Abstract
Background:
The efficacy and safety of potassium-competitive acid blockers (P-CABs) in the eradication of Helicobacter pylori (Hp) remains controversial when compared with proton pump inhibitors (PPIs).
Objectives:
The current study set out to compare the differences in the eradication rate and adverse reactions between eradication regimens based on P-CAB or PPI drugs and the differences between the vonoprazan-based and the tegoprazan-based regimens to explore the efficacy and safety of different Hp eradication regimens.
Data sources and methods:
Databases including PubMed, EMBASE, Cochrane Library, and WOS were searched from the inception of these databases up to July 2023, and eligible randomized controlled trials (RCTs) were included. The outcome measures were the eradication rate and the incidence of adverse reactions of different regimens in treating Hp. The results were estimated as relative risk (RR) and its 95% confidence interval (CI), and R 4.2.1 software was used to perform the network meta-analysis (NMA).
Results:
A total of 20 studies were included in the analysis, involving 5815 patients with Hp. In terms of eradication rate, the 2-week vonoprazan-based triple regimen (V-Tri-2w) was the best, which was superior to the 2-week PPI-based quadruple regimen [P-Qua-2w, RR = 0.9, 95% CI: (0.85–0.95)] and the 1-week tegoprazan-based triple regimen [T-Tri-1w, RR = 0.79, 95% CI: (0.64–0.97)]; the 2-week tegoprazan-based quadruple regimen (T-Qua-2w) was superior to the 1-week PPI-based triple regimen [P-Tri-1w, RR = 0.82, 95% CI: (0.67–0.99)], and there was no difference between the remaining tegoprazan-based regimens and the PPI-based or vonoprazan-based regimens. In terms of the incidence of adverse reactions, the 2-week vonoprazan-based binary regimen (V-Bi-2w) was lower than that of the 2-week PPI-based quadruple regimen [P-Qua-2w, RR = 1.98, 95% CI: (1.57–2.52)]; there was no significant difference between 1 and 2 weeks for each regimen, such as the vonoprazan-based triple regimen [RR = 1.11, 95% CI: (0.82–1.52)].
Conclusion:
In the eradication treatment of Hp, the efficacy and safety of vonoprazan-based regimens are generally better than those of PPI-based regimens. Among them, the V-Tri-2w regimen has the highest eradication rate and may be the preferred choice for Hp eradication.
Introduction
Helicobacter pylori (Hp) is a microaerophilic, Gram-negative bacterium. Around 75% of gastric cancer in the world results from inflammation and injury caused by Hp. 1 It is estimated that nearly half of the world’s population is infected with Hp, and the infection rate is 90% in developing countries with a high recurrence rate. 2 In China, 700 million people have been infected with Hp. 1 In addition, Hp also causes other gastrointestinal diseases, such as chronic gastritis, digestive tract ulcers, gastric adenocarcinoma, and mucosa-associated lymphoid tissue lymphoma.3,4 Sufficient inhibition of gastric acid secretion is a necessary condition for Hp eradication. 5 The triple or quadruple schemes mainly based on proton pump inhibitors (PPIs) are currently used for eradicating Hp, 6 but in recent years, there has been increasing research on PPI-related adverse events,7,8 which involve multiple organ systems.
Characteristic changes in gastrointestinal mucosa, such as fundic gland polyps and hyperplastic polyps, 9 as well as increased risk of fracture and kidney disease in older people have often been reported in long-term use of PPIs. 10 However, some studies have shown that after short-term use of PPIs, adverse reactions, such as rebound acid hypersecretion, may also occur. 11 In addition, some patients cannot tolerate a PPI-based regimen, making it necessary to find an alternative to PPI-based regimens for the eradication of Hp.
Potassium-competitive acid blockers (P-CABs) are a novel class of acid suppressants that produce stronger and more sustained gastric acid suppression than PPIs. 12 In recent years, there have been an increasing number of clinical studies on Hp eradication by P-CABs, confirming the therapeutic effect of P-CABs in Hp patients. Because the metabolism of PPIs is dependent on cytochrome P450 (CYP) 2C19, the effects of PPIs can be affected by genetic polymorphisms of CYP2C19 drugs.13,14 Vonoprazan and tegoprazan are newly developed P-CAB drugs and have significant effects on Hp eradication.15,16 Studies have shown that vonoprazan is not affected by the genetic polymorphism of CYP2C19 drugs, and therefore, it can fully inhibit gastric acid secretion. 17 At present, there are many Hp eradication regimens, but there are different opinions about the efficacy of various Hp eradication regimens that are based on P-CAB or PPI. Zhang et al. 18 found that the P-CAB-based triple regimen was superior to the PPI-based triple regimen, while the study by Du et al. 19 showed that the vonoprazan combined with amoxicillin duo regimen might be the preferred first-line Hp eradication regimen in clinical use. The efficacy of multiple Hp eradication regimens that are based on tegoprazan or vonoprazan could not be compared due to the limited evidence.
