope, a multibending dual-channel endoscope has two bending sections and two instrument insertion channels. This scope allows easy suctioning, even after inserting the knife through one channel and inserting the injection needle through the other channel, thereby enabling a quick procedure without the need to exchange equipment. Additionally, positioning the cutting plane and knife parallelly in difficult areas is easier when using a multibending endoscope than when using a conventional endoscope, which helps lower complication risks and allows quicker, more stable dissections to be performed. 12 In a randomized controlled study, performing ESD using a multibending endoscope resulted in a shorter procedure time and less muscle damage compared with using a conventional endoscope. 13 Because of these advantages, a multibending endoscope was used selectively when the lesion was located in a difficult area.
To our knowledge, the learning curve in gastric ESD using a multibending endoscope has not been investigated before. Therefore, this study aimed to investigate the learning curve for gastric ESD using a large volume of ESD data from two operators who performed over 700 procedures during a similar period. We performed an additional risk-adjusted cumulative sum (RA-CUSUM) analysis to adjust for the factors affecting each operator since we used CUSUM analysis for the analysis and data from two operators.
Methods
Study design and patients
This retrospective, single-center study was conducted at Ajou University Hospital (Suwon, Republic of Korea) between March 2016 and June 2023. The data used consisted of ESD cases performed by two operators. One operator performed ESD consecutively from March 2016 to June 2023, whereas the other operator performed ESD consecutively from March 2019 to June 2023. Patients diagnosed with early gastric neoplasms (gastric adenoma and early gastric cancer (EGC)) who underwent ESD were enrolled. All patients provided written informed consent to undergo gastric ESD before the procedure. The exclusion criteria were multiple lesions, subepithelial lesions, use of a needle-type knife only, and a change of operator midway through the procedure. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Operators
Both operators completed a 1-year fellowship training, during which they performed approximately 1500 upper endoscopies, 700 colonoscopies, and 300 colonic polypectomy procedures, including endoscopic mucosal resection. Subsequently, the operators performed 300 endoscopic ultrasonographies and 50 gastric endoscopic mucosal resections. As preclinical training for ESD, they observed an expert perform ESD for a month (approximately 40 cases) and performed ESD on ex vivo models (5 times) and live pig models (6 times). Then, they participated in a portion of 30 ESD procedures under the intensive supervision of experts. Following this training process, the operators participated in the entire ESD process in human patients, and data accumulated from this point onward were used for our analysis. No fellow was involved in any of the ESD procedures, and a single operator performed the entire procedure.
Endoscopic submucosal dissection
All ESD procedures were performed in the endoscopy unit under moderate sedation, which was achieved by intravenous injection of a balanced sedative agent (midazolam and propofol). A two-channel and multibending endoscope (GIF-2TQ260M; Olympus, Tokyo, Japan) was used in conjunction with an electrosurgical unit (VIO 300D; Erbe, Tübingen, Germany). Prior to the procedure, a distal cap (4 mm; D-201; Olympus) was attached to the endoscope. After identifying the lesion, its boundaries were estimated using image-enhanced endoscopy with narrow-band imaging and chromoendoscopy with indigo carmine, and markings were made 5 mm outside these boundaries using a needle-type knife (Dual knife, KD-650; Olympus) in the coagulation mode (swift coagulation mode, effect 4, 40 watts). A mixture of normal saline, glycerol, epinephrine, and indigo carmine was injected outside the marked dots for submucosal lifting. Before making a circumferential incision, a pre-cutting (dry-cut mode, effect 5, 50 watt) was performed 5 mm outside the marking dot using a needle-type knife to create an entry point for the insulated-tip (IT) knife (IT-2 knife, KD-611; Olympus). The pre-cutting was performed on the anal side of the lesion in cases involving dissection in the forward view and on the oral side in cases involving dissection in the retroflexion view. A circumferential incision (Endocut I mode, effect 3, cut interval 3, cut duration 3) was made by inserting the IT knife at the entry point (pre-cutting site), and dissection was performed in the dry-cut mode (effect 5, 50 watts). Bleeding that occurred during this process was controlled using hemostatic forceps (Coagrasper, FD-410LR; Olympus; soft coagulation mode, effect 6, 80 watts). Following tumor removal, prophylactic hemostasis was applied to visible vessels at the resected site using hemostatic forceps and argon plasma coagulation (APC; Erbe, Tübingen, Germany, pulsed APC mode, Effect 2, flow 2.0 L/min). The resected specimen was retrieved using a Swirl Net (ET 2011; Olympus). Next, all samples were fixed onto a plate with pins with the gastric mucosa facing upward. We measured the long and short diameters using a caliper. Finally, the specimen was placed in a 10% buffered formalin solution for transport to the Department of Pathology. After completing the procedure, all patients underwent chest and abdominal X-ray to check for free air. The traction method was not used in any procedure. At our center, all patients underwent follow-up endoscopy 1 day after ESD. No traction device was used in any of the cases.
