Abstract
Background:
Current eradication regimens are not ideal for Helicobacter pylori infected patients who have difficulty choosing antibiotics due to penicillin allergy or antibiotic resistance.
Objective:
To explore the efficacy and safety of a simple vonoprazan-minocycline dual therapy (VM dual therapy) in H. pylori eradication.
Design:
Clinical data of patients who were prescribed 14-day VM dual therapy were retrospectively collected. The included patients were 18–70 years old and positive for 13C urea breath test (13C-UBT). They were allergic to penicillin or had a history of repeated antibiotic exposure or had failed eradication with multiple regimens containing amoxicillin. VM dual therapy consists of vonoprazan 20 mg twice daily and minocycline 100 mg twice daily. 13C-UBT was repeated 4–6 weeks after treatment to evaluate the therapeutic outcome.
Results:
Of the 88 enrolled patients, 54 were treatment naïve, 13 had one prior eradication failure, and 21 had multiple eradication failures. The overall intention to treat (ITT) and per-protocol (PP) eradication rates were 90.9% (95% CI: 82.4–95.7) and 95.2% (95% CI: 87.6–98.5). The ITT eradication rates were 90.7% (95% CI: 78.9–96.5) in treatment-naïve patients, 84.6% (95% CI: 53.7–97.3) in patients with one prior treatment failure, and 95.2% (95% CI: 74.1–99.8) in patients with multiple failures. The PP eradication rates were 94.2% (95% CI: 83.1–98.5), 91.7% (95% CI: 59.8–99.6), and 100%, respectively. The overall incidence of adverse events was 23.0%. The common adverse reactions were nausea and mild dizziness, which could be resolved without intervention.
Conclusion:
Simple VM dual therapy exhibited a good eradication rate, low incidence of adverse effects, and good adherence. It is a potential new regimen for both first-line and rescue therapy.
Plain language summary
Current eradication regimens are not ideal for Helicobacter pylori infected patients who have difficulty choosing antibiotics due to penicillin allergy or antibiotic resistance. We explored a simple vonoprazan-minocycline dual therapy for these patients. We retrospectively collected 88 Helicobacter pylori infected patients' clinical data. They were allergic to penicillin or had a history of repeated antibiotic exposure, or had failed eradication with multiple regimens containing amoxicillin. VM dual therapy consists of Vonoprazan 20mg twice daily and minocycline 100mg twice daily. In the 54 treatment naïve patients, the eradication rate was 90.7%. In the 13 patients with on prior failure and the 21 patients with multiple failures, the eradication rates was 84.6% and 95.2%. 23% of the patients reported adverse events, which was mailnly nausea and mild dizziness. Our study suggested that the simple VM dual therapy exhibited a good eradication rate, low incidence of adverse effects, and good adherence. It is a promising new regimen.
Introduction
Helicobacter pylori infects approximately 50% of the world’s population and is a major cause of gastrointestinal diseases, such as peptic ulcer, chronic active gastritis, gastric mucosa-associated lymphoid tissue lymphoma, and gastric cancer. 1 Eradication of H. pylori is an important measure to prevent and treat these diseases.
The Chinese National Clinical Practice Guideline for H. pylori eradication treatment recommended bismuth-containing quadruple therapies as the preferred regimen in China. 2 Five antibiotic combinations were recommended, of which four included amoxicillin, and the other consisted of tetracycline and metronidazole. However, penicillin allergy is not rare in the population, with a reported incidence of 5%–15%. 3 For these patients, quadruple therapy containing tetracycline and metronidazole was nearly the only option. However, since the resistance rate of metronidazole was reported to be as high as 75%–90% in China, 4 a high dose of 1.2–1.6 g metronidazole per day was recommended to ensure effective eradication. 2 This often leads to intolerant adverse reactions that affect patient compliance. Furthermore, tetracycline is difficult to obtain in most regions of China. 5 Therefore, bismuth quadruple regimen with tetracycline plus metronidazole is not widely used in China. Once allergic to penicillin, patients will face difficulties in selecting appropriate antibiotics.
