Abstract
Background:
Potassium-competitive acid blocker (P-CAB)-based therapies are emerging as promising alternatives for eradicating Helicobacter pylori infection. However, the comparative efficacy of P-CAB-based therapy versus proton-pump inhibitor (PPI)-based therapy in treating H. pylori infection remains uncertain.
Objectives:
This meta-analysis evaluated the efficacy and safety of P-CAB-based therapies, including Vonoprazan (VPZ) and Tegoprazan (TPZ), compared to PPI-based therapies for H. pylori infection. Subgroup analysis assessed the influence of drug history, experimental drug, treatment duration, combination therapies, and geographic regions on treatment outcomes.
Design:
Meta-analysis.
Data sources and methods:
Comprehensive searches were conducted in major databases, including PubMed, Embase, the Cochrane Library, and Web of Science, up to January 1, 2024. The primary outcome was the eradication rate, analyzed by intention-to-treat (ITT). Secondary outcomes included adverse events. Heterogeneity among studies was assessed using the χ2 test and the I2 test. I2 > 50% or p < 0.05 indicated significant heterogeneity.
Results:
The analysis totally included 28 randomized controlled trials (RCTs) comprising 37 studies and 8818 patients diagnosed with H. pylori infection. Of these, 14 RCTs, including 20 studies and 4286 patients, compared P-CAB-based therapy with 14-day bismuth-based quadruple therapy (BQT). P-CAB-based therapy exhibited superior eradication rates compared to both 14-day BQT and PPI-based therapy (ITT analysis: 87.0% vs 79.8%, risk ratio (RR) = 1.08, 95% CI: 1.04–1.12, p < 0.0001; and 85.6% vs 77.8%, RR = 1.09, 95% CI: 1.05–1.12, p < 0.00001, respectively). This enhanced efficacy was particularly pronounced in patients with clarithromycin-resistant infections (73.7% vs 41.5%, RR = 1.53, 95% CI: 1.07–2.20, p = 0.02). Subgroup analysis demonstrated higher eradication rates with P-CAB-based therapy in treatment-naïve participants, VPZ recipients, and those receiving 7- or 14-day regimens (dual, triple, or quadruple therapy). However, no significant differences were observed in treatment-experienced subgroups, TPZ recipients, or those on 10-day regimens. In addition, P-CAB-based therapy showed a lower incidence of adverse events than PPI-based treatments (RR = 0.73, 95% CI: 0.63–0.86, p < 0.0001).
Conclusion:
P-CAB-based therapies are more effective than traditional PPI-based treatments for eradicating H. pylori infection, with a reduced incidence of adverse events.
PROSPERO registration:
CRD42024503665.
Introduction
The prevalence of Helicobacter pylori (H. pylori) infection represents a significant public health challenge, affecting approximately half of the global population. 1 This bacterium is associated with various gastrointestinal disorders, including chronic gastritis, peptic ulcer disease, gastric mucosa-associated lymphoid tissue lymphoma, and gastric cancer. 2 Recognizing its deleterious impact, the World Health Organization (WHO) classified H. pylori as a Group 1 carcinogen in 1994. 3 H. pylori infection stands out as a notable, modifiable risk factor for gastric cancer. Eradication of this pathogen can mitigate gastric inflammation, promote mucosal healing, resolve peptic ulcers, and reduce the risk of gastric cancer.4–6 Consequently, H. pylori eradication has garnered widespread support from numerous international guidelines and consensus forums,5–8 barring specific contraindications.
Currently, the most commonly used treatment options for H. pylori infection consist of two regimens. The first is bismuth-based quadruple therapy (BQT),5,6,9–11 which incorporates a proton-pump inhibitor (PPI), bismuth, and two antibiotics administered over a 10- to 14-day period. The second approach involves a triple therapy regimen combining a PPI with clarithromycin and either amoxicillin or metronidazole, typically administered for 14 days.5,9,11–13 Notably, Japan diverges from this approach, advocating a 7-day triple therapy regimen as the first-line treatment. 14 However, the efficacy of PPI-based regimens has gradually diminished owing to widespread antibiotic resistance 15 and insufficient acid suppression. 16 Rokkas et al. highlighted that many PPI-centered strategies fail to achieve the desired 90% eradication benchmark. 17 The short elimination half-lives of PPIs, their inadequate acid suppression, and pharmacokinetic variations across ethnicities may compromise their global efficacy in H. pylori management. 18 These challenges have catalyzed the pursuit of more effective and innovative solutions for addressing H. pylori infections. According to the recently published ACG guideline, 10 for treatment-naïve patients with H. pylori infection and unknown antibiotic susceptibility profiles, 14-day BQT with optimized dosing is recommended as the preferred first-line treatment. Alternative empiric therapies include 14-day rifabutin triple therapy or potassium-competitive acid blocker (P-CAB) dual therapy. In addition, in patients with no history of macrolide exposure or penicillin allergy and unknown antibiotic susceptibility, 14-day P-CAB-clarithromycin triple therapy is preferred over PPI-clarithromycin triple therapy when other first-line treatment options are not feasible. 10
P-CABs have emerged as promising alternatives to traditional PPIs. These novel acid-suppressive agents function by competitively inhibiting the binding of potassium ions to H+/K+-ATPase in gastric cells. 19 P-CABs have demonstrated superior and prolonged gastric acid suppression compared to PPIs, with the added advantage of being unaffected by cytochrome P450 2C19 (CYP2C19) genotype. 20 By enhancing acid suppression and antimicrobial activity, P-CABs could potentially augment H. pylori treatment efficacy. 21 The principal P-CABs currently employed in H. pylori eradication therapy are Vonoprazan (VPZ) and Tegoprazan (TPZ). VPZ was first introduced in Japan 22 in 2014 for H. pylori infection treatment and has subsequently been approved in other Asian countries. Recently, it received FDA approval and is now available in North, Central, and South America. 23 TPZ was initially launched in the Korean market in 2019 and has since been introduced in other Asian and Central/South American Countries.24,25 The Maastricht VI/Florence consensus report advocates for VPZ in combination with antibiotics as first- and second-line treatment, particularly for patients with antibiotic-resistant strains. 5 Studies conducted by Kim et al. 21 and Huang and Lin 26 suggested that TPZ-based and VPZ-based therapies achieve higher eradication rates with similar adverse events profiles than traditional PPI-based quadruple therapies.
