Abstract
Background:
Inflammatory bowel disease (IBD) is characterized by periods of remission and relapses, and treatment is based on phenotype, risk factors, and disease severity. Treatments include 5-aminosalicylates (5-ASA), thiopurines, methotrexate, calcineurin inhibitors, corticosteroids (CS), biological therapy (BxT), and, more recently, small molecules.
Objective:
To determine the baseline demographics and clinical characteristics, treatment patterns, and disease status of patients in Mexico with a history of moderate/severe IBD returning for hospital follow-up (Index Day).
Design:
This was a non-interventional, cross-sectional study.
Methods:
Socio-demographics, clinical characteristics, and prescribed treatments were collected from a retrospective review (3 years) of each patient’s medical records.
Results:
A total of 326 patients with a diagnosis of moderate/severe IBD at least 6 months before the Index Day were included in the analysis: 95 patients (29.2%) had Crohn’s disease (CD) and 231 (70.9%) ulcerative colitis (UC). In the CD group, 45.3% (n = 43) had a Harvey Bradshaw Index score ⩾8 or Crohn’s Disease Activity Index ⩾220; 10 patients had a B1-non-stenosing, non-penetrating phenotype and 17 had stenosis (B2). In the UC group, 18.2% (n = 42) had moderate/severe disease and the most frequent presentation was pancolitis (n = 56). Regarding treatment over the previous 3 years: for CD, 62 (65.3%) received CS and 20.0% (n = 19) were CS-dependent; 30.5% received 5-ASA + IMS; 27.4% BxT + IMS; and 38.9% 5-ASA + IMS + BxT. In the case of UC, 74.9% (n = 173) received CS and 32.9% (n = 76) were CS-dependent; 64.5% received 5-ASA + IMS; 2.2% BxT + IMS; and 31.6% 5-ASA + IMS + BxT.
Conclusion:
In Mexico, 45.3% of CD patients and 18.1% with UC presented with moderate/severe disease activity. Conventional therapy was used to treat the majority of patients, and the availability of more advanced therapies and a personalized treatment approach is needed to improve clinical outcomes in the future.

Introduction
Inflammatory bowel disease (IBD), comprising ulcerative colitis (UC) and Crohn’s disease (CD), is a chronic immune-mediated disorder of the gastrointestinal tract with a complex multifactorial etiology involving genetic, immunological, environmental factors, and gut dysbiosis.1 –10 It is a prevalent disease associated with significant morbidity and is a major public health burden worldwide. In Mexico, a recent study identified 9953 patients with CD and 33,060 with UC on specific databases of the Mexican National Healthcare System. 11 The authors reported that 35.1% of patients with CD and 31.6% of patients with UC were ⩾50 years of age, showing that IBD affects patients of all ages. In a nationwide cohort study in Mexico between the years 2000–2017, the prevalence rates for IBD, UC, and CD were 1.83, 1.45, and 0.34 cases per 100,000-person-years, respectively. The highest incidences were recorded in the final statistics reported for the year 2015, and the study found a significant increase in the incidence and prevalence of IBD in Mexico over 15 years, and this was predominantly for CD over UC. 12
For patients with moderate/severe CD, clinical practice guidelines recommend biologic drugs, either as monotherapy or combined with an immunosuppressant to induce and maintain remission for steroid-refractory disease or in steroid-dependent patients.13 –16 Regarding treatment of moderate/severe active UC is an immunosuppressant and/or a biologic drug is generally recommended.14,15 Oral or rectal mesalazine (5-aminosalicylate (5-ASA) or mesalamine)-like agents are also indicated for induction and maintenance treatment of UC. 16 Corticosteroids are recommended for short-term use (⩽3 months) during relapses of both CD and UC, but not for maintenance therapy.13,15,16 In Europe, physicians often try a number of different treatment options and sequences for the management of IBD, both as monotherapy or in combination regimens. 13 In Mexico, current guidelines support the use of biological therapies such as anti-TNF-α agents, vedolizumab, and ustekinumab in IBD, as well as the small-molecule Janus kinase inhibitor, tofacitinib in patients with ulcerative colitis. 17
Real-world studies are important to identify gaps in clinical management and might be particularly relevant for developing countries in Latin America such as Mexico, where the prevalence of IBD is increasing but access to advanced therapy may be restricted. 18 RISE-BR is a real-world study that assessed IBD in Brazil and found that in a real-world setting, the management of IBD was different from guideline recommendations due to difficulties in accessing advanced therapies, especially in a public healthcare system. 19 Based on those findings, and the lack of information related to Mexican IBD patients’ disease characteristics and treatments, we undertook this real-world non-interventional study in patients with IBD in Mexico (RISE-MX).
