Abstract
Background:
There is no consensus on whether proton pump inhibitor (PPI) or vonoprazan (VPZ) is superior in preventing delayed bleeding (DB) after endoscopic submucosal dissection (ESD) of the stomach.
Objectives:
This study aimed to compare the efficacy of combined intravenous and oral PPI versus oral VPZ alone therapy in preventing DB after gastric ESD in a consecutive and large case series.
Design:
Retrospective study.
Methods:
This study included consecutive patients who underwent gastric ESD at Chiba University Hospital from January 2017 to July 2023. Before 2019, patients received intravenous omeprazole 20 mg in the morning and evening on the day of ESD and the day after. Thereafter, esomeprazole 20 mg was administered orally once daily, which was continued for generally 28 days (defined as the PPI group). From 2020 onward, patients received oral VPZ 20 mg once daily starting on the day of ESD, also typically continued for 28 days (defined as the VPZ group). DB rates between the PPI and VPZ groups were compared using propensity score matching.
Results:
There were 720 cases (856 tumors) of gastric ESD during the study period, of which 352 (409 tumors) were in the PPI group and 368 (447 tumors) in the VPZ group. Propensity score matching for 9 covariates related to DB rates for gastric ESD ultimately produced 329 best matches. There was no significant difference in DB rates between the two groups (4.3% vs 3.6%, p = 0.84).
Conclusion:
Though further prospective studies are needed to draw definitive conclusions, it was suggested that the easily administered oral VPZ can be an important option for acid suppression after gastric ESD.
Introduction
Gastric cancer is the world’s fourth most common cancer among men and sixth most common among women, and is still considered a major cancer.1,2 Endoscopic submucosal dissection (ESD) for gastric adenoma or cancer has been reported to be highly effective and has become the mainstream endoscopic treatment.3,4
ESD is sometimes associated with complications such as delayed bleeding (DB) and perforation. 5 DB occurs in 4.1%–8.5% of patients undergoing gastric ESD.6–9 Proton pump inhibitor (PPI) has been widely used for the treatment of gastric ulcers caused by ESD to prevent DB and promote healing.10–12 In 2015, vonoprazan (VPZ) was launched in Japan. VPZ is an active potassium-competitive acid blocker that, like PPI, inhibits the gastric H+/K+-ATPase. Unlike PPI, however, VPZ inhibits the enzyme in a K+-competitive and reversible manner. In addition, the gastric acid secretion inhibitory effect of VPZ is hardly affected by the surrounding pH and does not require enteric coating. As a result, VPZ is more rapid, potent, and longer lasting than PPI.13,14 Furthermore, while PPI is metabolized by cytochrome P450, leading to individual differences in efficacy due to cytochrome P450 polymorphisms, VPZ is metabolized by multiple liver enzymes and is less affected by cytochrome P450 polymorphisms. 15 However, there is no consensus on the efficacy of PPI and VPZ in preventing DB after gastric ESD, with some literature claiming a significant difference16,17 and others claiming no significant difference.18–24 There are also several cases where intravenous PPI is used in combination,16,19,20,22,23 but intravenous administration has the drawback of being somewhat more complicated compared to oral administration.
This study aimed to compare the efficacy of combined intravenous and oral PPI versus oral VPZ alone therapy in preventing DB after gastric ESD in a consecutive and larger case series than previously reported.
Methods
Study design
In this study, we performed a retrospective review of gastric ESD cases at Chiba University Hospital. Consecutive patients who underwent gastric ESD at our institution from January 2017 to July 2023 were included, and their post-ESD course was reviewed from clinical records. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 25 We present the study flow diagram (Figure 1).

The study flow diagram.
Inclusion and exclusion criteria
Patients who underwent gastric ESD during the study period. Since this study involved consecutive cases, there were no exclusion criteria.
Helicobacter pylori status
To assess Helicobacter pylori status, we evaluated the history of H. pylori eradication therapy for all patients and performed H. pylori tests (antibody test, urea breath test, or stool test).
