Abstract
Background:
Immune checkpoint inhibitor (ICI)-induced colitis is a significant adverse event associated with ICI therapy, known to be linked to increased cytotoxic T-cell activity.
Objectives:
To compare T-cell subsets based on the endoscopic features of ICI-induced colitis and to compare these findings with those of inflammatory bowel disease (IBD).
Design:
Prospective cohort study.
Methods:
We analyzed patients with ICI-induced colitis, confirmed through both endoscopic and histological evaluation. Biopsy specimens were examined using multiplex immunohistochemistry to assess their immune cell profile. Clinical outcomes were analyzed. Immune cell profiles were compared based on their endoscopic features and contrasted with those of patients with IBD.
Results:
Seventeen patients with ICI-induced colitis were included in the study. All patients showed clinical improvement after treatment, and steroids were administered to 11 patients (64.7%). Based on endoscopic features, the patients were classified as Crohn’s disease (CD)-like (n = 3, 17.6%), ulcerative colitis (UC)-like (n = 9, 52.9%), or microscopic colitis (MC)-like (n = 5, 29.4%). In ICI-induced colitis, cytotoxic T cells (Tc cells) were more predominant than helper T cells (Th cells) (p = 0.053), and this trend was most pronounced in the MC-like subtype (p = 0.020). When comparing the number of CD8+ cells infiltrating the crypts, both the UC-like and MC-like subtypes had significantly more infiltrating cells than the CD-like subtype (p = 0.008 and p = 0.016, respectively). In comparison to IBD, IBD exhibited a Th-dominant profile, whereas CD-like ICI-induced colitis had a lower Th cell density than CD (p = 0.032) and UC-like ICI-induced colitis had a higher Tc density than UC (p = 0.045).
Conclusion:
Analysis of T-cell subsets of ICI-induced colitis revealed a Tc-dominant profile, contrasting with the Th dominance observed in patients with IBD. The Tc-dominant profile was evident in UC-like and MC-like subtypes, with significant crypt infiltration by CD8+ cells. Tc may play an important role in the pathophysiology of ICI-induced colitis.
Introduction
Immune checkpoint inhibitors (ICIs) are medications that target specific proteins, such as cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) or programmed cell death protein-1 (PD-1)/programmed cell death ligand 1 (PD-L1). They work by preventing cancer cells from evading T-cell responses, thereby enhancing the immune response of the patient and exerting anti-neoplastic effects.1,2 These therapies are being used to treat an increasing range of malignancies, changing the way cancer is treated.3,4
Immune-related adverse events are a range of inflammatory side effects that can occur in various organs as a result of increased immune system activity from using ICIs. 5 One of the most common immune-related adverse events is colitis, which can significantly impact a patient’s quality of life. In severe cases, this condition may even require the delay or discontinuation of ICI therapy. 6 The pathophysiological mechanisms of ICI-induced colitis and inflammatory bowel disease (IBD) may be similar, involving common types of T cells and inflammatory cytokines. 7 Therapeutic strategies commonly employed in IBD, such as administration of corticosteroids, infliximab, and vedolizumab, have demonstrated efficacy in the management of ICI-induced colitis. Pan-JAK inhibitor administration is also emerging as a potential therapeutic approach. 8 Increased activity of CD8+ T cells is considered to play a role in ICI-induced colitis.9 –11 In biopsy samples from patients with ICI-induced colitis, high accumulation of CD8+ T cells, as well as enhanced cytotoxic activity, was observed. 9 Another study provided evidence that CD8+ T cells were significantly elevated in ICI-induced colitis compared to patients with UC or those without colitis associated with ICI therapy. 10
The diagnosis of ICI-induced colitis is established by evaluating the timing of ICI administration, excluding other potential causes of colitis, and considering both endoscopic and histologic findings. ICI-induced colitis presents a range of endoscopic findings, often resembling those of IBD. Normal mucosa can also be observed.12,13 These findings can be classified into Crohn’s disease (CD)-like, UC-like, and microscopic colitis (MC)-like patterns based on their endoscopic appearance. 12 However, there have been no analyses of T-cell profiles based on these endoscopic features. In addition, few studies have compared the T-cell profiles between ICI-induced colitis and IBD.