Study outcomes and definitions
The primary outcome was the number of cases performed by the two operators to reach the existing benchmarks 14 using the multibending endoscope. The secondary outcomes were the new target resection speed to be used as the benchmark and the number of ESD cases required to achieve this target. We used the following commonly used benchmarks for ESD proficiency: (1) >90% en bloc resection rate, (2) >80% R0 resection rate, and (3) resection speed >9 cm2/h. 14 The ESD procedure time was defined as the time between submucosal injection and specimen retrieval. 11 Surface area was calculated using the following equation under the assumption that the specimen is oval-shaped: long diameter × short diameter × π × 1/4. Therefore, the ESD resection speed was defined as the surface area of the resected specimen (cm2) divided by the procedure time (h). 14 Difficult areas included the fundus, cardia, great curvature of the body, angle, and lesser curvature of the antrum including the prepyloric area. En bloc resection was defined as obtaining a single-piece specimen without mucosal defects. R0 resection was defined as a negative resection margin in both the horizontal and vertical margins. Curative resection was defined as tumor-free status in both the horizontal and vertical margins in gastric adenoma, and the final pathology was satisfied with eCura A and B in accordance with the Japanese gastric cancer treatment guidelines regarding EGC. 1 Acute bleeding was defined as any melena, hematochezia, or hematemesis event during the hospital stay, whereas delayed bleeding was defined as such event within a month after discharge. Micro perforation was defined as follows: (1) no gross perforation observed during endoscopy; (2) no symptoms suggesting peritonitis, such as fever or abdominal pain, after the procedure; and (3) free air observed on X-ray after the ESD. Recurrence was classified into four types: residual disease, recurrence at the resected site within 1 year after ESD; local recurrence, recurrence at the resected site more than 1 year after ESD; synchronous lesion, new lesion detected at the other site from the previous ESD site within 1 year after ESD; and metachronous lesion, new lesion detected at the other site from the previous ESD site more than 1 year after ESD.
Statistical analysis
Data are presented as numbers (n) and percentages (%) for categorical data or as means and standard deviations for continuous variables. Differences between the operators and among the phases were compared using a t-test or analysis of variance for continuous variables and Chi-square test for categorical variables. The p-value was adjusted using Bonferroni correction if necessary. Trends in en bloc, R0, and curative resection rates were explored over sequential blocks of 50 cases. The box plots were drawn to show resection speed distributions by each block of 50 cases, tumor location (upper, middle, and lower thirds), presence of severe submucous fibrosis, subcutaneous fat, depression morphology, ulcer, lesion size (<3 and ⩾3 cm), and difficult areas. Specifically, the trend in the resection speed across the block of 30 cases was examined in difficult areas.
Moving average
A 50-case simple moving average was calculated for the sequential 50-case blocks. These 50-case moving averages showed an uptrend or downtrend and a sign of movement change in the resection speed. Therefore, we identified the point at which the statistical properties of a sequence of moving average speeds changed. Such change detection can occur where the average speed statistically changes and is considered to reach the proficient phase from the learning phase. We performed a change-point analysis with the moving average speed and identified its change point (case) and benchmark speed at this change point. Logistic regression analysis was performed to identify the factors associated with a resection speed greater than the targeted speed during gastric ESD.
