Abstract
Background:
The association between inflammatory bowel disease (IBD) activity and poor sleep quality is reported. However, most research subjectively investigated this issue and lacked long-term follow-up.
Objectives:
Our study aimed to investigate the prevalence of sleep disturbance in IBD patients across disease activity and evaluate the long-term correlation between disease activity, sleep quality, and quality of life.
Design:
This prospective observational study assessed sleep quality in patients with IBD.
Methods:
Patients with IBD were categorized into groups based on clinical activity scores. The sleep questionnaire (Pittsburgh Sleep Quality Index (PSQI)) and IBD questionnaire (IBDQ) were evaluated monthly for 12 months. Seven-day sleep data from wrist actigraphy (Actiwatch®) were collected at initiation and completion. Longitudinal correlation was analyzed.
Results:
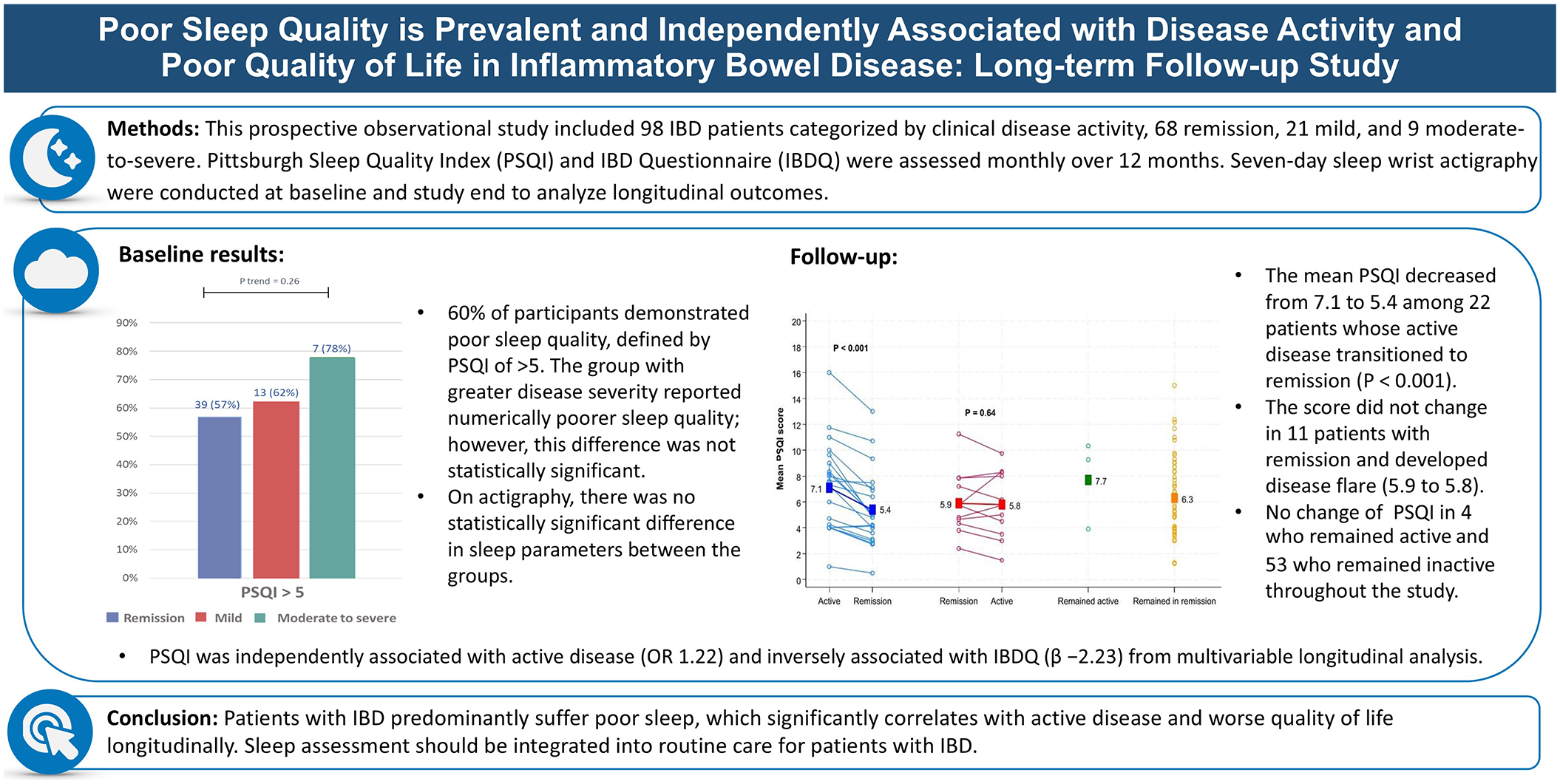
A total of 98 participants were enrolled, consisting of 68 remission, 21 mild, and 9 moderate-to-severe disease activities. At baseline, 60% of participants demonstrated poor sleep quality, defined by a PSQI of >5. The group with greater disease severity reported numerically poorer sleep quality; however, this difference was not statistically significant. On actigraphy, there was no statistically significant difference in sleep latency, wake after sleep onset, or sleep efficiency between the groups. During follow-up, 90 patients responded to questionnaires. The mean PSQI decreased from 7.1 to 5.4 among 22 patients whose active disease transitioned to remission (p < 0.001). However, the score did not change in 11 patients with remission and developed disease flare (5.9–5.8). The mean PSQI was 7.7 and 6.3 in 4 and 53 patients whose disease remained active and inactive during follow-up, respectively. Multivariable longitudinal analysis revealed that PSQI was independently associated with active disease (odds ratio = 1.22) and inversely associated with IBDQ (β = −2.23). Sleep latency was evaluated by PSQI, and actigraphy was significantly correlated.