Details of ESD
The operators were divided into experts and non-experts. Experts were those who were certified by the Japan Gastroenterological Endoscopy Society (JGES) and with at least 10 years of experience in endoscopic procedures, while non-experts were those other than experts. Preoperative and postoperative antithrombotic drugs were administered according to JGES guidelines.26,27 Mainly, ESD was performed using a conventional singe-channel endoscope with water supply function (GIF-H290T or GIF-Q260J; Olympus, Tokyo, Japan). After local injection of saline to confirm the location of the submucosa, a mixture of 0.4% sodium hyaluronate (MucoUp; Boston Scientific, Marlborough, MA, USA) and normal saline in a 1:1 ratio was injected locally into the submucosa. DualKnife J (Olympus) or ITknife 2 (Olympus) was mainly used for cutting instruments. An electrosurgical current was applied with the aid of an electrosurgical generator (VIO3, VIO300D, or 200D; ERBE Elektromedizin GmbH, Tubingen, Germany). The decision to use the traction method 28 was left to the operator’s discretion. If there were visible vessels after ESD, preventive coagulation was performed using hemostatic forceps (RAICHO2; Kaneka Medix, Tokyo, Japan, or Coagrasper; Olympus). Basically, a second look was not performed.
Medicine
Prior to 2019, omeprazole 20 mg was given intravenously in the morning and evening on the day of ESD and the day after. Thereafter, esomeprazole 20 mg was administered orally once daily, which was continued for generally 28 days (this patient group is hereafter defined as the PPI group). For cases post-2020, VPZ 20 mg was administered orally once a day starting on the day of ESD and continued for generally 28 days (this group of patients is hereafter defined as the VPZ group; Figure 2).
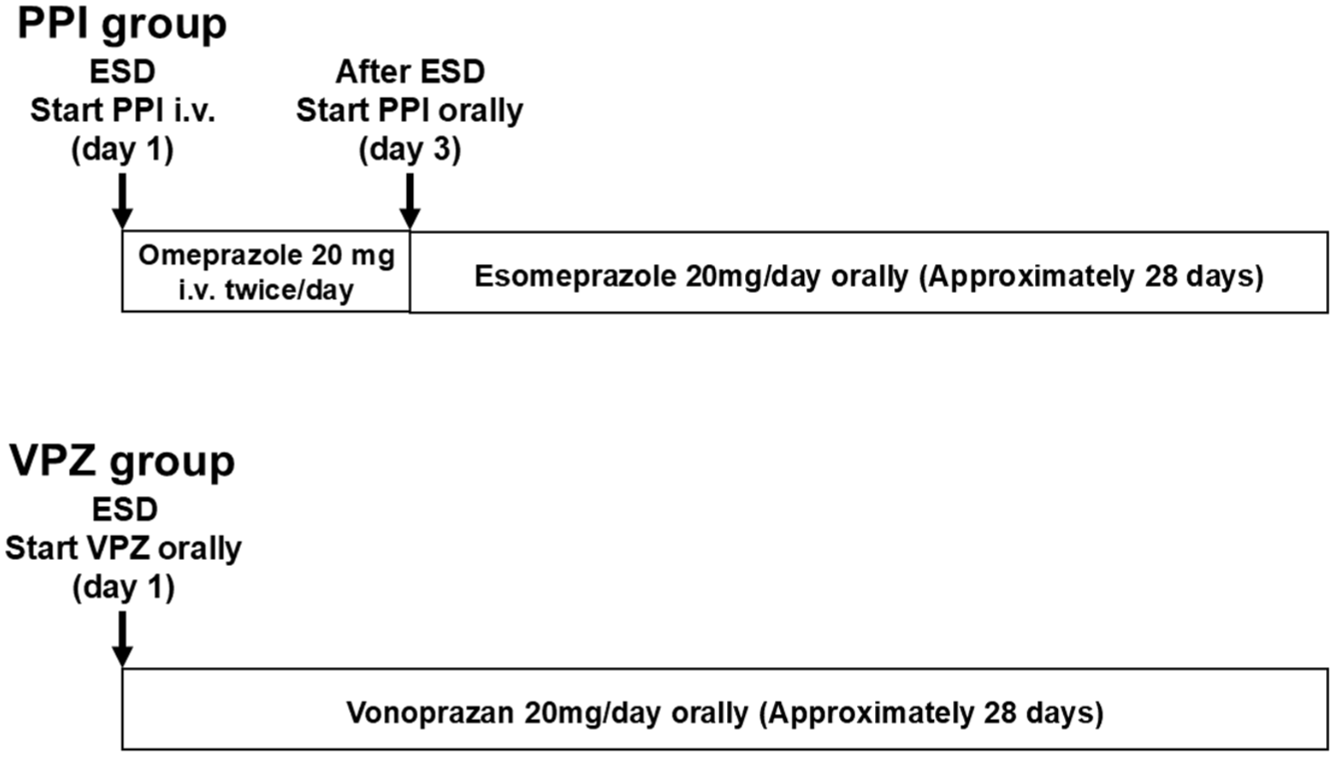
Administration method for medicine by group.