CUSUM analysis
The learning curve of the resection speed was computed using the CUSUM analysis. The CUSUM was calculated as follows:
where
RA-CUSUM analysis
CUSUM analysis is based on observations. However, poor resection speed can occur in certain patients with higher risk factors, and this random chance is unfairly distributed across the series of cases. This can lead to a bias in measuring the learning curve of resection speed, regardless of the operator’s maturity and proficiency. Thus, the learning curve can be further verified using RA-CUSUM analysis. This extension of the CUSUM analysis is a valuable method for illustrating trends in sequential differences between the observed and expected speeds after adjusting for other risk factors associated with the resection speed.15,16 All data from the operators were combined, and univariable logistic regression analysis was performed using age, sex, tumor location, histology, lesion size, fibrosis, depression, ulcer, subcutaneous gat, difficult location, muscle damage, clipping, and propofol. The stepwise algorithm was used to select the final multivariable logistic model and was confirmed using the lowest Akaike information criterion. The RA-CUSUM was calculated as follows:
where
Results
Study population and procedure outcomes
In total, 1574 patients underwent ESD for gastric neoplasms performed by the two operators, and 1491 patients were finally enrolled (Figure 1). The two operators performed a substantial number of ESDs in the first 3 years, with averages of 195.6 and 164.0 cases, respectively (Figure 2(a)). Overall, en bloc, R0, and curative resection rates were 97.7%, 96.0%, and 92.3%, respectively. There were 644 patients with EGC (43.2%), 515 (80.0%) with eCura A, and 82 (5.5%) with submucosal involvement. Forty-three (2.9%) bleeding events (both acute and delayed) occurred, and micro perforation occurred in 5 cases. However, there was no gross perforation. There were 102 (6.8%) cases of recurrence after ESD. Lesion characteristics and procedural outcomes are shown in Table 1. The cases were divided into 3 phases of 250 cases each, with both operators showing decreased procedure time, increased resection speed, and decreased propofol dose with advancing phases (p for all <0.05). The occurrence of muscle damage, which can contribute to potential or actual perforation, decreased (operator 1: 41–9–5, p < 0.001; operator 2: 24–16–14, p = 0.358; Supplemental Table 1). An analysis of proficiency parameter changes over the consecutive 50-case blocks revealed that both operators reached the existing proficiency benchmarks in the first block. The en bloc, R0, and curative resection rates remained consistent (Figure 2(b) and (c)).

Flow chart of the enrolled patients and procedure outcomes.

(a) Annual case volume for the two operators. (b) Trends of en bloc, R0, and curative resection rates over sequential blocks of 50 procedures for operator 1 and (c) operator 2. (d) The overall trend of resection speed over sequential blocks of 50 cases. (e) Trends of resection speed over sequential blocks of 50 cases in operator 1 and (f) operator 2.
Baseline characteristics and procedure outcomes.

Negative in both lateral and deep margin in histopathology.
Including both adenoma and cancer.
Melena or hematochezia.
Event occurred in 30 days after discharge.
CR, curative resection, EGC, early gastric cancer, ESD, endoscopic submucosal dissection, SD, standard deviation, SM, submucosal.
Resection speed for gastric ESD
The overall mean ESD speed was 19.8 ± 10.0 cm2/h, increasing gradually as the sequential 50-case block increased (Figure 2(d)–(f)). The median resection speed in the various conditions (tumor location in the upper and middle thirds, presence of submucosal fibrosis, subcutaneous fat, surface ulcer, lesion size <3 cm, and difficult areas) was lower than the median resection speed (18.5, interquartile range (IQR) 12.30, 25.55; Supplemental Figure 1). The resection speed was lower than the benchmark speed (9 cm2/h) in the presence of fibrosis (7.8 cm2/h, IQR 5.6, 11.0). We speculated that large lesions would be linked to a longer ESD time; however, the resection speed was the fastest for lesions sized >3 cm (23.6 cm2/h, IQR 17.8, 31.6). This speed was consistent between the operators. The most stressful situation for novice ESD operators was when tumors were present in difficult areas. Nevertheless, both operators increased in speed, even in difficult areas, with an increasing number of procedures performed (Supplemental Figure 2).
