Abstract
Background:
The Crohn’s Disease (CD) Activity Index (CDAI), Inflammatory Bowel Disease (IBD) Questionnaire (IBDQ), and IBD-Fatigue (IBD-F) scale are useful patient-reported outcome (PRO) tools for assessing the treatment benefits of vedolizumab (VDZ) beyond clinical trial endpoints in patients with CD.
Objectives:
To assess clinical response, clinical remission, steroid-free remission, changes from baseline for PROs, and safety in a real-world cohort of patients with moderate-to-severe active CD treated with VDZ.
Design:
POLONEZ II was a multicenter, observational, prospective study across 10 Polish centers from April 2020 to October 2023 for 54 weeks in patients with CD eligible for reimbursed VDZ.
Methods:
Primary endpoints at week 54 (W54) were clinical response (⩾70-point reduction in CDAI and >25% reduction vs baseline), remission (CDAI score ⩽150), and steroid-free remission. Other outcomes were changes in PROs (CDAI score and health-related quality of life) and safety. Kaplan–Meier survival analyses were performed.
Results:
Of 98 patients with CD, the mean age was 35.2 years, 57.1% were male, and 72.4% had an ileocolonic disease. At W54 (n = 98), 63.3% of patients achieved clinical response, 48.0% remission, and 36.0% steroid-free remission. The durability of clinical response, remission, and steroid-free remission (W14–W54) were 68.9%, 62.9%, and 57.1%, respectively. By W54, a significant reduction in the PROs, such as the total CDAI score (p < 0.001), stool frequency (p < 0.001), abdominal pain score (p < 0.001), IBDQ (p < 0.001), IBD-F (p < 0.05), and fatigue impact on daily activities (p < 0.001), was observed. During VDZ treatment, arthralgia (23.7%–8.7%) and anemia (22.6%–15.9%) decreased between baseline and W54. Non-serious adverse events (SAEs; 12.2%), SAEs (7.1%), and VDZ-related rash (1.0%) were reported. Mean CD-related hospitalization duration decreased from baseline (10.2 days) to the end of the study (5.3 days).
Conclusion:
POLONEZ II demonstrated long-term real-world benefits of VDZ toward effectiveness, safety, and improved PROs and patients’ quality of life.
Trial registration:
ENCePP (EUPAS32716).
Plain language summary
The goal of this study was to thoroughly assess the effectiveness and safety of vedolizumab (VDZ), a monoclonal antibody, as a treatment for Crohn’s disease (CD), a weakening inflammatory gut condition. This study used a selection of assessment tools to measure patient-reported outcomes (PROs), which are commonly used to evaluate the benefits of a treatment in terms of features important to patients. These tools included the CD Activity Index (CDAI), which measures disease activity; the Inflammatory Bowel Disease (IBD) Questionnaire (IBDQ), which evaluates patients’ quality of life; and IBD-Fatigue (IBD-F), which assesses fatigue. Medical records of patients treated with VDZ for moderate to severely active CD for 54 weeks were examined. Treatment outcomes showed marked improvements, with 63.3% of patients showing a favorable response and fewer CD symptoms. Approximately 48.0% of patients achieved clinical remission, showing better control of their symptoms and a long period of symptom improvement. Notably, the benefits of the treatment continued during the treatment period, and a total of 57.1% of patients were corticosteroid-free while maintaining good outcomes. From the start of the trial until W54, the CDAI, IBDQ, and IBD-F scores decreased, likely owing to a reduction in disease symptoms. Patients also described decreased abdominal pain and a lower stool frequency. Furthermore, VDZ treatment reduced fatigue, a common problem in CD. Side effects were reported in 7.1% of patients with CD, with only one patient reporting a VDZ-related rash. Although some patients had to stop using VDZ due to the ineffectiveness of treatment, the majority of patients (67.3%) continued their treatment. Overall, VDZ effectively reduced CD symptoms and improved the patients’ quality of life while showing clinical effectiveness and a safety profile consistent with the approved label.
Keywords
Introduction
Inflammatory bowel disease (IBD) has two primary components: Crohn’s disease (CD) and ulcerative colitis (UC).1,2 As of 2020, the prevalence of CD in Poland was 61.6 per 100,000 individuals, with a total of 23,574 patients diagnosed. 3 CD is a chronic, relapsing, and remitting IBD2,4 that can affect any part of the gastrointestinal tract and may involve all layers of the gut.4,5 Symptoms include abdominal pain, diarrhea with mucus passage, 2 intestinal blockages resulting in bowel wall thickening, and nutritional deficiencies. 5 However, the precise etiology and pathogenesis of CD remain unclear. 4
Unraveling the complexity of disease manifestations in patients with CD can be challenging. 6 Patient-reported outcomes (PROs) are gaining prominence in clinical practice, as they encompass disease activity signs, symptoms, 7 and health-related quality of life (HRQoL). 8 PROs are valuable tools for evaluating disease activity and the impact of CD on patients. 7 Clinical trials often rely on the CD Activity Index (CDAI), a promising candidate as a PRO measure to continuously assess the effectiveness of new medical therapies. 9 CDAI is widely used within clinical practice in the clinical trial setting as a comprehensive measure for assessing disease activity in patients with CD. 6 Furthermore, the IBD Questionnaire (IBDQ) is commonly used to evaluate various domains related to 32 items and assesses HRQoL, including intestinal symptoms, systemic symptoms, social aspects, and emotional aspects of IBD. 10 In addition, the IBD-Fatigue (IBD-F) scale is designed to assess fatigue levels, particularly in patients with IBD. 11 Understanding fatigue is crucial as it is a burdensome symptom 11 affecting approximately 48%–62% of patients with CD 12 and, therefore, impacts patients’ quality of life and overall well-being. 13 It is crucial to understand symptoms in specific patient groups to develop outcome measures that can quantify changes in disease burden over time.6,14
Vedolizumab (VDZ) is a humanized monoclonal antibody that inhibits the trafficking of lymphocytes into the gut mucosa to regulate inflammation and can be used to treat moderately to severely active CD.15,16 This targeted mechanism of action suggests a favorable safety profile, with no increased risk of serious infections or malignancy usually associated with systemic immunosuppression.17 –19 VDZ received approval for the treatment of moderate-to-severe UC and CD from the European Medicines Agency and United States Food and Drug Administration in 2014, 16 based on the positive outcomes of three pivotal clinical trials: GEMINI 1 in patients with UC 20 and GEMINI 2 21 and GEMINI 3 22 in patients with CD. In the previous POLONEZ study, we assessed the real-world effectiveness and safety of 54-week VDZ treatment for induction and maintenance therapy in patients with UC in Poland. 23
In the POLONEZ II study, we evaluated the real-world effectiveness, including PROs and safety, of VDZ in patients with CD treated in a reimbursement program in Poland. Previously, we reported the interim results of the POLONEZ I study at week 14 (W14) and week 54 (W54).24,25 Here, we describe the long-term results and outcomes, including clinical response, clinical remission, steroid-free remission, changes from baseline in PROs including fatigue, and safety evaluation after W54 of VDZ treatment in patients with moderately to severely active CD.