Conclusions:
Patients with IBD frequently experience poor sleep quality, which significantly correlates with active disease and worse quality of life longitudinally.
Plain language summary
Inflammatory bowel disease (IBD) can lead to poor sleep quality, but most studies have only looked at sleep over short periods. In this study, we divided 98 people with IBD into three groups based on how active their disease was: remission, mild, and moderate-to-severe and followed them for one year to better understand the connection between disease activity and sleep quality. We used both a sleep questionnaire (Pittsburgh Sleep Quality Index or PSQI) and a wrist device (Actigraphy) to monitor their sleep. At the beginning of the study, 60% of participants had poor sleep. Over the course of the study, 90 participants continued to provide data. Among patients whose IBD improved and went into remission, their sleep also improved, with PSQI scores dropping from 7.1 to 5.4. However, for those whose disease flared up, there was no significant change in sleep quality. We found that poor sleep was closely linked to active IBD and worsened quality of life. Overall, people with IBD often experience poor sleep, especially when their disease is active. Poor sleep also negatively affects their quality of life, highlighting the importance of managing both IBD symptoms and sleep problems.
Introduction
The International Organization for the Study of Inflammatory Bowel Disease (IOIBD) has recently incorporated restoring quality of life (QoL) and reducing disability as one of the formal long-term treatment targets in STRIDE II. 1 This acknowledgment emphasizes the importance of addressing well-being concerns, including sleep disturbance in patients with inflammatory bowel disease (IBD). The association between IBD and sleep may be bidirectional. Normal sleep patterns are frequently disrupted due to exacerbated abdominal pain and nocturnal diarrhea in patients with active IBD. Conversely, several studies indicated that sleep deprivation may act as a risk factor for active IBD inflammation, potentially associated with the increasing inflammatory cytokines.2–5 Both patients with active and inactive IBD reported poor sleep quality, with a noteworthy correlation between disease activity.6–13 The prevalence of poor sleep quality based on sleep questionnaires in previous cross-sectional studies was 50%–100% in active disease and 13%–72% in inactive disease.6–10,14–19 Various approaches for sleep evaluation have been utilized, mostly with subjective measures, such as validated sleep questionnaires (e.g., Pittsburgh Sleep Quality Index (PSQI), Patient-reported Outcomes Measurement Information System (PROMIS)),9,20–22 whereas some studies used objective tools, including polysomnography23–25 or actigraphy.7,26 The results have been conflicting possibly due to studied populations, different measures, and interest outcomes. Some studies revealed a significant association between poor sleep quality and disease activity,6,8,9,16,18,27 whereas others reported no significant association.14,15,17,19,26 Furthermore, most studies are cross-sectional.
Sleep quality is a dynamic parameter that fluctuates over time and is influenced by factors such as disease activity and lifestyle changes. Long-term follow-up with repeated measurements enables a more accurate assessment of sleep patterns and the temporal association with IBD activity, capturing trends, fluctuations, and the interplay between disease severity and sleep quality. However, to date, only one long-term study has been conducted, relying solely on subjective measures without incorporating objective assessments. 22 Our study aimed to investigate the prevalence of sleep disturbance in patients with IBD across different disease activity levels using subjective and objective methods. In addition, we evaluated the long-term correlation between disease activity, sleep quality, and QoL to guide the care of patients with IBD.
