Abstract
Background:
Few studies have investigated the use of endoscopic submucosal dissection (ESD) for cecal tumors extending into the appendiceal orifice. Herein, we assessed the feasibility and safety of ESD for cecal tumors extending into the appendiceal orifice.
Methods:
We retrospectively examined the outcomes of ESD for 78 patients with 78 cecal tumors (male/female ratio, 40/38; mean [standard deviation, SD] age, 67 [9] years; mean [SD] tumor size, 32 [15] mm), who underwent ESD at the Hiroshima University Hospital between October 2008 and March 2016. The indication for ESD in cecal tumors extending into the appendiceal orifice was recognition of the distal edge of the lesion in the appendix. They were classified into two groups: patients with cecal tumors extending (Group A: 29 patients, 29 tumors) and not extending (Group B: 49 patients, 49 tumors) into the appendiceal orifice. We compared the outcomes of ESD between both groups.
Results:
No significant differences in clinicopathological characteristics were observed between both groups. The rate of severe submucosal fibrosis in Group A (48%) was significantly higher than that in Group B (24%) (p < 0.05). The mean (SD) procedure speed in Group A (14 [10] mm2/min) was significantly slower than that in Group B (23 [16] mm2/min) (p < 0.01). The en bloc resection rates in Groups A and B were 90% and 96%, respectively. There were no significant differences in adverse events reported between both groups.
Conclusions:
ESD for cecal tumors with extension into the appendiceal orifice is effective and safe.
Introduction
Endoscopic submucosal dissection (ESD) has been established as a reliable method for the treatment of superficial colorectal tumors.1–9 Currently it is possible to completely resect large tumors and to evaluate tumors pathologically in detail using ESD. Colorectal ESD is technically more difficult than gastric ESD because of the thin intestinal wall, bending of the intestinal tract, the presence of folds, and poor scope operability. However, recently improved tools have made ESD more feasible and safer than before. Therefore, some high-volume centers have reported on the feasibility and safety of colorectal ESD for lesions with difficult location such as anorectal tumors with hemorrhoids close to the dentate line, 10 colorectal tumors involving a diverticulum,11,12 or cecal lesions involving the ileocecal valve. 13
In particular, ESD for cecal tumors is difficult because both the scope and tumor are almost perpendicularly oriented, and scope operability is often poor when deep colonic ESD is performed due to the bending of the sigmoid colon. In addition, the working space is narrow and the procedure cannot be performed with the inversed operation of scope. Moreover, for cases with tumor extension into the appendiceal orifice, the ESD procedure is technically more difficult to perform and laparoscopic surgery is often selected as the standard therapy for those tumors. 14 Data are scarce regarding ESD for cecal tumors extending into the appendiceal orifice.15,16 Here, we assessed the feasibility and safety of ESD for cecal tumors with extension into the appendiceal orifice.
Methods
Patients
We retrospectively examined the outcomes of colorectal ESD performed at the Hiroshima University Hospital between October 2008 and March 2016. A total of 886 tumors in the colon and rectum in 873 patients, including 78 cecal tumors in 78 patients (male/female ratio, 40/38; mean age, 67 [9] years) were treated by ESD. These patients with cecal tumors were classified into two groups: patients with cecal tumors extending into the appendiceal orifice (Group A: 29 patients, 29 tumors) and patients with cecal tumors not extending into the appendiceal orifice (Group B: 49 patients, 49 tumors) (Figure 1). The definition of cecal tumors extending into the appendiceal orifice was tumors of which the distal edge in the appendix could not be recognized without using transparent tip hood. We compared the clinicopathological findings and outcomes between the two groups.

Flow chart of the study.
The indications for ESD were defined using the criteria proposed by the Japan Gastroenterological Endoscopy Society 17 and the Japanese Society of Gastroenterology as follows. 18 ESD was indicated for tumors that required en bloc resection and for which en bloc resection using snare endoscopic mucosal resection (EMR) would be difficult, 19 which included laterally spreading tumors of the nongranular type, particularly, the pseudodepressed type, tumors with a type VI pit pattern, carcinomas with submucosal shallow invasions of less than 1000 μm, large depressed tumors, and large elevated tumors that were probably malignant, including large nodular lesions such as laterally spreading tumors of the granular type. In addition, ESD was indicated for intramucosal tumors with fibrosis caused by biopsies or peristalsis, local residual early stage carcinomas that developed after endoscopic resection, and sporadic localized tumors associated with chronic intestinal inflammation conditions, including ulcerative colitis. In addition, in our institution, the indication for ESD for cecal tumors with extension into the appendiceal orifice was the recognition of the distal edge of the lesion in the appendix.