In 2023, numerous randomized controlled trials (RCTs) comparing VPZ/TPZ-based therapies with PPI-based therapy for H. pylori eradication have been published, yielding inconsistent and controversial results. Existing meta-analyses indicate the superior efficacy of VPZ-based eradication regimens over conventional PPI-based ones.27,28 However, these analyses are limited either by the number of trials included or by focusing only specific treatments on VPZ-based treatments, thus lacking comprehensiveness. To date, no meta-analyses incorporating studies about TPZ for H. pylori infection have been found. In response to this gap, we conducted a systematic review and meta-analysis of RCTs examining the efficacy and safety of P-CAB-based therapies (including both VPZ and TPZ) compared to PPI-based therapy in individuals infected with H. pylori. In addition, we performed subgroup analyses to elucidate the impact of various factors on treatment outcomes, including drug history, experimental drug used, duration of treatment, combination treatment regimens, and country of study.
Methods
This review was registered on the PROSPERO platform (https://www.crd.york.ac.uk/PROSPERO/, registration number: CRD42024503665) and conducted in accordance with the latest PRISMA guidelines. 29 Detailed information is presented in Supplemental Material 1.
Data sources and literature search strategy
A comprehensive literature search was performed across multiple electronic databases, including PubMed, Embase, the Cochrane Library, and Web of Science, with the search timeline extending up to January 1, 2024, and without any restrictions on language. The search strategy incorporated both MeSH terms and keywords: “Helicobacter pylori,” “Helicobacter nemestrinae,” “Campylobacter pyloridis,” “Campylobacter pylori,” “Campylobacter pylori subsp. Pylori,” “1-(5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine,” “TAK 438,” “TAK438,” “TAK-438,” “Vonoprazan,” and “Tegoprazan.” To ensure comprehensive coverage, an additional manual search was conducted by reviewing the bibliographies of pertinent reviews and selected studies to unearth further applicable research. An elaborate description of the search methodologies employed for each database is documented in Table S1 (Supplemental Material 2), accessible online.
Study selection
The selection of studies was undertaken independently by researchers T.J. and Li Zhong. After removing duplicate entries, the researchers conducted a preliminary evaluation of the study abstracts and titles to ascertain their relevance to the research focus; non-relevant studies were discarded. A thorough examination of the full texts was then carried out, guided by pre-determined inclusion criteria.
Following the PICOS framework, studies were included if they met the following criteria:
P (participants): Adults identified with H. pylori infection.
I (intervention): Therapies based on VPZ or TPZ.
C (comparator): Therapies utilizing PPIs.
O (outcomes): (a) Primary outcome: H. pylori eradication rate; (b) Secondary outcomes: Incidence of adverse effects.
S (study design): RCTs.
Exclusion criteria were as follows: (i) studies in the forms of meta-analyses, reviews, letters, commentary, trial protocols, conference abstracts, or case reports; (ii) studies with incomplete or inaccessible data; (iii) studies lacking available full text; and (iv) studies not published in English.
Data extraction
Data extraction was independently performed by two researchers, T.J. and Li Zhong. Any discrepancies encountered during the process were resolved by consensus, with W.W. acting as the mediator. For each selected study, the extracted data included the first author’s name, publication year, country, study design, participant demographics, diagnostic tests employed for H. pylori infection and its eradication, total sample size, therapeutic approaches, treatment duration, timing of post-treatment confirmation tests, eradication rates based on intention-to-treat (ITT) analysis, and commonly reported adverse events.
Study quality assessment
To evaluate the reliability and risk of bias in each study, the Cochrane Risk of Bias Assessment tool was utilized. 30 This appraisal was independently conducted by T.J. and Li Zhong, with any disagreements resolved through discussion. The assessment framework covered several aspects: random sequence generation, allocation concealment, blinding of participants and staff, blinding in the evaluation of outcomes, completeness of outcome data, risk of selective outcome reporting, and other potential biases. Based on these criteria, studies were classified as “low,” “high,” or “uncertain” risk levels.