Methods
Study design
POLONEZ II was a multicenter, observational prospective study of patients with moderately to severely active CD eligible for VDZ treatment in 10 centers in Poland between April 2020 and October 2023. Physicians prescribed VDZ according to local prescribing information and the reimbursement criteria, authorized by the Ministry of Health. Data were collected from patient’s medical records as documented in case report forms. VDZ was administered initially at W0, W2, and W6, followed by an additional dose at W10, if there was no response. If a response was observed, it was given every 8 weeks over a total of five study visits. Treatment had to be stopped after 2 years of use according to the predefined reimbursement criteria.
Study population
Eligibility for reimbursed treatment included patients aged ⩾18 years with moderately to severely active CD, indicated by a CDAI score of >300 points. Furthermore, these patients needed to have shown non-responsiveness, contraindication, or intolerance to previous treatment with corticosteroids, immunosuppressive drugs, or antitumor necrosis factor-α (anti-TNF-α) inhibitors. The study exclusion criteria, based on a local summary of VDZ characteristics and reimbursement criteria, were hypersensitivity to VDZ or its excipients; severe active or opportunistic infections; chronic heart, kidney, liver, or respiratory failure; demyelinating disease; precancerous conditions or malignancy diagnosed within the past 5 years; pregnancy; and breastfeeding.
Study procedures
We enrolled a total of 98 patients with CD who initiated VDZ treatment. The study protocol was approved by the Bioethics Committee at the Medical University of Lublin (Approval No.: KE-0254/1/2020). All patients provided written informed consent for participation in the study and the publication of de-identified details. Disease activity assessment encompassed the CDAI subscales, which included soft/liquid stool frequency (SF), and abdominal pain score (APS) derived from daily ratings over the previous week (score scale: 0 = none, 1 = mild, 2 = moderate, 3 = severe). The study considered disease phenotype classification using the Montreal classification scale, extraintestinal manifestations (EIMs), concomitant treatments involving steroids and immunomodulators, prior biologic therapies, CD-related hospitalizations, comorbidities, current VDZ treatment, and scores from the IBD-F and IBDQ assessments. The safety evaluation covered serious infections (as serious adverse events (SAEs)), other clinically significant infections (classified as moderate or severe, requiring antibiotic treatment but not SAEs), malignancies, infusion-related reactions, all other adverse events (AEs), SAEs, AEs of special interest, and pregnancy outcomes (full-term, pre-term, fetal loss/stillbirth, miscarriage, induced abortion). If a patient became pregnant during the study period, they discontinued the treatment and, as a consequence, the study.
Outcomes and safety
The primary endpoints assessed at W54 were clinical response (defined as a reduction of ⩾70 points in the CDAI and >25% compared with that at baseline), remission (defined as a CDAI score of ⩽150), and steroid-free remission (defined as a CDAI score of ⩽150 in patients who were using steroids at baseline but not at the time of assessment). Treatment groups were stratified by treatment history: biologic naïve (patients who were not subject to any prior biologic treatment due to CD at any time in the past), biologic exposed (patients who were previously exposed to biological treatment), and biologic failure (patients who had to terminate previous biological treatment for any reason, including treatment intolerance, primary lack of response to treatment according to the Drug Program, or loss of previous response to treatment). The secondary endpoints evaluated at W54 included changes from baseline in PROs, which included changes in HRQoL (measured using the IBDQ total score and subscales for bowel symptoms, emotional function, social function, and systemic symptoms), fatigue levels (assessed using IBD-F), SF, and APS. Safety outcomes were assessed by monitoring and coding AEs using the Medical Dictionary for Regulatory Activities (MedDRA; version 25.0).
Statistical analysis
The analysis incorporated data from the full analysis set (FAS), which included all participants who were recruited in the study. Descriptive statistics were employed to summarize the data, including the number of patients, mean, standard deviation (SD), minimum, median, and maximum for continuous variables, as well as frequencies for categorical variables. The study assessed selected secondary and exploratory endpoints. The study groups were confirmed using the paired Wilcoxon signed-rank test with Bonferroni corrections for multiple comparisons and the Chi-square test (or Fisher’s test) for qualitative variables, with the significance level set to 0.05.
The durability of response was determined by calculating treatment persistence, which refers to the maintenance of sustained response, remission, or steroid-free remission from W14 to W54. VDZ treatment persistence was assessed using Kaplan–Meier survival analysis.
Safety endpoints were evaluated through incidence rates utilizing person-time analyses. The safety analysis set comprised all VDZ-treated patients. AEs were coded using the MedDRA dictionary and reported as preferred terms and system organ classes. When applicable, 95% confidence intervals (CIs) were calculated. All statistical analyses were conducted using R version 4.0.3 or higher software (The R Foundation for Statistical Computing, c/o Institute for Statistics and Mathematics Wirtschaftsuniversität Wien, Vienna, Austria). 26
This manuscript was prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 27
Results
Patient disposition and baseline characteristics
In the overall population of 98 patients enrolled in the study, 66 (67.3%) were continuing VDZ treatment at W54 (Supplemental Figure 1). The primary reason for treatment discontinuation was lack (n = 2) or loss (n = 19) of response. Other reasons for discontinuation included lost contact with the patient (n = 4), surgical intervention (n = 2), AE (n = 1), participant withdrawal (n = 1), started biological treatment (n = 1), terminated after last dose due to occurrence of paraspinal abscess (n = 1), diagnosis of other disease (n = 1), and pregnancy (n = 1). The mean age (SD) at baseline was 35.2 (12.2) years. Of the total patient population, 57.1% (n = 56/98) were male and 42.9% (n = 42/98) were female (Table 1). Among all patients, 71.4% (n = 70/98) had never smoked, 16.3% (n = 16/98) were current smokers, and 12.2% (n = 12/98) were former smokers. The mean time (SD) from diagnosis to VDZ treatment initiation was 9.1 (6.3) years. According to the Montreal classification of current disease extent by age at diagnosis, 17.3% of patients were categorized as A1 (⩽16 years old), 69.4% as A2 (17–40 years old), and 13.3% as A3 (>40 years old). The most common disease location was ileocolonic, affecting 72.4% (n = 71/98) of patients, while only 1.0% (n = 1/98) had isolated upper disease. Approximately half of the patients (45.9%, n = 45/98) exhibited an inflammatory phenotype with a nonstricturing, nonpenetrating disease behavior. At baseline, 61.2% (n = 60/98) of patients did not have any EIMs, while 38.8% (n = 38/98) had EIMs. The most common EIMs at baseline were arthralgia (23.5%, n = 23/98) and anemia (23.5%, n = 23/98). At baseline, 51.0% (n = 50/98) of patients were prescribed corticosteroids, and 50.0% (n = 49/98) received immunomodulatory drugs.