To sum up, the existing studies only compared the advantages and disadvantages of P-CAB and PPI drugs, lacking a comparison between different P-CAB drugs, as well as a comparison between regimens of different drug combinations and different courses of treatment. Network meta-analysis (NMA) is considered the highest evidence in evidence classification described in the treatment guidelines. 20 This is because the NMA includes direct and indirect comparisons, 21 providing a comprehensive understanding of the advantages and disadvantages of different regimens regarding the eradication rate and adverse reaction rate in eradicating Hp. With the Bayesian method, one can use the posterior probability distribution to rank all the analyzed interventions, better reflecting the real effect of each intervention, and avoiding the biased results by the frequentist approach, which estimates the maximum likelihood function through continuous iteration and is prone to instability, to make the estimated value more accurate. In this systematic review and NMA, we evaluated the advantages and disadvantages of P-CAB and PPI drugs in eradicating Hp and compared the P-CAB drugs of vonoprazan and tegoprazan. In addition, this study also compared different regimens and different courses of treatment to explore the best regimen for Hp eradication.
Data and methods
This study has been registered with the International Prospective Register of Systematic Reviews (PROSPERO) according to the protocol (ID: CRD42023445345) and followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Supplemental Material 1).
Literature retrieval strategy
Randomized controlled trials (RCTs) on P-CAB drugs in treating Hp were searched in PubMed, EMBASE, Cochrane Library, and Web of Science from the inception of these databases to July 2023. The search strategy was formulated based on different databases, and the main terms used to construct the search strategy included ‘Helicobacter pylori’, ‘potassium competitive acid blocker’, ‘vonoprazan’, ‘tegoprazan’, and ‘proton pump inhibitor’. The specific search strategy is provided in Supplemental Material 2.
Eligibility criteria
Inclusion criteria: Studies meeting the following criteria were included. Type of study: RCTs, without restrictions on language. Participants: Patients diagnosed with Hp infection. Interventions: The treatment group included patients receiving P-CAB-based (vonoprazan or tegoprazan) regimens, and the control group included patients receiving PPI-based (omeprazole, esomeprazole, rabeprazole, pantoprazole, etc.) regimens, without restrictions on the number of combined drug type in both groups. Outcome measures: eradication rate and incidence of adverse reactions. For the eradication rate, a value less than 4 in the carbon-13 breath test or a value below 100 in the carbon-14 breath test is considered negative, indicating that Hp has been eradicated. The incidence of adverse reactions included at least one of the following symptoms: abdominal discomfort, nausea and vomiting, constipation, dizziness, headache, rash, dry mouth, bitter taste, halitosis, etc. Exclusion criteria were set as follows: Overlapping publications, conference papers, reviews, guidelines, literature from animal experiments, etc.; studies on Hp patients with comorbidities of gastrointestinal cancer; literature with obvious errors in data. In our registration with PROSPERO, the use of PPI or P-CAB 2 weeks before the start of the trial was listed in the exclusion criteria. However, most of the included studies did not report this, and we did not exclude such studies.
Literature screening, quality assessment, and data extraction
Duplicated studies were removed automatically or manually using Endnote X9, and literature screening was conducted according to the inclusion and exclusion criteria. Two researchers independently extracted the literature, and any disagreement was solved through consultation with a third researcher. RevMan 5.4 software, The Cochrane Collaboration was employed for bias evaluation and heterogeneity test. The risk bias assessment tool recommended by Cochrane was used for assessing the risk of bias from the following domains: randomization sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting of study results, and other biases. The quality of the literature was rated as low risk, moderate risk, and high risk. Data were extracted in Excel tables, mainly including author, publication time, age, gender, sample size, interventions, and outcome measures.
Statistical analysis
The relative risk (RR) was used as the effect index for dichotomous variables, and the 95% confidence interval (CI) without invalid value (0 for continuous data and 1 for dichotomous data) indicated statistical difference. I2 was adopted to evaluate the heterogeneity among studies. I2 ⩽ 50% indicated that the heterogeneity among studies was small. The NMA was then conducted. I2 > 50% indicated the presence of heterogeneity, and the sources of heterogeneity were analyzed. Subgroup analysis or sensitivity analysis without data from the study that had significant differences was conducted in case of obvious clinical heterogeneity to explore the source of heterogeneity. If the sources of heterogeneity could not be found, descriptive analysis was adopted. The GeMTC package in R software was used to create network plots. The GeMTC software was used to perform the NMA and plot a probability ranking plot. The software utilizes the Markov Chain Monte–Carlo Bayesian inference (MC–MC) approach for analysis. The inconsistency test was conducted using a node-split model, with p > 0.05 for each study within a subgroup indicating no significant inconsistency. The convergence of the model was reflected by the potential scale reduction factor (PSRF). The PSRF of 1 or around 1 indicated better convergence efficiency and more reliable analysis results of the model.