Mexico’s healthcare system consists of three subsystems. INSABI (now IMSS-bienestar) provides cover for the poor and uninsured population (43.5%), while social insurance institutions (i.e., IMSS and ISSSTE) provide healthcare cover for 40.4% of formally employed individuals. Finally, there is a private system, primarily composed of small firms, providing cover for some large national corporations including the hospital and pharmaceutical sectors. The country has 4341 hospitals in total, with 30% (1381) in the public sector and 2960 (68%) in the private sector. Public hospitals primarily serve the uninsured, while the majority of private hospitals are concentrated in urban areas, particularly in Mexico City, Nuevo León, and Jalisco. In 2016, there were 1.9 physicians per 1000 inhabitants according to ENOE (National Occupation and Employment Surveys), below the OECD (Organisation for Economic Co-operation and Development) average of 3.3; however, the OECD adjusted this figure to 2.4 due to issues like double counting. This situation underscores the significant challenges in achieving equitable healthcare access throughout Mexico. 20
The aim was to quantify the number of IBD patients attending 12 major clinics in Mexico during a 9-month period, to determine the type, characteristics, and severity of the disease at the time of entry. In addition, a retrospective analysis of medical records was performed to define each patient’s IBD journey prior to entry, evaluating parameters relevant to the disease such as changes in severity, treatment patterns, and comorbidities.
Methods
Study design
RISE-MX study was a national, multicenter, non-interventional study designed to better understand the clinical management of IBD patients in Mexico. It involved a cross-sectional evaluation of all consecutive IBD outpatients attending 10 gastroenterology private practice clinics covering Mexico’s most populated regions between August 2021 and May 2022 (from 12 planned sites only 10 sites enrolled patients). This included a few (approximately 10%) patients from public hospitals when such patients agreed to a request for their medical records in compliance with local regulations (this variable was not collected for each patient, but there were at least two sites that provide private healthcare services to patients that also get them in public services). At each center, eligible subjects were identified consecutively as they attended a scheduled routine clinical appointment with their physician (Day 1). This was followed by retrospective data collection referring to the 3 years prior to Day 1, regarding the previous IBD treatments based on a review of their medical records for the previous 3 years.
Study objectives
Primary objective
To evaluate the disease activity on the day of enrollment (Day 1; Index Day) among UC and CD patients diagnosed with moderate/severe disease (i.e., to evaluate the proportion of patients with active CD defined as Harvey Bradshaw Index (HBI) score ⩾8 or Crohn’s Disease Activity Index (CDAI) ⩾220, and with active UC defined as 9-point partial Mayo (pMayo) ⩾5).
Secondary objectives
To characterize socio-demographic and clinical features of UC and CD patients, regardless of the disease activity, for patients with moderate to severe and for patients with mild or non-active(remission) by disease activity on the Index Day.
To characterize treatment patterns for UC and CD during the 3 years prior to the Index Day, including the use of biological therapy (BxT) and conventional therapies, and responses to these therapies (by disease activity on the Index Day).
Study population
A sample size of 335 patients was calculated considering the absence of data on the control of IBD in Mexico, we have assumed a control rate of 50% for both UC and CD. This assumption allows us to estimate the prevalence with 95% confidence and a margin of error of less than 5.5%. To achieve this level of precision, we needed a sample size of 318 patients, regardless of whether they have UC or CD.
Patients were included if they were aged 18 years or older and had a confirmed diagnosis of moderate-to-severe CD or UC established at least 6 months prior to the Index Day (based on clinical, endoscopic, or imaging criteria).21,22 This was irrespective of the severity of disease activity (see Study variables section for definition) at the time of enrollment as the objective of this study was to describe the disease activity on Day 1 (Index Day), and the journey of patients (disease activity changes and changes in treatment) during the previous 3 years. Patients were excluded if they presented with indeterminate/unclassified colitis, were unwilling to participate in the study, had a mental illness or language difficulties which in the view of the investigator precluded their ability to fully understand and cooperate with the study protocol; or had participated in a clinical trial within the last 3 years.
All patients provided written informed consent, and the study was conducted in accordance with the Declaration of Helsinki and the International Conference on Harmonization Guidelines for Good Clinical Practice, 23 and in accordance with all applicable laws and regulations. It was approved by the Local Ethics Committee at each study site according to national regulations and was registered in clinicaltrials.gov (NCT04873700).
Study variables
Based on an interview on the Index Day at the time of enrollment and a retrospective review of the patient’s medical records for the previous 3 years, the following information was recorded: sociodemographic characteristics (age, sex, educational level, professional status, and personal income); anthropometric data (weight and height) on the Index Day; disease features such as type of IBD, date first diagnosed, diagnostic criteria considered to characterize IBD (abnormalities according to the diagnostic criteria at the time); the extent, severity and location of CD or UC (Montreal classification); and response to corticosteroid therapy. With regards to corticosteroid usage: steroid-dependent disease was defined as, being unable to reduce steroids below the equivalent of prednisolone 10 mg/day (or budesonide <3 mg/day) within 3 months of starting steroids, without recurrent active disease; or, having a relapse within 3 months of stopping steroids. Steroid-refractory disease was defined as, patients with active disease despite prednisolone of up to 0.75 mg/kg/day over a period of 4 weeks.
Other relevant clinical information including the presence of extra-intestinal manifestations; all treatments used to manage the patient’s IBD including corticosteroids, BxT, 5-ASA, and immunosuppressants.
CD activity was evaluated with the HBI 24 and/or CDAI. 25 Moderate/severe activity CD was defined as patients with an HBI ⩾8 or a CDAI ⩾220, while mild/non-active CD disease was defined as an HBI <8 or a CDAI <220.24 –26 UC disease activity during follow-up was assessed with the 9-item pMayo score; moderate/severe active UC was defined as pMayo ⩾5, and mild/non-active UC was defined as pMayo <5. 27 The listing of all the sociodemographic, clinical, disease activity, and treatment variables which were collected in the study are shown in Supplemental Tables 1–10.