Learning curve analysis
The 50-case moving average resection speed based on the number of cases for each operator and overall speed are shown in Figure 3(a). Because both operators surpassed the benchmark resection speed (9 cm2/h) in the first 50-case block (operator 1: 18.3 cm2/h, operator 2: 18.9 cm2/h), we established a new target speed to analyze the learning curves. Hence, we attempted to examine the changes in speed by visualizing the 50-case moving average speed for the two operators and estimate the point at which the statistical properties of a sequence of moving average speed changed (Supplemental Figure 3). Consequently, we set 17.9 cm2/h as the target speed for the learning curve analysis. The CUSUM analysis indicated that at least 166 ESD cases were required to achieve a target speed of >17.9 cm2/h and to maintain a resection speed higher than the target speed subsequently (Figure 3(b)). Table 2 presents a comparison of the two operators among the three phases. The mean ESD speeds of the two operators increased by 1.5 and 1.2 times, respectively (phase I vs II; 13.7 ± 6.7 vs 21.0 ± 10.9 cm2/h, p < 0.001 and 17.1 ± 9.8 vs 20.2 ± 9.7 cm2/h, p < 0.001, respectively). Operator 1 showed a significant reduction in muscle damage occurrences after performing 166 procedures (phase I vs II vs III; 17.5% vs 8.9% vs 1.5%, p < 0.001), which led to fewer use of prophylactic clipping (6.6% vs 3.6% vs 1.5%, p = 0.006). However, operator 2 showed increased R0 resection rates (91.6% vs 96.9% vs 95.5%, p = 0.049) and a lower dose of propofol (107.5 vs 85.5 vs 84.4 mg, p < 0.001) despite the increased number of tumor cases located in the middle- to upper-third region (33.1% vs 48.6% vs 44.4%, p = 0.032). Favorable factors for resection speed were lower-third tumor location, larger tumor size, and increasing learning phase, whereas the negative factors were severe submucosal fibrosis, ulceration, and difficult area. The most potent factors were lower-third tumors (odds ratio (OR), 7.23, 95% confidence interval (CI), 4.347–12.467; p < 0.001) and submucosal fibrosis (OR, 0.05, 95% CI, 0.008–0.168; p < 0.001). The learning period was associated with a rapid resection speed in the progressive phases (Table 3).

(a) Fifty-case moving average of resection speed (red line: overall, green line: operator 1, and blue line: operator 2). (b) CUSUM analysis plot of cases required to reach the new target resection speed of 17.9 cm2/h. (c) RA-CUSUM analysis plots of cases required to reach the new target resection speed of 17.9 cm2/h in operator 1 and (d) operator 2. The gray line was drawn by curve fitting. The degree of curve fitting was determined on the basis of adjusted R-squared values. The gray line in (b) represents the fitted curve generated from the regression model (curve fitting).
Comparison of different phases according to the result of CUSUM analysis.

Negative in both lateral and deep margin in histopathology.
Including adenoma and cancer.
Melena or hematochezia.
Event occurred in 30 days after discharge.
CR, curative resection, CUSUM, cumulative sum, EGC, early gastric cancer, ESD, endoscopic submucosal dissection, SD, standard deviation, SM, submucosal.
Analyses of factors associated with fast resection speed (⩾17.9 cm2/h).