On the other hand, because of the low resistance rate and few adverse reactions, amoxicillin is one of the most commonly used antibiotics in first-line eradication of H. pylori. 6 It is generally believed that amoxicillin is not easily induced to develop secondary resistance and can be reused to treat H. pylori infection. However, recent studies have shown that the resistance rate of H. pylori to amoxicillin is increasing.2,7,8 With the increase in the number of treatment failures with amoxicillin-containing regimens, the resistance rate and minimum drug concentration (MIC) value of amoxicillin increased significantly.6,8 Xie et al. showed that after one single eradication failure with an amoxicillin-containing regimen, the secondary resistance rate exceeded 20%. 8 Therefore, in addition to patients with penicillin allergy, those with multiple amoxicillin exposures or those who have failed multiple amoxicillin regimens may no longer be suitable for empirical rescue therapies containing amoxicillin. It is of great practical importance to explore new eradication regimens.
High-dose dual therapy (HDDT) was also recommended as initial and rescue therapy for H. pylori eradication according to the Chinese National Clinical Practice Guideline, 2 with an eradication rate not inferior to bismuth quadruple therapy. A double dose of proton pump inhibitors (PPIs) strongly inhibits gastric acid secretion, which promotes H. pylori to enter a replicating state and increases its sensitivity to antibiotics. 5 As a pH-dependent antibiotic, amoxicillin can fully exert its bactericidal activity in the intragastric environment. 9 Vonoprazan, a novel potassium-competitive acid blocker (P-CAB), exerts a strong inhibitory effect on gastric acid secretion through reversible K+-competitive binding with active and resting H+, K+-ATPase. Pharmacodynamic data showed that the intragastric pH was >4.0 at 4 h after the first administration of vonoprazan 20 mg and was sustained for up to 24 h. 10 Administration of vonoprazan (20 mg twice daily) for seven consecutive days could maintain intragastric pH above 5 for almost the entire day. 11 The strong acid-inhibiting ability of vonoprazan greatly improves the bactericidal effect of antibiotics. A meta-analysis has shown that the eradication rates of P-CAB-containing regimens were superior to PPI-containing regimens. 12 Vonoprazan-amoxicillin dual therapy, in which high dose PPI was replaced by vonoprazan, perpetuated the effectiveness of HDDT and was reported to achieve satisfactory eradication rates. 13 Similar to amoxicillin, the bactericidal activity of tetracycline antibiotics is also affected by pH. 14 Minocycline, a second-generation semi-synthetic tetracycline, has been used for the eradication of H. pylori for more than 10 years 15 and is also recommended by the Chinese National Clinical Practice Guideline. 2 The resistance rate of H. pylori to minocycline was reported to be less than 1%.16 –19 Therefore, it is reasonable to speculate that vonoprazan-minocycline (VM) dual therapy, similar to vonoprazan-amoxicillin (VA) dual therapy, may also be a simple, safe, and effective new regimen for H. pylori eradication.
This study aims to retrospectively analyze the safety and efficacy of VM dual therapy in H. pylori eradication, particularly in patients who face challenges with antibiotic selection, to provide a basis for future research.
Materials and methods
Study design and participants
This was a pilot study conducted in the Department of Gastroenterology of Peking University First Hospital. We retrospectively searched the outpatient electronic medical record system from July 2023 to March 2024 and consecutively enrolled patients who met the inclusion criteria: (1) patients who were aged 18–70 years and were eligible for H. pylori eradication 2 ; (2) patients were treated with VM regimen. Exclusion criteria included patients: (1) with a history of gastrointestinal surgery; (2) in clinical conditions that could affect the evaluation of the treatment (hepatic/renal/cardiac dysfunction, malignancy, or mental diseases); and (3) administered with PPIs, antibiotics, anticoagulants, or steroids within 8 weeks, or concomitant use of these drugs. Medical information, such as general characteristics, treatment outcome, adverse effects, compliance, and combination medications, were extracted from the electronic medical records. Good compliance was defined as >80% adherence to treatment. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 20
Diagnosis and treatment of H. pylori infection
H. pylori infection was diagnosed by 13C urea breath test ( 13 C-UBT) in all patients. The 75 mg 13C-urea capsules were purchased from Shenzhen Zhonghe Headway BioSci & Tech Co., Ltd (Shenzhen, China). A HeliFANplus FANci3 device set (Fischer Analysen Instrumente GmbH, Leipzig, Germany) was used to obtain the delta over baseline (DOB) value. Exhaled air was collected at 30 min. A sample was considered positive if DOB was above 4‰, which indicated active H. pylori infection.