The objective of this study was to investigate the immune cell profile, specifically T-cell subsets, in patients with ICI-induced colitis. We aimed to determine whether the distribution of these subsets varies according to endoscopic features and to compare them with the T-cell profiles observed in patients with IBD.
Methods
Study population
This study was conducted within a prospective cohort of patients with ICI-induced colitis. We consecutively identified patients diagnosed with ICI-induced colitis at Asan Medical Center between January 2021 and April 2024. We included patients who met the following criteria: (1) diagnosed with malignancy, (2) received treatment with ICIs (CTLA-4 inhibitors and/or PD-1/PD-L1 inhibitors), (3) presented with abdominal pain or diarrhea, (4) diagnosed with ICI-induced colitis through both endoscopic and histological evaluation, and (5) agreed to participate in this prospective cohort study. Patients who (1) did not undergo endoscopy or (2) were not histologically confirmed to have ICI-induced colitis were excluded from the analysis.
Data for this study were collected including patient demographics, type of malignancy, type of ICI used, endoscopic findings, and dates of prescribed medications. After obtaining informed consent for participation in the cohort study, biopsy samples were collected from areas of severe inflammation, which were identified during endoscopy or normal-looking mucosa. Patients were subsequently followed up regularly and prescribed appropriate medical treatment as needed. Based on the review of endoscopies by the authors (M.K.K. and S.W.Hwang), patients were classified into CD-like, UC-like, and MC-like groups according to their endoscopic features. The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement. 14
Multiplex immunohistochemistry
All tissue samples from the included patients were obtained through endoscopic biopsies, with informed consent secured from all participants. Biopsies were conducted at the most active sites identified during endoscopy. For patients without active lesions, biopsies were taken from normal-appearing mucosa. The analysis of multiplex immunohistochemistry (IHC) was based on previous publications from our team.15,16 Tissue samples were obtained from formalin-fixed paraffin-embedded blocks and sectioned into 4 μm-thick slices. Slides were heated for at least 1 h in a dry oven at 60°C, followed by multiplex immunofluorescence staining with a Leica Bond Rx™ Automated Stainer (Leica Biosystems, Nussloch, Germany). Briefly, the slides were dewaxed with Leica Bond Dewax solution (#AR9222; Leica Biosystems, Nussloch, Germany), followed by antigen retrieval with Bond Epitope Retrieval 2 (#AR9640; Leica Biosystems, Nussloch, Germany) for 30 min. The staining proceeded in sequential rounds of blocking buffer (#C0103; TheraNovis, Seoul, Repulic of Korea), followed by primary antibody incubation for 30 min and Mouse/Rabbit HRP 2nd antibody (#C0105; TheraNovis, Seoul, Repulic of Korea) incubation for 10 min. The primary antibodies used for each protein were as follows: anti-CD8 (ab192847; Bio-Rad, Hercules, CA, USA), anti-CD4 (ab133616; Abcam, Cambridge, UK), anti-FOXP3 (ab20034; Abcam, Cambridge, UK), anti-CD20 (Ab9475; Abcam, Cambridge, UK), anti-CD68 (Ab192847; Abcam, Cambridge, UK), and anti-PD-1 (ab137132; Abcam, Cambridge, UK). Visualization of antigen was accomplished using Astra-dye (TheraNovis, Seoul, Repulic of Korea) for 10 min, after which the slide was treated with Bond Epitope Retrieval 1 (#AR9961; Leica Biosystems, Nussloch, Germany) for 20 min to remove bound antibodies. Finally, nuclei were stained with DAPI (62248; Thermo Scientific, Waltham, MA, USA) for counterstaining after the last round of antigen retrieval.
Image acquisition and quantitative data analysis
Multiplex-stained slides were scanned at 20× magnification using a PhenoImager™ HT (Akoya Biosciences, Marlborough, MA, USA) device. The multispectral images were then unmixed in the inForm software using the spectral library. Next, each single cell was segmented and phenotyped based on the expression compartment and intensity of each marker, using DAPI staining as a reference. The analysis was performed on designated regions of interest on the tissue slide, and the same algorithm was applied and batch-run.