Adverse event
DB was defined as hemorrhage with clinical symptoms and confirmed by emergency endoscopy within 28 days after ESD, similar to a previous study on the BEST-J score. 7 Clinical symptoms were defined as hematemesis or melena, or a decrease in Hb greater than 2 g/dL from the most recent laboratory test. 7 For DB cases, we evaluated the Forrest classification 29 and the presence or absence of blood transfusion. When hematemesis or melena was observed, patients were instructed to contact the medical staff (if it occurred after discharge, mainly by phone). In addition, patients are scheduled to return for an explanation of pathological results within 28 days after ESD, during which the presence of DB was assessed. Where possible, follow-up endoscopy for recurrence assessment was performed between 3 months and 1 year after ESD. Perforations were divided into intraoperative and delayed perforations. Intraoperative perforation was defined as an apparent perforation that occurred during ESD. Delayed perforation was defined as a perforation that occurred after ESD, not during the procedure, and in which free air was observed on imaging studies. Typically, symptoms such as abdominal pain, fever, and peritoneal signs develop hours to days after the procedure.
Pathological evaluation of the resected specimen
All tissue specimens underwent histological analysis with standard hematoxylin–eosin staining and were independently evaluated by pathologists. The pathologist made histopathological diagnoses according to Japanese diagnostic standards. In this context, cancer refers to both intramucosal carcinoma and submucosal invasive cancer.
Endpoint
The primary endpoint was the comparison of DB rates between the two groups.
Statistical methods
EZR version 1.68 was used for statistical analysis. For patient and tumor characterization analysis, continuous variables were summarized by median and compared using the Mann–Whitney U test. Categorical variables were summarized by n (%) and compared using the Chi-square test (Fisher’s exact test if one or more expected values were less than 5). Propensity score generation and 1:1 matching of patients between the PPI and VPZ groups were performed to minimize selection bias in treatment assignment. Matching was based on variables in the BEST-J score, including whether patients were taking warfarin, direct-acting oral anticoagulant (DOAC), P2Y12 receptor antagonist, aspirin, or cilostazol, whether it was chronic kidney disease (CKD) with hemodialysis, a tumor size >30 mm, lower-third in tumor location, and the presence of multiple tumors. 7 The incidence of DB in the PPI and VPZ groups was compared between the two groups using the Chi-square test after propensity score matching. Data were considered statistically significant at p < 0.05.
Results
Study flow
Figure 3 shows the study flow. There were 720 cases (856 tumors) of gastric ESD during the study period, of which 352 (409 tumors) were in the PPI group and 368 (447 tumors) were in the VPZ group.

Patient flowchart.
Baseline features and clinical outcomes
Baseline characteristics and clinical outcomes are described in Table 1. There were no significant differences between the PPI and VPZ groups except in the pathology result, expert, traction method, device, and R0 resection. Tumors with R0 resection in patients with current H. pylori infection were 156/172 (90.7%), while tumors with R0 resection in patients with past infection or negative status were 642/684 (93.9%). There was no significant difference (p = 0.19).
Patient and tumor characteristics.

This item applies to patients who are positive on H. pylori tests (antibody test, urea breath test, stool test) and have no history of H. pylori eradication, or to patients who have failed H. pylori eradication therapy.
Refers to ESD of two or more tumors at the same time.
If there are multiple tumors, at least one of them should be applicable.
Experts were those who were certified by the Japan Gastroenterological Endoscopy Society and with at least 10 years of experience in endoscopic procedures.
When multiple devices are used, the main device used will be applied.
Enbloc resection rate with free vertical and horizontal margins.
Only DB cases were evaluated.
BEST-J, bleeding after ESD trend from Japan; CKD, chronic kidney disease; DB, delayed bleeding; DOAC, direct-acting oral anticoagulant; IMC, intramucosal cancer; PPI, proton pump inhibitor; SM, submucosa; VPZ, vonoprazan.