The components of VM dual therapy were vonoprazan (Vonoprazan Fumarate tablets 20 mg/tablet; Takeda Pharmaceutical Co, Osaka, Japan) 20 mg, b.i.d. and minocycline (Minocycline Hydrochloride Capsules 50 mg/capsule; Hanhui Pharmaceuticals Co., Ltd., Hangzhou, China) 100 mg, b.i.d. Patients were instructed to take both drugs 30 min after breakfast and dinner. Patients were required to repeat 13C-UBT 4–6 weeks after the end of therapy to confirm successful eradication.
Statistical analysis
Eradication rates were evaluated using intention to treat (ITT) and per-protocol (PP) analysis. ITT analysis included patients who received at least one dose of VM dual therapy. Patients who took more than 80% of the drug and repeated the 13C-UBT were included in the PP analysis. The Wilson method with continuous correction was used to calculate 95% CI of the eradication rate. Statistical analyses were performed using SPSS (version 29.0; SPSS, Inc., Chicago, IL, USA). Quantitative data of normal distribution were expressed as mean ± standard deviation. Categorical variables were expressed as numbers and percentages.
Results
Patients enrolled and baseline characteristics
A total of 88 patients who were prescribed VM dual therapy were included in this study, of which 32 (36.4%) were males and 56 (63.6%) were females. The mean age was 46.4 ± 14.5 years. Three patients did not return for breath test and one patient lacked compliance information. All patients were either allergic to penicillin or had difficulty in choosing antibiotics (previous frequent exposure to amoxicillin, macrolides, quinolones and metronidazole, or intolerance to these drugs). Of all the patients, 54 were treatment-naïve, 13 had one prior failure, and 21 experienced multiple eradication failures. Demographic and clinical data were presented in Table 1.
Baseline characteristics and demographic data of the included patients.

BMI, body mass index; SD, standard deviation.
H. pylori eradication rate
Among all the 88 eligible patients, 80 got H. pylori successfully eradicated and 4 failed. Eradication outcomes for three patients and compliance information for one patient were not available. The ITT eradication rate was 90.9% (80/88, 95% CI: 82.4–95.7), and the PP eradication rate was 95.2% (80/84, 95% CI: 87.6–98.5).
Of the 54 treatment-naïve patients, 49 got H. pylori successfully eradicated and 3 failed. Two patients did not return for breath test. The ITT eradication rate was 90.7% (49/54, 95% CI: 78.9–96.5), and the PP eradication rate was 94.2% (49/52, 95% CI: 83.1–98.5). All treatment-naïve patients were allergic to penicillin and had previously used macrolides, quinolones, and metronidazole. All three patients who failed eradication took their medication as prescribed and reported no side effects.
Among the 13 patients with one prior failure, 11 got H. pylori successfully eradicated and 1 failed. Compliance information was not recorded for one patient at the return visit. The ITT eradication rate was 84.6% (11/13, 95% CI: 53.7–97.3), and the PP eradication rate was 91.7% (11/12, 95% CI: 59.8–99.6). Of these patients, seven had used bismuth quadruple therapy with amoxicillin and clarithromycin, one had used standard triple therapy, and one had used VA dual therapy, and three failed to provide the detailed previous eradication regimen (Table 2). All patients either had a history of frequent antibiotic use because of chronic infectious diseases or were intolerant to some commonly used antibiotics.