Outcomes and statistical analysis
The primary outcome of the study was the eradication rate by ITT analysis. The secondary outcomes were adverse events. First, we screened studies that compared the P-CAB-based regimens with the 14-day BQT and performed a comparative analysis. Second, to enhance the clinical applicability of our findings, we further conducted comparative analyses between P-CAB-based regimens and all PPI-based regimens.
Statistical analyses were conducted using Stata 15.0 version (Stata Corp, College Station, Texas, USA) and Review Manager 5.4 version (Cochrane Collaboration, Oxford, UK), with p values less than 0.05 considered statistically significant. The comparative efficacy between P-CAB-based and PPI-based treatments was evaluated through risk ratios (RRs) alongside their 95% CI. Heterogeneity among studies was assessed using the χ2 test (with p < 0.05 denoting significant heterogeneity) and the I2 test (I2 > 50% reflecting significant heterogeneity). For outcomes demonstrating low heterogeneity, a fixed-effect model was employed, whereas a random-effect model was utilized for outcomes with substantial heterogeneity. Sensitivity analyses were performed by sequentially omitting individual studies to evaluate result reliability. The potential for publication bias was explored through the visual inspection of funnel plots and Egger’s test.
Subgroup analysis
Comprehensive subgroup analyses were performed to identify potential contributing factors and sources of heterogeneity. These analyses considered variables such as prior treatment exposure (treatment-naïve or treatment-experienced), experimental drug (VPZ or TPZ), treatment duration (7, 10, or 14 days), therapeutic regimen (dual, triple, or quadruple therapy), and geographical distribution of studies (China, Japan, Korea, Pakistan, Singapore, Thailand, United States, and Europe).
Results
Study selection and their characteristics
As depicted in Figure 1, our initial search identified 1489 records across various databases. Following the removal of duplicates, 883 records remained. A thorough review of titles and abstracts reduced this number to 38, from which 28 studies19,21,26,31–55 were included in our comprehensive analysis following full-text assessments.

Flowchart of the study selection process.
These studies are cataloged in Table 1, detailing the study characteristics and therapeutic approaches employed. Notably, nine articles19,26,34,35,37,43,44,49,54 reported on two studies with independently controlled designs comparing P-CAB-based therapy with PPI-based therapy. Owing to their rigorous randomization, these were treated as separate studies for assessing efficacy and potential bias. Consequently, this meta-analysis included 28 RCTs encompassing 37 studies and involved a total of 8818 H. pylori-infected patients, with 4417 assigned to the P-CAB group and 4401 to the PPI group. Among the included RCTs, 13 were conducted in China, 5 in Japan, 4 in Korea, 2 in Pakistan, 1 in the United States and Europe, 1 in Singapore, 1 in Thailand, and 1 spanned multiple regions (China, South Korea, Taiwan, and the Philippines). In addition, 14 RCTs19,21,26,34,40–44,46,48,52–54 compared P-CAB-based regimens with 14-day BQT, comprising 20 studies and 4286 patients; of these RCTs, 11 were conducted in China, 2 in Korea, and 1 was a multicenter study spanning East Asian regions (China, South Korea, Taiwan, and the Philippines).
Main characteristics of included studies in the meta-analysis.

AB, anti-Helicobacter pylori IgG antibody testing; Amo, amoxicillin; bid, twice daily; C, culture; Cla, clarithromycin; Dox, doxycycline; Eso, esomeprazole; Fur, furazolidone; Hp, Helicobacter pylori; Lan, lansoprazole; MNZ, metronidazole; Ome, omeprazole; P-CAB, potassium-competitive acid blocker; Pan, pantoprazole; PPI, proton-pump inhibitor; qd, once daily; qid, four times daily; Rab, rabeprazole; RCT, randomized controlled trial; RUT, rapid urea test; SAT, stool antigen test; Sit, sitafloxacin; Tet, tetracycline; tid, three times daily; TPZ, Tegoprazan; UBT, urea breath test; VPZ, Vonoprazan.
Study quality assessment
The quality assessment of the included RCTs is summarized in Figure 2. Reassuringly, the evaluation indicated that risks related to incomplete outcome data, selective reporting, and other potential biases were generally low.

Quality assessment of the RCT studies: (a) risk of bias graph and (b) risk of bias summary.
Comparative analysis with 14-day BQT
Eradication rates
A total of 20 studies19,21,26,34,40–44,46,48,52–54 with 4286 patients compared P-CAB-based regimens with 14-day BQT for H. pylori eradication. The ITT analysis revealed superior eradication rates in the P-CAB group compared to the 14-day BQT group (87.0% vs 79.8%; RR = 1.08, 95% CI: 1.04–1.12, p < 0.0001), despite moderate heterogeneity (p = 0.003, I2 = 53%), as depicted in Figure 3(a). Sensitivity analysis confirmed the robustness of these findings (Figure 4(a)). The funnel plot analysis (Figure S1(a); Supplemental Material 2) and Egger’s test (p = 0.377) indicated no significant publication bias.