Assessment and definition
The immune cells included in the analysis were cytotoxic T cells (Tc cells), helper T cells (Th cells), regulatory T cells (Treg cells), B cells, and macrophages. Tc cells were defined as CD4−/CD8+/FOXP3−/CD20−/CD68− cells, Th cells as CD4+/CD8−/FOXP3−/CD20−/CD68− cells, and Treg cells as CD4+/CD8−/FOXP3+/CD20−/CD68− cells. In addition, PD-1+ cells within each T-cell subset were analyzed. PD-1+ Tc cells were defined as CD4−/CD8+/FOXP3−/CD20−/CD68−/PD-1+ cells, PD-1+ Th cells as CD4+/CD8−/FOXP3−/CD20−/CD68−/PD-1+ cells, and PD-1+ Treg cells as CD4+/CD8−/FOXP3+/CD20−/CD68−/PD-1+ cells. Cell density was calculated as the number of cells per area (cells/mm2). The proportion of each T-cell subset (Tc, Th, and Treg cells) within the total T-cell population was also determined. In addition, the number of CD8+ cells infiltrating the crypt area was counted. The visible crypt area in the field was measured using ImageJ software, and the total number of CD8+ cells infiltrated in crypts was normalized to this surface area. To compare with our ICI-induced colitis cohort, we performed multiplex IHC on samples from 13 patients with IBD, including 5 with CD and 8 with UC, using the same methodology.
Statistical methods
Nonparametric tests were conducted for comparisons. Differences in continuous variables between the two groups were analyzed using the Mann–Whitney U test, while those among the three groups were analyzed using the Kruskal–Wallis test. The median value and interquartile ranges were calculated for each continuous variable. The p-value interpretation was two-tailed, and a p-value of less than 0.05 was considered statistically significant. All statistical analyses were conducted using R version 4.3.0 (R Foundation for Statistical Computing, Vienna, Austria), and the figures were generated using GraphPad Prism (version 10.3.0, Boston, MA, USA).
Results
Study population and clinical outcomes
A total of 25 patients were enrolled in the prospective cohort study. Eight patients were excluded from the analysis for the following reasons: three did not undergo colonoscopy, four underwent colonoscopy but were not histologically confirmed as having ICI-induced colitis, and one had colitis due to consuming herbal material. Therefore, a total of 17 patients were included in the analysis. Most of these patients presented with common terminology criteria for adverse events (CTCAE) grade 2 or higher adverse events, specifically diarrhea (14/17, 82.4%) and colitis (12/17, 70.6%). Non-small-cell lung cancer was the most prevalent malignancy in the cohort, affecting 6 out of 17 patients (35.3%). All patients received a regimen that included a PD-1/PD-L1 inhibitor, with two patients receiving combination therapy (CTLA-4 inhibitor + PD-1 inhibitor). Eleven patients (64.7%) received oral or intravenous steroid therapy (excluding beclomethasone), while six patients (35.3%) were prescribed beclomethasone. Among those treated with beclomethasone, four patients experienced symptom improvement with beclomethasone alone, without the need for additional steroid therapy. Five patients (29.4%) were prescribed 5-aminosalicylate (5-ASA); among them, two patients achieved symptom resolution with 5-ASA alone, while the remaining three ultimately required steroid treatment. Two patients (11.8%) did not receive specific treatment. One patient experienced spontaneous symptom resolution, while the other showed symptom improvement after delaying ICI therapy. Although all patients eventually achieved symptom resolution, one patient experienced an initial improvement followed by a relapse 3 months later, after which symptoms improved following treatment with vedolizumab. Seven patients (41.2%) discontinued ICI due to colitis and were either switched to an alternative regimen or were unable to continue additional anticancer therapy. Table 1 provides a summary of the baseline characteristics of the included patients and their clinical outcomes.
Baseline characteristics and clinical outcomes of included patients.

CD, Crohn’s disease; CTCAE, common terminology criteria for adverse events; CTLA-4, cytotoxic T-lymphocyte associated protein 4; HCC, hepatocellular carcinoma; ICI, immune checkpoint inhibitor; IQR, interquartile range; MC, microscopic colitis; NSCLC, non-small-cell lung cancer; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; SCLC, small-cell lung cancer; UC, ulcerative colitis.