Results
Literature screening process
The included studies were conducted in Asia, Europe, and America, covering 1374 articles retrieved preliminarily. A total of 798 articles were left after removing duplicates; we excluded 374 articles that did not meet P(Population); I(Intervention); C(Control); O(Outcome); S(Study) (PICOS) requirements, 2 studies from animal experiments, 112 conference records, 145 reviews, 13 patents, and 106 websites, and then obtained the full texts of the remaining 46 articles. After reading the full texts, we excluded 26 articles with missing data and those on subjects having clarithromycin resistance. Finally, 20 articles were included. The detailed process is shown in Figure 1.
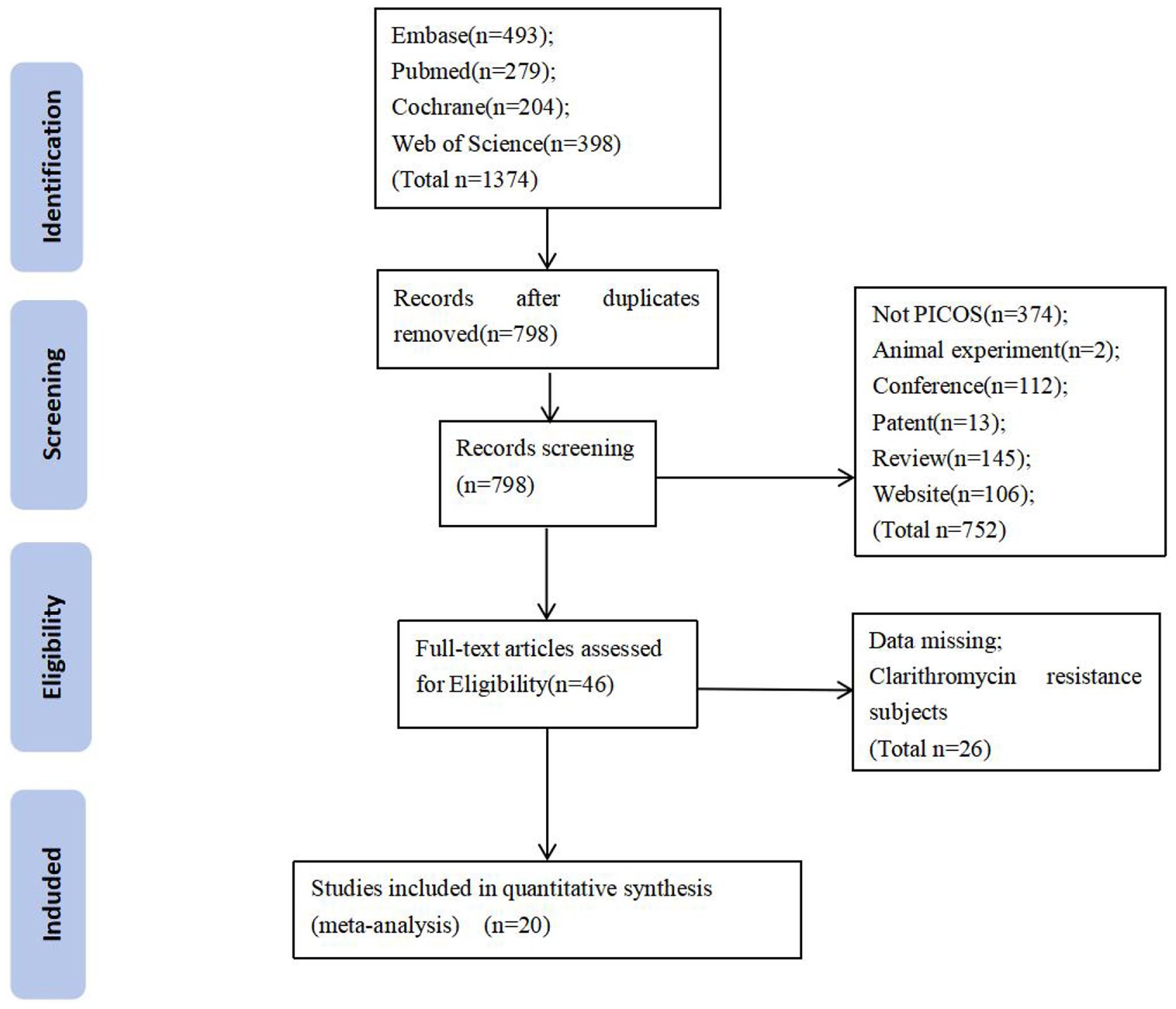
Diagram of literature screening.