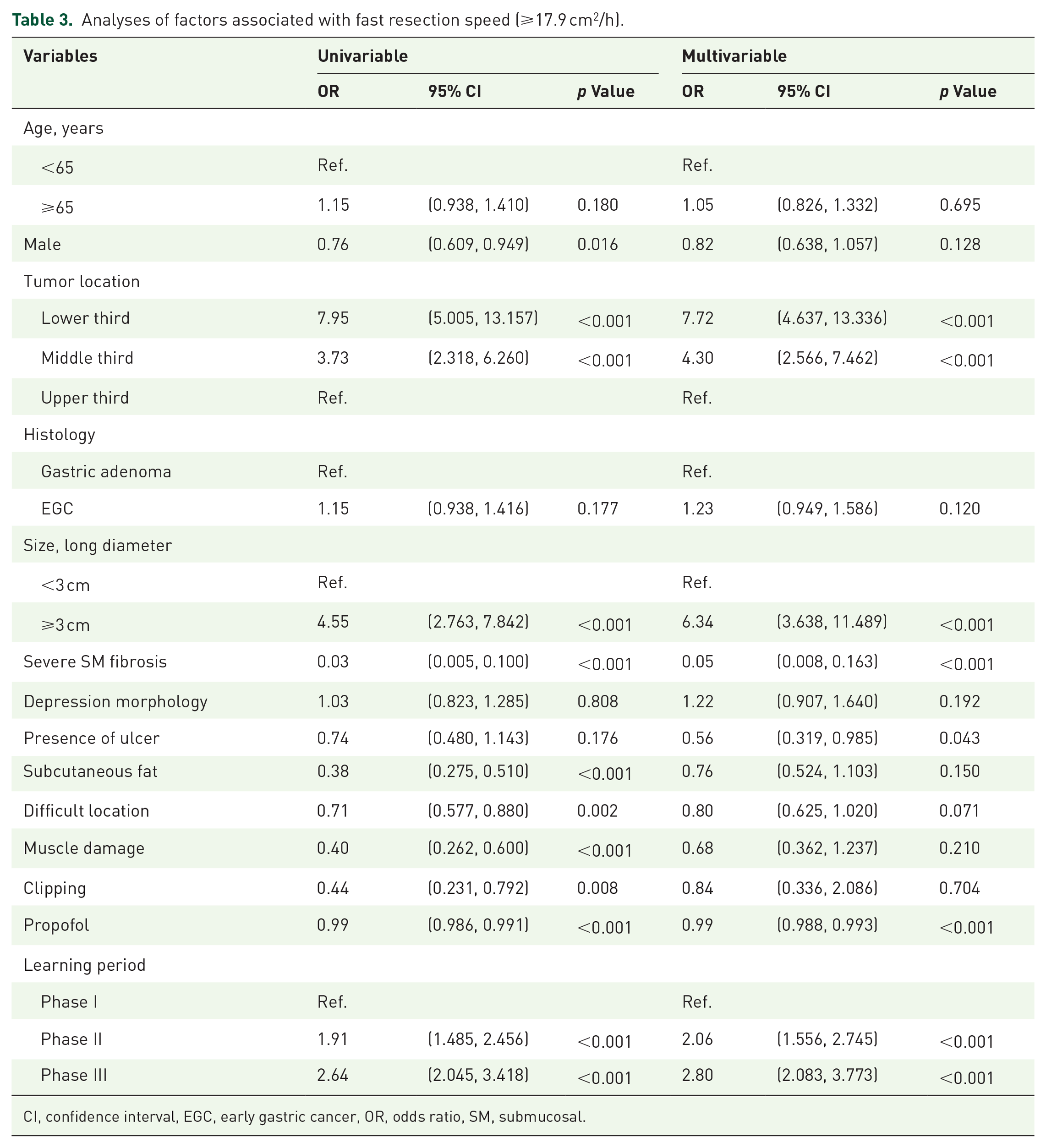
CI, confidence interval, EGC, early gastric cancer, OR, odds ratio, SM, submucosal.
Because the competency levels of the two operators and patients’ baseline characteristics differed, we identified the factors that affected speed through a risk-adjusted regression model of ESD speed and performed RA-CUSUM analysis (Supplemental Table 2). Consequently, 153 and 69 cases were required to achieve the target speed of 17.9 cm2/h in the RA-CUSUM analysis for operators 1 and 2, respectively (Figure 3(c) and (d)). Notably, the required cases for operator 2 to achieve the target speed was markedly fewer than those identified in the CUSUM analysis. A comparison of the different phases according to the results of the RA-CUSUM analysis for the two operators is shown in Supplemental Table 3. Both operators exhibited significantly better ESD proficiency and improved complication-related factors after achieving the target speed.