Differences in T-cell profiles according to endoscopic appearances in patients with ICI-induced colitis
Multiplex IHC was performed on 14 samples from 11 patients to categorize them based on their endoscopic findings. The samples were divided into CD-like, UC-like, and MC-like groups. Cell density (cells/mm2) was compared across these groups according to their endoscopic features; however, no statistically significant differences were observed (Table 2).
Comparison of cell density based on endoscopic features in ICI-induced colitis.

CD, Crohn’s disease; ICI, immune checkpoint inhibitor; IQR, interquartile range; MC, microscopic colitis; PD-1, programmed cell death protein 1; UC, ulcerative colitis.
To determine the dominant T-cell subset, we compared the proportions of each T-cell subset in the entire cohort of ICI-induced colitis patients, as well as within each subgroup. We assessed whether there were significant differences in the proportions of Tc and Th cells. Although Tc cells showed a tendency to be more prevalent than Th cells in the overall cohort (p = 0.053), this difference was not statistically significant. Similarly, in the CD-like and UC-like subgroups, Tc cells tended to be less prevalent (p = 0.061) and more prevalent (p = 0.068) than Th cells, respectively; however, neither reached statistical significance. Despite this, in the MC-like subgroup, the proportion of Tc cells was significantly higher compared to Th cells (p = 0.020, Figure 1(a)).

Proportion of each T-cell subset in (a) immune checkpoint inhibitor-induced colitis and (b) inflammatory bowel disease.
We also independently counted and compared the number of intraepithelial CD8+ cells (Figure 2(a)). The number of intraepithelial CD8+ cells within the field was significantly higher in the MC-like group compared to the CD-like group (p = 0.016) and similarly elevated in the UC-like group compared to the CD-like group (p = 0.008). No significant difference was observed between the UC-like and MC-like groups (p = 0.413; Figure 2(b)). In addition, when the intraepithelial CD8+ cells infiltrating into crypts were separately counted, the MC-like and UC-like groups had higher counts than the CD-like group (p = 0.016 and 0.008, respectively). No significant difference was found between the UC-like and MC-like groups (p = 0.730; Figure 2(c)). The relative densities of each cell type, as a proportion of the total cell population across all 14 samples, are presented in Figure 3.

Number of CD8+ cells according to endoscopic features in immune checkpoint inhibitor-induced colitis. (a) Representative multiplex immunohistochemistry image. (b) Number of intraepithelial CD8+ cells per field. (c) Number of intraepithelial CD8+ cells inside each crypt.

Proportion of each type of immune cell in all biopsy specimens included in the analysis.
Comparison of cell density between ICI-induced colitis and IBD
We conducted a comparative analysis of T-cell density between patients with ICI-induced colitis and those with IBD. In general, IBD showed a higher frequency of Th cells compared to Tc cells (p = 0.077). Specifically, in CD, the proportion of Th cells was significantly higher than that of Tc cells (p = 0.006, Figure 1(b)). Although overall cell density did not significantly differ between the entire ICI-induced colitis cohort and the IBD cohort, subtype-specific comparisons revealed that the CD-like ICI-induced colitis group had a lower density of Th cells compared to CD (median 315.2 vs 1051.5 cells/mm2, p = 0.032). In addition, this group also had a reduced density of Treg cells (median 50.4 vs 197.8 cells/mm2, p = 0.008). On the other hand, the UC-like ICI-induced colitis group exhibited a higher density of Tc cells compared to UC (median 391.5 vs 243.9 cells/mm2, p = 0.045; Table 3).
Comparison of cell density between patients with ICI-induced colitis and IBD.