Baseline characteristics of the included studies and quality assessment
A total of 20 RCTs were included,15,16,22–39 involving 5815 patients with Hp infection, with 3153 patients in the treatment group, and 2662 patients in the control group. The basic characteristics of the included studies are shown in Tables 1 and 2. Randomization was reported in all studies, and the specific randomization methods were reported in 16 studies,16,22–27,29,31–38 including random sequence generated by computer software programs in 7 studies,16,27,31–33,36,37 random numbers table in 2 studies,22,23 sequence generated by randomization system in 2 studies,25,26 blocking and stratification randomization method in 2 studies,29,38 random sequence generated by computer SAS software in 1 study, 35 odd–even number of a case number for randomization in 1 study, 34 and the order of patients in 1 study. 24 Of all the 20 articles, only 7 mentioned the blinding method,22,23,26,28–30,38 4 of which mentioned placebo22,26,29,38 and were rated as having low risk, while the remaining 13 mentioned open-label studies without using the blinding method, and therefore, were rated as having high risk. All studies had complete data and did not mention other risks and were therefore rated as having moderate risk. The details are shown in Figure 2.
Basic characteristics of the included studies.

Outcome measures: ① eradication rate; ② adverse reactions; ③ CYP2C19 genotype; ④ patient compliance; ⑤ antibiotic resistance rate; ⑥ intestinal flora; ⑦ pharmacokinetics evaluation; ⑧ cost–benefit analysis.
A, amoxicillin; B, bismuth; C, clarithromycin; D, doxycycline; E, esomeprazole; F, furazolidone; M, metronidazole; O, omeprazole; R, rabeprazole; T, tegoprazan; t, tetracycline; V, vonoprazan.
Basic information on adverse reactions in the included studies.

A, amoxicillin; ALT, Alanine aminotransferase; B, bismuth; bid, bis in die; C, clarithromycin; D, doxycycline; E, esomeprazole; F, furazolidone; M, metronidazole; O, omeprazole; qid, quarter in die; R, rabeprazole; T, tegoprazan; t, tetracycline; tid, ter in die; V, vonoprazan.

Quality assessment results of the included studies.
Network meta-analysis
Eradication rate
A total of 19 studies were included, involving 10 interventions. The evidence network diagram is shown in Figure 3(a). The included regimens involved vonoprazan (V), tegoprazan (T), and PPIs (P). The types of drug combinations were binary (Bi), triple (Tri), and quadruple (Qua) regimens and the courses of treatment included 1 week (1w) and 2 weeks (2w). The results of the forest plot showed that V-Tri-2w had the best eradication rate, and P-Tri-2w was better than P-Tri-1w, as shown in Figure 3(b) and (c). According to the results of the league table, V-Tri-2w was superior to P-Qua-2w [RR = 0.9, 95% CI: (0.85–0.95)], P-Tri-1w [RR = 0.76, 95% CI: (0.68–0.85)], P-Tri-2w [RR = 0.9, 95% CI: (0.84–0.97)], and T-Tri-1w [RR = 0.79, 95% CI: (0.64–0.97)]; P-Qua-2w was superior to P-Tri-1w [RR = 1.18, 95% CI: (1.05–1.34)] and inferior to V-Qua-2w [RR = 0.94, 95% CI: (0.89–0.98)]; P-Tri-1w was inferior to P-Tri-2w [RR = 0.84, 95% CI: (0.77–0.92)], T-Qua-2w [RR = 0.82, 95% CI: (0.67–0.99)], and V-Bi-1w [RR = 0.85, 95% CI: (0.77–0.94)], V-Bi-2w [RR = 0.81, 95% CI: (0.72–0.91)], V-Tri-1w [RR = 0.81, 95% CI: (0.76–0.86)], V-Tri-2w [RR = 0.76, 95% CI: (0.68–0.85)], and V-Qua-2w [RR = 0.79, 95% CI: (0.7–0.9)]. The remaining pairwise comparisons were not statistically significant, as shown in Figure 3(c).

Summary of the Bayesian meta-analysis of eradication rate. (a) Network plot. (b) Forest plot comparing eradication rates between eradication regimens. (c) Network meta-analysis of eradication rates.