Materials and methods
This prospective observational study was conducted at Siriraj Hospital, Bangkok, Thailand, from February 2021 to November 2023. All participants signed written informed consent. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 28
Participants
Eligible criteria were individuals aged ⩾18 years, diagnosed with IBD, either ulcerative colitis (UC) or Crohn’s disease (CD), based on compatible clinical, biochemical, endoscopic, and histologic findings. Participants routinely attended follow-up appointments at Siriraj Hospital IBD clinic, and the diagnosis was confirmed by at least partial responses to IBD treatments. Exclusion criteria were patients with non-IBD-related conditions that may affect sleep quality, including (1) patients with known psychiatric diseases, including major depressive disorder, (2) patients unable to withhold sedative drugs, (3) pregnant patients, (4) patients with obesity having a body mass index (BMI) of >35 kg/m2, and (5) patients who refused to provide consent or had cognitive impairment.
Study protocol
Eligible participants were selectively recruited from the IBD outpatient clinic. A psychologist (L.T.) used the Mini-International Neuropsychiatric Interview 30 in the Thai version to screen for psychiatric conditions before enrollment. A psychiatrist (K.W.) further assessed those with possible significant psychiatric diseases who were then excluded from the study if the significant psychiatric diseases were confirmed. The risk for obstructive sleep apnea (OSA) was assessed using the STOP-Bang questionnaire (score range: 0–8), with a score ⩾5 indicating a high risk for OSA. 29
Demographic and clinical data were collected at enrollment, including disease activity assessment using the Harvey–Bradshaw index (HBI) 31 for CD and Mayo clinical score for UC. 32 HBI was categorized into 0–4, 5–7, and ⩾8 for remission, mild, and moderate-to-severe CD, respectively. Similarly, Mayo clinical score was categorized into 0–1, 2–4, and ⩾5 for remission, mild, and moderate-to-severe UC. Serum C-reactive protein (CRP) and fecal calprotectin (EliA Calprotectin 2, Phadia AB, Uppsala, Sweden) were collected to ensure the disease activity. In addition, endoscopy was performed on the patients who agreed. A simple endoscopic score for CD (SES-CD) 33 and Mayo endoscopic subscore (MES) 32 were used for CD and UC, respectively. Sleep quality was subjectively evaluated with the Thai version of PSQI34,35 and objectively assessed by a 7-day actigraph (Actiwatch®, CamNtech, Cambridge, UK). Mental health status was investigated through the Thai version self-questionnaire, the Depression Anxiety Stress Scale (DASS-21). 36 The IBD Questionnaire (IBDQ) was used to assess the QoL. 37
Participants were assessed monthly for PSQI and QoL through computer-based questionnaires during a 1-year follow-up. Clinical activities were assessed following standard clinical practice—usually every 4 weeks in active and every 12 weeks in inactive state. Serum CRP, fecal calprotectin, and wrist actigraphy were reassessed at the end of the study except for patients who transitioned from remission to active disease, in which the serum CRP, fecal calprotectin, and wrist actigraphy were collected during clinical flare.
Sleep assessments
Sleep quality evaluation involves both subjective and objective methods. Subjective measurement included participants who completed the Thai version of PSQI, which is a validated self-sleep questionnaire. The PSQI consists of 19 self-rated questions that evaluated seven sleep components, including sleep duration, sleep latency, sleep disturbance, habitual sleep efficiency, subjective sleep quality, use of sleeping medications, and daytime dysfunction, with a total score of >5 indicating poor sleep quality.
Objective sleep measurement used wrist actigraphy, with participants wearing an actigraph on their non-dominant wrists for seven consecutive nights at home. The actigraph used a piezoelectric accelerometer to record the integration of movement intensity, amount, and duration in all directions. The epoch was scored as wake if the number of counts exceeds the threshold; otherwise, the epoch was scored as sleep. Participants were instructed to document their sleep diaries, noting moments when the light turned on or off, and to apply the device’s button at the beginning and end of sleep. This study used Actiware® (Mini Mitter, Respironics company), the Windows®-based software, to retrieve clinical data from devices and analyze sleep parameters. The terms defined sleep quality consist of time in bed, total sleep time, sleep onset latency, sleep efficiency, and wake after sleep onset (WASO). Figure 1 explains definitions. Software technicians, confirmed by sleep physicians, cross-referenced data with sleep diaries to ensure software accuracy and error detection.