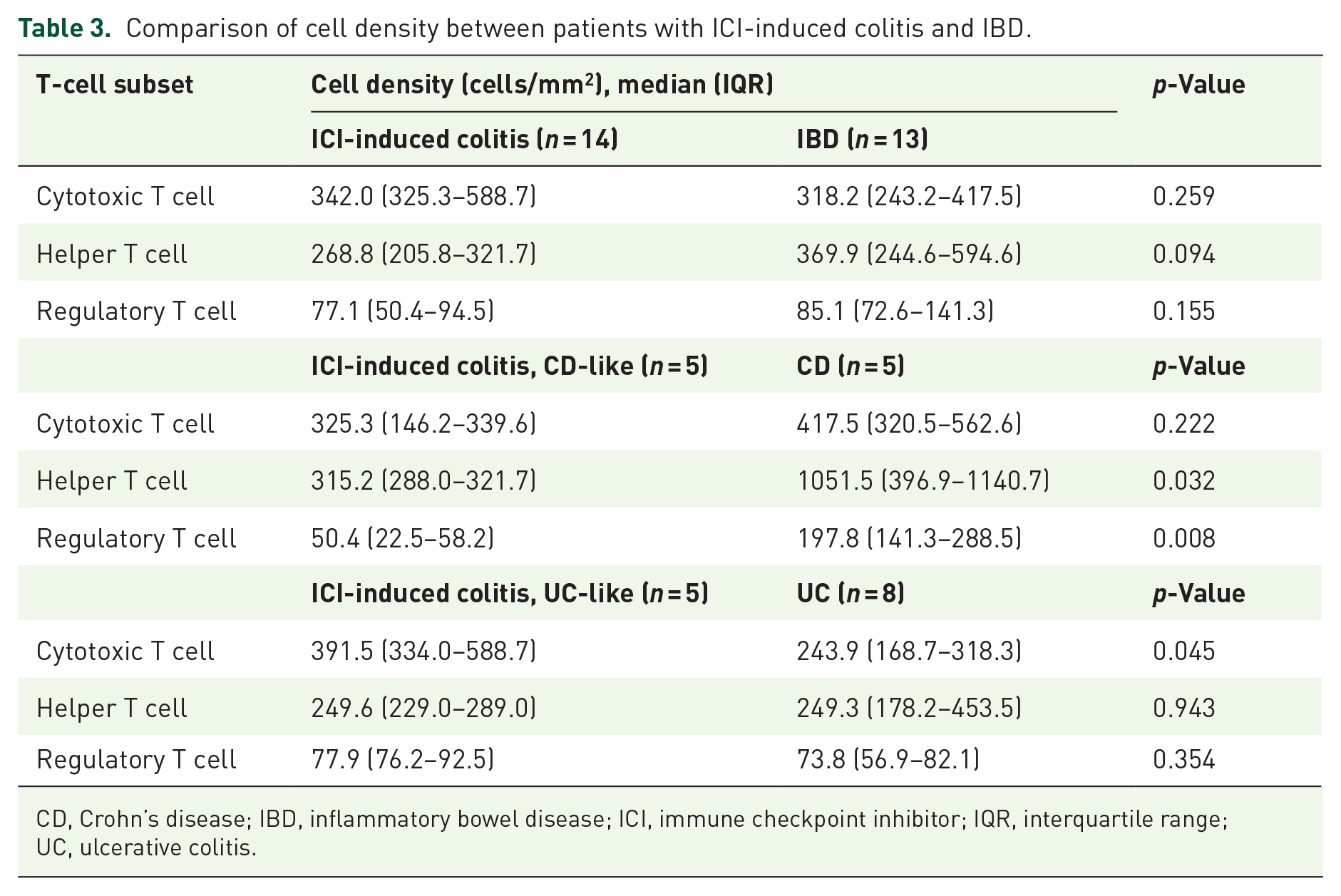
CD, Crohn’s disease; IBD, inflammatory bowel disease; ICI, immune checkpoint inhibitor; IQR, interquartile range; UC, ulcerative colitis.
Discussion
In this study, we first summarized the clinical outcomes of patients with ICI-induced colitis. Second, we compared the immune cell composition within biopsy specimens based on endoscopic features. Although there was no difference in overall cell density, including Tc cells, we observed a higher density of intraepithelial CD8+ cells within the crypts in the UC-like and MC-like groups compared to the CD-like group. When comparing the T-cell subsets in ICI-induced colitis to those in IBD, ICI-induced colitis exhibited a Tc-dominant profile, in contrast to the Th-dominant profile observed in IBD. Specifically, Th cell density was lower in CD-like ICI-induced colitis compared to CD, while Tc cell density was higher in UC-like ICI-induced colitis compared to UC.