Adverse reactions
A total of 19 studies were included, involving 10 interventions. The evidence network diagram is shown in Figure 4(a). The results of the forest plot showed that V-Bi-2w had the lowest incidence of adverse reactions, followed by V-Bi-1w. There was no significant difference between the 1-week eradication regimens based on P-CAB or PPI and their 2-week regimens, as shown in Figure 4(b) and (c). According to the results of the league table, V-Bi-2w had lower incidence of adverse reactions than P-Qua-2w [RR = 1.98, 95% CI: (1.57–2.52)], P-Tri-1w [RR = 1.75, 95% CI: (1.25–2.47)], P-Tri-2w [RR = 1.37, 95% CI: (1.13–1.67)], T-Qua-2w [RR = 1.78, 95% CI: (1.2–2.67)], and T-Tri-1w [RR = 1.93, 95% CI: (1.33–2.82)]; V-Bi-1w had lower incidence of adverse reactions than P-Qua-2w [RR = 1.67, 95% CI: (1.04–2.66)], P-Tri-1w [RR = 1.48, 95% CI: (1.08–2.03)], and T-Tri-1w [RR = 1.62, 95% CI: (1.14–2.31)]; P-Tri-2w had lower adverse reaction than P-Qua-2w [RR = 1.44, 95% CI: (1.09–1.92)], and T-Tri-1w [RR = 0.71, 95% CI: (0.52–0.98)]; V-Tri-1w had lower adverse reaction than P-Tri-1w [RR = 1.18, 95% CI: (1.03–1.36)] and T-Tri-lw [RR = 1.3, 95% CI: (1.05–1.61)]; V-Tri-2w had lower incidence of adverse reaction than P-Qua-2w [RR = 1.48, 95% CI: (1.16–1.92)]; V-Qua-2w had higher incidence of adverse reaction than P-Qua-2w [RR = 0.88, 95% CI: (0.79–0.98)], P-Tri-2w [RR = 0.61, 95% CI: (0.45–0.82)], V-Bi-1w [RR = 0.53, 95% CI: (0.32–0.85)], V-Bi-2w [RR = 0.44, 95% CI: (0.34–0.57)], V-Tri-1w [RR = 0.66, 95% CI: (0.45–0.97)], and V-Tri-2w [RR = 0.59, 95% CI: (0.45–0.77)]. The remaining pairwise comparisons were not statistically significant, as shown in Figure 4(c).
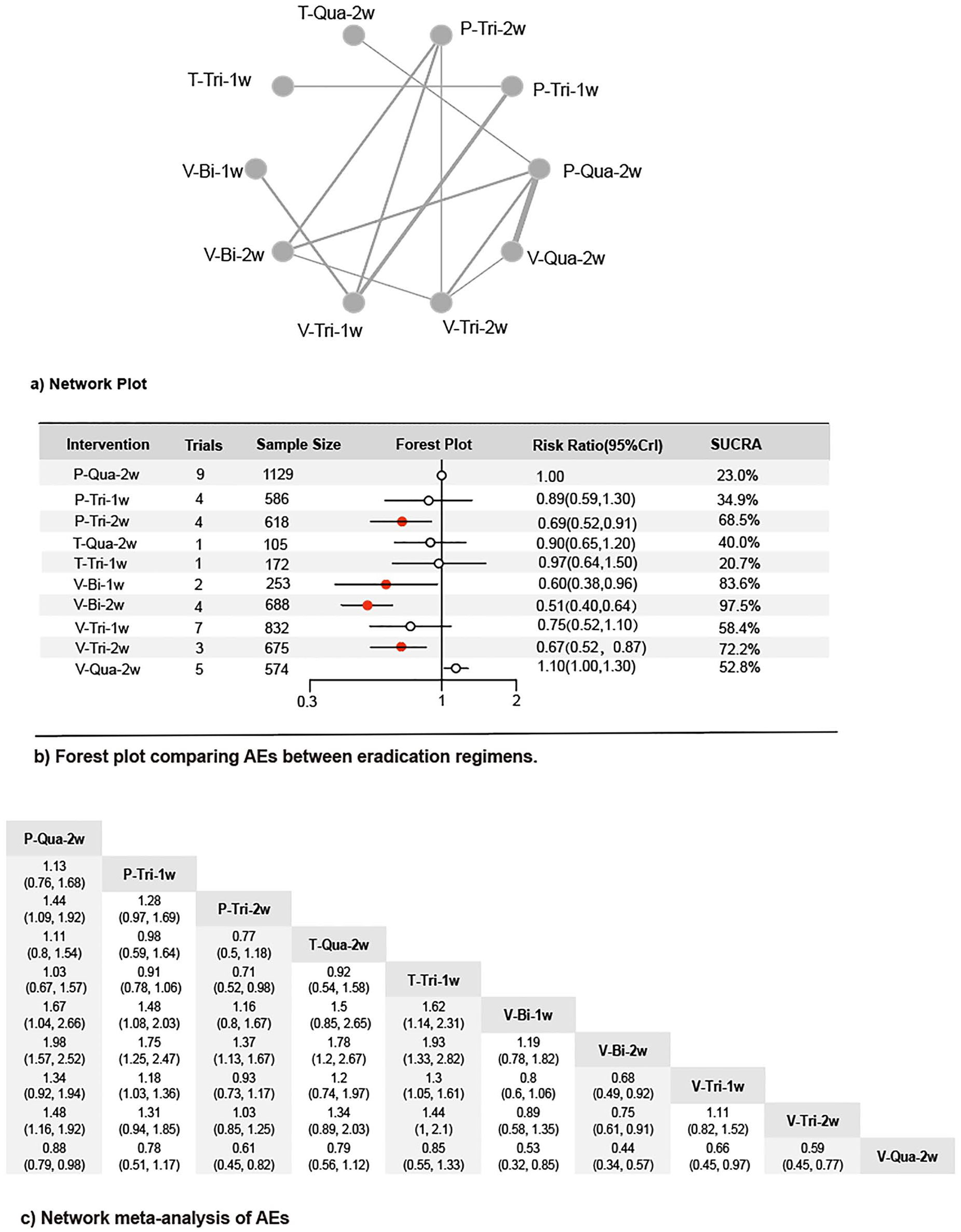
Summary of the results of Bayesian meta-analysis of AEs. (a. Network plot; b. Forest plot comparing AEs between eradication regimens; c. Network meta-analysis of AEs.)