Statistical analysis
Continuous variables were summarized using mean and standard deviation (SD), and for categorical variables, percentages were used. Initially, descriptive statistics summarized the treatment patterns for CD and UC over the previous 3 years, including the variables of interest mentioned earlier. In the CD and UC groups, we compared patients with moderate-to-severe disease activity with patients with mild or no activity regarding socio-demographic variables. We used the Chi-squared or Fisher exact test to compare categorical variables such as sex, smoking habits, professional status, and family history of IBD, among others. We employed the student’s test for the comparison of continuous variables such as age, time since diagnosis, and duration of treatment, among others. When applicable, a logistic regression model (for CD and UC patients) was used to identify independent variables associated with disease control on day 1, and odds ratios with 95% confidence intervals were presented. This analysis controlled for all risk factors for binary outcomes and was adjusted for moderate-to-severe versus mild/non-activity on day 1. Regression models were run on complete cases, that is, where data were not missing for any of the predictors. Missing data were not replaced with imputed values unless stated otherwise. All statistical tests were two-tailed, with a significance level of 0.05, using SAS® (version 9.4, SAS Institute Inc., Cary, NC, USA).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Results
Patient disposition and clinical characteristics
During the recruitment period, 335 patients from private practice centers were enrolled in the study, but nine were excluded because they did not meet the eligibility criteria (IBD type/activity not specified). Thus, 326 patients comprised the study population on the Index Day and represented the Analysis Set (Figure 1). Of these, 95 patients (29.1%) had CD and 231 (70.9%) UC. In the CD group, 45.3% (n = 43) of patients had moderate/severe disease activity and 54.7% (n = 52) had mild/non-active disease at the index date. In the UC group, 18.1% (n = 42) had moderate/severe disease activity and a much higher percentage had mild/non-active disease (81.9%; n = 189) at the index date. Figure 1 shows the distribution of the enrolled participants in the study in relation to gender and disease type/severity.
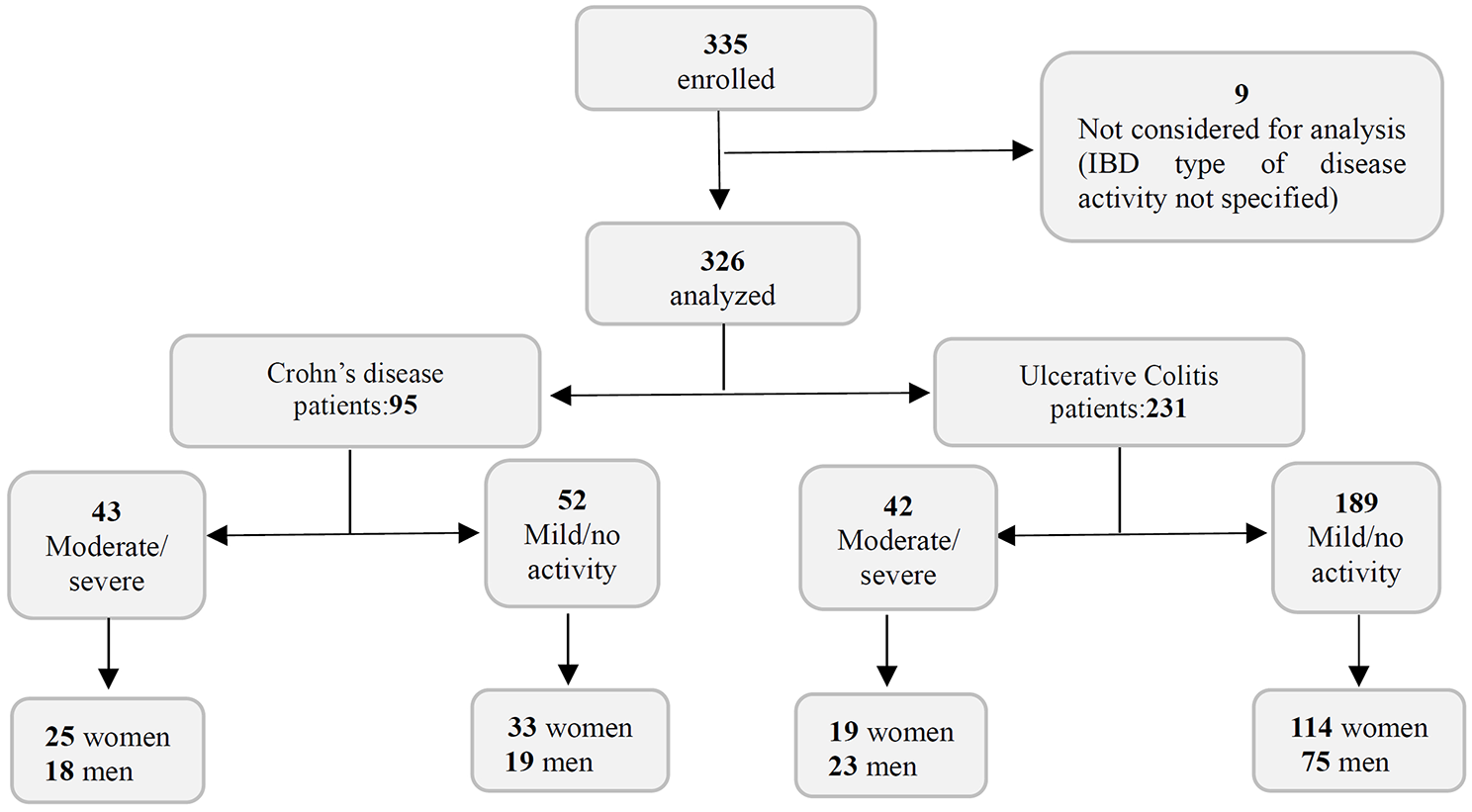
Flow chart of the distribution of the enrolled participants with Crohn’s disease or ulcerative colitis in the RISE-MX study.