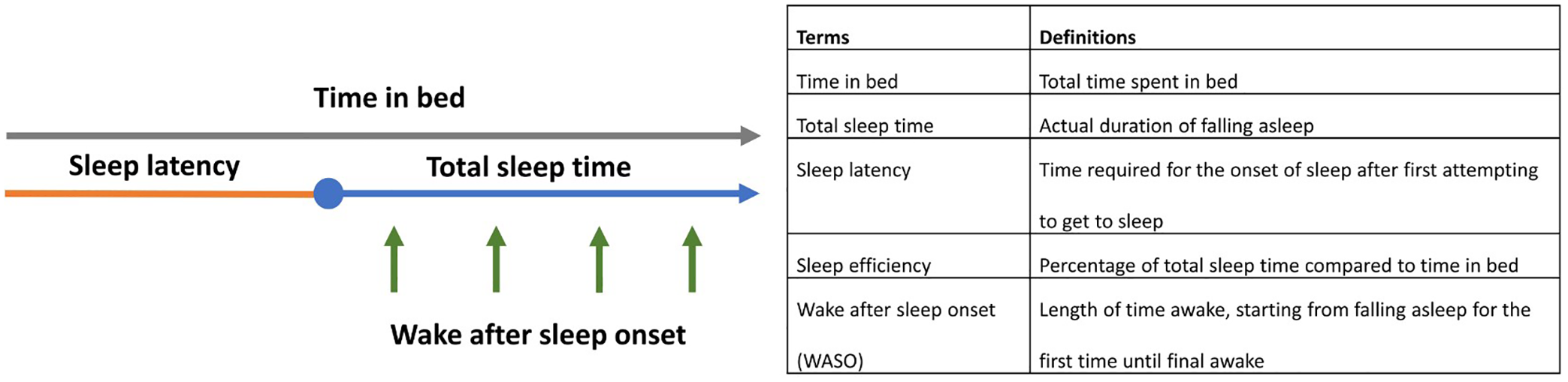
Terms and definitions for sleep quality used in this study.
Data analysis
At enrollment, we compared the mean PSQI, the proportion of participants who had PSQI of >5, and the value of each parameter from actigraphy among the group with remission, mild, and moderate-to-severe disease activities.
The participants were classified into four groups during follow-up according to clinical disease activity, including the groups (1) with active disease and could obtain disease remission, (2) with inactive disease who developed disease flare, (3) with active disease that remained, and (4) with inactive disease that remained. The mean PSQI during active and inactive disease was compared in individuals in groups 1 and 2. Furthermore, longitudinal correlations between PSQI and disease activity and between PSQI and QoL were assessed.
Sample size calculation
A study by Sobolewska-Wlodarczyk et al. 9 found that 78% of clinically active IBD patients and 35% in remission reported poor sleep quality. To determine the sample size of our study, we employed the proportion comparison sample size formula using a statistical power of 80% and a significance level of 0.05, adjusting for multiple comparisons to an alpha of 0.025. Assuming a ratio of 2:1:1 for remission, mild, and moderate-to-severe clinical disease activity, 44 participants for the remission group and 22 for both the remission and moderate-to-severe groups were required. We increased these numbers by 10% for potential dropouts, resulting in sample sizes of 48, 24, and 24, respectively.
Statistical analysis
Continuous variables were presented in median and range or mean ± standard deviation as appropriate, whereas categorical variables were expressed as the number of participants and percentages. Standard comparison methods, including one-way analysis of variance with multiple comparison tests or the Kruskal–Wallis test for continuous data and the chi-square test for non-continuous data, were used for baseline comparison among patients with different disease severities. The chi-square test for trend was utilized to compare the proportion of participants with poor sleep quality. The paired t-test was used to compare the mean PSQI within individuals with disease activity change during follow-up. The logistic mixed model and linear mixed model with random intercept by the patient were performed for longitudinal correlations between PSQI and disease activity and between PSQI and QoL, respectively. A two-tailed p-value of < 0.05 indicated significance for all analyses. SAS for academic (SAS Institute Inc., North Carolina, USA) and Stata MP/18 (StataCorp, 2023, College Station, TX, USA) were used for all analyses.