Probability ranking
The 10 interventions were ranked for their probability of being the best treatment. The probability of being the best treatment in terms of eradication rate was as follows: V-Tri-2w (94.7%), V-Qua-2w (76.4%), V-Bi-2w (66.2%), V-Tri-1w (64.9%), T-Qua-2w (59.2%), V-Bi-1w (41.4%), P-Tri-2w (41.0%), P-Qua-2w (39.2%), T-Tri-1w (13.0%), and P-Tri-1w (3.9%), as shown in Figure 3(b). The probability of being the best treatment in terms of the incidence of adverse reactions was as follows: V-Bi-2w (97.5%), V-Bi-1w (83.6%), V-Tri-2w (72.2%), P-Tri-2w (68.5%), V-Tri-1w (58.4%), V-Qua-2w (52.8%), T-Qua-2w (40.0%), P-Tri-1w (34.9%), P-Qua-2w (23.0%), and T-Tri-1w (20.7%), as shown in Figure 4(b).
Model convergence assessment, heterogeneity analysis, inconsistency analysis, and sensitivity analysis
Overall, model convergence assessment was performed based on the eradication rate, and the PSRF of the clinical eradication rate was 1, indicating good convergence and fitting of the model, and the results of NMA under the model were reliable. According to the results of heterogeneity analysis, the eradication rate and the risk of adverse reactions had an overall I2 < 50%, indicating that the homogeneity assumption was met. Based on the results of the inconsistency test, the eradication rate and the risk of adverse reactions had a main p value >0.05, indicating that the consistency hypothesis was met. The results of the heterogeneity analysis and inconsistency test are shown in Supplemental Material 3. In the sensitivity analysis, we found that the study by Kim et al. was significantly different from other studies in the composition of its regimen (including bismuth, metronidazole, tetracycline, and lansoprazole). Therefore, we conducted sensitivity analysis without inclusion of the study by Kim et al. and compared the sensitivity analysis results with the previous results. No significant changes were found in any of the outcome measures, indicating that the difference in the study by Kim et al. did not affect the stability of the results. The sensitivity analysis results are shown in Supplemental Material 3. In addition, we also added Supplementary Network meta-analysis, including the NMA of the treatment regimen without regard to the treatment courses, the NMA of the 2-week regimen, and the NMA excluding Kim, and the results were consistent with those described above, as detailed in Supplemental Material 4.
Discussion
The results of this study showed that the vonoprazan-based regimen was better than the PPI-based regimen for Hp eradication. Among the P-CAB drugs, V-Tri-2w, V-Qua-2w, V-Bi-2w, and V-Tri-1w were superior to the tegoprazan-based regimens, and V-Tri-2w had the best eradication rate. In terms of adverse reactions, with the increase in the number of combined drug types, the incidence of adverse reactions of each eradication regimen presented an upward trend, and the V-Bi-2w regimen had the least adverse reactions. There was no significant difference in safety between 1 week and 2 weeks for each regimen.