Differences in DB by specific factors
For specific factors (expert, traction method, device) that showed significant differences between the PPI and VPZ groups, we investigated the DB rates for each factor (Table 2). Divided into the PPI group, VPZ group, and overall group, we compared the DB rates between specific factors, but there was no significant difference.
Differences in DB by specific factors.

Experts were those who were certified by the Japan Gastroenterological Endoscopy Society and with at least 10 years of experience in endoscopic procedures.
When multiple devices are used, the main device used will be applied.
DB, delayed bleeding; PPI, proton pump inhibitor; VPZ, vonoprazan.
Patients’ baseline characteristics after propensity score matching
In the original cohort, there were no significant differences between the two groups in whether patients were taking warfarin, DOAC, P2Y12 receptor antagonist, aspirin, or cilostazol, whether it was CKD with hemodialysis, a tumor size >30 mm, lower third in tumor location, or the presence of multiple tumors (Table 1). After matching for nine covariates, the best final match of 329 patients was generated. The baseline characteristics of the propensity-score-matched patients are summarized in Table 3. The distribution of the nine covariates was balanced between the two groups. There was no significant difference in DB rates between the two groups (Table 3).
Patients’ baseline characteristics after propensity score matching.

If there are multiple tumors, at least one of them should be applicable.
Refers to ESD of two or more tumors at the same time.
CKD, chronic kidney disease; DB, delayed bleeding; DOAC, direct-acting oral anticoagulant; PPI, proton pump inhibitor; VPZ, vonoprazan.
Case presentation
A case of gastric ESD with DB is shown in Figure 4. The patient was a male in his 50s, not taking any antithrombotic treatment, and had no renal dysfunction. ESD was performed for a 10 mm depressed tumor using ITknife 2 with GIF-H290T. The next day, the patient had hematemesis and underwent emergency endoscopy, which revealed DB. Hemostasis was performed using RAICHO2 with GIF-H290T and VIO3. Thereafter, no recurrence of bleeding was observed. Pathological results showed intramucosal cancer with R0 resection (Figure 4).

A case of DB. (a) 10 mm depressed tumor in the lower third. (b) Post-ESD ulcer was about 30 mm. (c) The next day, the patient had hematemesis and underwent emergency endoscopy, which revealed DB in the area indicated by the white arrow. (d) Hemostasis was performed on the area indicated by the white arrow.
Discussion
This study compared a total of as many as 720 patients in the PPI group and the VPZ group using propensity score matching and found no significant difference in the DB rates after gastric ESD. The advantage of this study is that it used propensity matching for data on hundreds of patients, whereas previous reports have reported dozens of units, with the largest number being about 200 patients.16–24 By using propensity score matching with a large sample size, it is expected that more accurate results can be obtained.
The study included cases from 2017, conducted in accordance with the updated 2017 guidelines for gastroenterological endoscopy in patients undergoing antithrombotic treatment. 27 In the 2017 guidelines, for gastric ESD, warfarin can now be administered without discontinuing treatment, provided that the prothrombin time-international normalized ratio is within the therapeutic range, rather than being replaced with heparin. 27 In addition, guidelines for DOAC have been added, stating that for gastric ESD, DOAC should be discontinued from the morning of the procedure. 27 It focused on cases where gastric ESD was performed following the current protocols for antithrombotic drugs.26,27 Therefore, the use of antithrombotic drugs was standardized within the study, enabling unbiased comparisons.
Since August 2023, our institution has been performing suturing of the ulcer after gastric ESD, which may reduce DB. Therefore, cases from this period onwards were excluded from this study. Furthermore, because the first half of the study period was the PPI group and the second half was the VPZ group, the trend in ESD changed over time, and there were significant differences between the two groups for specific factors (expert traction method, device). However, there was no difference in the DB rates due to specific factors. The impact of specific factors on the DB rate was considered to be minimal.