Discussion
Complications, as opposed to outcomes, are a major concern for beginners performing ESD. Once proficiency is achieved, good outcomes become a priority. Considering complications as unavoidable or criticizing endoscopic beginners who are worried about their procedures owing to the risk of complications is an outdated perspective. Furthermore, therapeutic endoscopists should perform procedures with the mindset that complications should never occur during any procedure. Using a multibending endoscope makes it easier to access lesions in difficult locations compared to that with a conventional endoscope, and the dissection plane and knife can be positioned parallelly. 12 We hypothesized that the existing proficiency benchmarks would be achieved more quickly when performing ESD training with a multibending endoscope than with a conventional endoscope. We performed learning curve analysis for gastric ESD training using a multibending endoscope by evaluating the progress and outcomes of two operators who underwent intensive ESD training and performed an average of over 160 cases each for the first 3 years. Notably, both operators reached the benchmarks after performing the first 50 procedures; specifically, the two operators achieved high procedural speed and en bloc resection and curability rates. We also analyzed their growth data and performed CUSUM analysis to propose new benchmarks for beginners in ESD. On the basis of a change-point analysis of the moving average speed, we identified 17.9 cm2/h as the change point in the average speed, and 166 procedures were required to reach this speed. Reaching this benchmark ensured that the operators were able to achieve high-quality outcomes while maintaining high resection speeds. Because we analyzed data from two operators, we additionally performed RA-CUSUM analysis after adjusting for factors affecting the speed. The results showed that the number of cases required to achieve the threshold proficiency level varied across individual operators. The novelty of our study lies in the evaluation of the learning curve for gastric ESD using a multibending endoscope, which to our best knowledge, has not been investigated before. Our results show that multibending endoscope enables trainees in gastric ESD with high resection speed, safety, and favorable outcomes, despite a similar number of learning cases with conventional endoscope.
In the present study, both operators reached a proficiency benchmark speed of 9 cm2/h in the first 50 cases. Thus, we established and set a new target speed of 17.9 cm2/h by analyzing 50 cases of moving average in CUSUM analysis. Therefore, it is inappropriate to apply the standards recommended for first-generation ESD operators in the current era. We established a new target speed on the basis of our data, which was approximately two times higher than the previous target of 9 cm2/h. The resulting resection speed in our study was higher than that reported in previous studies for the following reasons: (1) We used a two-channel multibending endoscope for all ESD cases. (2) The two operators underwent intensive training with a large number of ESD cases in the first 3 years. During the first year, operators 1 and 2 performed 218 and 109 procedures, respectively. In terms of the learning curve, operators who performed 100 procedures in a year would become more skillful than those who performed 100 procedures in over 10 years. (3) Our center features 3 highly skilled experts ESD operators (S.G.L., S.J.S., and K.M.L.) who have performed more than 1000 cases of ESD, and the operators of our study had the ability to seek advice from these experts and directly and indirectly learn from them. Repeated case reviews and discussions with these experts may have served as valuable aids in their learning. (4) Recently, many training programs are available for ESD beginners in Korea. The two operators in our study also underwent thorough ESD training on live pig models and observed the procedures performed by highly skilled experts.
Two factors were required to identify the learning curve: the target speed and the case number required to achieve the target speed. Our study represents the first attempt at a learning curve analysis using a multibending endoscope. To apply the 17.9 cm2/h target speed identified in our results to beginners in ESD, further research with a larger number of operator data is needed. If endoscopists do not reach the 17.9 cm2/h target speed even after performing over 150 ESD cases, it would be necessary to evaluate the speed appropriate to the situation through change-point analysis with the moving average speed. However, we expect that the target speed presented can be a guide for endoscopists who want to start ESD in a new era.