Demographic, baseline, and clinical characteristics (FAS).

p Value for differences between groups.
Total number of soft/liquid stools in the last 7 days.
Sum of daily ratings from the past 7 days. Scale used for rating: 0 = none, 1 = mild, 2 = moderate, 3 = severe.
APS, abdominal pain score; BMI, body mass index; CDAI, Crohn’s Disease Activity Index; CRP, C-reactive protein; FAS, full analysis set; hs-CRP, high-sensitivity CRP; IBD, inflammatory bowel disease; N, number of patients; SD, standard deviation; SF, stool frequency.
Effectiveness outcomes
The median total CDAI score (range) decreased from 336.5 (301.0–483.0) to 108.0 (20.0–321.0) at W14 and to 110.0 (0.0–358.0) at W54 (p < 0.001; Table 1, Figure 1). For SF, the mean baseline score (from the CDAI subscales) (SD) for the 98 patients was 37.3 (19.5; Table 1). This score decreased to 14.4 (15.0) for 93 patients at W14 and further reduced to 14.1 (16.0) for 66 patients at W54 (p < 0.001). For APS, the mean baseline score (from the CDAI subscales) (SD) was 15.5 (5.5) for 98 patients (Table 1). At W14, this score reduced to 4.0 (4.9) for 93 patients and decreased further to 3.6 (4.7) for 66 patients at W54 (p < 0.001).
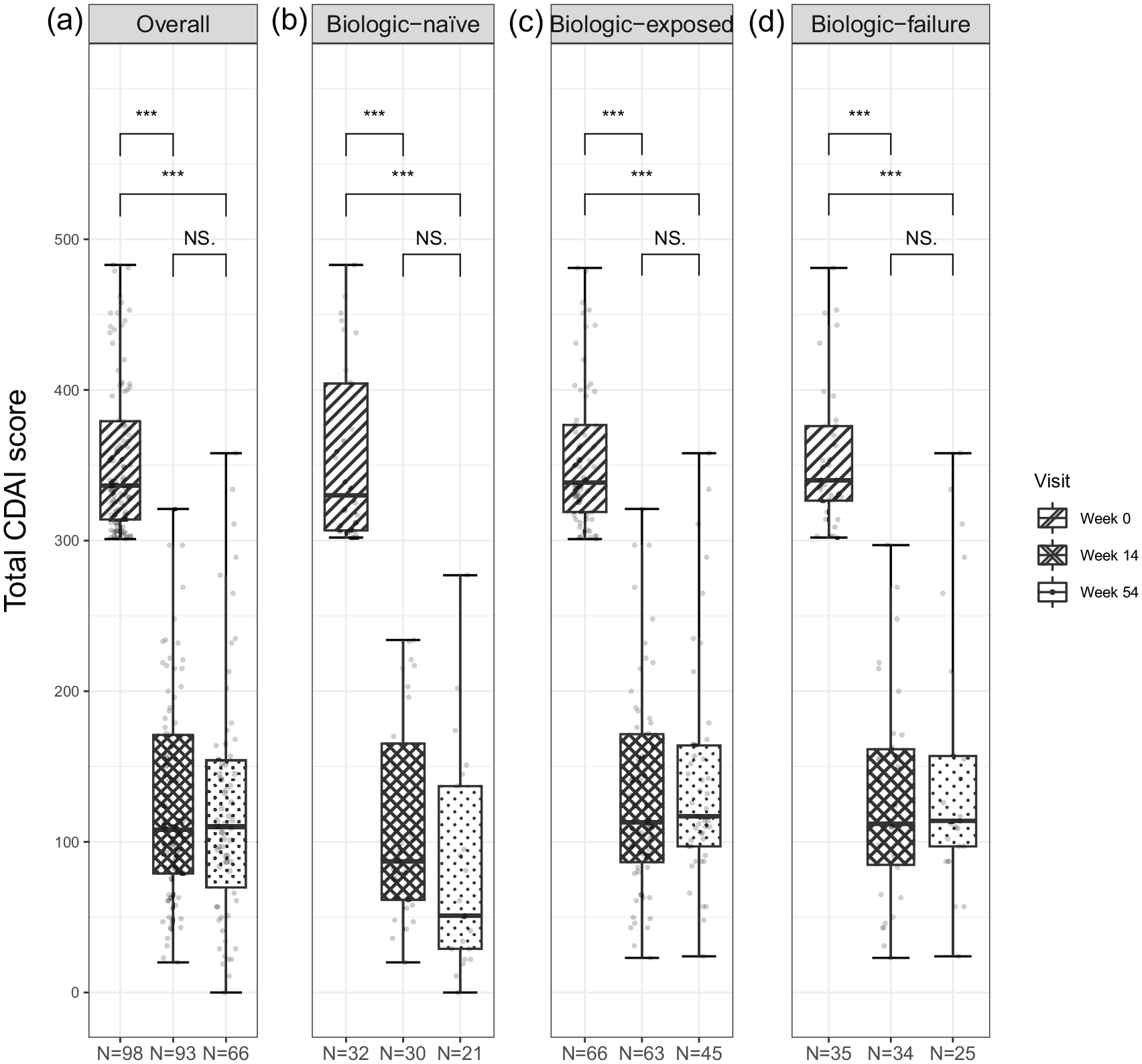
The CDAI total score for disease activity and clinical response assessment with CDAI (FAS). (a) Overall. (b) Biologic naive. (c) Biologic exposed. (d) Biologic failure.
At W0, the mean C-reactive protein (CRP)/high-sensitivity (hs)-CRP level (SD) for the 98 patients was 12.5 (22.9) mg/L (Table 1). This level decreased to 10.6 (14.8) mg/L for 93 patients at W14 and remained relatively stable at 10.4 (15.5) mg/L for 66 patients at W54.
EIMs during VDZ treatment decreased from W0 values of 38.7% (n = 36/93) to 25.8% (n = 17/66) at W54 (Supplemental Table 1). Furthermore, arthralgia decreased from 23.7% (n = 22/93) at W0 to 8.7% (n = 6/66) at W54, and anemia decreased from 22.6% (n = 21/93) to 15.9% (n = 11/66).
Clinical response and remission
At W14, clinical response was 91.8% (n = 90/98), and clinical remission was achieved in 63.3% (n = 62/98) of patients (Table 2, Figure 2(a) and (b)). By W54, clinical response decreased to 63.3% (n = 62/98) of patients, and the number of patients in clinical remission was 48.0% (n = 47/98, Supplemental Figure 1). Durability was defined as the maintenance of sustained response, remission, or steroid-free remission from W14 to W54. The durability of clinical response from W14 to W54 was reported in 68.9% (n = 62/90) of patients, while the durability of clinical remission was reported in 62.9% (n = 39/62) of patients.
Clinical response and remission at W14 and W54 (FAS).