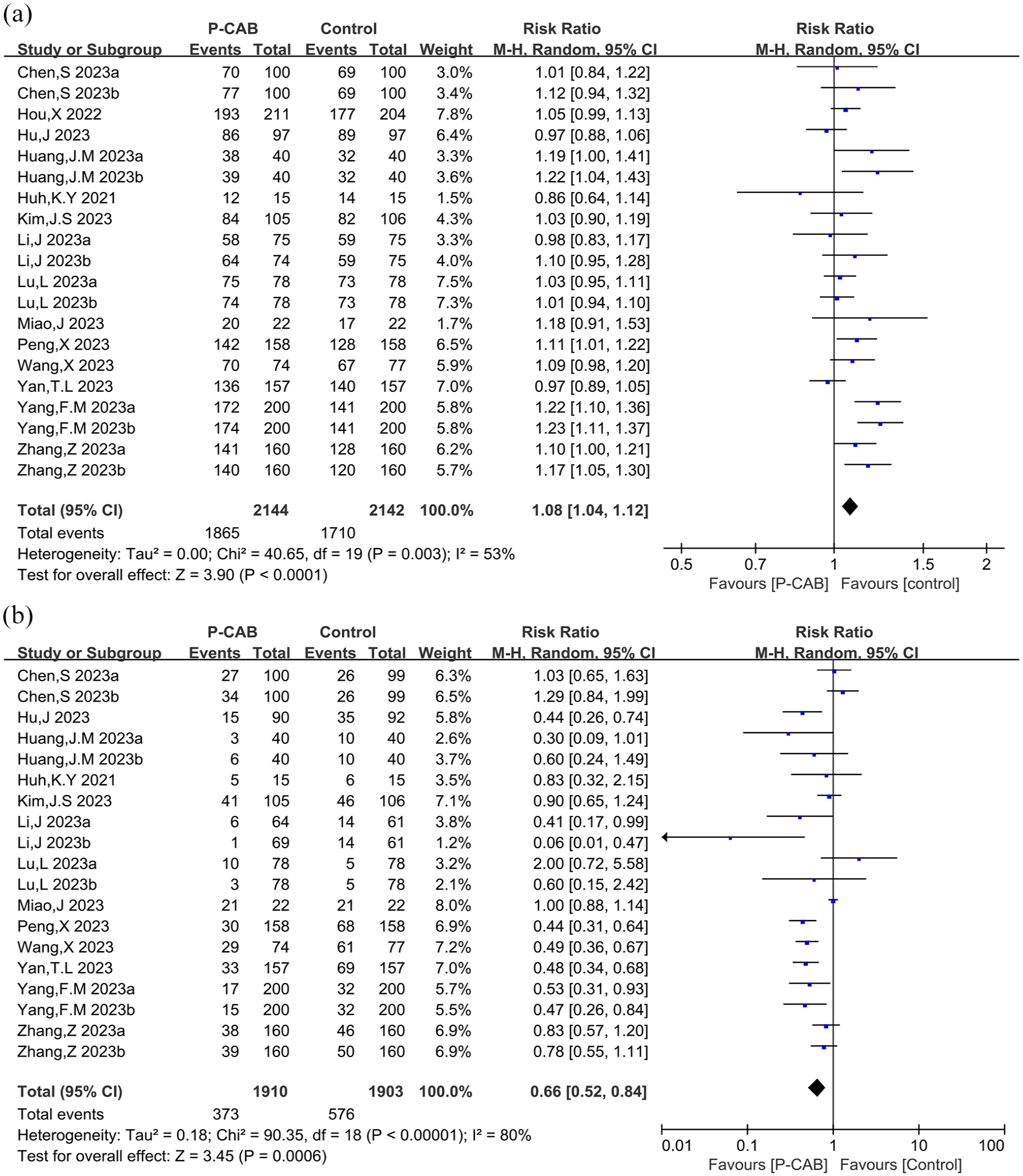
Forest plot of Helicobacter pylori eradication rate (a) and adverse events (b) in comparison with 14 days BQT.

Sensitivity analysis of Helicobacter pylori eradication rate (a) and adverse events (b) in comparison with 14 days BQT.
Adverse events
The safety analysis included 19 studies19,21,26,34,41–44,46,48,52–54 involving 3813 patients. The P-CAB-based treatment demonstrated a significantly lower incidence of adverse events compared to the 14-day BQT group (RR = 0.66, 95% CI: 0.52–0.84, p = 0.0006), despite substantial heterogeneity (I2 = 80%, p < 0.00001), as illustrated in Figure 3(b). Sensitivity analysis confirmed the stability of these results (Figure 4(b)). The funnel plot is presented in Figure S1(b) (Supplemental Material 2), and Egger’s test suggested potential publication bias (p = 0.029).
Comparative analysis with all PPI-based regimens
Eradication rates
A comprehensive analysis of studies,19,21,26,31–55 encompassing a total of 8818 patients, revealed H. pylori eradication outcomes. Pooled data demonstrated eradication rates of 85.6% in the P-CAB treatment group compared to 77.8% in the PPI-based treatment group, according to ITT analysis. A notable advancement in eradication efficacy was observed in the P-CAB cohort (RR = 1.09, 95% CI: 1.05–1.12, p < 0.00001), despite significant study heterogeneity (p < 0.0001, I2 = 57%), as depicted in Figure 5(a). The symmetry detected in the associated funnel plot (Figure S2(a); Supplemental Material 2) and Egger’s test (p = 0.403) indicates no significant publication bias. Of these, 10 RCTs,26,33,40,44–47,49,52,55 comprising 12 studies and involving 2418 patients, reported eradication rate exceeding 90% for P-CAB regimens.