Table 1 details the sociodemographic and anthropometric characteristics of the studied population and is presented in relation to disease activity on the Index date. The only age-related statistically significant difference was in the CD group in which patients with moderate/severe disease tended to be older than patients with mild/non-active disease (49.5 ± 15.4 years vs 42.8 ± 16.2 years; p = 0.027). Across the total cohort, there was a higher proportion of females to males (191 (58.6%) vs 135 (41.4%)) with approximately 70% having UC. As expected, there was a higher proportion of patients with moderate/severe disease in the CD group compared with the UC group (45.3% vs 18.2%) and the most noticeable gender-related trend was for more female than male patients with UC to have mild/non-active disease (85.7% (114/133) vs 76.5% (75/98)). In the CD group, the number of female patients with mild/non-active disease was only slightly more than the number with moderate/severe disease (63.5% vs 58.1%). There were no statistically significant differences related to gender in either of the UC or CD groups. A greater number of patients with moderate/severe CD and mild/non-active UC had completed higher levels of education, but the differences in relation to personal income were only significant for CD (p = 0.018) (Table 1).
Sociodemographic characteristics for patients with Crohn’s disease and ulcerative colitis by disease activity at day 1.

Obtained from Chi-square/Fisher’s exact or t-test.
Table 2 summarizes the medical history, including extra-intestinal manifestations, of the enrolled patients on the Index Day. A relevant medical history or extra-intestinal manifestation was reported by 69.8% of patients with moderate/severe CD, and 53.8% with mild/non-active disease. The comparable numbers for patients with moderate/severe and mild/non-active UC were 38.1% and 47.6%, respectively. In both the CD and UC groups, the two most common extraintestinal manifestations were arthralgia and arthritis (Table 2).
Summary of medical history/extraintestinal manifestations for Crohn’s disease and ulcerative colitis patients by disease activity at day 1.

Obtained from Chi-square/Fisher’s exact t-test.
Categories are not mutually exclusive, and proportions were calculated based on all the patients across the categories.
Information pertaining to IBD for all 326 patients eligible for the study on the Index Day is presented in Table 3 and includes disease type/severity; age at time of diagnosis and duration of disease from diagnosis to Index Day; and all criteria considered for the diagnosis of IBD. The mean ± SD age at the time of diagnosis ranged from 36.3 ± 13.7 years (mild/non-active UC) to 41.4 ± 15.5 years (moderate/severe CD) and the mean ± SD disease duration ranged from 71.8 ± 57.8 months (mild/non-active CD) to 119.9 ± 100.1 months (moderate/severe CD). For all groups of IBD patients, the main criteria for severe-moderate diagnosis were clinical (83%–100% of cases), endoscopic (57.7%–81.0% of cases), and histologic (35.7%–48.1% of cases), with imaging and biomarkers used far less frequently. It is important to emphasize that results come from multiple answer selection, patients were diagnosed using a combination of clinical, endoscopic, or histologic criteria.
Summary of inflammatory bowel disease type by disease activity at day 1.

Incidence of manifestations on the Index day in patients with a diagnosis of IBD. Categories are not mutually exclusive, and proportions were calculated based on all the patients across the categories.
Patients with mild disease.
IBD, inflammatory bowel disease.
On the Index Day, 40.5% of patients presented a major steroid-dependent behavior in the UC moderate/severe group compared with 31.2% in the mild/non-active UC group; in contrast, it was less frequency observed in the CD groups (25.6% for moderate/severe patients and 15.4% for mild/non-active patients). A small number of patients with IBD were considered to have steroid-refractory disease (4.9%) with a slightly higher proportion in the moderate/severe group (10.6%) versus the mild/non-active group (2.9%) (Table 3).
A summary of the available data for the clinical characteristics of patients with CD on the Index Day in relation to disease activity, disease location according to the Montreal classification, 28 phenotypes, and perianal/ileal disease are presented in Table 4. Likewise, a summary of the available data on the clinical characteristics of patients with moderate/severe and mild/non-active UC on the Index Day in relation to disease location and behavior (severity) is presented in Table 5. It should be noted that these data are very limited since such information was not routinely collected in all hospitals.
Summary of clinical characteristics of Crohn’s disease patients by disease activity at day 1.

GI, gastrointestinal.
Summary of clinical characteristics of UC patients by disease activity at day 1.

UC, ulcerative colitis.