Guidelines for ESD and endoscopic mucosal resection for early gastric cancer issued by JGES recommend the administration of acid secretion inhibitors after gastric endoscopic resection, but do not describe the type of acid secretion inhibitors. 30 In addition, the guidelines for ESD for the European Society of Gastrointestinal Endoscopy and the American Society for Gastrointestinal Endoscopy do not mention acid secretion inhibitors.31,32 The results of this study suggest that the more easily administered oral VPZ alone may be a viable option for DB prevention in gastric ESD. The stable effect of PPI is said to be achieved approximately 5 days after oral administration, and it is considered difficult to raise the intragastric pH above 4 within 24 h of administration.33–35 PPIs are membrane-permeable, acid-unstable weak bases that are formulated as enteric-coated tablets in oral preparations to prevent premature activation and degradation by gastric acid. Once PPIs pass through the stomach, they are absorbed in the proximal small intestine and transported via the bloodstream to activated gastric wall cells, where they exert their acid-suppressive effects. 36 By contrast, injectable PPI formulations are administered directly into the bloodstream and may allow for an immediate acid-suppressive effect, expecting that PPI intravenous and oral combined therapy could bring more effectiveness compared to PPI oral alone.
VPZ, on the other hand, can raise the intragastric pH to 4 or higher 6 h after administration and suppresses gastric acid more potently and rapidly than PPI, resulting in more immediate and sustained inhibition of gastric acid secretion.13,14,37 In addition, unlike PPI, VPZ is acid stable and does not require enteric coating, and VPZ is metabolized by multiple hepatic enzymes, making it less susceptible to the effects of Cytochrome P450 polymorphism. 15 In fact, it has been suggested that VPZ may be more effective than PPI in the treatment of reflux esophagitis.38,39 However, there is limited evidence for the superiority of VPZ over PPI in the prevention of DB of peptic ulcers after hemostasis.40,41 There are also several publications comparing the speed of ulcer shrinkage after gastric ESD, with some claiming faster ulcer shrinkage with VPZ compared to PPI,24,42 while others found no difference,16,18–22 so a unified view has not been reached. Further investigation is needed to evaluate the relationship between ulcer healing and the effectiveness of DB prevention.
Physiological studies have shown that acute ulcer rebleeding is dependent on intragastric pH.43–46 Gastric acid not only inhibits thrombus formation and platelet aggregation43,44 but also promotes thrombolysis by activating pepsin, and suppression of gastric acid may inhibit fibrinolysis.45,46 Therefore, VPZ, which rapidly inhibits gastric acid secretion, may be more suitable for treating gastric ulcers than PPI. However, contrary to these expectations, the results of this study showed that VPZ and PPI combined therapy were equally effective in preventing DB after gastric ESD. Considering these previous reports and our results, it is suggested that important factors in preventing DB after gastric ESD are not limited to gastric acid and ulcer healing speed but also include other factors such as bile reflux and peristalsis.
This study has several limitations. First, it is a retrospective study conducted at a single institution. Second, while the impact of specific factors such as device and traction method on the DB rate was considered minimal, the timing of ESD implementation differed between the PPI group and the VPZ group. Third, ESD has been performed at our institution since 2005, and the study period from 2017 to 2023 corresponds to the latter half of the learning curve. However, since both groups were divided between the first and second halves of the study period, it was not possible to completely rule out differences in learning effects.
Conclusion
In conclusion, there was no significant difference between the PPI intravenous and oral combined therapy and the oral VPZ alone in the prevention of DB after gastric ESD. Though further prospective studies are needed to draw definitive conclusions, it was suggested that the easily administered oral VPZ can be an important option for acid suppression after gastric ESD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251386760 – Supplemental material for Comparison of the preventive effects of proton pump inhibitors and vonoprazan on delayed bleeding after gastric endoscopic submucosal dissection
Supplemental material, sj-docx-1-tag-10.1177_17562848251386760 for Comparison of the preventive effects of proton pump inhibitors and vonoprazan on delayed bleeding after gastric endoscopic submucosal dissection by Shohei Mukai, Kenichiro Okimoto, Tomoaki Matsumura, Tsubasa Ishikawa, Yuhei Oyama, Hayato Nakazawa, Yukiyo Mamiya, Satsuki Takahashi, Chihiro Goto, Ryosuke Horio, Akane Kurosugi, Michiko Sonoda, Tatsuya Kaneko, Yuki Ohta, Takashi Taida, Keisuke Matsusaka, Jun-ichiro Ikeda and Jun Kato in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Declarations
Permission to reproduce material from other sources
The material was created by the authors and does not require permission.
Clinical trial registration
Our study was a retrospective study. Clinical trial registration was not performed.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