ICI-induced colitis typically manifests several weeks to several months after initiation of ICI therapy17,18 and demonstrates a favorable response to corticosteroids, resulting in rapid symptom resolution. Consequently, current clinical guidelines recommend steroids as the first-line treatment for this condition.19 –22 In our study, all patients achieved resolution of symptoms, with or without corticosteroids. This outcome can be attributed to the predominance of patients treated with PD-1/PD-L1 inhibitors, which are associated with a milder presentation of ICI-induced colitis compared to CTLA-4 inhibitors.23,24 Furthermore, the relatively short duration from symptom onset to colonoscopy, with a median of 44 days, resulted in timely therapeutic intervention and contributed to favorable clinical outcomes.
The endoscopic presentation of ICI-induced colitis is heterogeneous. Among the studies reported to date, the study by Abu-Sbeih et al., 12 which included the largest cohort of 182 patients with ICI-induced colitis, classified endoscopic findings into CD-like and UC-like categories. Normal-appearing mucosa can also reveal features of MC upon histopathologic evaluation. 25 However, to our knowledge, no previous studies have compared the immune cell composition within biopsy specimens based on these endoscopic features. Our study demonstrated that within ICI-induced colitis, both UC-like and MC-like subtypes exhibited a predominance of the Tc cell subtype. In addition, intraepithelial CD8+ cells within the crypts were significantly higher in the UC-like and MC-like types compared to the CD-like type. This finding is consistent with previous studies linking ICI-induced colitis to CD8+ T-cell activation. 10 Overall, ICI-induced colitis appears to be driven by CD8+ T cells, with active inflammatory responses occurring within the crypts.
While both ICI-induced colitis and IBD share similarities in terms of endoscopic findings and treatment options, there are key differences between these conditions. One of the main differences is the chronic, relapsing, and progressive nature of inflammation observed in IBD. In patients with IBD, the gastrointestinal mucosa is characterized by an abundance of Th cells, which produce pro-inflammatory cytokines that contribute to the ongoing inflammation. 26 On the other hand, a previous study indicates that patients with ICI-induced colitis have a Tc-dominant profile in their mucosa. 10 In our analysis, we found that this Tc dominance was particularly evident in ICI-induced colitis cases with features resembling MC and UC. In addition, in these two subtypes, significant infiltration of CD8+ cells was observed within the crypts. In ICI-induced colitis, histopathologic changes are known to occur predominantly in the crypts.13,27 –29 Notably, previous studies have reported the presence of CD8+ T cells in the epithelium and lamina propria of specimens taken from MC patients without the use of ICIs. 30
Our study had some limitations which should be acknowledged. First, the analysis was limited by small cohort size, and we did not include control groups such as healthy individuals or ICI-treated patients who did not develop colitis. These limitations restrict the ability to draw definitive conclusions. Second, although we analyzed cell distribution using cell surface markers, we did not thoroughly explore cell-to-cell interactions or analyze inflammatory cytokines. Despite these limitations, this study is notable for being, to the best of our knowledge, the first to investigate the distribution of T-cell subsets in ICI-induced colitis based on endoscopic classifications and compare these findings with specimens from patients with IBD.
Conclusion
In the analysis of T-cell subsets from biopsy specimens of patients with ICI-induced colitis, a dominant Tc feature was identified. This is in contrast to the dominant Th feature observed in patients with IBD. The dominance of Tc cells was particularly pronounced in cases with UC-like and MC-like endoscopic features, where there was clear evidence of significant infiltration of CD8+ cells within the crypts. This suggests that Tc cells may play a central role in the pathophysiology of ICI-induced colitis, with this trend being especially prominent in cases exhibiting MC-like or UC-like endoscopic features.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241309445 – Supplemental material for CD8+ cell dominance in immune checkpoint inhibitor-induced colitis and its heterogeneity across endoscopic features
Supplemental material, sj-docx-1-tag-10.1177_17562848241309445 for CD8+ cell dominance in immune checkpoint inhibitor-induced colitis and its heterogeneity across endoscopic features by Min Kyu Kim, Hye-Nam Son, Seung Wook Hong, Sang Hyoung Park, Dong-Hoon Yang, Byong Duk Ye, Jeong-Sik Byeon, Seung-Jae Myung, Suk-Kyun Yang, Shinkyo Yoon and Sung Wook Hwang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