Details of patients with one prior treatment failure.

BMI, body mass index; NA, not available; RBAC, quadruple therapy with rabeprazole, bismuth potassium citrate, amoxicillin, and clarithromycin.
Among the 21 patients with previous multiple failures, 20 got H. pylori successfully eradicated, and 1 patient did not return for reexamination. The ITT eradication rate was 95.2% (20/21, 95% CI: 74.1–99.8), and the PP eradication rate was 100% (20/20). They had previously experienced two, three, four, or even as many as eight eradication failures. The most commonly used antibiotics were amoxicillin (13 patients), clarithromycin (13 patients), metronidazole (9 patients), levofloxacin (9 patients), furazolidone (4 patients), tetracycline (2 patients), and moxifloxacin (1 patient), as shown in Table 3. There was no statistically significant difference in the ITT and PP eradication rates among the overall, first-line, and rescue treatment patients (Figure 1).
Details of patients with multiple treatment failures.
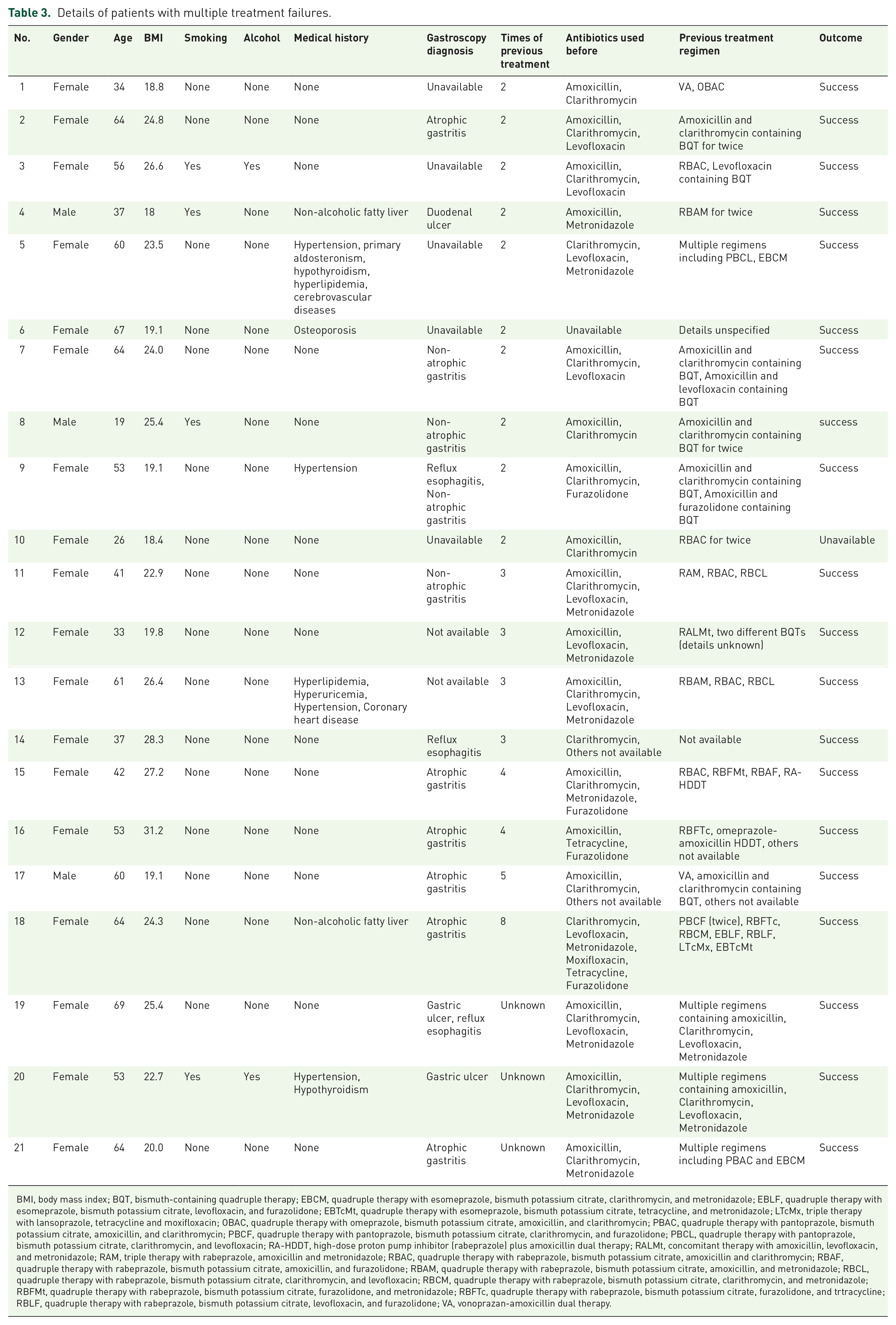
BMI, body mass index; BQT, bismuth-containing quadruple therapy; EBCM, quadruple therapy with esomeprazole, bismuth potassium citrate, clarithromycin, and metronidazole; EBLF, quadruple therapy with esomeprazole, bismuth potassium citrate, levofloxacin, and furazolidone; EBTcMt, quadruple therapy with esomeprazole, bismuth potassium citrate, tetracycline, and metronidazole; LTcMx, triple therapy with lansoprazole, tetracycline and moxifloxacin; OBAC, quadruple therapy with omeprazole, bismuth potassium citrate, amoxicillin, and clarithromycin; PBAC, quadruple therapy with pantoprazole, bismuth potassium citrate, amoxicillin, and clarithromycin; PBCF, quadruple therapy with pantoprazole, bismuth potassium citrate, clarithromycin, and furazolidone; PBCL, quadruple therapy with pantoprazole, bismuth potassium citrate, clarithromycin, and levofloxacin; RA-HDDT, high-dose proton pump inhibitor (rabeprazole) plus amoxicillin dual therapy; RALMt, concomitant therapy with amoxicillin, levofloxacin, and