The study was performed in accordance with the Declaration of Helsinki. All patients were informed of the risks and benefits of ESD, and each provided written informed consent for the use of patients’ data. This study protocol was approved by the Institutional Review Board of Hiroshima University Hospital (Approval number: E-591, Institutional Review Board registration date: 14 November 2016).
ESD procedure for cecal tumors with extension into the appendiceal orifice
ESD was performed by the two experts (ST or SO) in this series. Carbon dioxide insufflation was used routinely. We performed ESD using a high-resolution video endoscope (CF-H260AZI, PCF-Q260AZI, CF-Q260JI, Olympus, Tokyo, Japan, CF-Y0047 [prototype], or CF-Y0006 [prototype]). In order to overcome the difficulty of ESD for cecal tumors with extension into the appendiceal orifice, we devised some measures: (a) the use of a bell-shaped, small-caliber-tip, transparent tip hood; (b) the use of a splinting tube with a balloon; (c) the use of Dual knife needle-in technique and SB knife Jr.
An endoscope was attached to a bell-shaped, small-caliber-tip, transparent tip hood (ST Hood, Fujifilm, Tokyo, Japan or ST Hood short type, Fujifilm) in order to facilitate good field visualization and allow stable dissection. We used a splinting tube with a balloon (ST-CB1, Olympus) to improve scope operability if scope operability was poor due to the bending of the sigmoid colon. 20 We mainly used a Dual knife (Olympus) or Dual knife J (Olympus) with an electrosurgical generator (ESG-100, Olympus). The needle length of a Dual knife is 1.5 mm in a needle-out state and the tip measures only 0.3 mm in a needle-in state. With a Dual knife J, it is 0.1 mm, in the needle-in state, thereby ensuring even safer dissection. In a situation where the scope and muscular layer are almost perpendicularly oriented, the risk of perforation is high in a needle-out state, so we dissected safely in a needle-in state. When the lesion moves with the act of breathing or the scope operability is poor, we used the SB knife Jr (Sumitomo Bakelite, Tokyo, Japan), which allows safe dissection simply by opening and closing the scissors without moving the knife itself. We mixed equal volumes of 0.4% sodium hyaluronate (Muco Up, Johnson & Johnson, New Brunswick, NJ, USA) and 10% glycerin solution, and added a small amount of indigo carmine (0.2 ml per 20 ml sodium hyaluronate + glycerin). Endoscopic hemostasis was achieved with hemostatic forceps (Coagrasper, Olympus). The pulse-cut slow-mode setting (25 W) was used for mucosal incisions, and the forced coagulation mode (25 W) was used for submucosal dissection. We used the pulse-cut fast-mode setting (30 W) and soft coagulation (40 W) with the SB knife Jr. Basically, mucosal incision and initial dissection were performed from the side of the appendiceal lumen to prevent very deep dissection into the appendiceal orifice. When there was inadequate space for mucosal incision on the side of the appendiceal lumen, the mucosal incision and the submucosal dissection were started from the periphery of the tumor, and finally continued into the appendiceal orifice sufficiently. These procedures are shown in Figure 2. This tumor was a cecal tumor of size 10 mm and growth type 0–Is. Although a part of the tumor extended into the appendiceal orifice, its edge could be recognized in the appendix. Therefore, we performed an ESD after explaining the risks and benefits of the procedure to the patient. The histological features of the tumor are shown in Figure 2(m). The tumor was an intramucosal carcinoma. Although the tumor extended into the appendix, the horizontal and vertical margins were negative.

Endoscopic submucosal dissection procedure.
Outcomes of ESD
We evaluated the clinicopathological characteristics of cases, procedure time, en bloc resection rate, histological complete resection rate, curative (R0) resection rate, and adverse events.