Forest plot for Helicobacter pylori eradication rate in total population (a), clarithromycin-resistant population (b), and comparison with high-potency PPI therapies (c).
Furthermore, six studies31,35,36,47,52 focusing on clarithromycin-resistant infections underscored the superior efficacy of P-CAB treatments. These treatments achieved eradication rates of 73.7%, markedly surpassing the 41.5% efficacy rate in PPI-based interventions (RR = 1.53, 95% CI: 1.07–2.20, p = 0.02). This divergence was accompanied by substantial heterogeneity (p < 0.0001, I2 = 84%; Figure 5(b)). However, the funnel plot (Figure S2(b); Supplemental Material 2) and Egger’s test (p = 0.915) confirmed the absence of significant publication bias among these studies.
In addition, a subanalysis comparing P-CAB with high-potency PPI therapies (rapeprazole or esomeprazole) was conducted, including seven studies.26,34,38,39,44,46 In this subanalysis, the types and dosages of antibiotics or bismuth used in combination were identical in both groups, differing only in the acid inhibitor, while the treatment duration for P-CAB group was less than or equal to that of the PPI group. Results revealed a higher eradication rate with P-CAB-based therapy compared to high-potency PPI-based therapy (88.9% vs 84.0%, RR = 1.06, 95% CI: 1.02–1.10, p = 0.007), with no significant study heterogeneity (p = 0.33, I2 = 14%), as depicted in Figure 5(c). The funnel plot (Figure S2(c); Supplemental Material 2) and Egger’s test (p = 0.433) reassured the absence of significant publication bias.
Adverse events
An assessment spanning 30 studies,19,21,26,34–39,41–49,52–54 involving 7605 patients, was conducted to evaluate the frequency of adverse events across groups. Results significantly favored P-CAB-based therapies, which exhibited a reduced incidence of overall adverse events compared to PPI-based treatments (RR = 0.73, 95% CI: 0.63–0.86, p < 0.0001). This analysis noted significant heterogeneity (I2 = 71%, p < 0.00001), as illustrated in Figure 6(a).

Forest plot for total adverse events (a), nausea and/or vomiting (b), dysgeusia/bitter taste (c), and diarrhea (d).
All studies19,21,26,34–39,41–49,52–54 listed detailed adverse reactions, and we carried out statistical analysis of specific adverse events. Our meta-analysis underscored a lower occurrence of nausea/vomiting (RR = 0.7, 95% CI: 0.58–0.84, p = 0.0002), dysgeusia or bitter taste (RR = 0.44, 95% CI: 0.27–0.73, p = 0.001), and diarrhea (RR = 0.77, 95% CI: 0.66–0.90, p = 0.0009) in the P-CAB group relative to the PPI group (Figure 6(b)–(d)). Conversely, for adverse symptoms like abdominal distension, abdominal pain, constipation, skin rash, dyspepsia, dizziness, and headaches, no significant disparities were discerned between the two cohorts (Figure 7). Notably, dysgeusia/bitter taste presented considerable heterogeneity (p < 0.00001, I2 = 85%). The synthesized funnel plots for overall and specific adverse occurrences are visualized in Figures S3 and S4 (Supplemental Material 2), respectively. Application of Egger’s test revealed potential publication bias concerning the totality of adverse events, specifically in the case of dysgeusia/bitter taste (Egger’s test p = 0.016 and p = 0.006, respectively).
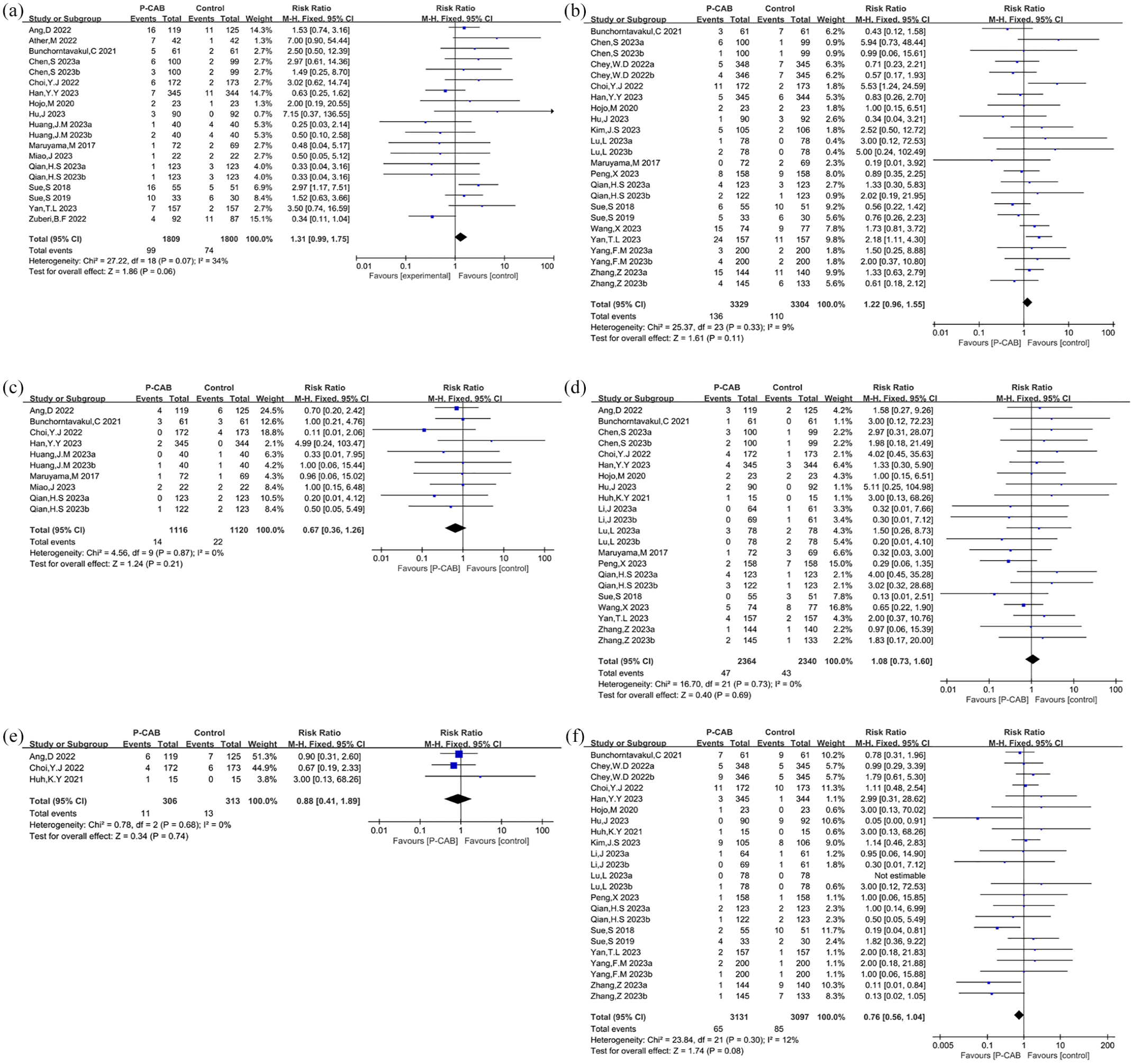
Forest plot for abdominal distension (a), abdominal pain (b), constipation (c), skin rash (d), dyspepsia (e), and dizziness and headaches (f).
Sensitivity analysis
In the sensitivity analysis, data were dissected to investigate the influence of individual studies on the pooled RR. The leave-one-out test, which systematically excludes one study at a time, confirmed the stability and reliability of our findings regarding the H. pylori eradication rate, the total incidence of adverse events, and the specific adverse event of dysgeusia or bitter taste anomaly (Figure 8(a), (c), and (d)). However, variability emerged within the results concerning the eradication rate in the clarithromycin-resistant group, indicating some degree of instability in this subset of the analysis (Figure 8(b)). Publications by Chey et al. 35 and Murakami et al. 47 contributed to the observed instability in the study outcomes.