metronidazole; RAM, triple therapy with rabeprazole, amoxicillin and metronidazole; RBAC, quadruple therapy with rabeprazole, bismuth potassium citrate, amoxicillin and clarithromycin; RBAF, quadruple therapy with rabeprazole, bismuth potassium citrate, amoxicillin, and furazolidone; RBAM, quadruple therapy with rabeprazole, bismuth potassium citrate, amoxicillin, and metronidazole; RBCL, quadruple therapy with rabeprazole, bismuth potassium citrate, clarithromycin, and levofloxacin; RBCM, quadruple therapy with rabeprazole, bismuth potassium citrate, clarithromycin, and metronidazole; RBFMt, quadruple therapy with rabeprazole, bismuth potassium citrate, furazolidone, and metronidazole; RBFTc, quadruple therapy with rabeprazole, bismuth potassium citrate, furazolidone, and trtracycline; RBLF, quadruple therapy with rabeprazole, bismuth potassium citrate, levofloxacin, and furazolidone; VA, vonoprazan-amoxicillin dual therapy.

ITT and PP eradication rates of different patients.
Compliance and adverse effects
Of the 88 patients, 84 (96.4%) had good compliance. One treatment-naïve patient discontinued treatment on day 9 because of fatigue and fever, but the patient was later confirmed to have influenza A. Another treatment-naïve patient missed seven doses. One patient on rescue treatment was discontinued on day 8 because of pruritic rash, and the compliance information for another patient was not available. H. pylori was successfully eradicated in all four patients.
Adverse events (AEs) were reported in 20 patients (20/88, 23.0%) during treatment. The most common AE were nausea (8/88, 9.1%) and dizziness (5/88, 5.7%). Three patients reported acid reflux, two reported headache, and one reported loose stools. All of the above symptoms were relieved spontaneously without drug discontinuation. One patient (female) developed urinary tract infection on day 12 of the treatment and discontinued the medications. Three patients who complained of poor appetite before treatment reported significant improvement after taking the drugs.