The procedure speed was calculated by dividing the area of the resected specimen by the procedure time (mm2/min). The approximate area of the resected specimen (mm2) was calculated as follows: 3.14 × 0.25 × long axis diameter (mm) × short axis diameter (mm).21,22 Poor scope operability was defined as situations in which paradoxical movement of the endoscope, poor control with adhesions, and lesion motion with heart beats or breathing occurred, as reported previously. 23 Endoscopically, the degree of submucosal fibrosis was classified as no fibrosis, mild fibrosis, and severe fibrosis. 24 Delayed bleeding was defined as a decrease in hemoglobin levels by 2 g/dL or more compared with the last preoperative level, or any apparent bleeding or massive melena. 25 A histologically complete resection was defined as a histopathologically complete en bloc resection with negative tumor margins. A curative (R0) resection was determined using the Japanese Society for Cancer of the Colon and Rectum guideline criteria, which involved satisfying all four of the following characteristics: a well/moderately differentiated or papillary carcinoma; no vascular invasion; a submucosal invasion depth < 1000 μm; grade 1 budding. 26 At 1 year after ESD, follow-up colonoscopies were performed on the patients who underwent histologically complete resections of high-grade dysplasias and T1 carcinomas that met the curative criteria. For patients with piecemeal high-grade dysplasias and those with tumors that had histologically positive horizontal margins, follow-up colonoscopies were performed at 3–6 months after ESD, and further colonoscopies were performed 1 year later. For patients who had undergone ESD for T1 carcinomas that did not meet the curative criteria, blood tests, including the carcinoembryonic antigen level, and computed tomography of the abdomen and pelvis were performed every 6 months postoperatively for the first 3 years and every 12 months thereafter, and follow-up colonoscopies were performed every year.
Statistical analysis
The Student’s t test and the Mann–Whitney U test were used to compare the distribution of continuous variables by outcome, and the Pearson chi-squared test or Fisher’s exact probability test were used to examine the association between categorical variables and outcomes. p < 0.05 was considered to be statistically significant. All statistical analyses were performed using JMP version 10 (SAS Institute Inc., Cary, NC, USA).
Results