Risk factors
Odds ratios for risk factors associated with moderate/severe IBD are presented in Table 6. Even though there appeared to be some risk factors during the retrospective period such as age, smoking habits, and steroid behavior amongst others, only immunosuppressor treatment in the UC group demonstrated statistical significance based on the 95% Confidence interval (CI). However, the overall CIs were generally very wide and any associations based on this analysis involving relatively small numbers of patients should be viewed cautiously.
Summary of odds ratios for risk factors associated with severe/moderate Crohn’s disease or ulcerative colitis.

Reference: Reference category used to compare the other categories for each section.
Obtained from an adjusted logistic regression model with the binary outcome of moderate/severe versus mild/no disease activity at Day 1 and controlling for all risk factors shown. The model is based on complete cases (i.e., patients with data available for all predictors (N = 94 (CD) and N = 231 (UC)).
Includes azathioprine, mercaptopurine, methotrexate, cyclosporine, or tacrolimus.
Includes infliximab, adalimumab, vedolizumab, certolizumab, golimumab, or ustekinumab.
Biologic therapy includes the same regimens as noted in footnote c; 5-Aminosalicylates include Sulfasalazine, mesalazine/mesalamine, or mesalazine/mesalamine extended release; Other includes hydrocortisone, prednisone, prednisolone, budesonide, or methylprednisolone, azathioprine, mercaptopurine, methotrexate, cyclosporine, tacrolimus, metronidazole, ciprofloxacin, enteral nutrition, or parenteral nutrition or other.
CD, Crohn’s disease; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Previous treatments
Figure 2 provides details of previous treatments or regimens used by patients with IBD. For both the CD and UC groups, mesalazine (5-ASA) was one of the most used therapies, with 47.4% for CD and 76.2% for UC. Other commonly used treatments included azathioprine (used in 34.7% for CD and 40.3% for UC), infliximab (23.2% for CD and 23.8% for UC), and adalimumab (45.3% for CD and 13.0% for UC). The most used treatment in the three previous years was: aminosalicylates + immunosuppressants 30.5% for CD, and 64.5% for UC; 5-ASA aminosalicylates + immunosuppressants + biologic therapy 38.9% for CD, and 31.6% for UC; and biologic therapy + immunosuppressants 27.4% for CD, and 2.2% for UC.
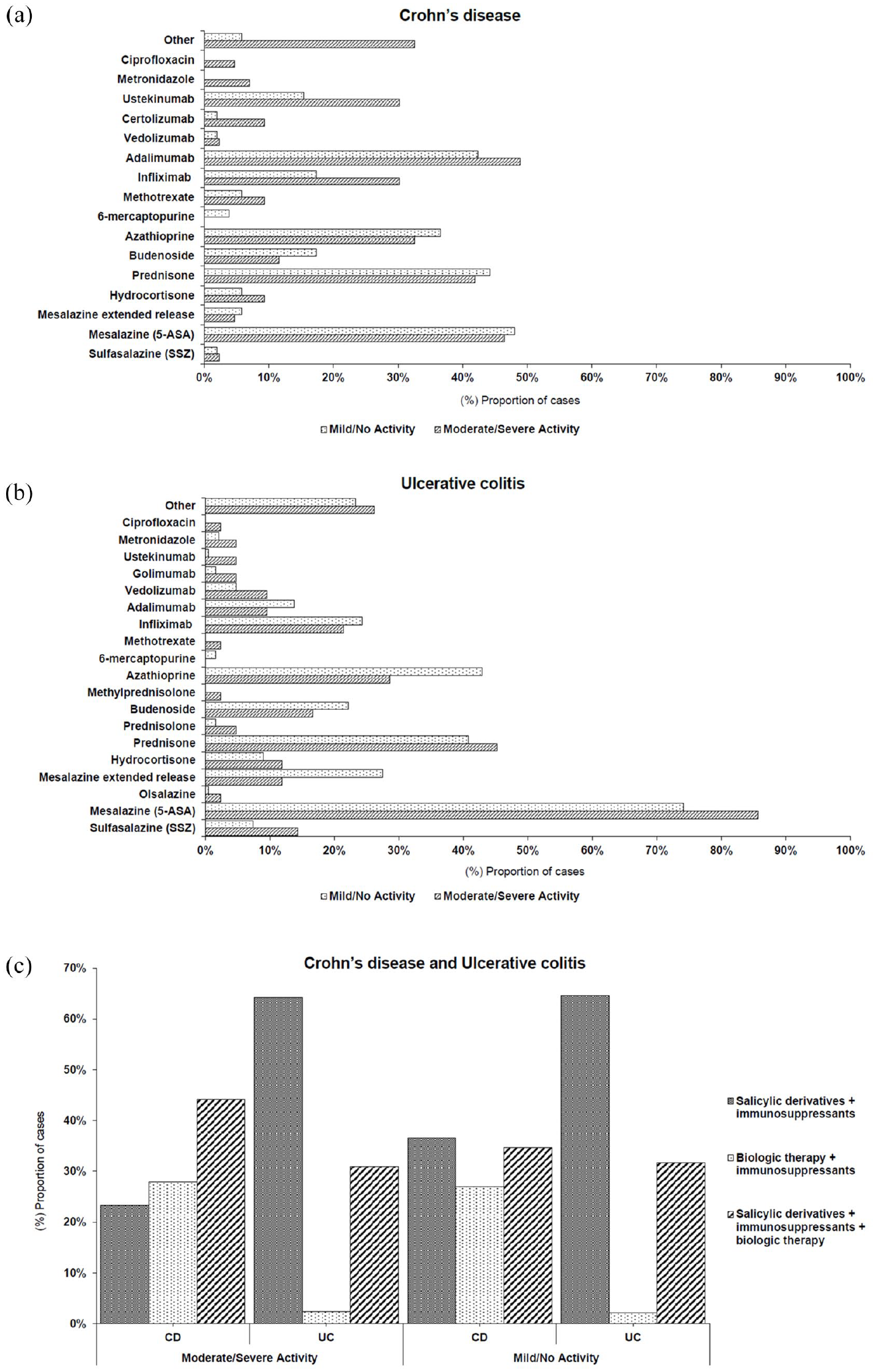
Previous treatments or regimens over 3 years before day 1 for CD (a) and UC (b); patients sorted by disease activity at day 1. All categories are not mutually exclusive. 5-ASA + immunosuppressants include sulfasalazine, mesalazine/mesalamine, olsalazine, mesalazine/mesalamine extended release, azathioprine, mercaptopurine, methotrexate, cyclosporine, or tacrolimus. Biologic therapy + immunosuppressants includes infliximab, adalimumab, vedolizumab, certolizumab, golimumab, ustekinumab, azathioprine, mercaptopurine, methotrexate, cyclosporine, or tacrolimus. Previous treatment or regimen in CD and UC patients’ treatment schemes (c). Salicylic derivatives + immunosuppressants + biologic therapy includes any of the regimens mentioned previously. Other corresponds to any other treatment not mentioned on the list.
Discussion
This study was designed to update the status of IBD in real-world everyday practice in Mexico. A total of 326 eligible patients with a diagnosis of moderate/severe IBD within 6 months of enrollment into the study comprised the analysis set. A cross-sectional evaluation of this cohort found that on the Index Day, the majority of patients had UC (70.9% (231/326)) compared with 29.1% (95/326) with CD, and across both cohorts, 73.9% had mild/non-active disease. According to available epidemiological data in Mexico, UC is three times more prevalent than CD and our results broadly correspond with this rate.12,29 These findings also suggest that the prevalence ratio for UC and CD in Mexican IBD patients has remained constant since 2017.