Patients with sustained clinical response/remission/steroid-free remission maintained from W14 to W54.
CI, confidence interval; FAS, full analysis set; N, number of patients; W, week.

Primary endpoints of disease activity and assessment at W14 and W54. (a) Clinical response based on the pMayo score. (b) Clinical remission based on the pMayo score. (c) Steroid-free remission assessment using CDAI (FAS). Clinical response was defined as a reduction in the CDAI score ⩾70 points and >25% compared with the baseline value at W14 (or any of the following visits). Clinical remission was defined as achieving a CDAI total score of <150 points. Steroid-free remission was defined as achieving a CDAI total score <150 points in patients not using steroids at the time of assessment but who were using them at baseline.
Among the 50 patients analyzed for steroid-free remission, 28.0% (n = 14/50) achieved this outcome at W14 (Table 2, Figure 2(c), Supplemental Figure 1). By W54, the percentage of patients in steroid-free remission had increased to 36.0% (n = 18/50). The durability of steroid-free remission was reported in 57.1% (n = 8/14) of patients.
Treatment patterns
The mean number of VDZ doses (SD) was 7.5 (3.0) in the overall population (N = 98). Eleven patients (11.8%) received an additional induction dose at W10, and only 10 patients required intensification during maintenance therapy (Supplemental Table 2). Additional doses of VDZ at W10 were received by 13.3% of biologic-naïve patients, 11.1% of biologic exposed patients, and 11.8% of biologic-failure patients. Following the additional doses of VDZ at W10, response and remission were 100.0% (n = 11/11) and 63.6% (n = 7/11) in the overall population, 100.0% (n = 4/4) and 50.0% (n = 2/4) in biologic-naïve patients, 100.0% (n = 7/7) and 71.4% (n = 5/7) in biologic exposed patients, and 100.0% (n = 4/4) and 75.0% (n = 3/4) in biologic-failure patients, respectively. Compared with the induction phase, the need for intensification during maintenance therapy was higher in the overall population (14.5%), biologic exposed patients (19.1%), and biologic-failure patients (20.0%) and lower in biologic-naïve patients (4.5%). Response and remission following an additional dose at W10 during both phases ranged from 80.0% to 100.0% and from 0% to 33.3%, respectively.
The total analysis showed that the percentage of patients without concomitant corticosteroid use increased by ~1.6-fold from baseline to W54 (49.0%, n = 48/98 vs 78.8%, n = 52/66). The proportion of patients (responders) on corticosteroid treatment over time is illustrated in Figure 3(a). For responders, the percentage of patients without concomitant corticosteroid use increased by 1.5-fold from W0 to W54 (51.1%, n = 46/90 vs 78.8%, n = 52/66). Of those responders (n = 36) with a corticosteroid dose reduction (83.3%, n = 30/36), 73.3% (n = 22/30) had a 100% dose reduction at W54 (corticosteroid-free; Figure 3(b)). Current nonbiologic treatment from baseline to W54 (corticosteroids (prednisone, methylprednisolone, and budesonide), immunomodulatory drugs (azathioprine, mercaptopurine, and methotrexate), and sulfasalazine/mesalazine drug intake) and doses are shown in Supplemental Table 3.

(a) Corticosteroid intake up to W54 and (b) CS dose reduction at W54 (responders).
Kaplan–Meier curves demonstrated the probability of VDZ persistence over 54 weeks (Figure 4). Initially, a slight decline in persistence was observed during the early weeks (discontinuation); however, it stabilized thereafter and remained generally consistent until W54. The number of patients at risk of relapse decreased from W0 to W54.

Kaplan–Meier analysis of treatment persistence (FAS).
Patient-reported outcomes
Median changes from the baseline IBDQ total score were statistically significant at both W14 and W54 (p < 0.001; Figure 5(a)). Median changes from the baseline IBDQ total score were also statistically significant in all the subgroups of biologic-naïve, biologic exposed, and biologic-failure patients at W14 (Figure 5(b)–(d)) and in biologic-naïve and biologic exposed patients, but not in biologic-failure patients at W54 (Figure 5(b)–(d)). Similarly, median changes from baseline in overall IBDQ subscales (bowel symptoms, systemic symptoms, emotional function, and social function) were statistically significant at both W14 and W54, (p < 0.001; Figure 6 and Table 3).

Effect of VDZ on the IBDQ scores (overall FAS). The effect of VDZ induction therapy on IBDQ total score. (a) Overall. (b) Biologic naïve. (c) Biologic exposed. (d) Biologic failure.
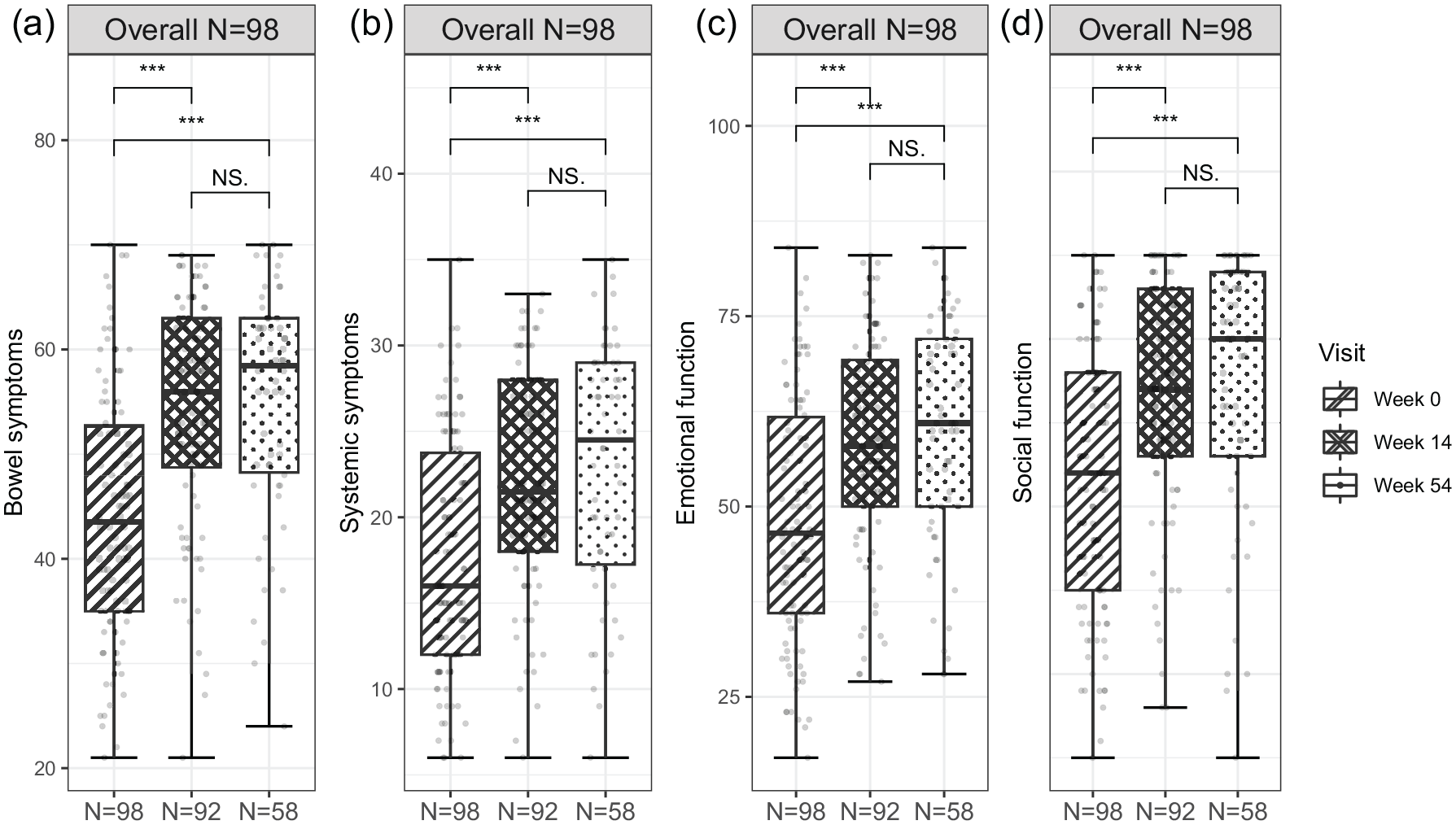
Effect of VDZ on the IBDQ scores (overall FAS). The effect of VDZ induction therapy on (a) bowel symptoms, (b) systemic symptoms, (c) emotional function, and (d) social function. Boxes correspond to median values and interquartile ranges, and error bars represent minimum−maximum values.