Comparison of clinicopathological characteristics between Group A and Group B are shown in Table 1. The mean and standard deviation (SD) tumor size in Group A was 32 (16) mm and that in Group B was 33 (15) mm. Seven (24%) patients in Group A and five (10%) patients in Group B had undergone appendicectomy in the past; the difference between both groups was not significant. Likewise, there were no significant differences between the two groups in sex ratio, mean age, ratio of anticoagulants and/or antiplatelets use, growth type, and histology.
Comparison of clinicopathological characteristics between Group A and Group B.
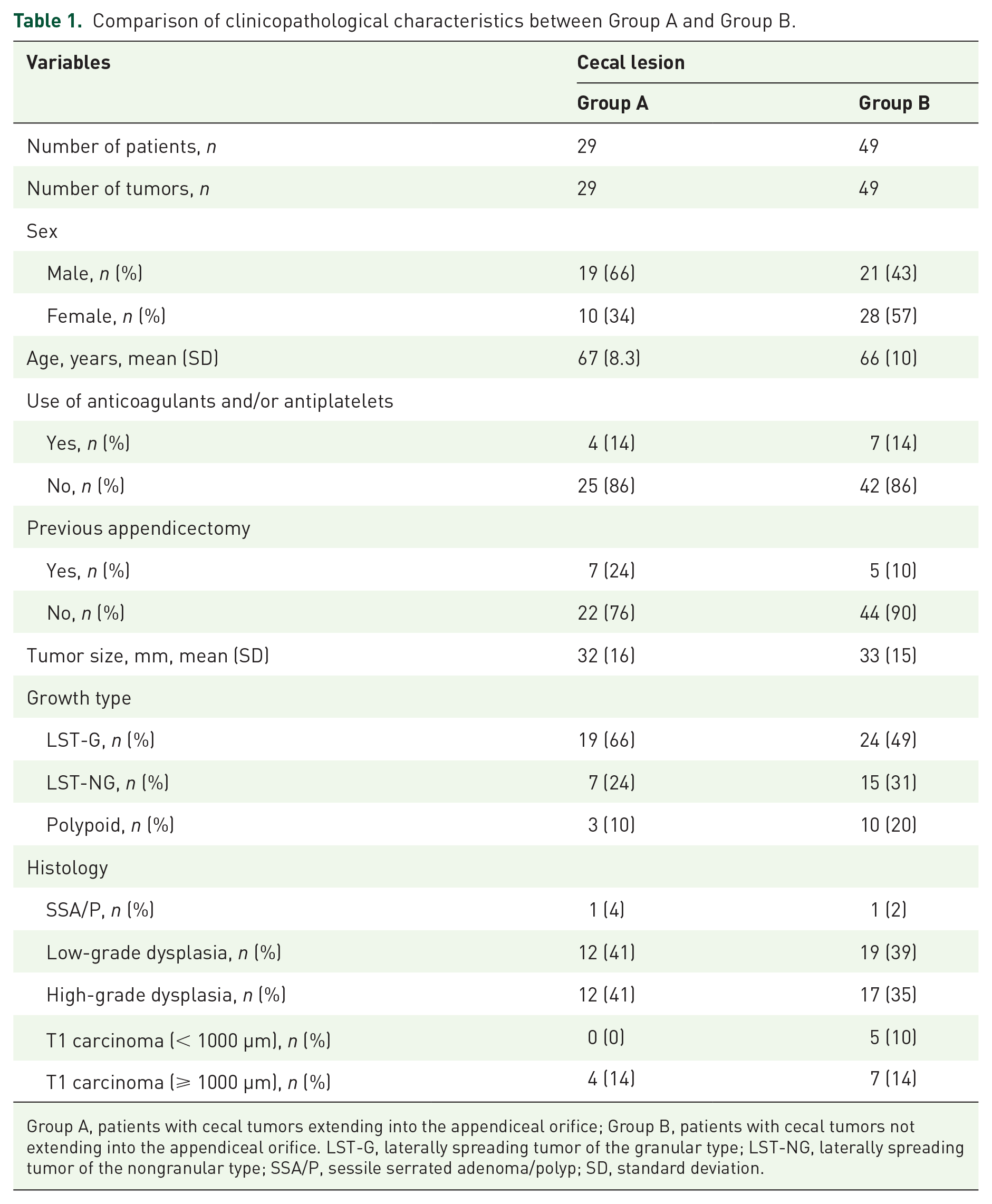
Group A, patients with cecal tumors extending into the appendiceal orifice; Group B, patients with cecal tumors not extending into the appendiceal orifice. LST-G, laterally spreading tumor of the granular type; LST-NG, laterally spreading tumor of the nongranular type; SSA/P, sessile serrated adenoma/polyp; SD, standard deviation.
Comparison of outcomes related to ESD between Group A and Group B are shown in Table 2. The rate of severe submucosal fibrosis in Group A (48%) was significantly higher than that in Group B (24%) (p < 0.05). The mean procedure time in Group A was 117 (127) min and that in Group B was 82 (52) min, and the difference was not significantly different. The mean (SD) procedure speed in Group A (14 [10] mm2/min) was significantly slower than that in Group B (23 [16] mm2/min) (p < 0.01). The en bloc resection rate, histological complete resection rate, and R0 resection rate were 90%, 86%, and 86%, respectively, in Group A and 96%, 88%, and 84%, respectively, in Group B, and were not significantly different between both groups. Two (7%) patients in Group A and five (10%) patients in Group B underwent additional surgery. Perforation during the procedure occurred in two (7%) patients in Group A and one (2%) patient in Group B. All cases underwent ESD for tumors with severe submucosal fibrosis in the early period of the introduction of ESD and none of the patients required surgery. Delayed bleeding occurred in one (3%) patient in Group A and two (4%) patients in Group B. None of them required transfusion. There were no significant differences in adverse events between the two groups. The mean follow-up duration was 27 (14) months (range 7–62 months) and no local recurrence was found.
Comparison of outcomes related to endoscopic submucosal dissection between Group A and Group B.