Sensitivity analysis for Helicobacter pylori eradication rate in total population (a), CLA-resistant population (b), total adverse events (c), and dysgeusia/bitter taste (d).
Subgroup analysis
To achieve a deeper understanding of our findings, subgroup analyses were conducted based on various criteria, including patients’ drug history (treatment-naïve or treatment-experienced), experimental drug (VPZ or TPZ), treatment duration (7, 10, or 14 days), P-CAB-based combination treatment regimen (dual, triple, or quadruple therapy), and the geographical location of the studies. Subgroup analysis indicated no significant difference between P-CAB-based and PPI-based therapy in treatment-experienced groups, TPZ, 10-day treatments, and participants from Korea, Singapore, and Thailand. In addition, subgroup analyses for adverse events were performed. These detailed results are presented in Table 2.
Results of subgroup analyses of eradication rates and adverse effects.

P-CAB, potassium-competitive acid blocker; RR, risk ratio; TPZ, Tegoprazan; VPZ, Vonoprazan.
Discussion
In the current context of increasing antibiotic resistance and diminishing success rates of H. pylori eradication worldwide, effective and sustained acid suppression is increasingly seen as a pivotal strategy for enhancing antibiotic efficacy and improving H. pylori eradication rates. 56 The introduction of P-CABs offers a novel and effective method of acid suppression, with VPZ leading the class due to its prompt, potent, and long-lasting effects. TPZ is another promising P-CAB currently under development. In Japan, the use of VPZ in H. pylori treatment protocols surged from 45.6% in 2015 to 95.5% in 2018, resulting in improved eradication rates. 57 Currently, VPZ is recommended in multiple international guidelines as a first-line or second-line treatment of H. pylori.5,6,10,14,58 A review 59 emphasized the efficacy of VPZ with eradication rates approaching or exceeding 90%, which is a benchmark for satisfactory H. pylori treatment outcomes. 60
Current international guidelines and consensus statements consistently recommend BQT as the preferred first-line treatment for H. pylori infection.5,6,9–11 Recent meta-analyses have provided compelling evidence supporting alternative treatment approaches. Zhou et al. 61 demonstrated that VPZ-amoxicillin dual therapy achieves comparable eradication rates to BQT while exhibiting a superior safety profile and equivalent compliance. Furthermore, a network meta-analysis by Ouyang et al. 62 revealed that VPZ-based regimens, including high-dose dual therapy, triple therapy, and quadruple therapy, achieved significantly higher eradication rates compared to traditional PPI-based combinations in ITT analyses. Our meta-analysis, incorporating 20 studies comparing P-CAB-based regimens with 14-day BQT, corroborates these findings. The results demonstrated significantly higher eradication rates with P-CAB-based regimens (ITT analysis: 87.0% vs 79.8%, RR = 1.08, 95% CI: 1.04–1.12, p < 0.0001) and lower adverse event rates (19.5% vs 30.3%, RR = 0.66, 95% CI: 0.52–0.84, p = 0.0006). The recently published ACG Guidelines 10 advocate for optimized BQT as the first-line treatment in treatment-naïve patients, specifically recommending bismuth (300 mg four times daily), metronidazole (1.5–2 g daily in 3–4 doses), tetracycline (500 mg four times daily), and standard-dose PPI (twice daily) for 10–14 days, preferably the latter. However, further research is warranted to definitively establish whether P-CAB-based regimens demonstrate superior efficacy compared to optimized BQT.