Concomitant medications
Fourteen patients received concomitant medications during treatment. The most commonly used drugs were antihypertensive agents (five patients), followed by nonsteroidal anti-inflammatory drugs (three patients), statins (three patients), aspirin (two patients), levothyroxine (two patients), and acarbose (one patient). All of the above patients had got H. pylori successfully eradicated.
AEs were reported in four (4/14, 28.6%) patients taking combination medications, including dizziness (two patients), nausea (one patient), and acid reflux (one patient). There was no significant difference in the incidence of AEs between patients with and without concomitant medications (28.6% vs 22.9%, p=0.73).
Discussion
To our knowledge, this is the first study to explore the efficacy and safety of VM dual therapy as a first-line and rescue treatment for H. pylori infection. The results revealed that the simple VM dual therapy achieved good eradication rates in both naïve and rescue patients. This regimen not only provided a new option for patients with penicillin allergy, but also showed satisfactory efficacy for patients who had difficulty in choosing antibiotics, such as those with multiple treatment failures or an unknown history of prior antibiotic use.
In recent years, the effectiveness of conventional treatment regimens has declined due to increased resistance of H. pylori to antibiotics. Amoxicillin, with a relatively low resistance rate, is one of the most commonly used and effective antibiotics for H. pylori eradication, but is not suitable for patients with penicillin allergy or positive skin test. It is noteworthy that the secondary resistance rate of amoxicillin increases with the number of eradication courses. 8 For other commonly used antibiotics, such as clarithromycin, metronidazole, and levofloxacin, drug resistance is more serious. Studies in China reported that the primary resistance rates for these antibiotics were as high as 50%–70%, whereas the secondary resistance rates increased to 80%–90% after one treatment failure, and nearly 100% after two or three treatment failures. 6 Resistance to tetracycline and furazolidone is rare; however, the relatively high incidence of AEs and low availability limit their use. In our study, VM dual therapy achieved a satisfactory eradication rate in both first-line and rescue therapy. Remarkably, the eradication rate was over 90% in patients with one previous failure and over 95% in patients with multiple failures. In the context of increasing antibiotic resistance worldwide, VM dual therapy may be a promising regimen that deserves attention.
Minocycline is the second generation of semi-synthetic tetracycline antibiotics and has been used for more than 30 years. Tetracycline enters bacteria through outer membrane protein channels, binds to the ribosome 30S subunit, and inhibits bacterial protein synthesis. 21 Minocycline improves its affinity with ribosome 30S subunit through D ring modification of carbon 7–9, and acquires higher antibacterial activity. 22 Previous studies reported that the primary resistance rate of H. pylori to minocycline was low,16,17 and that the secondary resistance was rare. 23 Thus, eradicating H. pylori with minocycline is less affected by previous exposure to commonly used antibiotics and does not require susceptibility testing. However, previous studies showed that the eradication rate of triple therapy with semi-synthetic tetracycline was not satisfactory. Minocycline-based triple therapy could only achieve an eradication rate of less than 40%. 24 Therefore, minocycline has not been a commonly used drug in H. pylori eradication for a long time. Recent studies have indicated that the eradication rate of quadruple therapy with minocycline is satisfactory, not inferior to other control regimens, or superior to regimens without tetracycline.24,25 However, like other quadruple therapies, minocycline quadruple therapy is complex and has a relatively high incidence of adverse effects, which limits its application.