Group A, patients with cecal tumors extending into the appendiceal orifice; Group B, patients with cecal tumors not extending into the appendiceal orifice. NS, not significant; SD, standard deviation.
Discussion
ESD for cecal tumors with extension into the appendiceal orifice is technically difficult because the field visualization and scope operability are often poor and the scope and tumor are almost perpendicularly oriented. Thus, laparoscopic surgery has often been selected as the standard therapy for those tumors. 14 Recently, there have been reports about endoscopic full-thickness resection for appendiceal adenoma.27–29 However, the number of cases was small and the feasibility and safety were unclear. Moreover, endoscopic full-thickness resection cannot achieve en bloc resection of large tumors. However, our data revealed that ESD for cecal tumors with extension into the appendiceal orifice was clinically effective and safe. An endoscope attached to a bell-shaped, small-caliber-tip, transparent tip hood such as the ST Hood or ST Hood short type facilitated good field visualization even in the appendiceal lumen. We also used a single-use splinting tube with a balloon to improve poor scope operability due to the bending of the sigmoid colon in deep colonic ESD. 20 We used single-use splinting tubes for five cases in this study. In the situations where the scope and muscular layer were almost perpendicularly oriented, we used the Dual knife needle-in technique or SB knife Jr. 30 Recently, Ritsuno and colleagues 31 reported that S-O clip-assisted ESD was safe and fast for en bloc resection of large superficial colorectal tumors because the S-O clip allows the direct visualization of the cutting line during ESD and can be used at any location without withdrawing the endoscope.
In the present study, there was no local recurrence during the follow-up duration. However, Eun Mi Song and colleagues 32 reported that the recurrence rate after endoscopic resection for cecal polyps involving appendiceal orifice was high (15.6%), and that polyps involving 75% or more of the appendiceal orifice circumference were an independent risk factor for recurrence in their study regarding endoscopic resection for cecal polyps involving the appendiceal orifice. Although it was mainly caused by the high rates of endoscopic piecemeal mucosal resection in their series (23.7%) due to the difficulty of procedure, it was also possible that a part of tumor remained in the appendiceal lumen. It is important to detect the minute amounts of residual tumor tissue that surround the resected ulcer or ulcer bed after ESD, and to evaluate and confirm the pathological horizontal margin carefully even when an en bloc resection has been achieved.
No patients in this study experienced acute appendicitis after ESD. However, there are some case reports about acute appendicitis after EMR.33,34 Furthermore, Jacob and colleagues 15 reported that 2 out of the 76 patients who underwent ESD for tumors near the appendiceal orifice experienced acute appendicitis after ESD, and that one case might have been caused by the clips used for closure of the ulcer bed during ESD. Therefore, it is important to explain to patients before ESD about the possibility of appendicitis and to prevent unnecessary clipping for the ulcer bed around the appendiceal orifice after ESD. Moreover, in the event of increased inflammatory response after ESD, the use of antibiotics should be considered.
Jacob and colleagues 15 classified the lesions into four groups according to the relationship between the tumor and appendiceal orifice. ESD was not performed on tumors that deeply entered the appendiceal orifice with edges that could not be observed unless appendicectomy was performed prior to ESD in their series. In our institution the indication for ESD in cecal tumors extending into the appendiceal orifice is the recognition of the distal edge of the lesion in the appendix, and this must be definitively observed before ESD. The ST Hood and ST Hood short type are useful in facilitating good visualization and ascertaining how deep the tumor extends into the appendix. If the distal edge of the tumor cannot be observed despite the use of the ST Hood or ST Hood short type, the tumor should be resected by surgery. Five cases underwent surgery for this reason during the same period in the current study. Histologically, in all of these cases, the tumor extended deeply into the appendiceal lumen and surgical resection was reasonable.
This study has some limitations. First, it was a retrospective study of clinical records in a single center. Second, the study data were only from experts in ESD. Third, the number of cases that underwent ESD for cecal tumors extending into the appendiceal orifice was relatively small. Thus, more of these cases are needed to clarify further the indications of ESD for cecal tumors. Finally, this study was not designed to compare ESD with conventional surgery or EMR.
In conclusion, ESD for the cecal tumors with extension into the appendiceal orifice can be performed effectively and safely.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