Our comprehensive meta-analysis, encompassing 28 RCTs with 37 studies and 8818 patients, presents a robust evaluation of VPZ/TPZ-based therapies compared to conventional PPI regimens for both H. pylori eradication efficacy and adverse events profiles. The findings underscore the statistical superiority of P-CABs over PPIs in eradicating H. pylori and highlight fewer associated adverse events—a pattern in line with prior studies. A meta-analysis by Sun et al. in 2023, which included 8 RCTs, reflected our findings, showing eradication rates of 83.5% for VPZ regimens compared to 72.6% for PPI regimens, 28 closely aligning with our results of 85.6% and 77.8%, respectively. Adverse events, often encountered during eradication therapy, have critical implications for patient adherence and treatment success. Our analysis found that P-CAB regimens are associated with a significantly lower incidence of total adverse events and specific adverse events, including nausea, vomiting, dysgeusia, and diarrhea, compared to PPI regimens. Given the variability across different populations, medications, treatment durations, combination regimens, and countries in the studies reviewed, we performed several subgroup analyses. These analyses confirmed the superiority of P-CAB therapy over PPIs, especially in treatment-naïve patients (n = 25), although such benefits were not observed in the treatment-experienced cohort (n = 2)—likely due to the limited number of studies and smaller sample sizes. While TPZ exhibits non-inferiority to PPI therapies, the underlying reasons for its lack of clear superiority warrant further investigation with larger cohorts. The advantages of P-CAB therapy were more pronounced in China, Japan, Pakistan, the United States, and Europe, but were less in Korea, Singapore, and Thailand, possibly due to the small number of sample sizes included.
Intriguingly, our subgroup analysis revealed that 7- and 14-day P-CAB-based therapies demonstrated significantly higher efficacy than PPI-based therapies, reflected by the pooled RR of 1.11 (95% CI: 1.02–1.21, p = 0.01) and 1.11 (95% CI: 1.08–1.14, p < 0.00001), respectively. Conversely, in the subgroup undergoing 10-day therapy, no statistical difference in efficacy was observed between the P-CAB and PPI groups (pooled RR = 1.04, 95% CI: 0.97–1.10, p = 0.25). This discrepancy prompted speculation regarding potential underlying reasons. Specifically, among the 10 studies on 7-day P-CAB-based therapy, 5 were conducted in Japan where all participants received triple therapy (VPZ plus two antibiotics). Japan predominantly employs a 7-day triple therapy regimen for H. pylori eradication, which includes a PPI or VPZ, amoxicillin, and clarithromycin as a first-line treatment. 14 By contrast, the six studies examining 10-day P-CAB-based therapy were all conducted in China, where most patients received dual therapy with VPZ plus amoxicillin (VA). While the 7-day VA dual therapy regimen has shown satisfactory outcomes in Japan, 63 it resulted in a notably lower eradication rate when used in China, falling below 80%. Remarkably, extending the therapy duration to 14 days in China enhanced its efficacy, achieving eradication rates exceeding 90%.41,48,52,64–66 This underscores the necessity to delve into the optimal treatment duration, dosage, and intervals for VA dual therapy, tailored to different geographical regions. Although a previous network meta-analysis identified VPZ triple therapy as the most effective among various initial treatments, with an eradication rate above 90%, 17 our analysis demonstrates that P-CAB-based dual, triple, and quadruple regimes exhibit similarly superior effectiveness.