With respect to disease location, an Asian consortium reported that the most frequent CD presentation was L3: ileocolic (43%) with low rates of perianal disease (8%). 30 In patients with UC, the disease was predominantly distal (proctitis/left-sided in 67%). A total of 9.4% of perianal disease was observed, in contrast, two studies involving Chinese patients with IBD reported higher rates of perianal disease of between 15% and 40%,31,32 this could be related to the fact that no coloproctologists participated in the study sites reflecting only the complications for treated patients by gastroenterologist. Unfortunately, data from our study are very limited regarding IBD disease location since such information was not routinely collected in all hospitals.
The mean duration of IBD disease was 10.0 and 6.0 years in patients with moderate/severe and mild/non-active CD, respectively; and 7.6 and 9.4 years in patients with moderate/severe and mild/non-active UC, respectively. Our results correspond to the results published in the Brazilian population where it was found to be a median of 10 years. 19 In an Argentinian population, the median duration of IBD following diagnosis was 6 years. 33 The effects of disease duration on the risk of symptoms and comorbidities is a topic of ongoing research.34,35 Interestingly, Bernstein and colleagues assessed the impact of disease duration on the cost of managing IBD and found that CD was significantly more costly to treat than UC, and for both diseases treatment costs were greatest within the first year of diagnosis. 36 Cost of treatment is an important consideration in Mexico and an interesting area for future research in our patient population.
In our study, the median age of diagnosis was 39 and 41 years for mild/non-active and severe/moderate Crohn’s disease, respectively; and 34 and 36 years for mild/non-active and severe/moderate ulcerative colitis, respectively. This corresponds with data reported from Western Europe, North America, and Oceania where the average age of onset in adult incident cases was a median of 31–34 years, whereas in Asia it was 34 and 42 years for CD and UC, respectively. 37
Previous immunosuppressant treatment appeared to be a protective factor in patients with UC but was not so effective in CD. There was no statistical significance for the rest of the variables, however, it is important to note that the analysis has wide confidence intervals, indicating that these results may not be reliable due to the small sample size. Caution should be exercised when interpreting these findings. Yamamoto-Furusho et al. evaluated data from more than 2600 patients with IBD in Mexico over a 15-year period and found that smoking habits were a protective factor in adults with UC, while in CD patients, a family history of autoimmune disease was found to be a risk factor; however, patients with UC should nonetheless be dissuaded from smoking. These authors demonstrated a significant increase in the incidence and prevalence of IBD in Mexico over a 15-year period, predominantly for CD over UC. The clinical and sociodemographical characteristics of Mexican patients appeared to be like other geographical regions 12 and UC was diagnosed four times more frequently than CD in this population. However, the trend over time was for the incidence of UC to stabilize while the incidence of CD was increasing, and they forecast that the difference between UC and CD would decrease over time. Our data support this suggestion, and we found that the prevalence ratio in our cohort was 2.4 for UC versus CD (70.9% vs 29.1%).