Results
Baseline characteristics
We enrolled 100 participants with IBD during the study period. Two individuals were subsequently excluded due to a suspicion of major depressive disorder in a psychological interview. Among 98 participants (48 CD and 50 UC), 68 (69.4%) were in remission, 21 (21.4%) had mild, and 9 (9.2%) had moderate-to-severe disease activities. The number of participants in the active disease groups was fewer than our plan despite data collection over 2 years, including during the COVID-19 pandemic. Table 1 shows baseline demographic data stratified by disease activity. The average age was 46.2 years, with 55% being male. Age, gender, and smoking history were comparable across all groups. Participants with moderate-to-severe disease activity exhibited a trend of lower BMI compared to the less severe groups (p = 0.08). There were 8 (11.8%) in the remission group and 3 (14.3%) in the mild group, identified as high risk of OSA based on the STOP-Bang scores. The disease phenotypes, including location and behavior, were comparable among each group. A significantly higher rate of concurrent corticosteroid use was found in the mild compared to the remission group (p < 0.001), and also in the moderate-to-severe compared to the remission group (p < 0.001), whereas no difference was observed in the rate of biologic usage. Significantly lower hemoglobin and albumin levels were observed in the mild group compared to the remission group (p = 0.03 and p < 0.001, respectively), as well as in the moderate-to-severe group compared to the remission group (p < 0.001 for both). Similarly, higher serum CRP and fecal calprotectin levels were noted in the mild group compared to the remission group (p = 0.004 and p < 0.001, respectively), and the moderate-to-severe group also showed elevated levels compared to the remission group (p = 0.08 for CRP, and p < 0.001 for fecal calprotectin). Endoscopic assessment was available in 85 (87%) patients. A significant difference in MES was observed within the UC subgroup (p < 0.001), while no significant difference in SES-CD was found in the CD subgroup (p = 0.13). QoL, assessed by the IBDQ, demonstrated a significant decline corresponding to increasing disease activity (p < 0.001). Mental health assessment using DASS-21 revealed no significant differences in anxiety, depression, and stress across each group.
Baseline demographic data of the cohort categorized by disease activity at enrollment.

5-ASA, 5-aminosalicylic acid; DASS-21, Depression Anxiety Stress Scale; IBDQ, Inflammatory Bowel Disease Questionnaire; SD, standard deviation; SES-CD, Simple Endoscopic Score for Crohn’s Disease.
Subjective and objective sleep assessment at baseline
The PSQI was evaluated at baseline to identify the quality of sleep stratified by IBD activity. Figure 2(a) shows the mean PSQI in each group of participants following their disease activity status. PSQI scores >5 were found in 59 (60%) participants, 39/68 (57%) in remission, 13/21 (62%) in mild, and 7/9 (78%) in the moderate-to-severe group. However, there was no statistical significance between the three groups (p = 0.26). Acknowledging the discordance between clinical disease activity and objective assessment, we evaluated sleep quality in patients within the remission group who had subclinical disease activity, defined by either endoscopic evidence (SES-CD > 2 or MES > 1) or elevated biochemical markers (fecal calprotectin > 150 g/kg or CRP > 5 mg/L) if endoscopy was unavailable. There was no significant difference in poor sleep quality (PSQI > 5) between those with and without subclinical disease activity (59% vs 54%, p = 0.69). Wrist actigraphy data were available in 85 patients because the data retrieval failed for nine patients, and four patients did not wear the actiwatch for the required duration. As shown in Figure 2(b), the moderate-to-severe group demonstrated numerically longer sleep latency, more frequent awakenings after sleep, and lower sleep efficiency than the less severe groups; however, these differences were not statistically significant.

Sleep parameters were evaluated by sleep questionnaires (a) and wrist actigraphy (b) in participants with IBD in remission, mild, and moderate-to-severe disease activity.
Longitudinal evaluation of sleep quality and health-related QoL
A total of 90 participants underwent monthly assessments and had complete 12-month follow-up data. The groups with mild and moderate-to-severe disease activity at baseline were re-defined as the active group to facilitate comparisons, comprising 26 participants, while the remission or inactive group at baseline included 64 participants. As shown in Figure 3(a), during follow-up, 22 of 26 patients with active disease achieved clinical remission posttreatment, with the median time to remission of 12 (4–44) weeks. The mean PSQI in this group was 7.1 ± 3.5 while the disease was active and significantly decreased to 5.4 ± 2.9 after remission (p < 0.001). Of 64 patients with inactive disease, 11 developed disease flares during the study period, with the median time to flare of 24 (16–44) weeks. The mean PSQI in this group was 5.9 ± 2.4 and 5.8 ± 2.6 before and after flare (p = 0.64), respectively. There were 4 (4.4%) participants who remained active, whereas 53 (58.9%) remained in remission throughout the study, with mean PSQI scores of 7.7 ± 2.8 and 6.3 ± 3.1, respectively. Longitudinal trends in PSQI scores across different disease activities over time are demonstrated in Figure 3(b), which showed the durability of sleep improvement in patients who transitioned from active to inactive disease during follow-up. Furthermore, the persistently inactive group demonstrated relatively lower PSQI scores and less fluctuations compared to the persistently active group throughout the follow-up period. Multivariate long-term analysis revealed that PSQI was independently associated with active disease (odds ratio: 1.22, 95% CI: 1.07–1.40, p = 0.003) after adjusting for age, gender, disease type, and corticosteroid use. Furthermore, PSQI was significantly inversely correlated with IBDQ (β = −2.23, 95% CI: −2.81 to −1.65, p < 0.001) after adjusting for age, gender, disease type, disease activity, and corticosteroid use (Table 2).