Impact of therapy with vedolizumab on patients’ quality of life measured with IBDQ score (FAS).

p-Value for Wilcoxon signed-rank test comparing W0 with the following visits.
95% CI for the mean difference was calculated using the bootstrap method for patients with results available on both compared visits.
P values were adjusted using the Bonferroni–Holm approach: *p < 0.05. **p < 0.01. ***p < 0.001.
CI, confidence interval; FAS, full analysis set; IBDQ, Inflammatory Bowel Disease Questionnaire; SD, standard deviation; W, week.
Statistically significant improvements were also observed from baseline to W14 in the IBD-F severity and frequency scores in the overall population (Figure 7(a); p < 0.001) as well as in the subgroups of biologic-naïve, biologic exposed, and biologic-failure patients (p < 0.05; Figure 7(b)–(d)). At W54, the median change from baseline in the IBD-F severity and frequency scores remained statistically significant in the overall population but was no longer significant in the three subgroups.

Effect of VDZ on fatigue severity and frequency (IBD-F Questionnaire). (a) Overall. (b) Biologic naïve. (c) Biologic exposed. (d) Biologic failure.
Improvement in IBD-F impact on daily activities from baseline to W14 was significant in the overall population (p < 0.001) as well as in the subgroups of biologic-naïve (p < 0.05), biologic exposed (p < 0.001), and biologic-failure (p < 0.01) patients (Figure 8(a)–(d)). At W54, the change from baseline in IBD-F impact on daily activities remained significant in the overall population (p < 0.001) and biologic exposed (p < 0.001) and biologic-failure (p < 0.01) subgroups but was no longer significant in the biologic-naïve subgroup (Figure 8(a)–(d)).

Effect of VDZ on fatigue impact on daily activities (IBD-F Questionnaire). (a) Overall. (b) Biologic naïve. (c) Biologic exposed. (d) Biologic failure (FAS). Boxes correspond to median values and interquartile ranges, and error bars represent minimum−maximum values.
Safety outcomes
Overall, 12.2% (n = 12/98) of patients experienced 26 AEs with 16 nonserious AEs (non-VDZ-related), and 7.1% (n = 7/98) experienced 10 SAEs (Supplemental Table 4). One patient (1.0%, n = 1/98) experienced rash as a serious VDZ-related AE (incidence of 1.3 per 100 patient-years). Furthermore, a decrease in hemoglobin levels was observed in 6.1% (n = 6/98) of patients, with an incidence of 10.2 per 100 patient-years.
Hospitalizations 12 months before VDZ treatment (FAS) and emergency department (ED) visits and hospitalizations during VDZ treatment (FAS) are shown in Table 4. In the 12 months prior to the VDZ treatment, 38.8% (n = 38/98) of patients were hospitalized once due to exacerbation of CD. This number decreased to 2.2% (n = 2/93) and 1.5% (n = 1/68) at W14 and W54, respectively. Data for patients with two or more than two CD-related exacerbations before starting VDZ treatment, at W54, and at the end of the interim study are shown in Table 4. The mean (SD) total duration of hospitalizations due to CD exacerbations decreased in the total population from 10.2 (9.3) days at baseline to 5.3 (3.4) days until the end of the interim study.
Hospitalizations and ED visits due to CD exacerbations (up to 12 months before enrollment to NDP and during VDZ treatment (FAS)).

Hospitalizations from W0 until the end of the Interim period (W54 or Early termination). Data on hospitalizations, ED visits, or length of hospitalization for this category are aggregated per patient. If a Subject had an event reported at a W14 and W54 visit, this category presents the sum of events during the Interim period for the patient.
Tested differences between all groups, biologic-naïve, biologic exposed, and biologic-failure patients.
Presented as the sum of the length of all hospitalizations for a given patient per visit, if there was more than one hospitalization reported on a given visit.
CD, Crohn’s disease; ED, emergency department; FAS, full analysis set; NDP, National Drug Program; SD, standard deviation; VDZ, vedolizumab; W, week.
Discussion
Within the framework of POLONEZ II, we studied the clinical outcomes and treatment patterns of patients with moderately to severely active CD. To the best of our knowledge, this is the first study to examine the PROs and HRQoL of VDZ for CD in a Polish real-world cohort for 54 weeks. The clinical response showed statistically significant improvements across all patient groups (biologic naïve, biologic exposed, and biologic failure), independent of prior treatment response or exposure at W54. At W54, approximately two-thirds of patients responded to VDZ treatment, with 48.0% achieving clinical remission and durability of clinical response of 68.9%. This study not only provides valuable insights into the durability of treatment response but also addresses the long-term durability and safety of VDZ. These findings offer valuable information for Poland and other regions around the world.