Prednisone was the most common corticosteroid used to treat IBD; in 43.2% and 41.6% of CD and UC patients, respectively. Mesalazine was the most common 5-ASA administered, 47.4% in CD and 76.2% in UC. In terms of biological agents, anti-TNF therapy (infliximab, adalimumab, certolizumab pegol, and golimumab) was more frequently used than non-anti-TNF therapy (vedolizumab, ustekinumab). Infliximab was administered to 23.2% of CD patients and 23.8% of UC patients; adalimumab was administered to 45.3% (CD) and 13.0% (UC); certolizumab pegol was only administered to 5.3% of the CD group and golimumab to 2.2% of the UC group. By contrast, vedolizumab was administered to 2.1% of CD patients and 5.6% of UC patients, while ustekinumab was administered to 22.1% (CD) and 1.3% (UC). These data need to be interpreted cautiously since they are largely influenced by the dates of approval and availability within the Mexican healthcare system (vedolizumab and ustekinumab received market approval in 2018 while infliximab, adalimumab, certolizumab pegol, and golimumab have been available since 2000, 2003, 2012, 2014, respectively). 38 Most of these drugs are used to treat both CD and UC; however, certolizumab is only used to treat CD and golimumab is indicated for patients with UC. The most common drug regimens used in CD patients included 5-ASA + immunosuppressants + biologic therapy (38.9%), and in the UC group, it was 5-ASA + immunosuppressants (64.5%). Yamamoto et al. conducted a chart review in 10 countries from Asia-Pacific, Latin America, Russia, and the Middle East regions involving patients diagnosed with IBD and who initiated anti-TNF therapy between 2010 and 2015. Suboptimal responses to anti-TNF therapies were common in these newly industrialized countries, emphasizing the importance of earlier recognition of treatment failures to allow timely alternative treatment decisions. 39 Our results confirmed that there is a high utilization of anti-TNF therapies in Mexico; it is important to ascertain the use of newer biological therapies such as vedolizumab and ustekinumab with different mechanisms of action. Both have achieved market approval by COFEPRIS in Mexico. As might be expected, given the relatively recent approval of these agents (2018 for both therapies), our results show that only small proportions of patients with IBD are treated with vedolizumab and ustekinumab in Mexico. 38 This contrasts with the higher level of usage of these therapies in the United States, 40 our results showed that biologic therapy in UC is lower than CD, probably due to the multifactorial reasons to get access to biologic therapies; and considering the level of corticoid use in this population, UC patients with severe–moderate disease are not getting benefits from biological therapy, this could be a future opportunity for Mexican physicians to address some of the unmet needs of patients with UC and CD.
In our study, we found a high prevalence of corticosteroid use. Almost one-third of the IBD patients were dependent on corticosteroids (n = 95; 29.1%). Corticosteroids are an important tool to induce remission, but they have no role in the continued management of patients to maintain remission, as there is clear evidence, that they lack efficacy in this respect. 41 The significant adverse side effect profile of these drugs means their longer-term use should be avoided whenever possible, or measures taken to avoid corticosteroid excess or abuse. In the USA, Raine et al. analyzed the trend in corticosteroid usage, and they concluded that despite advances in IBD treatment over the past 10 years, the use of corticosteroids had not decreased. The excessive use of corticosteroids was higher in the first year after treatment and slightly decreased in the second year. 42 In a Brazilian population included in the RISE-BR study, corticosteroid use was 47.5% in CD patients and 58.3% in UC patients over a 12-month period. The proportion of patients receiving corticosteroids tended to decrease over time. 19 Clinical recommendations in Mexico allow the use of corticosteroids in all IBD patients to induce clinical remission, but they are not recommended for maintenance therapy. 17 A previous study in Mexico reported using prednisone as induction therapy in 21% of patients with mild CD and 23% with moderate/severe CD, and budesonide was used in 27% of CD patients. In the case of UC, systemic corticosteroids were used to induce remission in 76% of patients with severe disease. 11 In Mexico, there are no reports about the proportion of patients who have dependence or intolerance to corticosteroids and this probably highlights the need to define the correct use of corticosteroids in these populations. In our study, we observed that nearly one-third of patients continued to use corticosteroids long term, and this could be a signal of low-quality clinical management and follow-up of some patients with respect to treatment outcomes. We believe that the origin of this practice might relate to multiple factors such as a lack of experience and limited access to biological agents, and this needs to be investigated further based on the fact that these more recently approved treatments are used routinely in many countries around the world.