(a) Mean PSQI scores during long-term follow-up in each group of patients categorized by disease activity status, highlighting sleep quality changes following transitions in disease status; squares represent the group means, while each dot and line represents an individual patient’s data. (b) Longitudinal trends in PSQI scores across different disease activities over time.
Long-term multivariable analysis for the correlation between PSQI and disease activity a , as well as PSQI and IBDQ.
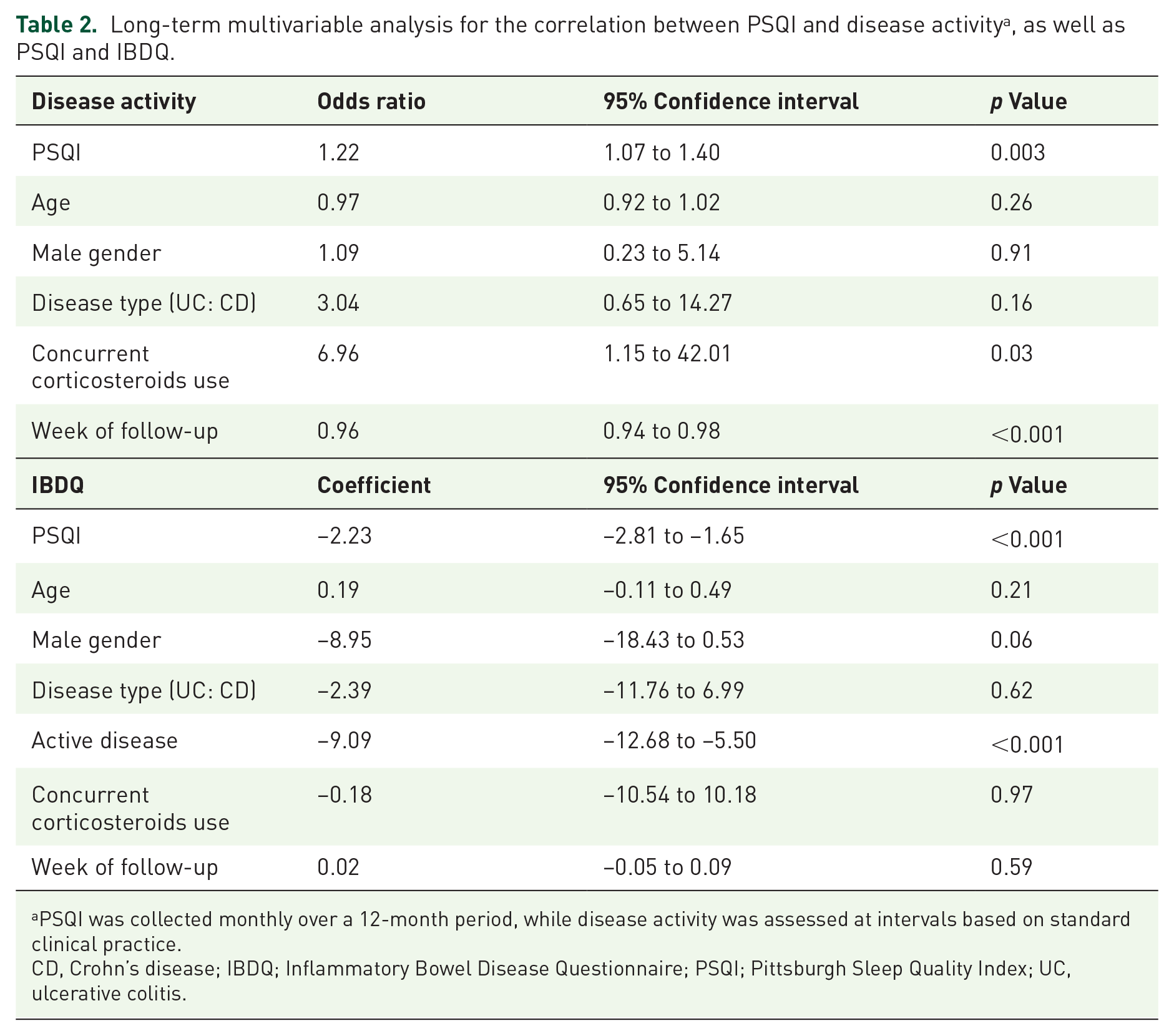
PSQI was collected monthly over a 12-month period, while disease activity was assessed at intervals based on standard clinical practice.