Clarithromycin is a critical drug for the treatment of H. pylori worldwide; however, its primary resistance rate has significantly increased, ranging from 17.2% to 19.7% or even reaching 27.2%, 67 which has led to a decline in H. pylori eradication rates. 68 The classification of clarithromycin-resistant H. pylori as a high-priority pathogen by the WHO 69 underscores the global urgency for efficient eradication strategies. Current guidelines recommend that PPI-clarithromycin-based triple regimens should only be used in patients without prior macrolide use who live in regions where the prevalence of clarithromycin resistance is known to be less than 15%.5,9,11,12 In areas where the resistance rate is unknown or exceeds 15%, most guidelines recommend first-line treatment options such as BQT for 10–14 days, typically consisting of PPI, bismuth, and dual antibiotics.5,6,9,11–13 Alternatively, non-bismuth concomitant quadruple therapy for 10–14 days, comprising a PPI, amoxicillin, clarithromycin, and a nitroimidazole administered concurrently, is recommended if bismuth is unavailable.5,11–13 Furthermore, in clinical settings where bismuth, tetracycline, or Pylera® (a single-capsule formulation containing bismuth subcitrate potassium, metronidazole, and tetracycline) is not readily available, rifabutin-based triple therapy or high-dose dual therapy represents viable empirical alternatives to BQT.5,10 Notably, P-CAB-based regimens may provide an effective alternative to PPI-based therapy. Many guidelines now recommend P-CAB-antimicrobial combination treatments for first-line and second-line treatment, especially for patients with evidence of antimicrobial-resistant infections.5,6,10,14 In Japan, even among patients with clarithromycin-resistant H. pylori, using VPZ in combination with amoxicillin and clarithromycin or metronidazole for 7 days as a first-line treatment has yielded favorable results.47,70–72 A similar trend was observed in a multicenter RCT conducted by Chey in the United States and Europe. 35 The study demonstrated enhanced effectiveness of both VPZ triple and dual therapies over PPI-based triple therapy in eliminating clarithromycin-resistant H. pylori, with eradication rates of 65.8% and 69.6% compared to 31.9%, respectively. Our meta-analysis corroborates these findings, indicating a substantial difference in eradication success between the two approaches against clarithromycin-resistant strains, with pooled eradication rates of 73.7% versus 41.5% (RR = 1.53, 95% CI: 1.07–2.20, p = 0.02).
In addition to antibiotic resistance and patient non-adherence, inadequate acid suppression is a significant factor contributing to H. pylori eradication failures.12,73,74 Utilizing higher dosages or more potent acid-suppressing drugs to increase gastric pH is a proven strategy for enhancing H. pylori eradication. It is documented that H. pylori enters a growth phase and becomes more susceptible to antibiotics at a gastric pH of 6–8. 56 However, the acid suppression achieved by currently available PPIs typically does not reach or maintain the required level or duration over a full 24-h period to achieve this optimal gastric environment. 5 The introduction of P-CAB has revolutionized the approach to gastric acid suppression, making it more straightforward and efficacious. P-CABs offer more rapid, potent, and enduring acid suppression compared to PPIs,22,47,75 which has been associated with higher H. pylori eradication rates. 76 Based on this theoretical foundation, we performed a subanalysis comparing P-CAB with the second-generation PPIs (esomeprazole and rabeprazole), known for more efficient acid inhibition than first-generation PPIs and are less affected by the CYP2C19 genotype, 77 under conditions where the type and dosage of the combined drug were consistent. The result showed a significant difference in eradication rate between the P-CAB-based and esomeprazole/rabeprazole-based therapies (pooled eradication rates: 88.9% vs 84.0%, RR = 1.06, 95% CI: 1.02–1.10, p = 0.007), supporting the proposition that the more profound suppression of gastric acidity achieved with P-CABs, compared to PPIs, enhances antibiotic effectiveness. 78
To our knowledge, this represents the most recent and comprehensive meta-analysis comparing VPZ/TPZ-based therapies with PPI-based therapies for H. pylori infection. Given that all included studies were RCTs, this meta-analysis provides high-quality evidence. It further substantiates the advantages of P-CABs in H. pylori eradication, offering alternative treatment options to enhance eradication rates and providing a theoretical foundation for further P-CAB research. However, several limitations should be considered. First, the inclusion of open-label RCTs might affect the assessment of objective outcomes, such as adverse events. Second, with only one included study conducted outside of Asia, the generalizability of our findings on a global scale may be limited due to variations in antibiotic resistance across regions. Third, some heterogeneity was noted after combining data from different studies, and subgroup analysis suggested that heterogeneity may arise from population, experimental drug, treatment duration, and country. Lastly, the limited number of studies investigating TPZ-based therapy and treatment-experienced subgroups restricts the scope of our conclusions. Future research on TPZ is anticipated, and we will update our findings accordingly.
Conclusion
In summary, our meta-analysis demonstrates that P-CAB-based therapy outperforms PPI-based therapy in eradicating H. pylori, with a lower incidence of adverse events. Future research should focus on optimizing antibiotic combinations and treatment durations in P-CAB-based therapies across various geographical settings to enhance eradication rates.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251314801 – Supplemental material for The efficacy and safety of Vonoprazan and Tegoprazan in Helicobacter pylori eradication: a comprehensive systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tag-10.1177_17562848251314801 for The efficacy and safety of Vonoprazan and Tegoprazan in Helicobacter pylori eradication: a comprehensive systematic review and meta-analysis of randomized controlled trials by Ting Jin, Wei Wu, Lei Zhang, Han Xuan, Haixiang Zhang and Li Zhong in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251314801 – Supplemental material for The efficacy and safety of Vonoprazan and Tegoprazan in Helicobacter pylori eradication: a comprehensive systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-tag-10.1177_17562848251314801 for The efficacy and safety of Vonoprazan and Tegoprazan in Helicobacter pylori eradication: a comprehensive systematic review and meta-analysis of randomized controlled trials by Ting Jin, Wei Wu, Lei Zhang, Han Xuan, Haixiang Zhang and Li Zhong in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