LOw countries VEdolizumab in CD (LOVE-CD) was a prospective, multicenter trial conducted in Belgium and the Netherlands that enrolled 110 patients with active CD who had a CDAI score of >220. 28 During the LOVE-CD trial, patients received VDZ treatment and assessments were made at W26 and W52. 28 At baseline, the median CDAI score was 261, and the trial demonstrated mean decreases in the CDAI score of 131.5 and 124.8 at W26 and W52, respectively. 28 The greater reduction in the CDAI score from baseline to W54 (356.3–123.3) observed in our study than in LOVE-CD (261) 28 may be attributed to the higher baseline CDAI score in the POLONEZ II study, which is indicative of a greater severity of disease at the beginning of the study. Furthermore, in the LOVE-CD trial, at W26, 32 patients (29%) were in corticosteroid-free remission (CDAI score < 150), and at W52, 34 patients (31%) achieved the same remission status. 28 In our study, in the overall population, steroid-free remission (CDAI score ⩽150) was similar (28.0%) at W14 but higher (36.0%) at W54.
In recent long-term real-world studies,29 –33 significant findings have been reported regarding the clinical outcomes of patients with CD. Notably, among these studies, a Swedish cohort of 286 patients with active CD (n = 169) treated with VDZ showed that the 52-week continuation rate of VDZ was 60% (n = 102/169), similar to that observed in our study (64.3% at W54). 33 Furthermore, at W52, 38 patients (22%) showed a clinical response, 69 (41%) were in remission (with no differences between biologic-naïve and biologic-failure groups), and 67 (40%) achieved corticosteroid-free remission, which were all lower than the rates reported in the POLONEZ II study. 33 In the Swedish cohort, 41% of the patients with CD had experienced at least one surgical resection and 75% had elevated CRP levels (CRP higher than the lower limit of detection, hs-CRP >2.87 mg/L). 33 In our study, at W0, the mean CRP/hs-CRP was 12.5 mg/L, also indicating elevated levels (CRP higher than the lower limit of detection, hs-CRP >2.87 mg/L). 33
In a retrospective Israeli cohort study of 193 patients with IBD, of whom 133 had CD, the observed clinical response and remission rates at W52 following VDZ treatment were lower than those observed in our study, at 46.6% (n = 62/133) and 21.1% (n = 28/133), respectively. 29
Regarding clinical response, our findings at W54 revealed a favorable response in 63.3% (n = 62/98) of patients, indicating positive progress in their condition. Within the same timeframe, clinical remission was favorable, with 48.0% (n = 47/98) of the overall population achieving remission at W54. An Italian study demonstrated a similar clinical benefit rate (rate of clinical steroid-free remission plus clinical response) of approximately 68%, with patients diagnosed with CD achieving a clinical benefit of 59.4% after the induction phase. 32 Similarly, our study showed an impressive durability of steroid-free remission at 57.1%. Among the 50 patients analyzed specifically for steroid-free remission, 36.0% (n = 18/50) achieved this outcome at W54. We also assessed the treatment patterns and found that the percentage of patients without concomitant corticosteroid use increased from 49.0% at W0 to 78.8% at W54. We defined steroid-free remission as achieving a CDAI total score of ⩽150 points in patients with steroid use but not at the time of assessment. Zingone et al. 32 reported improved steroid-free remission in patients with CD receiving VDZ (53.6%, n = 37/69), where steroid-free remission was defined as a partial Mayo (pMayo) score of <2 or Harvey–Bradshaw Index (HBI) score ⩽4 without steroid use.
An Italian multicenter, real-world study evaluated 172 patients with CD and UC with a median age of 66.0 years and a 58.8-week median follow-up. 31 At W52 of treatment, the clinical response to VDZ was 77.4% in patients with CD, which was higher than that reported in our biologic-naïve group (65.6%) at W54 (Table 2). 31 In a German study, clinical response was defined as a reduction of ⩾3 in the HBI score for CD, 31 whereas in our study, it was defined as ⩾70-point reduction in the CDAI and >25% versus W0. Notably, the median age of patients in the German study (66.0 years) 31 was higher than that of patients in our cohort (mean age, 35.1 years). Furthermore, regarding steroid-free remission, the German group reported that 59.7% of biologic-naïve patients with CD using VDZ achieved steroid-free remission, defined as HBI < 5. 31
An Italian study retrospectively collected data on 117 patients with IBD, of whom 69 had CD (median age 47 years). 32 The study demonstrated a clinical benefit rate (rate of clinical steroid-free remission plus clinical response) of approximately 68%, with patients diagnosed with CD achieving a clinical benefit of 59.4% after the induction phase. 32 A retrospective, multicenter medical chart review conducted in Germany evaluated the real-world effectiveness of VDZ or anti-TNF-α on clinical remission and symptom resolution in biologic-naïve patients with moderate-to-severe endoscopic disease. 30 By W26, estimated clinical remission rates were 14.4% for patients treated with VDZ and 32.8% for those on other anti-TNF-α therapies. 30 Similarly, another real-world prospective study in Germany based on the VEDOIBD registry demonstrated that clinical remission after 2 years of treatment in biologic-naïve patients with CD was significantly higher in patients treated with VDZ (74.2%) compared with patients treated with anti-TNF-α therapies (44.7%). 34
Remission rates were higher in biologic-naïve VDZ-treated patients compared with those with prior anti-TNF-α experience similar to our study.30,34 In the retrospective German study, remission rates in the biologic-naïve cohort were 25.0% for VDZ and 33.7% for patients treated with anti-TNF-α therapy, while in a subcohort, with a single prior anti-TNF-α experience, rates were 13.0% and 33.0%, respectively, by W26. 30 In comparison, our study reported higher clinical remission rates of 65.6% and 53.1% in biologic-naïve patients at W14 and W54, respectively. Interestingly, at baseline, the German patients with CD received VDZ treatment (14%, n = 69), whereas the remainder received anti-TNF-α. 30 Of the patients receiving VDZ, the majority were female (67%), with a median age and disease duration of 41.0 and 9.8 years, respectively. 30 By contrast, our patient cohort had a slightly lower mean age of 35.2 years, with 42.9% being female, and a mean time from diagnosis of 9.1 years. These variations in clinical remission rates between the two biologic-naïve cohorts may be attributed to factors such as sex, age, and timing of treatment initiation during the disease course. It is worth considering that treatment initiation earlier in the disease course, by approximately 1 year in our study, may have also contributed to improved outcomes. Furthermore, ileal and colonic CD may have distinct disease characteristics that influence treatment responsiveness. 35 In the German cohort, 50% of patients receiving VDZ had ileocolonic CD, 25% had stricturing disease, and 11% had penetrating disease. 30 In our Polish cohort, 72.4% of patients had an ileocolonic disease, 27.6% had a stricturing disease, and 26.5% had a penetrating disease. In the future, further categorizing different cohorts into subtypes of the disease, particularly distinguishing between ileal dominant (isolated ileal and ileocolonic) and isolated colonic disease, may provide additional insights into CDAI responses or other responses to CD therapies.