The poor efficacy of minocycline-containing triple therapy may be attributed to low stomach pH. The bactericidal activity of many antibiotics is closely related to pH. Sufficient acid suppression helps achieve a higher eradication rate. The Maastricht VI/Florence consensus report recommends that H. pylori eradication requires strong and sustained acid suppression to achieve a target stomach pH >6 and holding time ratio (HTR) of ⩾75% throughout the day. 26 To date, the bactericidal activity of minocycline under different pH conditions has not been reported. However, an in vitro experiment showed that MIC90 (the minimum drug concentration that inhibits 90% bacterial growth) of tetracycline against H. pylori was 0.5 mg/L at pH 5.0, and decreased to 0.125 mg/L at pH 6.0. 14 Similar results were reported by Njume et al. At pH 5.5, 6.0, and 7.5, MIC90 of tetracycline against H. pylori was 0.5, 0.25, and 0.12 mg/L, respectively. 27 Based on these results, it is reasonable to speculate that the bactericidal activity of minocycline is pH dependent. However, the acid-suppressing effect of PPIs is limited. When taken at a standard dose twice daily, the HTR of gastric pH ⩾6 rarely reaches 40%–50%, 28 which prevents minocycline from fully exerting its antibacterial activity. Vonoprazan, a novel P-CAB, has demonstrated potent and sustained acid-suppression ability. It has been reported that administration of vonoprazan 20 mg twice daily can achieve an HTR of gastric pH ⩾6 up to 85%. 29 This may be the pharmacological basis for the satisfactory eradication rate of VM dual therapy.
Minocycline has improved bioavailability and lipophilicity compared with tetracycline, and its absorption is not affected by food. After oral administration, minocycline is almost completely absorbed through the duodenum and upper jejunum, and is widely distributed in various tissues. 30 Minocycline is mainly metabolized by the liver and only 4%–9% is excreted in urine, which makes it well tolerated in patients with mild renal insufficiency. 31 The half-life of minocycline is 12–18 h, and its metabolites still have antibacterial activity, 31 so twice daily administration can maintain sufficient blood concentration and antibacterial activity. In the VM dosing regimen, minocycline can be taken together with vonoprazan, which makes the treatment more convenient and helps improve patient compliance.
Consideration of adverse effects is an essential issue in H. pylori eradication. Minocycline has a good safety record. Long-term administration at a dosage of up to 200 mg daily is generally safe and well tolerated. 22 The most common side effects of minocycline are dizziness, nausea, hepatotoxicity, and skin pigmentation. 24 A meta-analysis 25 indicated that the total incidence of adverse effects in quadruple therapy containing minocycline was 35.9%, which was similar to that without minocycline. Another meta-analysis 24 reported that minocycline-containing regimen had a higher incidence of nausea and dizziness, but the symptoms were mild and self-resolving. The total incidence of AEs in this study was 23%. And in accordance with previous studies, 24 The most common AEs in our study were also nausea and dizziness, with a relatively low incidence (9.5% and 5.7%). It was speculated that VM dual therapy could simplify the eradication regimen and thus reduce the adverse effects of bismuth and other antibiotics.
There are several limitations in this study. First, although our study presented promising results, it cannot be generalized due to its retrospective nature, the specific patient population, and the relatively small sample size. Well-designed randomized controlled trial should be performed to further validate the effectiveness in a broader patient population. Second, a previous history of antibiotic exposure was not comprehensively collected in this study; therefore, it was not possible to assess whether previous administration of tetracyclines affected the eradication outcomes. Third, antibiotic susceptibility information was not available in this study. In the future, well-designed randomized controlled studies with a large sample size and antibiotic susceptibility test should be conducted to further evaluate the clinical value of VM dual therapy.
Conclusion
Our retrospective study suggests that VM dual therapy may achieve a good eradication rate with a low incidence of adverse effects and high patient adherence. The regimen is simple, friendly to patients with penicillin allergy, may not require antibiotic susceptibility testing and may be a potential new protocol for both initial and rescue treatment. Well-designed prospective randomized controlled studies should be conducted to further evaluate the efficacy and safety of VM dual therapy in H. pylori eradication.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241299734 – Supplemental material for The efficacy and safety of a simple 14-day vonoprazan-minocycline dual therapy for Helicobacter pylori eradication: a retrospective pilot study
Supplemental material, sj-docx-1-tag-10.1177_17562848241299734 for The efficacy and safety of a simple 14-day vonoprazan-minocycline dual therapy for Helicobacter pylori eradication: a retrospective pilot study by Xiaolei Wang, Guigen Teng, Xinhong Dong, Yun Dai and Weihong Wang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