As evidenced by our data, the operators achieved a high resection speed within the first 50 procedures. They achieved a faster resection speed and improved benchmark parameters after approximately 166 cases, which was identified in the CUSUM analysis. Furthermore, we suspected that the rate at which an operator reaches the desired proficiency level would vary, even within the same institution and endoscopic environment, depending on the presence of other factors that affect speed. To test this hypothesis, we conducted RA-CUSUM analysis to identify the number of ESD cases required by each operator after adjusting for factors that affect resection speed. The results showed that the number of cases differed between the two operators, with operator 2 requiring a markedly fewer number of cases than operator 1. Hence, we speculate that operator 2 was subjected to fewer factors that affect resection speed than operator 1 and thus reached the benchmark target speed faster. Another possible reason may be that operator 2 had superior skills compared with operator 1. Of note, both operators achieved better outcomes after the inflection point identified in RA-CUSUM analysis.
We excluded ESD cases with needle-type knives alone and cases with multiple lesions from the analysis. This was aimed at preventing some specific cases from affecting the learning curve analysis results. In our study, operators 1 and 2 had 28 and 0 ESD cases with needle-type knives alone, respectively. This was 1.7% of the total number of cases. In multiple lesions, operators 1 and 2 had 59 (3.7%) and 26 (1.6%) cases, respectively. When performing ESD, we determined that the condition of the first lesion differed from that of the subsequent lesion. This is because the environment that occurs when the first lesion is removed (such as bleeding or complications) and the quality of the sedation status may affect the ESD procedure of the second lesion. Moreover, only 5 of the 85 multiple-lesion cases were within the 166 cases required for the operator to achieve the target speed. Therefore, the exclusion of these factors is estimated to have had minimal effect on the results.
This study had some limitations. First, we did not conduct a randomized trial to compare cases using a single-channel conventional scope; therefore, the superiority of the multibending endoscope could not be established in this study. Thus, we could not analyze the type of endoscope as a factor that may affect the resection speed. Second, the analysis was based on data from only two operators. This is because only two operators who had performed a sufficient number of ESD procedures for long-term learning curve analysis were included in the present study. Therefore, further study including a large amount of operator data is required to establish the target speed. Third, this was a single-center study involving data from two operators. Since the patient selection criteria were dependent on the physician’s discretion, selection bias may have influenced the results. However, the two operators made treatment decisions and patient selection based on the Japanese gastric cancer treatment guidelines to minimize these potential biases. Fourth, the outcomes were from two different operators; however, as shown in the RA-CUSUM analysis, the outcomes may vary depending on the conditions of the procedure and skill levels. Third, this was not a multicenter study. Finally, owing to the retrospective nature of the study, we could not randomize the participants on the basis of factors that affect resection speed or prospectively compare the two operators.
Conclusion
This study elucidates the process of growth from a beginner to an expert at a desirable proficiency level among two operators who performed more than 700 procedures each. The results of learning curve analyses, including ours, require caution when applied to any organization or individual because the organizational environment and individual skill levels differ. However, once an operator performs approximately 150 cases of ESD using a multibending endoscope, they should achieve a very high resection speed with excellent operative outcomes. Our study results will serve as a useful guide for beginners who are trying to use a multibending endoscope in ESD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251318861 – Supplemental material for Learning curve for endoscopic submucosal dissection in early gastric neoplasm using a multibending endoscope
Supplemental material, sj-docx-1-tag-10.1177_17562848251318861 for Learning curve for endoscopic submucosal dissection in early gastric neoplasm using a multibending endoscope by Gil Ho Lee, Eunyoung Lee, Sun Gyo Lim, Bumhee Park, Sung Jae Shin, Kee Myung Lee and Choong-Kyun Noh in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848251318861 – Supplemental material for Learning curve for endoscopic submucosal dissection in early gastric neoplasm using a multibending endoscope
Supplemental material, sj-pdf-2-tag-10.1177_17562848251318861 for Learning curve for endoscopic submucosal dissection in early gastric neoplasm using a multibending endoscope by Gil Ho Lee, Eunyoung Lee, Sun Gyo Lim, Bumhee Park, Sung Jae Shin, Kee Myung Lee and Choong-Kyun Noh in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