Meserve and Dulai 36 showed that positive predictors that influence clinical response and remission include baseline disease activity and demographic and baseline characteristics. In a German prospective study, a consecutive cohort of 212 adult patients with IBD together with active disease (HBI > 7/pMayo score > 4) who were newly receiving VDZ were recruited from academic and community centers. 37 Significant associations between clinical remission and indicators of lower disease activity or a less complicated disease course were reported. Factors such as absence of recent hospitalizations, low disease activity, a less complicated disease course (low HBI score at baseline), or no history of EIMs were identified as good predictors of clinical remission. 37
In an Israeli cohort study of 130 patients with CD, 116 (89.2%) completed a 14-week VDZ induction period. 38 Among the participants, 44 (33.5%) had moderate clinical disease activity and 41 (31.8%) had severe clinical disease activity. 38 Notably, the study highlighted that mild clinical disease activity at treatment onset was associated with increased clinical remission at W14. 38 It is important to note that the mean time from diagnosis to treatment in our study was 9.1 years, suggesting a significant gap between diagnosis and initiation of VDZ treatment, while the Israeli study reported a mean ± SD disease duration of 10 ± 6 years. 38 Our study shows that, in Poland, patients seem to encounter longer delays and present with more severe disease before commencing treatment. Collectively, these findings highlight a potential opportunity to review access to biological therapy in Poland and implement measures to improve patient outcomes.
Interestingly, the CALM study showed that a CRP-based treatment algorithm for monitoring inflammation and symptoms yielded improved outcomes compared with clinical management alone in early CD. 39 When examining the influence of VDZ treatment on CRP and hs-CRP levels, we found that the mean baseline CRP/hs-CRP level in our cohort of 98 patients was 12.5 mg/L, which decreased to 10.4 mg/L at W54 (n = 66). The decrease in CRP/hs-CRP levels observed in our study suggests a reduction in inflammation, supporting the positive clinical response observed in most patients at W14. Although a slight decline in response and remission rates was observed by W54, a considerable proportion of patients maintained an improved health status versus baseline with reduced inflammation over the long term. Therefore, VDZ is a viable option for patients with moderately to severely active CD who are refractory to corticosteroids, immunosuppressive drugs, or other TNF-α inhibitors. Because of its limited sensitivity in detecting active intestinal inflammation in CD, CRP is not considered a reliable primary endpoint 40 ; however, no equally strong alternative serum markers are currently available to replace CRP. 41
In our study, durability was defined as the maintenance of response/remission/steroid-free remission from W14 to W54. The durability of the clinical response was 68.9% (n = 62/90), remission was 62.9% (n = 39/62), and steroid-free remission was 57.1% (n = 8/14). The discontinuation rate of VDZ after 3 months of therapy was similar to that reported in other studies,32,42 mainly because of the loss of response (34.3%).
A retrospective study of 95 patients with IBD, of which 58 had CD, showed that VDZ discontinuation occurred at a median of 17.5 months. 43 After discontinuing VDZ therapy, approximately two-thirds of patients with IBD who achieved clinical remission experienced relapse within the first year. 43 However, when these patients (n = 24) were retreated with VDZ, steroid-free remission was achieved in 71% of patients. 43 Furthermore, following VDZ retreatment, a median follow-up of 11.0 months revealed that 62.5% (n = 15/24) of patients remained in clinical remission while receiving VDZ therapy. 43
In our study, from W0 to W14, 31.6% (n = 31/98) of patients did not respond to treatment. Other studies have reported different discontinuation rates. For example, the Israeli cohort reported that a high percentage of patients (55.0%, n = 33/60) discontinued VDZ treatment or did not respond to treatment. 29 In the French cohort, 22 patients with IBD discontinued VDZ at the start of treatment, and 20 patients with CD chose to discontinue VDZ treatment after the W17 follow-up. 44 The number of patients who discontinued therapy increased from W14 to W54, and reasons for discontinuation included lack of clinical efficacy, surgery, pregnancy, and loss of follow-up. 44
Our study showed that arthralgia was the most common EIM, and it decreased from 23.7% at W0 to 8.7% at W54, indicating an improved HRQoL. Another recent multicenter, descriptive study that evaluated the effect of VDZ on EIMs in a real-world cohort of patients with IBD reported arthralgia as the most frequent EIM. 45 The study demonstrated resolution of all EIMs in 25.3% of patients and improvement in 49.5% of all EIMs within 12 months of VDZ treatment. 45 Similarly, in another study that evaluated the real-world effectiveness of VDZ for EIMs in patients with IBD, arthralgia was the most common EIM. 46 At W54, 44.7% of patients had complete remission for inflammatory arthralgia/arthritis, which was shown to be associated with clinical remission of IBD and recent onset of inflammatory arthralgia/arthritis. 46
The IBDQ is the most commonly reported disease-specific PRO measure used in clinical trials (85%). 41 Our study encapsulates both traditional gastrointestinal symptoms and nontraditional symptoms, such as social and emotional function and fatigue, which are critical for patient well-being. Fatigue is a major concern in patients with CD, but data in this area are scarce. 47 In a study conducted in Norway, which included 440 patients with IBD, patients with active disease had significantly higher fatigue scores than those with quiescent disease. 47 Specifically, the fatigue score for patients with active CD was 17.5, whereas that for patients with quiescent disease was 13.3 (p < 0.001). 47 Interestingly, the fatigue scores of patients with quiescent disease were comparable with those of the reference population. 47 Factors associated with fatigue in patients with IBD include self-perceived disease activity, poor sleep quality, anxiety, and depression. 47
Our study demonstrated significant improvements from W0 in the IBDQ scores. Notably, there was an improvement from baseline to W14 in median IBDQ total overall scores after VDZ treatment, which was maintained until W54 in all subgroups apart from biologic-failure patients. Remission rates were higher in biologic-naïve VDZ-treated patients compared with biologic exposed patients during our study, but this did not appear to have negatively impacted the IBDQ total score, which was maintained at W54 in both these subgroups. As might have been expected, the positive impact of VDZ treatment on the IBDQ total score was no longer significant for biologic-failure patients at W54. However, due to the low patient numbers included in the subgroups, the results should be interpreted with caution.
IBDQ total subscale scores (bowel and systemic symptoms and emotional and social function) showed improvements from baseline to W14 and W54. This corresponded with the positive change in CDAI scores, which improved from baseline over the course of 54 weeks.
In addition, the assessment of disease-specific HRQoL incorporated IBDQ that targeted individual domains, 48 and we used IBD-F to particularly focus on fatigue. In our study, patients experienced notable improvements in fatigue severity and frequency scores, as measured using IBD-F, in combination with a significant reduction in impact on daily activities.
VDZ is a gut-selective monoclonal antibody directed against α4β7 integrin. The decrease in inflammatory activity observed after VDZ treatment in the current study, as demonstrated by a reduction in CRP levels, corresponded to a positive clinical response at W14. Small decreases in response and remission rates were observed by W54, but many patients maintained an enhanced health status compared with that at baseline, thereby exhibiting decreased long-term inflammation post-VDZ treatment. This positive clinical response may have translated into the improvements in PROs observed in our study. This is also in agreement with a recent post hoc analysis of patients with moderately to severely active UC and CD enrolled in the VISIBLE 1 and 2 phase III studies, where an early improvement in patient-reported symptoms was documented in patients with UC and CD receiving VDZ induction therapy. 49 The range of validated assessment tools used in the current study provides further evidence of the consistent positive disease-specific and global benefits on well-being provided by long-term VDZ therapy.