In the eradication treatment of Hp, an environment of pH >5.0 in the stomach is required to activate the proliferation of Hp for the maximum effect of antibacterial drugs. Therefore, the use of acid suppressants is crucial. Traditional PPI-based regimen has played an important role in the treatment of Hp in the past. However, the effectiveness of traditional PPI-based regimens dropped to 70% due to an increase in drug-resistant bacteria. 5 About two-thirds of patients with symptoms of gastroesophageal reflux disease do not have the reflux symptoms adequately controlled after the first dose of a PPI drug, and about half of the patients still have symptoms several days after starting the treatment. 40 In addition, more than half of patients taking PPIs were dissatisfied with the treatment, 41 and the nocturnal acid breakthrough is a major problem that is difficult to control.42–44 The P-CAB-based regimens are superior to PPI-based regimes possibly due to the following aspects: (1) The PPIs are acid-unstable and need to be dissolved in the small intestine in the form of enteric-coated preparations instead of in the stomach. However, the time to reach the small intestine changes due to the action of digestive tract peristalsis and gastric emptying. 45 After moving to the small intestine, the PPIs absorbed in the small intestine are metabolized by the liver to varying degrees due to CYP2C19 gene polymorphism, resulting in differences in drug exposure between individuals. P-CAB is a class of drugs that inhibit the H+/K+ ATPase enzyme by ion binding on the outer surface of cells and competing with K+ and is metabolized by CYP3A446–48 instead of CYP2C19. Besides, no evidence suggests that the genetic polymorphism of CYP3A4 will affect P-CAB drugs, and therefore there is no significant difference in acid inhibition among individuals. (2) PPIs have a slow onset of action, generally taking 3–5 days to show the best effect because of the short plasma half-life of PPIs (1–2 h), and the concentration of the active form of PPIs in the secretory tubules of gastric parietal cells drops in a short time. 49 However, the H+/K+ ATPase enzyme has a half-life of about 50 h, and 25% of this enzyme is newly synthesized every day. 50 As a result, the PPIs cannot achieve a long-term inhibition of the H+/K+ ATPase enzyme. Therefore, repeated dosing for 3–5 days is required to achieve a stable concentration for a maximum effect and consistent clinical effect.51–54 By contrast, P-CAB drugs work quickly and can achieve the maximum effect when they are first administered. Preliminary studies have shown that P-CAB drugs can achieve the maximum acid-suppressing effect after being administrated within 30 min. 55 P-CAB is a class of lipophilic weak bases with a high pKa value. P-CAB drugs are chemically stable in an environment with a low pH value, and these properties allow them to be highly concentrated in the acid environment of the parietal cells, resulting in rapid onset of action and a direct dose–response relationship. When combined with the H+/K+ ATPase enzyme, the P-CAB drugs stabilize the enzyme in the E2 conformation, thus preventing the hydrogen ions (H+) from entering the secretory tubules of the parietal cells. 56 These drugs inhibit the H+/K+ ATPase enzyme in a potassium-competitive reversible mechanism by ion binding at or near potassium-binding sites, thereby blocking gastric acid secretion through a direct and reversible mechanism.55,57 This leads to a fast onset of action. (3) The acid-suppressing effect of PPIs usually does not last for 24 h, and therefore PPIs cannot adequately inhibit nighttime acid secretion. The acid-suppressing effect of P-CABs lasts for 24 h or longer. 58 According to an earlier study, 59 the pH value in the stomach can reach 7.02 3 h after oral administration of 20 mg vonoprazan. Oral administration of 40 mg vonoprazan leads to an acid-suppressing effect lasting 24 h.12,17 (4) PPIs need to be converted into an active imine form via the acid activation process and irreversibly inhibit the H+/K+ ATPase enzyme via covalent binding.60,61 Therefore, PPI drugs need to be taken before meals to promote acid activation. 62 Besides, when PPIs accumulate in the secretion tubules of parietal cells, they are converted into active forms in the presence of acids. The H+/K+ ATPase enzyme is mainly activated by food, and PPIs block H+ in an active form through covalent binding with the H+/K+ ATPase enzyme. With a high degree of acid stability, P-CAB drugs have an acid-suppressing effect that is not affected by gastric acid secretion. 63 Therefore, the administration of P-CAB drugs is not affected by food. All this may explain why P-CAB drugs are superior to PPI drugs in the treatment of Hp.
This is the first study to make a comparison between P-CAB drugs. Although existing studies have shown that tegoprazan produces a more rapid, effective, and sustained acid suppression effect than vonoprazan, 64 the results of this study suggest that vonoprazan-based regimens are superior to tegoprazan-based regimens in terms of eradication rate. Choi et al. reported the reasons why tegoprazan-based regimens had a lower eradication rate than expected, including inadequate dosing (tegoprazan, 50 mg bid), distribution differences in minimal inhibitory concentration between clarithromycin-resistant strains, pharmacological differences between vonoprazan and tegoprazan, and inadequate duration of treatment. 65 Tegoprazan is a novel P-CAB based on benzimidazole structure and is highly selective for inhibition of the H+/K+ ATPase enzyme. It is characterized by rapid absorption and the maximum concentration can be reached in 0.5–1.5 h. The half-life of tegoprazan is short, with an average half-life of 3.7–5.4 h, while the plasma half-life of vonoprazan is longer. In healthy adults, the average terminal half-life of vonoprazan is 7.7 h, and this may be one of the reasons why the eradication rate of vonoprazan is better than that of tegoprazan. Due to the lack of relevant evidence, large RCT studies need to be carried out in the future to compare the efficacy and safety of vonoprazan and tegoprazan in eradicating Hp.