In this study, we do not know the proportion of patients who obtained their medications through public or private healthcare plans; however, all participating centers were from private practice. It is important to note that in Latin America, there is heterogeneity between the public and private healthcare sectors, and the literature continues to emphasize the role of the fragmentation of the health system in relation to inequalities in access to and quality of treatment. 43 This heterogeneity might enable some patients to have access through major medical insurance plans while others must seek mechanisms through public health insurance or through other institutions that have treatment restrictions related to the same health system. The list of the results of the previous treatments collected in the retrospective part of the study reflects that the drugs were accessed according to the National Authority that was providing approval in the market and that the doctors have been assigning the treatment to the patients according to the available therapies. There appears to be no standardization for access to high-cost therapies in Mexico and the private sector and the Secretariat of Health may cover some cases with approved agents, whereas the Social Security system only covers selected anti-TNF drugs for IBD. 44
For the medical treatment of UC, the First Latin American Consensus of PANCCO recommends a step-up approach. 30 Mild/moderate cases are initially managed with 5-ASA and corticosteroids. The concomitant use of azathioprine works as a corticosteroid sparing agent. In moderate/severe UC, biologic therapies are recommended to induce and maintain remission in patients who do not respond to conventional treatments irrespective of whether the patient is dependent or refractory to corticosteroids. On the other hand, for CD, the treatment strategy is planned according to phenotype and risk factors. Mild/moderate cases are initially managed with corticosteroids as induction therapy. In patients with moderate/severe CD who do not respond to initial treatment, immunomodulators such as azathioprine and methotrexate are considered. Biologic therapies play an important role in inducing and maintaining remission in refractory or severe CD. In both UC and CD, treatment decisions are individualized based on disease severity, patient characteristics, and response to previous therapies. Regular monitoring and close follow-up are essential to assess treatment efficacy and adjust the therapeutic approach as needed, to achieve and maintain remission, improve the quality of life, and prevent disease complications and long-term adverse effects. The AGA guide for medical management in CD suggests the early use of biologic therapy with or without an immunomodulator rather than delaying their use until after the failure of 5-ASA. 45 Indeed, systematic reviews showed no convincing benefits of 5-ASA over placebo either in the induction of remission or in the maintenance of medically induced remission in CD. 46 In Mexico, clinical recommendations allow the use of 5-ASA for the induction and maintenance of clinical remission just for mild UC patients, 29 and recently for mild CD patients. 47 Our results showed that 5-ASA has been used in a great part of CD patients in Mexico, highlighting that treatment could be improved in this population as guidelines suggest. These statements are important to consider when making decisions relating to the clinical management of patients with CD to help optimize treatment outcomes and decrease disease burden.
Limitations
This study collected real-world information from patients treated in everyday clinical practice which has the benefit of reflecting the influence of factors that are associated with day-to-day living (e.g., diet, concomitant medications) which are very important for diseases such as IBD. However, there are a number of limitations that need to be acknowledged. First, the main limitation of the study is its observational, uncontrolled design which is a prerequisite of such real-world studies. Another limitation is that the number of patients enrolled is relatively small, particularly in some groups, and this restricts the statistical robustness of the study (including wide ranges for CIs in some analyses) and this limits any conclusions that can be drawn from these findings. Furthermore, the study was retrospective in nature, and valuable information could be missing in the patient’s medical records, this was notably the case for information relating to disease location and clinical characteristics which were not routinely determined in some hospitals. The cross-sectional nature makes it challenging to interpret associations between IBD index information, and clinical and sociodemographic data. Also, it is important to recap that all centers that participated in this study were private clinics for the management of the disease in the major cities in Mexico and just a few publicly treated patients were enrolled (number not provided), as such, the information captured may not be generalizable to all patients in Mexico, especially those treated in public hospitals.
Conclusions
In this study, more patients presented with a diagnosis of UC compared with a diagnosis of CD. A relatively large proportion of patients with IBD presented with moderate/severe disease, particularly in the CD group (45.3%). Conventional therapy represented the most frequently used treatment strategy in Mexican patients with moderate/severe IBD, with high rates of corticosteroid dependence reported, particularly in patients with UC. In patients with CD, 5-ASA agents were used in Mexico (in almost 50% of cases) despite not being recommended by treatment guidelines indicating that treatment care could be improved. Indeed, the availability of more effective biologic treatments was identified as an area that needs to be reviewed to help improve healthcare delivery and address some of the unmet needs of Mexican patients with UC and CD. This could reduce the excessive prescription of corticosteroids and promote the use of biological therapies and small molecules in IBD to maintain long-term remission. Overall, these findings underscore the need to provide a more personalized approach to disease management to achieve optimal disease control and enhance the quality of life of patients with IBD in Mexico.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251318857 – Supplemental material for RISE-MX, a real-world study of patients with moderate/severe inflammatory bowel disease returning for hospital follow-up in Mexico: baseline demographics and clinical characteristics, treatment and disease status
Supplemental material, sj-docx-1-tag-10.1177_17562848251318857 for RISE-MX, a real-world study of patients with moderate/severe inflammatory bowel disease returning for hospital follow-up in Mexico: baseline demographics and clinical characteristics, treatment and disease status by Manuel Alejandro Martínez-Vázquez, Francisco J. Bosques-Padilla, Rosa María Miranda-Cordero and Jesus K. Yamamoto-Furusho in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251318857 – Supplemental material for RISE-MX, a real-world study of patients with moderate/severe inflammatory bowel disease returning for hospital follow-up in Mexico: baseline demographics and clinical characteristics, treatment and disease status
Supplemental material, sj-docx-2-tag-10.1177_17562848251318857 for RISE-MX, a real-world study of patients with moderate/severe inflammatory bowel disease returning for hospital follow-up in Mexico: baseline demographics and clinical characteristics, treatment and disease status by Manuel Alejandro Martínez-Vázquez, Francisco J. Bosques-Padilla, Rosa María Miranda-Cordero and Jesus K. Yamamoto-Furusho in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Editorial assistance, under the authors’ guidance, was provided by Content Ed Net (Madrid, Spain). Reprints Unlimited Mexico has performed editorial management and was funded by Takeda Mexico, S.A. de C.V.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