Several clinical trials assessing VDZ examined AEs and SAEs, with reported findings indicating comparable rates between the VDZ and placebo groups. 20 In a study undertaken by Sandborn et al., 21 during the maintenance phase, AE and SAE rates were 20.1% and 24.4% for VDZ and 21.6% and 15.3% for placebo, respectively. The phase III GEMINI LTS is the largest and longest clinical study of VDZ therapy. 50 The GEMINI LTS enrolled patients with UC (n = 894) or CD (n = 1349) from four previously conducted trials, including VDZ-naïve patients. 50 The study evaluated the long-term effectiveness of VDZ (300 mg IV every 4 weeks), with a focus on efficacy and PROs. 50 The median cumulative exposure to VDZ was 31.5 months for CD. Over the course of 8 years, AEs were observed in 96% of patients with CD, with CD exacerbations being most frequently reported (35%). 50 SAEs were reported in 41% of patients with CD, and VDZ was discontinued in 17% of patients. 50 No new trends were reported for infections, malignancies, infusion-related reactions, hepatic events, or progressive multifocal leukoencephalopathy. 50 The authors concluded that the safety profile of VDZ was favorable, with no unexpected or new safety concerns. 50
In accordance with the findings of GEMINI LTS, 50 our study revealed a low incidence of SAEs. Among the 98 patients included in the study, only 7 (7.1%) experienced serious AEs until the end of the interim analysis period. Notably, one patient (1.0%) encountered a rash judged as a VDZ-related SAE by the investigator, with an estimated incidence rate of 1.3 per 100 patient-years.
The progressive nature of CD often necessitates hospitalization and possibly surgery; however, this has changed over the past few decades owing to the advent of new and progressive therapies to manage CD.51,52 In the population-based “IBD South Limburg cohort” study, which followed 1162 patients with CD from 1991 and 2014, there was a notable increase in the percentage of patients receiving immunomodulators within the first 5 years of treatment (30.6%–70.8%). 51 In addition, the biologics use increased (3.1%–41.2%), the risk of hospitalization decreased (65.9%–44.2%), and the risk of bowel resection decreased (42.9%–17.4%) over the same 5-year period. 51 However, a nationwide study from Portugal showed that during 2000–2015, the CD-related hospitalization rates per 100,000 citizens increased to 8.4–11.2. 52 Zhao et al. 53 reported that in 2020, approximately 50% of patients with CD in Europe were hospitalized once within 5 years since diagnosis. Hospitalization rates for patients with CD differed significantly between European countries. For example, the mean (SD) medical hospitalization rate in Denmark was 4.8 (13.9) days per patient-year, which was 12 times higher than that in Norway at 0.4 (2.6) days per patient-year. 54 This variability highlights the subjective nature of hospitalization and emphasizes the need to interpret rates within local practices with caution. In our study, we observed a decrease in the number of patients visiting the ED due to exacerbations. Hospitalizations due to CD at W0, W14, and W54 showed that 38.8% (n = 38/98), 2.2% (n = 2/93), and 1.5% (n = 1/66) experienced one exacerbation and were hospitalized, respectively. By the end of the study, 3.2% (n = 3/93) experienced one hospitalization. In addition, the total duration of hospitalization due to exacerbations for patients with CD decreased from a mean of 10.2 to 5.3 days at the end of the study (p = 0.35). These findings indicate an improvement in the frequency of ED visits and exacerbations over the course of the study. These findings also highlight the positive outcomes and treatment patterns observed in our study, underscoring the efficacy and safety of VDZ in managing CD. Nevertheless, it is essential to acknowledge the inherent limitations of our research, including the specific study population.
This study provides data on the long-term use and safety of VDZ in a real-world clinical setting in Poland. Long-term follow-up allowed us to observe the dynamics of changes with relatively rapid clinical effects. This study marks the first instance of capturing PROs for VDZ in the context of CD. PROs offer a patient-centric perspective, capturing subjective experiences not captured by clinical assessment alone. Clinicians can better understand the treatment response, intervention effectiveness, and treatment satisfaction through PROs.
Due to sampling bias, the study population may not be representative of the broader population, limiting the generalizability of the study results. Limited external validity and selection bias were observed because only patients in the National Drug Program were enrolled. Therefore, this population may not reflect the diversity of patients with CD in terms of demographic characteristics, disease severity, comorbidities, and previous treatment experiences. No formal statistical calculations were performed. The small sample size (N = 98) could have affected the ability to detect significant differences. The potential response bias due to the selected population may provide socially desirable or biased responses.
Conclusion
The results of our study elucidate the real-world use of VDZ in patients with moderately to severely active CD treated for 54 weeks and offer encouraging evidence of the effectiveness of VDZ in improving PROs, including HRQoL, alleviating fatigue, and minimizing the effect of CD on daily activities. VDZ was effective in maintaining clinical remission in approximately half of the patients, with beneficial effects on disease activity. Moreover, VDZ was generally well tolerated, with a low incidence of serious AEs in patients with moderately to severely active CD. These findings contribute to our understanding of the role of VDZ in managing CD and provide a basis for further exploration and optimization of treatment strategies to benefit patients globally. To provide more comprehensive insights into treatment outcomes and ensure the generalizability of our findings, further research involving larger-scale studies and diverse patient populations is warranted.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241293938 – Supplemental material for Effectiveness and safety outcomes after long-term (54 weeks) vedolizumab therapy for Crohn’s disease: a prospective, real-world observational study including patient-reported outcomes (POLONEZ II)
Supplemental material, sj-docx-1-tag-10.1177_17562848241293938 for Effectiveness and safety outcomes after long-term (54 weeks) vedolizumab therapy for Crohn’s disease: a prospective, real-world observational study including patient-reported outcomes (POLONEZ II) by Ariel Liebert, Maria Kłopocka, Agata Michalak, Halina Cichoz-Lach, Renata Talar-Wojnarowska, Danuta Domz.ał-Magrowska, Łukasz Konecki, Aleksandra Filipiuk, Michał Krogulecki, Maria Kopertowska-Majchrzak, Kamila Stawczyk-Eder, Katarzyna Waszak, Piotr Eder, Edyta Zagórowicz, Izabela Smoła, Krzysztof Wojciechowski and Szymon Drygała in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors did not receive any direct or indirect compensation for the development of this manuscript. Writing, editorial support, and formatting assistance for this manuscript were provided by Reham M. Milhem, PhD, of Cactus Life Sciences (part of Cactus Communications), which was contracted and compensated by Takeda for these services.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