CD, Crohn’s disease; IBDQ; Inflammatory Bowel Disease Questionnaire; PSQI; Pittsburgh Sleep Quality Index; UC, ulcerative colitis.
Sleep actigraphy data after the study were available in 73, including active to remission in 20, remission to active in 10, remained active in 3, and remained in remission in 40. No significant changes in sleep parameters were observed between the first and second actigraphy measurements in any group, whether participants experienced a flare-up or achieved remission. Supplemental Table S1 in the supplementary document describes the details. The long-term correlation of the same parameters, including sleep latency and sleep efficiency, measured by PSQI and Actiwatch, was conducted. The results reveal a significantly positive correlation in sleep latency measured by Actiwatch and PSQI (β = 6.59, 95% CI: 0.27–12.91, p = 0.041). However, no correlation was observed in sleep efficiency (β = 0.18, 95% CI: −1.23 to 1.60, p = 0.801).
Longitudinal PSQI score of the patients who developed disease flare
To assess whether poor sleep quality triggered disease flare, 11 patients in remission at baseline and developed clinical flare during follow-up were evaluated. As shown in Figure 4, the PSQI scores did not increase before the disease became active.

Longitudinal PSQI scores of 11 patients in remission at baseline and developed clinical flare during follow-up. Blue dots represent the disease remission stage, whereas red dots represent the active stage. The number on top of each graph indicates the patient identification number.
Discussion
Our study is the first to subjectively and objectively assess sleep quality and disease activity with long-term follow-up. We found that poor sleep quality was prevalent, even in those with disease remission. Based on subjective assessment, the group with greater disease severity reported numerically poorer sleep quality; however, this difference was not statistically significant. The PSQI was significantly correlated with disease activity in longitudinal follow-up. However, we did not find that poor sleep quality triggered disease flares in participants who were initially in remission but later developed clinical flares. Nonetheless, the number of participants in this group was small. Importantly, our study revealed that poor sleep quality was significantly associated with poor QoL in longitudinal follow-up.
Poor sleep quality is common in IBD, with our study showing a prevalence of 60% based on a PSQI score >5. However, no significant differences were observed between the remission, mild, and moderate-to-severe groups. This is consistent with findings from previous cross-sectional studies that have subjectively assessed sleep quality, with some studies reporting an association between poor sleep quality and increased disease activity. Furthermore, the meta-analysis in 2022 reveals a higher prevalence of poor sleep in patients with inactive IBD compared to controls. 38 These data highlight the high prevalence of sleep problems evaluated by questionnaires among patients with IBD.
Our study selected wrist actigraphy for objective sleep assessment, considering its ability to monitor sleep at home, which is a familiar environment for the patients, for many days. Previous studies using wrist actigraphy reported inconsistent results. Some studies revealed significantly longer WASO and lower sleep efficiency in patients with active disease compared to control 26 or inactive disease. 8 Some studies demonstrated pronounced differences only in moderate-to-severe disease, without differences in mild disease and remission. 7 Some studies revealed no significant differences between patients and control and between patients with active and inactive disease.6,15 In our study, patients with moderate-to-severe disease demonstrated numerically longer WASO and sleep latency and lower sleep efficiency. However, the differences were not statistically significant.
The data on the long-term association between poor sleep quality and IBD activity are limited. Only the study by Sofia et al. 22 repeatedly evaluated sleep by questionnaire before our study and revealed a significant moderate correlation between the change in PSQI and HBI during follow-up. 22 Our study confirms these results. PSQI was longitudinally correlated with disease activity status after adjusting for age, gender, disease type (UC vs CD), corticosteroid use, and week of follow-up. This result further emphasizes the correlation between IBD disease activity and poor sleep quality. Furthermore, we revealed that poor sleep quality could be improved in patients with active disease by turning the disease into remission. This indicates another benefit of effective IBD treatment.