In terms of the incidence of adverse reactions, the vonoprazan-based regimen had the lowest incidence of adverse reactions. It is noteworthy that in all eradication regimens included in this study, the incidence of adverse reactions increased with the increase in the number of drug types. Among the included triple regimens, the vonoprazan-based triple regimen had the lowest incidence of adverse reactions, followed by the tegoprazan-based triple regimen. Among all the included quadruple regimens, the vonoprazan-based quadruple regimen had the highest incidence of adverse reactions, and the tegoprazan-based quadruple regimen had the lowest incidence of adverse reactions. Sapmaz et al. 66 have found that the incidence of adverse reactions in a binary regimen containing amoxicillin and PPI has a lower incidence of adverse reactions than the PPI-based triple or quadruple regimens, 67 while some other studies report that these regimens have an equal incidence of adverse reactions.68,69 Drug resistance also needs to be considered in clinical applications, but there are few discussions on drug resistance in the included studies. Therefore, we failed to extract enough data for drug resistance analysis. Antibiotic resistance analysis was reported in four of the included studies, including the resistance of amoxicillin, metronidazole, tetracycline, levofloxacin, and furazolidone. Antibiotic resistance is an important factor affecting the eradication rate of Hp, among which clarithromycin resistance is the most common. In the treatment of Hp patients with clarithromycin resistance, Chey et al. 25 found that the vonoprazan-based regimen was superior to the lansoprazole-based regimen (p < 0.001), and Kim et al. found that the tegoprazan-based regimen was superior to the lansoprazole-based regimen (p < 0.05). 29 Suzuki et al. 37 found that the binary regimen combining vonoprazan and amoxicillin had a higher eradication rate than the triple regimen combining vonoprazan, amoxicillin, and clarithromycin (p < 0.05). Therefore, whether P-CAB drugs can reduce the occurrence of drug resistance may be the future research direction. No studies are comparing the differences in adverse reactions between different regimens combined with various drug types. Therefore, multi-center RCTs with large samples can be carried out in the future to compare the incidence of adverse reactions of different regimens combined with various drug types, or animal experiments can be carried out to explore the molecular biological mechanisms of different regimens combined with various drug types in treating Hp.
The present study compared the eradication rate of P-CAB-based regimens and PPI-based regimens in eradicating Hp. Although the control group had different PPI drugs, studies have shown no differences in treating Hp with regimens based on omeprazole, lansoprazole, or rabeprazole. This study is the first to compare the eradication effects of P-CAB drugs including vonoprazan-based and tegoprazan-based eradication regimens. For the first time, this study compared different eradication regimens and different courses of eradication. However, this study still has some limitations: (1) Only a very few of the included studies are double-blind RCTs, and most are open-label studies, without blind methods or distribution concealment, and this may affect the reliability of the results. It is suggested that the research plan should be formulated and implemented in strict accordance with the SPIRIT statement in the future to effectively control bias. (2) Since there were only two included studies for tegoprazan and no evidence of direct comparison between tegoprazan and vonoprazan, the reliability of the inferences made about the drugs in this study might be reduced. Therefore, we recommend that more studies on tegoprazan be conducted in the future. (3) The safety of regimens is affected not only by P-CAB and PPI drugs but also by other factors such as antibiotics. However, this study has limitations due to the limited data of the included studies, making it impossible to evaluate the safety of each regimen with the limited evidence. It is necessary for future studies to further explore the safety of different regimens.
Conclusion
To sum up, we recommend the V-Tri-2w regimen as the first choice in clinical decision-making and the vonoprazan-based binary regimen as appropriate according to the adverse reaction tolerance of patients. Due to the limitations of the study, clinicians should be cautious in interpreting the ranking results based on clinical practice when choosing drugs. In the future, high-quality multicenter, double-blind RCTs on vonoprazan-based triple regimen should be carried out to validate the reliability of our results and provide a basis for clinical drug selection. In addition, since vonoprazan and tegoprazan have come into the market for just a short time, close attention should be paid to the adverse events in their clinical application in the future.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241241223 – Supplemental material for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848241241223 for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis by Yutong Jiang, Rongrong Zhang, Yuxuan Fang, Ruixia Zhao, Yu Fu, Pingping Ren, Qingqing Zhan and Mingyi Shao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848241241223 – Supplemental material for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis
Supplemental material, sj-docx-2-tag-10.1177_17562848241241223 for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis by Yutong Jiang, Rongrong Zhang, Yuxuan Fang, Ruixia Zhao, Yu Fu, Pingping Ren, Qingqing Zhan and Mingyi Shao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848241241223 – Supplemental material for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis
Supplemental material, sj-docx-3-tag-10.1177_17562848241241223 for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis by Yutong Jiang, Rongrong Zhang, Yuxuan Fang, Ruixia Zhao, Yu Fu, Pingping Ren, Qingqing Zhan and Mingyi Shao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848241241223 – Supplemental material for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis
Supplemental material, sj-docx-4-tag-10.1177_17562848241241223 for P-CAB versus PPI in the eradication of Helicobacter pylori: a systematic review and network meta-analysis by Yutong Jiang, Rongrong Zhang, Yuxuan Fang, Ruixia Zhao, Yu Fu, Pingping Ren, Qingqing Zhan and Mingyi Shao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