Not only correlation but also poor sleep quality may trigger IBD disease flare-ups. Poor sleep quality has been reported to activate the immune response39–41 and trigger disease flare-ups in many autoimmune conditions, such as systemic lupus erythematosus and rheumatoid arthritis, including IBD.11,14,22,42,43 Ananthakrishnan et al. 11 analyzed data from the Crohn’s and Colitis Foundation of America Partners study, which is a longitudinal and Internet-based cohort, and revealed that the participants with CD who had impaired sleep demonstrated a twofold increase in the risk of active disease at 6 months. This effect was not found in participants with UC. Uemura et al. 14 performed a 1-year follow-up study on 136 patients with IBD and revealed that 61% of patients with poor sleep quality, defined by PSQI at baseline, developed disease flare, while the rate was 34% in those with good sleep quality. Sofia et al. 22 conducted a 1-year follow-up study on 92 patients with CD and revealed that a PSQI of > 8 at baseline predicted surgery or hospitalization, with a hazard ratio of 5.37 during follow-up. Leal et al. 16 performed a 6-month follow-up study in 205 patients with IBD and revealed that PSQI of > 5 at baseline was independently associated with composite poor outcomes. However, Calvo et al. 44 revealed no association between baseline PSQI and the requirement for treatment optimization during follow-up. Uemura et al., 14 Sofia et al., 22 and Leal et al. 16 revealed that poor sleep quality triggers disease flare in long-term follow-up, but these three studies focused on sleep quality at baseline. Sleep can change over time; thus, we approached this problem by assessing patients’ sleep quality monthly. We considered the overall sleep quality and the closest point before developing a clinical flare to reveal poor sleep in patients before flaring. However, we did not detect this effect. Nonetheless, our cohort included 64 patients with disease in remission at baseline, and only 11 patients developed flare during follow-up. The effect of poor sleep quality as a trigger for disease flare warrants further investigation.
Many studies have revealed a cross-sectional association between poor sleep quality and poor QoL.8,14,17,18 Notably, our study is the first to reveal a significant longitudinal correlation between poor sleep quality and poor QoL. Multivariate longitudinal analysis revealed that PSQI exhibited an inversely significant correlation with IBDQ after adjusting for age, gender, disease type, disease activity, and corticosteroid use. These results further confirm the importance of sleep problems in patients with IBD.
The strength of this study is that this is the first prospective study to repeatedly subjectively and objectively evaluate sleep quality and disease activity with long-term follow-up. Our study confirmed the disease activity status by demonstrating lower hemoglobin and albumin levels, higher fecal calprotectin levels, and higher endoscopic activity scores in the group with more severe clinical disease activity. In addition, we confirmed sleep quality by wrist actigraphy and repeated sleep questionnaire assessment monthly. Longitudinal follow-up design helped us demonstrate the long-term association between poor sleep quality, active disease, and poor QoL and revealed that turning active IBD to remission helped to improve sleep quality, thereby accentuating the benefit of IBD treatment.
Our study has some limitations that should be acknowledged. First, it was a single-center study, and the results may not have been generalized to other populations with different backgrounds. In addition, some wrist actigraphy data were missing. Furthermore, the sample size, particularly in the active disease group, was relatively low, potentially impacting the overall results. Moreover, our reliance solely on clinical disease activity measures might overlook patients with subtle disease activity indicated by biochemical elevations or higher endoscopic scores, which do not align with the assigned severity group. Next, while our study excluded individuals with major depressive disorder as our objective was to investigate the association between sleep quality and IBD activity specifically, this may have inadvertently excluded some IBD patients who also experience poor sleep quality. However, only two patients failed screening due to psychiatric issues. Lastly, while wrist actigraphy is a useful ambulatory tool for assessing sleep quality, it has limitations. Actigraphy may have reduced accuracy in measuring sleep stages and can be influenced by movement, making it less reliable compared to polysomnography, the gold standard method for sleep assessment, providing detailed information on sleep architecture, respiratory function, muscle activity, and other vital parameters. In addition, given that some patients in this study were at high risk for OSA and sleep quality could be affected, confirmation with polysomnography may be warranted.
Conclusion
Our study revealed that poor sleep quality is common among patients with IBD, even in clinical remission, and is longitudinally independently associated with disease activity and poor QoL. Treating active IBD to remission improved sleep quality. Sleep should be evaluated in routine patients with IBD care. Future interventional studies to improve sleep quality, which should help improve QoL and possibly reduce flare, are warranted.
Supplemental Material
sj-doc-1-tag-10.1177_17562848241311142 – Supplemental material for Poor sleep quality is common and independently associated with disease activity and poor quality of life in inflammatory bowel disease: a long-term follow-up study
Supplemental material, sj-doc-1-tag-10.1177_17562848241311142 for Poor sleep quality is common and independently associated with disease activity and poor quality of life in inflammatory bowel disease: a long-term follow-up study by Thanaboon Chaemsupaphan, Chawanont Pimolsri, Nichcha Subdee, Phutthaphorn Phaophu, Marianee Salaemae, Parinya Permpim, Lakkana Thongchot, Kamonnet Wannasewok, Wattanachai Chotinaiwattarakul and Julajak Limsrivilai in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the Faculty of Medicine, Siriraj Hospital, Mahidol University, Thailand for the generous financial support, which made this study possible. We gratefully acknowledge Pimrapat Gebert, Institute of Biometry and Clinical Epidemiology, Charité—Universitätsmedizin Berlin, Germany, for outstanding support in statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
